Abstract
Hypoactive Sexual Desire Disorder (HSDD) is defined as a persistent or recurrent deficiency of sexual fantasies and desire for sexual activity, which causes marked personal distress or interpersonal difficulty, and is not better accounted for by another psychiatric disorder or the direct physiological effects of a substance (e.g., a medication) or medical condition. HSDD is believed to be the most common form of Female Sexual Dysfunction and is associated with emotional distress and relationship problems. No pharmacologic therapy is approved for the treatment of HSDD in premenopausal or naturally postmenopausal women. Flibanserin is a 5-HT1A agonist/5-HT2A antagonist that is under investigation as a treatment for HSDD in women. The aim of this article is to present an overview of the pharmacology, clinical efficacy and safety of flibanserin. Flibanserin is an investigational drug that is not licensed for any indication in any country.
Hypoactive Sexual Desire Disorder (HSDD) is defined by the American Psychiatric Association in its Diagnostic and Statistical Manual of Mental Disorders, 4th Edition, Text Revision (DSM-IV-TR) as a persistent or recurrent deficiency or absence of sexual fantasies and desire for sexual activity that causes marked distress or interpersonal difficulty and that is not better accounted for by another psychiatric condition, medical condition or substance (e.g., a medication) [1]. As with other types of Female Sexual Dysfunction (FSD), HSDD is further classified as being either lifelong (present from the onset of sexual functioning) or acquired (developed after a period of normal functioning) and as either situational (limited to certain partners, situations or types of stimulation) or generalized [1].
Diagnosis of HSDD is made by a clinician taking into account the biologic, psychosocial, relationship and cultural factors that may be contributing to the woman's lack of sexual desire, and whether the woman is troubled by her lack of desire [1,2]. It is essential that healthcare professionals adopt a patient-centered approach to the evaluation of sexual disorders and take a thorough medical, sexual and psychosocial history [2]. A number of checklists, screeners and diagnostic instruments have been developed to aid clinicians in the evaluation of women presenting with sexual problems [2]. For those presenting with low sexual desire, these include the Decreased Sexual Desire Screener, a brief diagnostic instrument for generalized acquired HSDD designed for use by clinicians with little or no expertise in the diagnosis of female sexual disorders [3]. Such instruments are particularly important in primary care, where time for patient consultations is limited.
Epidemiologic studies have found that approximately one in ten women report having low sexual desire associated with distress, which may be HSDD [4,5]. Occurring in pre-, peri- and post-menopausal women, HSDD is associated with emotional distress, lower sexual satisfaction for both the patient and her partner, and impairments in general health status and quality of life [5–10]. However, HSDD remains under-recognized, underdiagnosed and undertreated. The majority of physicians are reluctant to ask their female patients about their sexual health as they are poorly trained in female sexual health, have only a short time for consultations and lack effective treatment options [11–14].
At present, there are no approved pharmaceutical treatments for HSDD outside Europe. In Europe, the only approved agent is a testosterone patch (Intrinsa®; Procter & Gamble Pharmaceuticals), which is licensed solely for use in surgically menopausal women with HSDD who are receiving estrogen therapy. The aim of this article is to present an overview of flibanserin (Boehringer Ingelheim), a potential new treatment for HSDD in premenopausal women that is in late-stage clinical development. At present, flibanserin is an investigational drug that is not licensed for any indication in any country.
Overview of the market
There are no official treatment guidelines for HSDD. However, a committee of experts in FSD, convened as part of the 3rd International Consultation on Sexual Medicine (ICSM), and have published recommendations on the management of women with desire and arousal disorders [15]. Suggested treatments for low sexual desire include psychologic therapy, such as cognitive behavioral therapy [15]. However, while there is some evidence of benefit of cognitive behavioral therapy in women with desire problems [16–18], randomized controlled data supporting the efficacy of psychologic counseling in women with HSDD are lacking [15].
Testosterone has been used as a treatment for low sexual desire since the 1930s, but systematic investigation into its use in this setting has been relatively recent [15]. The benefits of testosterone therapy in women with low desire remain controversial. In December 2004, the US FDA refused to approve a testosterone patch for the treatment of HSDD in women owing to insufficient safety data on the long-term effects of testosterone treatment in women [101]. This safety concern was predominantly driven by the findings of the Women's Health Initiative, which found an increased risk of breast cancer and cardiovascular effects in women who took hormonal therapies combining progesterone with estrogen [19]. The same testosterone patch was approved in some European countries for the treatment of HSDD in surgically menopausal women who are undergoing estrogen therapy [102]; however, it is not widely used. Recent recommendations from the 3rd ICSM state that current data are not adequate to support the use of testosterone therapy in pre- and peri-menopausal women, and that additional safety data are required before the long-term use of testosterone therapy can be recommended [15,20]. Safety data on testosterone therapy continues to be collected and 4 years of safety data on the safety of the testosterone patch in surgically menopausal women have recently been presented [21].
The lack of licensed pharmacologic treatments for HSDD has led to significant off-label prescription of drugs that are not licensed for HSDD, particularly antidepressants and hormones. In addition, many women with desire problems use over-the-counter medications that have no proven efficacy [22].
