Abstract
Low sexual desire is a prevalent symptom, but not one frequently volunteered by women. When accompanied by distress, loss of libido is known as hypoactive sexual desire disorder, which can have a significant impact on a woman's wellbeing. The etiology of hypoactive sexual desire disorder is multifactorial and its management requires a combination of psychosocial and pharmacological interventions. This article outlines the assessment of patients presenting with the symptom of low sexual desire and discusses the evidence for pharmacological management.
 Medscape: Continuing Medical Education Online
Medscape: Continuing Medical Education Online
This activity has been planned and implemented in accordance with the Essential Areas and policies of the Accreditation Council for Continuing Medical Education through the joint sponsorship of Medscape, LLC and Future Medicine Ltd. Medscape, LLC is accredited by the ACCME to provide continuing medical education for physicians.
Medscape, LLC designates this Journal-based CME activity for a maximum of
All other clinicians completing this activity will be issued a certificate of participation. To participate in this journal CME activity: (1) review the learning objectives and author disclosures; (2) study the education content; (3) take the post-test with a 70% minimum passing score and complete the evaluation at www.medscape.org/journal/wh; (4) view/print certificate.
Learning objectives
Upon completion of this activity, participants should be able to:
Distinguish the most common age associated with the diagnosis of HSDD
Perform an adequate assessment of women with suspected HSDD
Analyze treatment options for women with HSDD
Low sexual desire is a common problem amongst women of all ages. When associated with distress, it is termed hypoactive sexual desire disorder (HSDD), which is estimated to affect approximately one in ten women [1]. Unfortunately, women are often reluctant to report the symptom of low sexual desire, despite the fact that it is a prevalent and distressing condition for many women, associated with a range of negative effects on women's health. This article aims to give a practical overview on the identification and management of this disorder by reviewing the most recent and relevant literature on the subject.
Female sexual function
Understanding of female sexual function has developed significantly from the traditional linear, biologically determined models of human sexual response by Masters and Johnson in 1966 [2]. Their model divided the sexual response into four distinct phases: excitement, plateau, orgasm and resolution and was later modified by Kaplan to incorporate desire [3]. Increased recognition of the complexity of female sexual function has led to the evolution of nonlinear models that recognise the importance of nonbiological factors, such as emotional intimacy, relationship satisfaction and sexual stimuli along with other psychosocial factors [4]. However, the question for clinical practice is: has this improved understanding led to improved management of low sexual desire?
Female sexual dysfunction is a term that covers several aspects of sexual health and has been classified by the American Psychiatric Association into distinct disorders of desire, arousal, orgasm or pain [5]. Although this article will focus on the diagnosis and management of low sexual desire, there is often considerable overlap between the sexual disorders, and their coexistence must be considered when assessing patients.
Defining low sexual desire
Hypoactive sexual desire disorder is currently defined by The American Psychiatric Association's Diagnostic and Statistical Manual of Mental Disorders 4 (DSM-IV) as a persistent or recurrent deficiency or absence of sexual fantasies and desire for sexual activity that causes marked distress or interpersonal difficulty [5]. The WHO also classifies low sexual desire with similar criteria [6].
Overlap between sexual disorders and criticism of the current definition of HSDD have led to many expert panels re-evaluating the definition of low sexual desire [7,8]. This has led to suggestions that desire and arousal should be reclassified in the future as a combined condition: sexual interest/arousal disorder in women [9,10].
Epidemiology
Of the female sexual disorders, low desire is the most common, although its true prevalence has been the subject of much debate over recent years. High prevalence estimates of over 30% [11] led to suggestions that low sexual desire was being pathologized for the benefits of the pharmaceutical market [12]. Early prevalence studies on HSDD suffered from a lack of standardized definitions and often failed to examine the degree of distress associated with low desire. Many subsequent studies have now looked at the prevalence of HSDD around the world using strict diagnostic criteria and with validated diagnostic tools. These studies enable us to estimate more accurately the prevalence of HSDD and highlight that, although many women will experience low sexual desire, it is often not perceived as a health problem, nor associated with distress.