Introduction to flibanserin
Chemistry & pharmacology
Flibanserin (BIMT 17 BS; 2H-benzimidazol-2-one, 1,3-dihydro-1-[2-[4-[3(tri-fluoromethyl) phenyl]-1-piperazinyl]ethyl]) is a nonhormonal, centrally acting molecule that acts as an agonist at postsynaptic 5-HT1A receptors and as an antagonist at 5-HT2A receptors [23,24]. In rats, flibanserin administration has been shown to lead to brain region-specific decreases in serotonin (5-HT) and increases in dopamine and norepinephrine [25]. Dopamine and norepinephrine are involved in the ‘excitement’ phase of the sexual response (e.g., desire and arousal) while 5-HT is involved in the ‘inhibitory’ phase (e.g., satiety or refractory period) [26]. A balance between excitatory activity driven by dopamine and norepinephrine and inhibitory activity driven by 5-HT is believed to be necessary for a healthy sexual response [26]. One hypothesis suggests that an imbalance between these systems may be present in sexual dysfunction. By selectively modulating these neurotransmitters in specific brain areas, flibanserin may act to re-balance these systems in women with HSDD.
Following chronic oral administration, flibanserin plasma concentrations rise rapidly, usually within the first hour, and decrease biexponentially thereafter (Cmax,ss = 469 ng/ml; Tmax = 45 to 60 min, AUCτ,ss = 2080 ng h/ml for 100 mg once-daily dose). A total of 90% of the dose is absorbed, primarily from the upper intestine, and 33% of flibanserin is available systemically due to first-pass metabolism. The basic pharmacokinetic properties of flibanserin demonstrate adequate exposure to flibanserin for once-daily administration due to its terminal half-life of approximately 10 h. Flibanserin exposure can be controlled as there is moderate accumulation (accumulation ratio for AUC 1.44 after once-daily 100 mg dosing), a steady state is established within 3 days and plasma levels do not change afterwards (as observed during 2 weeks of chronic dosing). Flibanserin exposure is proportional to dose [BI data on file].
As expected for a centrally acting drug, the plasma protein binding of flibanserin (98% to albumin) is high. Flibanserin is nearly completely metabolized with at least 35 metabolites formed in humans, most of which occur in low concentrations in plasma or urine. Two metabolites could be characterized that show plasma concentrations similar to flibanserin: flibanserin-6-sulfate and flibanserin-6,21-disulfate These two metabolites were thoroughly assessed in receptor assays and are not considered to be active [BI data on file].
Flibanserin metabolites are primarily excreted in urine (44.1%) and feces (50.9%). Hepatic impairment causes a pronounced increase in flibanserin exposure, but renal impairment does not. In regard to metabolic drug–drug interactions, flibanserin exposure can be affected by alterations in cytochrome P450 (CYP)3A4 activity, which is the major isoform involved in the metabolism of flibanserin, but not by impaired CYP2D6 function, which plays a minor role in flibanserin metabolism. No clinically relevant effect of flibanserin has been observed on the pharmacokinetics of simvastatin, a substrate of CYP3A4; bupropion, a substrate of CYP2B6; or a combination of levonorgestrel and ethinyl estradiol. Flibanserin can be given with or without food. Food increases flibanserin systemic exposure by up to 56%, but only has a minimal effect on the maximum plasma concentrations and typical flibanserin side effects [BI data on file].
Rationale for clinical development program
Acting as a cortical 5-HT1A agonist and 5-HT2A antagonist, flibanserin was initially expected to possess antidepressant characteristics [23]. Preclinical studies demonstrated that flibanserin displayed antidepressant-like activity in most animal models sensitive to antidepressant drugs; however, its activity appeared to be qualitatively different from that of known antidepressants [23].
When investigated as a potential treatment for Major Depressive Disorder (MDD) in Phase II/IIb trials, flibanserin was found not to be an effective antidepressant in active controlled studies [27]. However, in women whose depressive symptoms included decreased sexual desire at baseline, flibanserin was found to be superior to a placebo and the comparator antidepressant (paroxetine) on the Arizona Sexual Experiences Scale (ASEX) [28], particularly on the ‘sex drive’ item [27]. These findings provided a rationale for beginning a clinical development program in women with HSDD.
Clinical efficacy
Phase II proof-of-concept trials
Two proof-of-concept trials with identical design were conducted in premenopausal women with HSDD. Both were 12-week, randomized, double-blind, placebo-controlled trials. Flibanserin was initiated at 50 mg twice daily, with optional uptitration to 100 mg twice daily at the end of week 8. The trials included women aged 18–45 years with a diagnosis of HSDD (DSM-IV-TR) [1] of 24 weeks or longer. Study participants were also required to have an ASEX sex drive (desire) score [28] of 5 (very weak) or 6 (none), and to be in a stable, monogamous, heterosexual relationship with a sexually functional partner. Key exclusion criteria were sexual aversion disorder, sexual pain disorders, psychiatric disorders likely to affect sexual functioning (including MDD), a history of any serious clinical disorder and use of any medication known to induce sexual dysfunction during the past 6 months.
Since there were no established end points for trials in HSDD when these trials were planned, a number of exploratory efficacy measures were used, including the ASEX (chosen as the primary end point based on observations from the Phase II/IIb MDD trials) [27], the Female Sexual Function Index (FSFI) [29], a version of the Changes in Sexual Functioning Questionnaire-Female (CSFQ-F) [30] that was self-administered over the telephone using an automated system, the Female Sexual Behavior Questionnaire (FSBQ, a simple self-administered weekly questionnaire designed to gather counts of sexual encounters, Satisfying Sexual Events [SSE], number of days with sexual thoughts or sexual fantasies and number of days with desire to engage in sexual activity), the preliminary 17-item version of the Sexual Interest and Desire Inventory-Female (SIDI-F) [31], the sum of the 12 core items of the SIDI-F and the Clinical Global Impression of Improvement (CGI-I) [32].
In the two trials, 148 patients received placebo and 149 patients received flibanserin. The trials were completed by 106 (71.6%) patients on placebo and by 77 (51.7%) patients on flibanserin. Adverse events (AEs) led to discontinuation in 36.2% of patients treated with flibanserin, compared with 5.4% of placebo patients. Flibanserin AE dropouts were mainly due to sedative effects of the morning dose, which at the 50 mg twice daily level, and especially at the 100 mg twice daily level (used in the last 4 weeks of treatment by 51.7% of patients) tend to interfere with morning work for approximately 1 h [BI data on file].
In a
Summary of pooled analysis of Phase II proof-of-concept trials in premenopausal women with Hypoactive Sexual Desire Disorder.

Values shown are least squares mean (standard error) change from baseline, except for CGI-I ‘much improved’, for which values are n (%), for the full analysis set and the per protocol set (both Last Observation Carried Forward).
12 core items of the 17-item inventory.
ASEX: Arizona Sexual Experiences Scale; CGI-I: Clinical Global Impression of Improvement; CSFQ: Changes in Sexual Functioning Questionnaire; FLI: Flibanserin
FSBQ: Female Sexual Behavior Questionnaire; FSFI: Female Sexual Function Index; SIDI-F: Sexual Interest and Desire Inventory-Female; SSE: Satisfying Sexual Event.
Phase III clinical development program: the BOUQUET studies
Overview of Phase III trials
The Phase III trials of flibanserin in women with HSDD are collectively known as the BOUQUET studies. Five Phase III trials in premenopausal women have been completed and results reported:
Three North American, 24-week, randomized, placebo-controlled trials: DAHLIA (511.70) [33], VIOLET (511.71) [34] and DAISY (511.75) [35]
One European (13 countries), 24-week, randomized, placebo-controlled trial: ORCHID (511.77) [36]
A randomized withdrawal trial (ROSE [511.74]) in which patients received 24-weeks' open-label treatment with flibanserin and then participants who met prespecified enrichment criteria were randomized in a double-blind manner to receive flibanserin or placebo for 24 weeks, to assess potential withdrawal effects [37–40].
The BOUQUET program also includes two extension trials:
SUNFLOWER (511.84), an extension study in which patients who completed a North American trial received open-label treatment with flibanserin for 52 weeks
MAGNOLIA (511.118), an extension study in which patients who completed the ORCHID trial received open-label treatment with flibanserin for 28 weeks
Overall, the premenopausal program involves approximately 5000 patients.
Phase III randomized placebo-controlled trials
North American trials: DAHLIA, VIOLET, DAISY
The objectives of these trials were to establish the efficacy of flibanserin over 24 weeks of treatment to produce a clinically meaningful therapeutic response in premenopausal women with generalized acquired HSDD, and to document the safety profile of flibanserin administered to such women for 24 weeks. The three trials had an almost identical design

Design of pivotal trials.
The trials included women aged 18 years or over with a diagnosis of generalized acquired HSDD (DSM-IV-TR) [1] of a duration of 24 weeks or more, who were premenopausal according to Stages of Reproductive Aging Workshop criteria [42]. The diagnosis of generalized acquired HSDD was made by a clinician following a structured diagnostic interview. Participants were required to have a score of 15 or above on the Female Sexual Distress Scale-Revised (FSDS-R) [43], indicating that they were distressed about their sexual dysfunction, and to score a rating of ‘0’ or ‘1’ on item two (receptivity) of the SIDI-F [44–46], indicating little or no receptivity to approach by their partner
Description of measures used in Phase III flibanserin clinical trials.

FSDS-R: Female Sexual Distress Scale-Revised; FSFI: Female Sexual Function Index; HSDD: Hypoactive Sexual Desire Disorder; PBE: Patient Benefit Evaluation
PGI-I: Patient's Global Impression of Improvement; SIDI-F: Sexual Interest and Desire Inventory-Female; SSE: Satisfying Sexual Event.