The large Prevalence of Female Sexual Problems Associated with Distress and Determinants of Treatment Seeking (PRESIDE) study, a cross-sectional, population-based survey of 31,581 US adults, found that low sexual desire was the most common sexual problem across all age groups, with an unadjusted prevalence of 38.7% [1]. When associated with distress, as judged by a Female Sexual Distress Scale (FSDS) score of at least 15, the prevalence of HSDD was 8.9% in the 18–44-year-old age group, 12.3% in the 45–64-year-old age group, and 7.4% in the 65 years and over age group.
Another key study was the Women's International Study of Health and Sexuality (WISHeS) study which investigated the prevalence of HSDD across North America, the UK, France, Germany and Italy. The prevalence of HSDD in this cross-sectional survey ranged from 9 to 26% in America [13] and from 6 to 16% in Europe [14], depending on factors such as age and menopausal status.
Although the prevalence of low libido increases with age, the associated distress actually decreases [15] and so HSDD tends to be most common in middle age [1]. Menopausal status has a significant impact on the prevalence of HSDD with several studies showing that the prevalence of HSDD is greatest in the younger surgically menopausal women (16–26%) compared with premenopausal (7–14%) and naturally menopausal women (6.6–9%) [13,14,16].
Other factors that influence the prevalence of HSDD were identified in another cross-sectional study in the USA of over 1900 women aged 30–70 years. They found the overall prevalence of HSDD was 8.3% but varied depending on age, educational level, BMI, current smoking status, current depression, use of hormone therapy (in menopausal women) and, possibly, ethnicity [16].
In addition to being a common problem across all age ranges, HSDD has an important impact on physical and psychological wellbeing. Low sexual desire has been associated with low self esteem [14], depression [15] and emotional or psychological distress [13]. Women with low desire are also more likely to experience lower arousal and pleasure, orgasmic difficulties and suffer dissatisfaction with their sex life and partner relationship [14].
A telephone survey study of postmenopausal US women showed that, in the 1189 respondents, HSDD was a significant burden in health-related quality of life [17]. Women with HSDD experienced a greater health burden in terms of comorbidities, and were nearly twice as likely to report depression, fatigue, back pain and memory problems, compared with women without HSDD.
Etiology
Normal female sexual function is the result of a complex interaction between physiological, psychological and social factors. Stress, relationship difficulties, age, menopausal status, medical comorbidities and their drug treatments are some of the many underlying causes of low sexual desire