The eDiary is a handheld electronic diary that prompted the participants on a daily basis to answer questions on their sexual activity, whether this activity was satisfying to them and their most intense level of sexual desire. A sexual event was defined for the participants as sexual intercourse, oral sex, masturbation or genital stimulation by their partner. An SSE was recorded when a woman reported that a recorded sexual event was satisfying for her. The desire question in the eDiary was ‘Indicate your most intense level of sexual desire in the last 24 h’, with responses scored on a four-point scale of 0 (no desire), 1 (low desire), 2 (moderate desire) or 3 (strong desire)
The coprimary end points were change from baseline in number of SSE/month and sexual desire score, both measured using the eDiary
European trial: ORCHID (511.77)
The objectives and design of ORCHID were essentially the same as for the North American trials
Baseline characteristics
Across all four trials, there were no notable differences between the placebo and flibanserin treatment groups in baseline characteristics. The mean (SD) age of the North American participants was 35.4 (7.0) years and the mean (SD) age of the European population was 35.2 (7.2) years.
At baseline, the mean FSDS-R total score was 31.0 in all the North American trials and 28.2 in the European trial, approximately double the cutoff score of 15 for sexual distress [48]. The mean FSFI total score at baseline was 19.2 in all the North American trials and 18.8 in the European trial, well below the cutoff score of 26.55 for sexual dysfunction [49]. The mean FSFI desire domain score at baseline was 1.8 in all the North American trials and 1.9 in the European trial, in both cases below the cutoff score (≤3) established to distinguish women with HSDD from healthy controls [50]. At baseline, the mean SSE per month was 2.7 in all the North American trials and 2.3 in the European trial. Unlike criteria such as sexual distress or sexual dysfunction, SSE does not form part of the diagnostic criteria for HSDD. Consequently, no cutoff for a ‘dysfunctional’ level of SSE has been published so far. Indeed, a decrease in SSE in HSDD is considered to be an epiphenomenon or a ‘downstream’ consequence of a loss of sexual desire [51].
Patient disposition
Patient disposition differed according to the treatment group, and was primarily driven by dropouts due to AEs, which were dose- and regimen-related
Summary of patient disposition in the North American and European pivotal trials (DAISY, VIOLET, DAHLIA and ORCHID).

Results from randomized placebo-controlled trials
Flibanserin 100 mg once daily at bedtime was found to be efficacious and well tolerated [52]. Other doses of flibanserin were found to have inconsistent efficacy [33–36]. The Phase III statistical analysis plan included a prespecified pooled analysis of data on the flibanserin 100 mg once daily at bedtime dose from the two North American trials that investigated this dose (DAISY and VIOLET). The results of all doses tested in the individual trials have been presented elsewhere [33–36] and differed little from the pooled results. This paper summarizes the efficacy results of the 100 mg once daily at bedtime dose from the North American trials (DAISY and VIOLET), the European trial (ORCHID), the prespecified pooled analysis of the DAISY and VIOLET trials, and the
Efficacy data for flibanserin 100 mg once daily at bedtime
North American trial, DAISY: primary & secondary end points
The mean change from baseline to week 24 (end of trial) in SSE/month was 1.9 for flibanserin verses 1.1 for placebo (p < 0.01;
Summary of efficacy results for flibanserin 100 mg once daily at bedtime versus placebo.
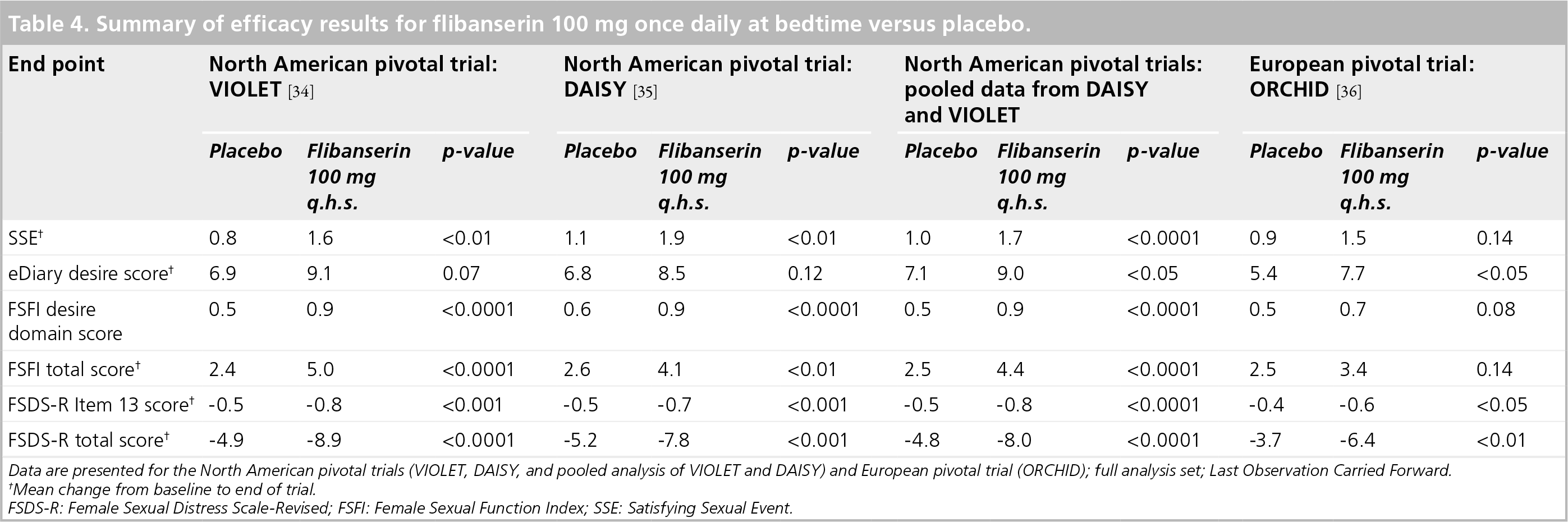
Data are presented for the North American pivotal trials (VIOLET, DAISY, and pooled analysis of VIOLET and DAISY) and European pivotal trial (ORCHID); full analysis set; Last Observation Carried Forward.