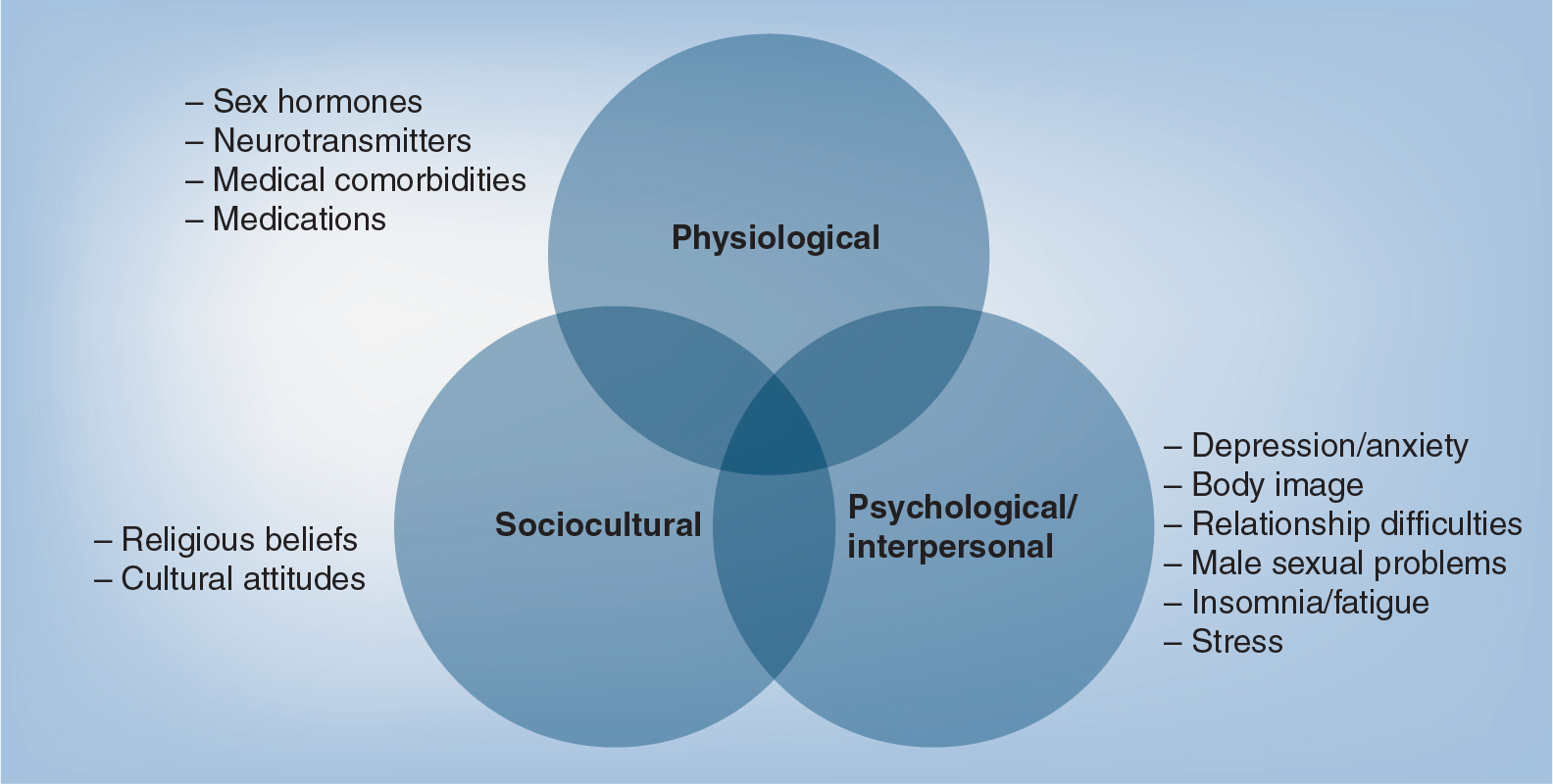
Factors modulating female sexual desire.
The sex hormones have been shown to interact with various neurotransmitters to modulate sexual desire [18]. A critical balance is required between the excitatory pathways, primarily controlled by the dopaminergic system, and inhibitory pathways of the serotonergic system, for normal sexual function. Animal models have suggested that dopamine acts to enhance sexual desire and excitement [19], while noradrenalin affects arousal and orgasm. Although serotonin may play a role in the resolution phase of the sexual response, an over-active serotonergic system can result in reduced desire and delayed orgasm, as observed from animal models and also in women using selective serotonin reuptake inhibitor (SSRI) antidepressants, who often experience loss of libido as a side-effect.
Many other factors are involved in the neurobiology of the sexual response and can, therefore, influence the balance between the excitatory and inhibitory pathways, including sex hormones, oxytocin (excitatory) and opioids or prolactin (inhibitory).
The prevalence of low sexual desire increases with age, which partly reflects the normal aging process but is also caused by the effect of the menopause on sexual function. The impact of the menopause transition results from the interplay between biological, psychosexual and sociocultural factors, which will be individual to each woman. From a pharmacological point of view, the role of the sex hormones in sexual desire must be considered as these provide the most common means of managing women with low sexual desire.
Role of sex hormones
Estrogens
Estrogens play a crucial role in female sexual function, particularly in the maintenance of healthy genital tissue. Vulvovaginal atrophy caused by postmenopausal estrogen deficiency results in thinning of the vaginal epithelium, loss of elasticity, reduced lubrication and changes in genital sensation, which can cause vaginal dryness and dyspareunia. Vaginal symptoms are a common problem after the menopause and a large European telephone survey to describe women's perceptions on the menopause found that symptoms of vaginal pain or dryness had been experienced by 29% [20]. Vaginal atrophy has a significant impact on sexual functioning and can affect all domains of sexual health including sexual desire [21]. Furthermore, the systemic effects of estrogen deficiency, such as hot flushes, poor sleep, and mood disturbance may also have a negative impact on sexual function in peri- or post-menopausal women.