Mean change from baseline to end of trial.
FSDS-R: Female Sexual Distress Scale-Revised; FSFI: Female Sexual Function Index; SSE: Satisfying Sexual Event.
North American trial, VIOLET: primary & secondary end points
The mean change from baseline to week 24 (end of trial) in SSE/month was 1.6 for flibanserin verses 0.8 for placebo (p < 0.01;
North American trials, prespecified pooled analysis, VIOLET & DAISY: primary & secondary end points
The mean change from baseline to week 24 (end of trial) in SSE/month was 1.7 for flibanserin verses 1.0 for placebo (p < 0.0001;
Flibanserin was significantly superior to placebo on FSFI desire domain score, FSDS-R Item 13, FSDS-R total score and FSFI total score (p < 0.01 for all)
European trial, ORCHID: primary & secondary end points
The mean change from baseline to week 24 (end of trial) in SSE/month was 1.5 for flibanserin verses 0.9 for placebo (p = 0.14;
Post hoc PGI-I-anchored responder analyses (pooled data from North American [DAISY & VIOLET] & European [ORCHID] trials)
For every end point, the mean change from baseline was calculated and ‘anchored’ to every level of the PGI-I. The difference between ‘minimally improved’ and ‘no change’ on the PGI-I was used as the responder criterion, that is, women with values greater than the responder criterion were considered to be responders. Using this methodology, responders had improvements of greater than 1.39 in SSE, 7.87 in eDiary desire score, 0.81 in FSFI desire domain score (requiring a 2-point or greater increase in the sum of the two desire items, which provide a raw score before weighting of between 2 and 10), 4.57 in FSFI total score, −0.46 in FSDS-R Item 13 score (requiring a 1-point or greater decrease in the item's scoring from 0 to 4), or −5.31 (a six-point or greater decrease) in FSDS-R total score [55].
Compared with the placebo group, approximately a third more women receiving flibanserin 100 mg once daily at bedtime responded on SSE (32.6 vs 42.6%; p < 0.0001), FSFI desire domain score (30.6 vs 42.0%; p < 0.0001), and FSFI total score (31.4 vs 42.3%; p < 0.0001). Approximately a quarter more women on flibanserin 100 mg once daily at bedtime responded on FSDS-R Item 13 score (39.4 vs 49.4%; p < 0.0001) and FSDS-R total score (41.4 vs 53.2%; p < 0.0001)

Results of Patient Global Impression of Improvement-anchored responder analyses conducted on pooled data for flibanserin 100 mg q.h.s. versus placebo from VIOLET, DAISY and ORCHID.
These results demonstrate that flibanserin 100 mg once daily at bedtime provides clinically as well as statistically meaningful benefits on sexual desire, overall sexual function, distress associated with sexual dysfunction, distress associated with low sexual desire and SSE.
Post hoc remitter analyses (pooled data from North American [DAISY & VIOLET] & European [ORCHID] trials)
Based on published cutoff points for differentiating women with and without sexual dysfunction, remitters were classified as patients who, at study end, had: FSDS-R total score less than 15 [48]; FSFI total score 26.55 or more [49]; or FSFI desire domain score (weighted score) of more than three [50].
This
ROSE (randomized withdrawal trial)
Design of ROSE study
The objectives of the ROSE trial were to determine if efficacy is maintained when premenopausal women with HSDD who have responded to 24 weeks of open-label treatment with flibanserin continue treatment for a further 24 weeks; to evaluate potential withdrawal effects from flibanserin over 24 weeks; and to assess the safety of flibanserin therapy over 48 weeks.
Inclusion and exclusion criteria for ROSE were the same as those used in the randomized placebo-controlled trials except that nonreceptivity (SIDI-F item two) was not required. The trial design is shown in

Design of randomized withdrawal trial (ROSE).
Results of the ROSE study
Patient population
The demographics and baseline characteristics of patients participating in the ROSE trial were very similar to those in the randomized placebo-controlled trials [BI data on file].
Of the 738 patients entering the open-label phase of the study, 525 (71.1%) completed it. Of the patients who did not complete the open-label phase, 83 (11.2%) discontinued due to AEs. A total of 333 patients (45.1%) met the predefined enrichment criteria and were randomized to flibanserin (n = 163) or placebo (n = 170). Overall, 132 (81.0%) flibanserin and 146 (85.9%) placebo patients completed 48 weeks of treatment. During double-blind treatment, four (2.5%) patients treated with flibanserin discontinued due to AEs, compared with seven (4.1%) on placebo [40].
Primary & secondary efficacy end points
Over 24 weeks of open-label treatment, SSE and eDiary desire score increased by twofold and there was a threefold increase in the number of days in which desire was rated as moderate or strong [37]. From week 29 (i.e., 4 weeks postrandomization) to week 48, flibanserin was significantly superior to placebo on eDiary desire score (p < 0.05) [37]. At the end of the double-blind treatment period (weeks 45–48), flibanserin was significantly superior to placebo on SSE, FSFI desire domain score, FSFI total score, FSDS-R Item 13 score FSDS-R total score, and PGI-I (p < 0.05 for all)
Results of ROSE randomized withdrawal trial (double-blind period).

170 for FSDS-R and FSDR-R Item 13.
FSDS-R: Female Sexual Distress Scale-Revised; FSFI: Female Sexual Function Index; PGI-I: Patient's Global Impression of Improvement; SSE: Satisfying Sexual Event.
Safety & tolerability
Exposure
Flibanserin has been tested in healthy volunteers (0.1–150 mg; n = 803 [432 women, 371 men]; mean age: 35.9 years; age range: 18–73 years), depressed patients (2–100 mg twice daily; n = 1210 [702 women, 508 men]; mean age of women patients: 41.4 years; age range of women patients: 18–45 years), and premenopausal women with HSDD (25–100 mg twice daily; n ≥ 4318). Over 6500 human subjects have received 1 dose or more of flibanserin in clinical trials to date. In the four randomized, placebo-controlled HSDD trials, 1729 women were exposed to flibanserin at a dosage of 100 mg/day (100 mg once daily at bedtime or 50 mg twice daily).
Proof of concept trial data
In the two proofs of concept trials, flibanserin was initiated at 50 mg twice daily, with optional uptitration to 100 mg twice daily at the end of week 8. Overall, 51.7% of patients in the flibanserin group uptitrated to 100 mg twice daily. The most frequently reported AEs in patients receiving flibanserin were somnolence (25.5 vs 2.0% for placebo), dizziness (25.5 vs 4.1%), nausea (24.8 vs 8.8%), sedation (18.8 vs 3.4%) and fatigue (18.1 vs 6.1%). There were no deaths or serious AEs, and other safety measures (i.e., blood pressure, pulse and laboratory tests) were unaffected by treatment [BI data on file].
Phase III data
Randomized controlled trials: DAHLIA, VIOLET, DAISY & ORCHID
Safety assessments included the evaluation of AEs, clinical laboratory parameters (i.e., hematology, chemistry, urinalysis and sex hormones), vital signs, physical findings and electrocardiogram data. There were no important differences in the frequencies of AEs reported in the North American and European trials. Across the four trials, AEs were reported by 66.2% of patients who received flibanserin and 57.7% of patients who received placebo [56]. The majority of AEs were of mild intensity, emerged during the first 14 days of treatment and resolved during the trial.
AEs were dose related
Summary of adverse events reported during flibanserin pivotal trials (DAHLIA, DAISY, VIOLET and ORCHID).
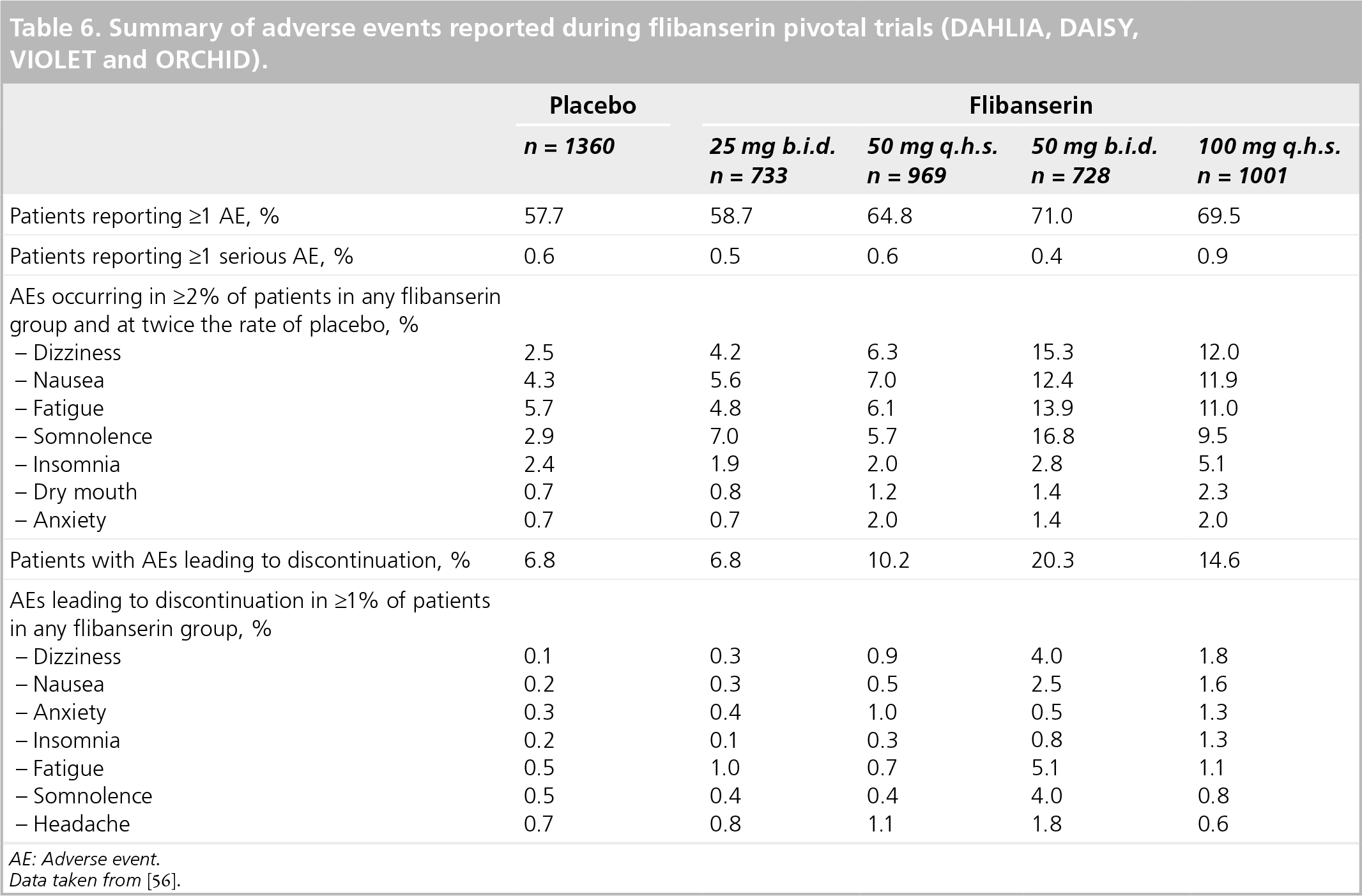
AE: Adverse event.
Data taken from [56].