Androgens
Androgens are produced in women by both the adrenal gland and the ovaries, and although the exact role of androgens in females remains poorly understood [22] they clearly have an important role in sexual desire and arousal [23]. The level of circulating androgens declines gradually with age, owing to a reduction in adrenal production, so the levels at the age of 40 years are around half that at the age of 20 years [24].
The adrenal gland produces dihydroepiandrosterone (DHEA) and its sulfate, which act as precursor hormones in the production of testosterone via peripheral conversion. Most testosterone circulates bound to sex hormone binding globulin (SHBG) and albumin, and only approximately 2% circulates freely in a biologically active form. Testosterone bound to albumin does so with relatively weak affinity and so is also sometimes considered to be bioavailable testosterone. Bioavailable testosterone can, therefore, be influenced by the many factors that can affect SHBG levels, including obesity or exogenous estrogens. Additionally, testosterone can be converted to estradiol by aromatase and so it is not exactly clear whether testosterone exerts its actions directly or through the actions of estradiol.
Testosterone appears to be the most important sex steroid in terms of modulating sexual desire, although no studies have consistently linked low circulating testosterone levels with low sexual desire. A significant contributing factor to this is the methodological flaws in testosterone assays [25]. Many of the assays used are not sensitive enough to pick up the low levels of free androgen seen in women and controversy exists regarding which fraction of the testosterone pool is most clinically relevant. In addition, the many different techniques used mean there are currently no standardized normal levels for women of different ages. Therefore, no clinical utility has been found in monitoring androgen levels in order to diagnose HSDD.
A further reason why studies have failed to correlate low testosterone levels with low sexual desire may be owing to the nature of androgen production. Recent studies have demonstrated that many androgens are synthesized from DHEA in peripheral tissue and exert their effects locally within these cells [26]. Therefore, these androgens are not released into the circulation in significant amounts, limiting the clinical utility of serum androgen levels.
The ovary is an important contributor to the androgen pool and even in postmenopausal women the ovary is a continued source of androgen production. Studies have shown that bilateral salpingo-oophorectomy can result in a decrease in circulating testosterone levels by approximately 40–50% [27,28]. This abrupt drop in androgen production may be a contributing factor as to the high prevalence of sexual dysfunction in younger, surgically menopausal women.
Women with premature ovarian failure (POF) have been shown to have lower androgen levels compared with controls [29,30], with a trend of those with lower androgen levels to have a lower sexual function scores on self-report questionnaires [31]. However, a recent study showed that although the risk of sexual dysfunction in a group with POF (62%, as measured using the Female Sexual Function Index questionnaire) was three-times that of a control group, with the POF group having worse arousal, lubrication and pain, there was no significant difference in desire between the groups. This highlights that many factors influence sexual desire in POF and that low desire is probably not explained by androgen deficiency alone [32].
Assessment
Women suffering from low sexual desire may present in a variety of healthcare settings, such as general practice, psychiatry, gynecology and sexual health clinics, however, many hurdles to diagnosis exist. Many women will not volunteer the information for fear of embarrassment to themselves or even to the medical staff and so symptoms often go unrecognized [33]. Unfortunately, many healthcare professionals feel either ill-equipped or lacking in time to deal with a patient complaining of low libido. One study found that 90% of practitioners in a primary care setting did not feel confident in making a diagnosis of HSDD [34]. Health professionals commonly perceive sexual problems to be outside their area of expertise, however, in many cases, simple measures such as identifying the SSRI they are taking, reassuring against any misconceptions regarding sex, or advice on stress reduction, are all that is required.
Crucial to the assessment is an adequate history
Physiological, psychological and sociological predisposing or perpetuating factors should then be sought. Medical comorbidities that have been associated with low sexual desire include thyroid disease, diabetes, pituitary disorders, neurological conditions, malignancy and cardiovascular disease. A gynecological history should identify menopausal status, particularly premature or surgical menopause and other chronic gynecological conditions such as endometriosis, vaginal atrophy or pelvic inflammatory disease, which may cause dyspareunia. In addition, various medications can influence sexual desire including antidepressants, antipsychotics, antihypertensives, hormones and corticosteroids. Psychological factors such as depression, anxiety, substance abuse and a history of sexual abuse should also be identified. Other vital parts of the history include any relationship difficulties, to identify those patients who would be best managed through relationship counseling, and the partner's sexual function. While many of these factors can contribute to low sexual desire, it is important to remember that for a formal diagnosis of HSDD the low desire must not be exclusively caused by another medical condition or the effects of concomitant medication.