Adverse events led to discontinuation of treatment in 14.6% of patients who received flibanserin 100 mg once daily at bedtime verses 6.8% of patients who received placebo
No deaths occurred in any of the trials except for a woman who died as a passenger in an airplane crash. The incidence of serious AEs was less than 1% in all of the trials and no serious AEs were considered related to flibanserin. No more clinically significant changes in blood pressure, pulse rate, physical examinations, electrocardiogram or laboratory parameters were observed than with placebo, nor did results reflect trends or clinical concerns. The rates of significant weight change (defined as a change of ≥7%) in patients associated with flibanserin treatment were 5.1% for weight loss and 0.8% for weight gain.
The following AEs were of special interest in flibanserin's development program due to findings in earlier studies or at the request of a regulatory authority: depression, suicide/self-injury, hemorrhage and ophthalmologic examination. Suicidality and depression (as it may relate to suicidality) are a potential concern for all centrally acting agents, including flibanserin. As other serotonergic agents (peripherally acting 5-HT2A inhibitors) have been tested as antiplatelet agents, the potential occurrence of hemorrhage was also of interest. Safety assessments showed that flibanserin was not associated with an increased risk of suicidality, depressive AEs, bleeding AEs or eye abnormalities, compared with placebo.
No excess compared with placebo was found with the use of flibanserin 100 mg once daily at bedtime in women with HSDD in overuse or withdrawal AE (abuse potential), hypersexuality, changes in sex hormones including indices of fecundity or adverse pregnancy outcomes (observations were limited to women whose treatment was stopped promptly upon discovery of pregnancy). Currently, results from adequate and well-controlled studies of flibanserin in pregnant women are unavailable.
Randomized withdrawal trial: ROSE
During the initial open-label phase of the ROSE trial, during which all patients received flibanserin, 511 out of 738 (69.2%) patients reported one or more AEs [40]. During the double-blind phase, the most frequently reported AEs (≥2% more patients in one group than the other) were diarrhea (3.7% for flibanserin vs 0.6% for placebo), dizziness (0.6% for flibanserin vs 2.9% for placebo), hypertension (0% for flibanserin vs 2.4% for placebo), nausea (2.5% for flibanserin vs 4.7% for placebo), sinusitis (4.9% for flibanserin vs 7.1% for placebo) and urinary tract infection (6.1% for flibanserin vs 3.5% for placebo). AEs leading to discontinuation occurred in 2.5 and 4.1% of patients in the flibanserin and placebo groups, respectively. No withdrawal reactions were reported upon switching from flibanserin to placebo after the first 24 weeks of exposure to flibanserin or following abrupt treatment discontinuation at week 48 [40].
Regulatory affairs
Flibanserin has not received marketing authorization or been approved for commercial use in any indication in any country.