There are various questionnaire-based tools for diagnosing low sexual desire, which often incorporate an assessment of distress to diagnose HSDD. The most commonly used include the Decreased Sexual Desire Screener (DSDS) [35], a short validated diagnostic aid for HSDD that can be used easily in clinical practice by health professionals without specific sexual health specialist knowledge. Other validated methods include the Female Sexual Function Index (FSFI) [36], which assesses all domains of female sexual function and the Profile of Female Sexual Function (PFSF) [37], which was designed to assess loss of sexual desire in postmenopausal women.
In some situations, further investigation may be required, for example to exclude other medical comorbidities, to assess amenorrhoea or oligomenorrhoea, and especially to identify sex steroid deficiency. The limitations in testosterone measurement make it difficult to correlate testosterone levels clinically with HSDD, but a baseline measurement could be considered in those being considered for testosterone replacement.
Management of HSDD
The complex nature of female sexual function requires a holistic approach to management, as low sexual desire is often not solely caused by hormone deficiency. The history should uncover any medical causes or other sexual dysfunction disorders that need to be addressed and highlight any psychological factors.
Generally, pharmacological measures should be combined with psychological interventions to achieve maximum benefit. There are a range of psychological therapies that may be helpful, including basic psychosexual counseling, cognitive behavioral therapy, relationship counseling and body awareness education [38] although there are limited data regarding the efficacy of many of these interventions [39]. Basic sexual counseling can include educating patients on basic anatomy and physiology, the normal sexual response and the normal changes in sexuality with aging. For patients with more complex psychological problems, cognitive behavioral therapy may be required to explore thoughts and behaviors more deeply and evidence exists to support the use of cognitive behavioral therapy in the management of HSDD [40,41].
Despite our advances in the understanding of sexual desire, pharmacological treatments remain limited. In those women with hormone deficiency, androgen replacement is recommended [42], however, there are currently no testosterone preparations licensed in the USA for treatment of HSDD, while in Europe only replacement via the transdermal patch is recognized as a valid treatment.
Patient assessment.
Desire/pain/arousal/orgasm (consider >1)
Lifelong or acquired
Situational or generalized
Associated distress
Gynecological/obstetric/genitourinary history including contraception and menopausal status
Medical comorbidities
Concomitant medications
Substance abuse
Intimacy, quality of relationship
Partner's sexual function
Sexual beliefs
Family issues
Depression/anxiety, psychiatric history
Body image/self-esteem
History of abuse
Life stressors
Hormonal management
Estrogens
Estrogens play a crucial role in modulating genital sensitivity and maintaining the function of the sexual organs. They act via vasodilatory pathways to increase vaginal, clitoral and urethral blood flow, resulting in genital congestion and vaginal lubrication.
Recent reviews on postmenopausal estrogen replacement have demonstrated the benefits of both local and systemic therapy on sexual function [43]. The authors also highlight how different estrogens and different routes of administration may impact treatment outcomes. Conjugated equine estrogens are effective for treating vulvovaginal atrophy but, by increasing SHBG levels, result in a reduction in circulating testosterone. Conversely, by avoiding first-pass hepatic metabolism, transdermal estradiol has minimal impact on circulating androgen levels and so may be preferred in women with sexual dysfunction who require systemic estrogen therapy [43]. The decision to use systemic or local treatment will be influenced by many individual factors, such as the presence or absence of vasomotor symptoms, requirement for osteoporosis protection, and individual risk of thrombosis or breast cancer.
For the management of vulvovaginal atrophy with local estrogen, several options exist including creams, pessaries and rings. A Cochrane review showed that all preparations are effective and have minimal systemic absorption [44]. Current guidelines recommend that patient preference should largely guide choice [45]. When treating postmenopausal vulvovaginal atrophy, symptoms are likely to return when the treatment course is completed, however, some preparations have now been granted a licence for long-term use.
When vaginal dryness is a contributor to low libido, other preparations that may have short-term benefit in relief of symptoms include vaginal moisturizers and vaginal lubricants. Water, oil or silicone-based lubricants are available but only play a role in the short-term relief of vaginal dryness during intercourse.
Testosterone
For many years exogenous testosterone has been recognized to play a role in improving sexual desire. Although older studies demonstrated a benefit from testosterone with estrogen replacement in postmenopausal women, these studies generally involved oral testosterone preparations administered in supraphysiological doses. These studies demonstrated that oral testosterone used in surgically and naturally postmenopausal women on estrogen-replacement therapy improved sexual desire [46,47]. However, oral testosterone, which is only licensed for use in men, is not usually used in women owing to concerns over adverse effects on lipid profiles due to first-pass hepatic metabolism. More recent research has concentrated on replacement via transdermal or subcutaneous routes, with serum levels closer to the physiological range.
The largest group of evidence to support the use of testosterone replacement for HSDD was generated following the development of the transdermal testosterone patch (TTP). The first of these studies was a randomized placebo-controlled study involving 75 surgically menopausal women also taking conjugated equine estrogens, who demonstrated improved sexual function as determined by scores on the Brief Index of Sexual Functioning for Women and the Psychological General Well-Being Index [48]. A second larger, randomized, placebo-controlled Phase II study [49], also examined the effects of the TTP in surgically menopausal women on concomitant oral estrogen. Women were randomized to receive placebo (n = 119) or testosterone patches in dosages of 150 μg/day (n = 107), 300 μg/day (n = 110), or 450 μg/day (n = 111) twice weekly for 24 weeks. Interestingly, women receiving the 300 μg/day patch had a significant increase in sexual desire and frequency of satisfying sexual activity, but there was no significant effect from either the 150 or 450 μg/day groups.
These pivotal studies were then followed by two larger Phase III studies (Investigation of Natural Testosterone in Menopausal women Also Taking Estrogen [INTIMATE] 1 and 2) [50,51] involving a total of 1094 women, which demonstrated that women receiving the 300 μg/day testosterone patch following total abdominal hysterectomy with bilateral salpingo-oophorectomy in addition to estrogen had significantly more satisfying encounters and increased sexual desire. Crucially, these improvements were felt to be clinically significant as they were associated with a reduction in associated distress.
Similar large, randomized, double-blind, placebo-controlled studies involving naturally menopausal women on HRT [52], naturally postmenopausal women predominantly not on HRT [53] and postmenopausal women not on concomitant HRT [54] have also demonstrated that the TTP led to a significantly increased frequency of satisfying sexual episodes, increased sexual desire and reduced distress.
Despite the latter studies, the TTP currently has only been approved by the European Medicines Agency for use in surgically menopausal women who are on estrogen therapy. Furthermore, the TPP is currently not approved for any indication by the US FDA. Although safety outcomes in all the aforementioned studies have been reassuring, uncertainty remains owing to the lack of long-term safety data, especially with regard to risk of breast and endometrial cancers and cardiovascular disease. The 4 year follow-up data from two of the studies demonstrated that in 967 surgically menopausal patients who were also on estrogen replacement, and each received at least one application of the TTP, there appeared to be no increased occurrence of breast cancer or disturbances of lipid profiles [55].
Common side effects of the TTP include hirsutism (7% compared with 5% in controls), acne (9% compared with 7% in controls) and patch site irritation in up to 30% of participants. The unwanted androgenic effects tend to be dose dependent and resolve following discontinuation of treatment.
Other less well studied routes for testosterone replacement include subcutaneous implant and transdermal gels. The testosterone implants have been used for over 30 years and early observational studies showed a significant improvement in libido in women receiving estradiol and testosterone implants [56]. Subsequent randomized studies demonstrated a benefit in sexual functioning in women receiving estrogen plus testosterone implants, compared with estrogen alone [57,58]. A more recent observational study examined the use of 75–160 μg testosterone implants, without concomitant estrogen, in 300 premenopausal and postmenopausal women reporting symptoms of androgen deficiency, such as sexual problems, fatigue, mood disturbance, headaches, insomnia, memory loss and hot flushes. Testosterone implants were found to be effective for the relief of psychological, somatic and urogenital symptoms, including improvements in sexual desire, as measured by the self-administered, validated health-related quality of life Menopause Rating Scale [59]. However, further randomized controlled trial data are required to support the use of testosterone implants in premenopausal women.