Conclusion
Flibanserin 100 mg once daily at bedtime, the highest dose tested extensively in Phase III studies, has been shown to be superior to placebo for treating premenopausal women with generalized acquired HSDD. The consistent therapeutic effect in two well-controlled trials of the 100 mg once daily at bedtime dose regimen, and its use by most patients in a 1-year randomized withdrawal (efficacy maintenance) trial, support the claim to treat premenopausal women with flibanserin. Flibanserin 100 mg once daily at bedtime has been shown to be safe and well-tolerated, with no evidence of a withdrawal effect. Thus, the overall risk/benefit of flibanserin can be considered positive.
In the pooled North American trials, the flibanserin 100 mg once daily at bedtime group consistently showed a statistically significant difference from placebo for all of the important end points (SSE, FSFI desire domain score, FSFI total score, FSDS-R total score, FSDS-R Item 13). The European trial results were supportive. Responder end points anchored to PGI-I showed that flibanserin provided clinically meaningful as well as statistically significant improvements.
Side effects reported most frequently, and in higher proportions of subjects taking 100 mg flibanserin once daily at bedtime compared with placebo, were dizziness, nausea, fatigue and somnolence, at approximately 10–12% each, and insomnia, at approximately 5%.
Future perspective
Over the next 5–10 years, the field of FSD is expected to evolve rapidly in areas such as diagnosis, clinical measures, pathophysiology, pharmacologic understanding and therapeutics. The 5th revision of the American Psychiatric Association's Diagnostic and Statistical Manual of Mental Disorders is expected in 2013 and some changes to the classification of female sexual disorders are expected. Since DSM-IV was published, large databases on women with sexual dysfunction have been collected, providing an opportunity to base diagnostic criteria on solid empirical evidence for the first time. Over the past year, the neurobiology of the sexual response has been elucidated in some biochemical and anatomic detail, and brain imaging studies are beginning to allow close correlation between animal and human pharmacology; in the future greater strides should be made. The 3rd International Consultation on Sexual Medicine conducted in 2009 proposed large changes to standards for clinical trials in FSD, which will lead to more rigorously conducted trials in the next several years. Over the next few years, we should see the first pharmacologic treatments licensed for the treatment of FSD in premenopausal and postmenopausal women. Combination therapy (medications plus sex/psychotherapy) may also become available and prove beneficial. All of the above, plus growing cooperation between medical and psychosocial researchers in sexual health, should mediate rapid advances in therapeutics in this field.
Over the next few years, our understanding of the therapeutic value of flibanserin should broaden considerably too. Clinical trials are in progress to evaluate flibanserin in postmenopausal women with HSDD, and in women with sexual dysfunction who are taking SSRI or SNRI antidepressants.
Information resources
The 3rd International Consultation on Sexual Medicine (ICSM) was conducted under the auspices of the International Consultation on Urological Diseases and the International Society for Sexual Medicine (ISSM). As part of this consultation, committees of experts in diverse areas of sexual medicine reviewed the scientific literature and developed their conclusions and recommendations. Summaries of the committees' findings were presented and discussed at the 3rd ICSM congress held in Paris in July 2009. Most of the committees' reports were published in the Journal of Sexual Medicine in January 2010 [57] and provide a valuable overview of the current literature. Several of the committees' reports are cited in this article.
Executive summary
Flibanserin is a novel 5-HT1A agonist/5-HT2A antagonist.
Flibanserin administration leads to brain region-specific increases in dopamine and norepinephrine (which are involved in the ‘excitement’ phase of the sexual response) and decreases in serotonin (5-HT) (which is involved in the ‘inhibitory’ phase).
It is hypothesized that by selectively modulating these neurotransmitters, flibanserin may act to rebalance systems that have become imbalanced in women with Hypoactive Sexual Desire Disorder (HSDD).
The absolute bioavailability of flibanserin after oral administration is 33.2%, and it is moderately distributed in body tissues, with a half-life of about 10 h.
Steady state is established within 3 days.
Flibanserin is nearly completely metabolized (mainly by CYP3A4); metabolites are primarily excreted via urine (44%) and feces (51 %).
In North American premenopausal women with generalized acquired HSDD, 24 weeks' treatment with flibanserin 100 mg once daily at bedtime is associated with significant improvements versus placebo on all relevant end points, that are:
– Number of satisfying sexual events
– Sexual desire
– Distress associated with sexual dysfunction
– Overall sexual function
– Patient's Global Impression of Improvement
– Patient benefit evaluation
Responder end points anchored to the Patient's Global Impression of Improvement show that flibanserin produces improvements that are clinically meaningful to patients. Furthermore, a significant percentage of patients remit (no longer meet scale cutoffs for HSDD).
Flibanserin is well-tolerated at doses up to 100 mg/day (the highest dose tested in Phase III) for 24 weeks.
The most frequent adverse events reported in patients taking flibanserin 100 mg once daily at bedtime are dizziness, nausea, fatigue, somnolence and insomnia.
The metabolism of flibanserin is significantly inhibited by strong CYP3A4 inhibitors and greatly increased by CYP3A4 inducers.
No clinically relevant effect of flibanserin is observed on the pharmacokinetics of simvastatin, bupropion or a combination of levonorgestrel and ethinyl estradiol.
Flibanserin is administered orally.
The minimal effective dose of flibanserin is 100 mg once daily at bedtime.
Footnotes