The use of testosterone gels has been investigated in two studies. A randomized, placebo-controlled, crossover study in 53 post-menopausal women with low libido on HRT demonstrated that 10 mg daily of testosterone gel was associated with significant improvements in desire, including frequency of sexual activity, fantasies and sexual interest [60]. A small randomized double-blind crossover pilot study, assessing the effect of a single dose of testosterone gel 4–8 h prior to intercourse in premenopausal women with HSDD, resulted in increased arousal [61]. Further data are required to confirm these findings. In the UK, testosterone gels remain unlicensed and no products developed for use specifically in women currently exist.
Tibolone
For menopausal women with vasomotor symptoms and low sexual desire, tibolone is another useful treatment option. Tibolone, licensed as HRT, is a synthetic steroid molecular with estrogenic, progestogenic and androgenic action. In addition to improvements in vasomotor symptoms and vaginal atrophy, tibolone has been shown to improve sexual desire compared with placebo [62,63]. These findings were confirmed on a more recent randomized double-blind active control trial with transdermal estradiol/norethisterone acetate over 24 weeks. Both treatments resulted in improved sexual function as assessed by the FSFI, however, the group on tibolone had significantly greater improvements on FSFI scores compared with the control group [64]. Although tibolone has also been shown to have a positive effect on the risk of breast cancer and fracture, concerns exists regarding the risk of stroke when used in older women [65].
Dihydroepiandrosterone
Dihydroepiandrosterone is an androgen that acts as a prohormone. It is produced by the adrenal gland and is metabolized in peripheral tissues such as breast, brain, bone and ovaries, into various different steroids including estrogen and testosterone. The levels of DHEA decline with age and it has been postulated to be responsible for a range of age-related symptoms [66]. DHEA is available without prescription in the USA and is often bought as an over-the-counter product. Although often touted as the ‘elixir of youth’ or the ‘hormone of all hormones’, it is poorly regulated and the safety of exogenous administration has not yet been clarified.
Recent reviews of the evidence have shown that many early studies used supraphysiological doses, or had methodological flaws such as small sample sizes, short duration or used non-validated tools for assessment of female sexual function [67]. Furthermore, more recent higher quality studies have failed to demonstrate a benefit in terms of libido or general wellbeing.
Although the use of systemic DHEA remains controversial, there is some evidence that vaginal DHEA may help libido through improvement of vaginal dryness. A prospective, randomized, double-blind, and placebo-controlled Phase III clinical trial has evaluated the effect of daily local intravaginal DHEA for 12 weeks in 218 postmenopausal women with moderate-to-severe symptoms of vaginal atrophy and the results have demonstrated beneficial effects on all four aspects of sexual function (desire, arousal, orgasm and pain) [68]. Further large-scale data are required to confirm these findings.
At present there is no evidence that DHEA supplements should be recommended for the management of low libido, especially as its exact final metabolites and the safety of use are unclear.
Other treatments
Flibanserin, has been investigated as a potential treatment for HSDD. It is a 5-hydroxytryptamine1A agonist and 5-hydroxytryptamine2 antagonist, that acts selectively on the inhibitory and excitatory neurotransmitter pathways involved in sexual desire [69]. Pooled analysis from a European and two North American Phase III, randomized, placebo-controlled studies in premenopausal women with HSDD showed that flibanserin 100 mg daily led to a significant improvement in FSDS-Revised scores and increased desire compared with placebo [70]. Despite these findings, the FDA recently declined the application as it was felt the benefits did not outweigh the side effects of nausea, dizziness, fatigue and insomnia. The manufacturers have now withdrawn the drug from further development [101].
Another agent that has been suggested to improve sexual desire is bupropion, an atypical antidepressant, which causes dopamine and noradrenalin reuptake inhibition and, therefore, acts on the excitatory pathways of sexual desire.
Earlier evidence for the use of bupropion came from trials in premenopausal women suggesting an improvement in sexual functioning [71]. In a recent randomized, placebo-controlled, double-blind study of 232 nondepressed, premenopausal women with HSDD, bupropion sustained release 150 mg daily for 12 weeks was associated with a significant improvement across all domains of sexual functioning and a reduction in distress [72].
In a survey of psychiatrists, bupropion is often used as a treatment for SSRI-induced low libido, either as an alternative antidepressant, or as an augmentative treatment [73]. This is supported by clinical data from another randomized, placebo-controlled study by Safarinejad who assessed the impact of bupropion sustained release 150 mg in 218 women between the ages of 25 and 45 years with SSRI-induced sexual dysfunction [74]. Treatment with bupropion was associated with a significant improvement in desire, arousal, lubrication, orgasm and satisfaction.
A recent review has examined the evidence for the use of bupropion and other pharmacological agents in modulating the monoamine neurotransmitters serotonin, dopamine and noradrenalin [75]. Overall, adequate randomized controlled trial data are lacking and current agents often have unacceptable side effects. However, these agents provide potential hope for future development of more selective agents that may be better tolerated.
Future perspective
Large gaps remain in our understanding of female sexual function and treatment options remain limited. In the future we need to better educate health professionals to help improve recognition of HSDD and help give confidence to patients to voice these symptoms.
More evidence is required regarding the current therapies in terms of efficacy and long-term safety. Data exist for the use of testosterone patches, however, further long-term studies and evidence to support the use of other routes of testosterone replacement are needed. Hopefully, continued research will lead to improved understanding of the neurobiological pathways involved in sexual desire and help identify potential therapeutic targets. Development of more selective agents targeting particular neurotransmitters will be highly beneficial to help improve efficacy and reduce adverse effects.
Executive summary
Female sexual dysfunction is commonly classified into distinct disorders of desire, arousal, orgasm or pain. In clinical practice, these disorders frequently overlap.
Hypoactive sexual desire disorder (HSDD) is defined as low sexual desire that causes distress or relationship difficulties.
HSDD affects 6–16% of women in Europe and menopausal status has a significant impact on its prevalence.
HSDD has important effects on women's wellbeing and is associated with low self-esteem, depression and relationship dissatisfaction.
Low sexual desire can result from a variety of physiological, psychological and sociocultural factors.
Estrogens and androgens play a crucial role and interact with various neurotransmitters to modulate sexual desire.
A thorough history will uncover many causes for sexual dysfunction that may be treated with the specialist psychosexual environment such as medication side-effects or medical comorbidities.
A greater understanding of the female sexual response has helped identify many potential biological and psychosocial targets for therapeutic intervention.
Management of HSDD requires an individual approach incorporating pharmacological and psychosocial interventions.
Menopause is a time particularly associated with sexual dysfunction and estrogen and testosterone replacement can be valuable in improving sexual function.
The transdermal testosterone patch is licensed in Europe for the treatment of HSDD in surgically menopausal women on concomitant HRT.
No pharmacologic therapy is currently approved for the treatment of HSDD in premenopausal or naturally postmenopausal women and further research is urgently needed in these populations.
At present there is no evidence that dihydroepiandrosterone supplements should be used for HSDD as its safety and efficacy are currently unclear.
Despite evidence supporting the use of flibanserin for HSDD, its development has been withdrawn owing to safety concerns.
Bupropion is associated with an improvement in sexual desire and may be particularly useful in selective serotonin reuptake inhibitor-induced low desire.
In the future, improved understanding of the neurochemical pathways of sexual desire may help aid development of more selective agents.
Footnotes
Managing low sexual desire in women
To obtain credit, you should first read the journal article. After reading the article, you should be able to answer the following, related, multiple-choice questions. To complete the questions (with a minimum 70% passing score) and earn continuing medical education (CME) credit, please go to www.medscape.org/journal/wh. Credit cannot be obtained for tests completed on paper, although you may use the worksheet below to keep a record of your answers. You must be a registered user on Medscape.org. If you are not registered on Medscape.org, please click on the New Users: Free Registration link on the left hand side of the website to register. Only one answer is correct for each question. Once you successfully answer all post-test questions you will be able to view and/or print your certificate. For questions regarding the content of this activity, contact the accredited provider,
