Abstract
Breast cancer is the most common malignancy in females. The origins and biology of breast carcinomas remain unclear. Cellular and molecular heterogeneity results in different distinct groups of tumors with different clinical behavior and prognosis. Gene expression profiling has delineated five molecular subtypes based on similarities in gene expression: luminal A, luminal B, HER2 overexpressing, normal-like and basal-like. Basal-like breast cancer (BLBC) lacks estrogen receptor, progesterone receptor and HER2 expression, and comprises myoepithelial cells. Specific features include high proliferative rate, rapid growth, early recurrence and decreased overall survival. BLBC is associated with ductal carcinoma in situ, BRCA1 mutation, brain and lung metastasis, and negative axillary lymph nodes. Currently, chemotherapy is the only therapeutic choice, but demonstrates poor outcomes. There is an overlap in definition between triple-negative breast cancer and BLBC due to the triple-negative profile of BLBC. Despite the molecular and clinical similarities, the two subtypes respond differently to neoadjuvant therapy. Although particular morphologic, genetic and clinical features of BLBC have been identified, a variety of definitions among studies accounts for the contradictory results reported. In this article the molecular morphological and histopathological profile, the clinical behavior and the therapeutic options of BLBC are presented, with emphasis on the discordant findings among studies.
Breast cancer is the most frequent female malignancy worldwide. According to the retrospective trend analysis of the WHO, breast cancer accounts for 23% of all cancers and is the second leading cause of cancer death with a mortality rate of 14% [1]. Human breast tumors are histologically complex, consisting of a variety of cell types besides cancer cells, and diverse in their biological behavior and response to therapy [2].
Breast cancer comprises a group of diseases with distinct molecular subtypes [3] and distinct etiologies [4]. Many studies have examined the associations between common breast cancer risk factors, race [5–11], clinical behavior and the prognostic value among the various molecular subtypes. However, traditional clinical and pathological prognostic factors such as age, estrogen receptor (ER) status, HER2 expression, tumor size and histological grade may miss the inherent heterogeneity of breast cancer, leading to different outcomes among patients with the same clinicopathological profile.
Gene expression profiling through cDNA microarrays opened a new field in research as an independent predictor by determining and classifying tumors into prognostically and predictively discrete groups in respect of local recurrence and survival. The recently recognized breast cancer with basal phenotype (BP), as defined either by immunohistochemical (IHC) [12] analysis or gene expression profile (GEP) arrays [13], comprises a subtype of triple-negative cancers associated with aggressive behavior and poor survival. Although basal-like breast cancer (BLBC) has been identified as a distinct entity with specific features, there is still discordance in definition and in clinical outcomes.
Because they are ER and HER2 negative, BLBCs do not benefit from antihormone receptor or anti-HER2 therapy, so chemotherapy remains the only available approach for systemic treatment, albeit with poor outcomes. Since over 50% of BLBCs express EGF receptor (EGFR), it is possible that anti-EGFR targeted therapy may be useful [12]. High levels of p53, a marker that is associated with poor prognosis, and BRCA1 mutations are typically expressed in BLBC [14]. Given that research increasingly reveals new detailed information on the origins of tumors, the correlation of genes and the significance of clinical biomarkers in tumor expression, the need for new biological targets for targeted therapies tailored to the specific features of BLBCs is undoubted.
The purpose of this review is to present the molecular profiling of breast cancer with an emphasis on the basal-like subtype, highlighting the therapeutic and prognostic implications. This review includes publications listed on the PubMed database before 31 August 2010.
Molecular profiling of breast cancer
The biology of breast cancer is not well characterized, with interactions between the different genes leading to carcinogenesis being unclear. It is evident that there is a large number of genes involved in cell growth, differentiation and apoptosis [15], leading to cellular and molecular heterogeneity in breast tumors. Gene microarray studies and other molecular approaches are beginning to reveal the cellular origins of breast cancer and consequent behavior of different types [16].
The breast comprises glandular tissue, adipose tissue and specialized mammary stroma [17]. Most breast diseases, either benign or malignant, originate from the functional terminal ductolobular units of the mammary gland [18]. Normally, ducts and lobules are lined by two cell layers: an inner/luminal and an outer/myoepithelial cell layer immediately under the basement membrane [19]. Luminal epithelial cells are characterized by low molecular weight luminal cytokeratins (CKs). Outer layer cells are morphologically and phenotypically heterogenous. Their behavior is affected by the location and hormonal status of the tissue and they combine features of both epithelial and smooth muscle cells [20]. Myoepithelial cells express basal CKs as well as myoepithelial-associated proteins.
Using gene expression patterns in cell lines of solid human tumors, Perou et al. found two distinct types of epithelial cell in the human mammary gland that are conveniently distinguished immunohistochemically – basal (and/or myoepithelial) cells and luminal epithelial cells – allowing a distinct ‘molecular portrait’ to be produced of each tumor [3]. The gene expression cluster characteristic for basal epithelial cells consisted of keratin 5, keratin 17, integrin-β4 and laminin. For the luminal epithelial cells the cluster comprised ER-α, GATA-binding protein 3, X-box-binding protein 1 and hepatocyte nuclear factor 3a. A hierarchical clustering method was used to group genes on the basis of similarity in pattern with which their expression varied over all samples. A subset of 496 genes from the 8102 examined was selected that demonstrated significantly greater variation in expression between different tumors than between paired samples and was termed ‘intrinsic’. Four clearly different molecular phenotypes were identified: ER+/luminal, basal-like, Erb-B2+ and normal-like breast cancers [3].
Subsequently, Sørlie et al. refined their findings in a larger number of tumors, and divided the luminal ER+ group into two or possibly three subgroups: luminal A showed the highest expression of the ER-α gene, GATA-binding protein, X-box binding protein 1, trefoil factor 3, hepatocyte nuclear factor 3a, estrogen-regulated LIV-1 and no expression of CERBB2; luminal B showed low to moderate expression of the luminal-specific genes but no expression of CERBB2; and luminal C was distinguished from the other subgroups by the high expression of a novel set of genes with unknown function [13]. In addition, the correlation between different subtypes of tumors and prognosis was evaluated and a highly significant difference in overall survival (OS) between the subtypes was shown: the basal-like and CERBB2+ subtypes were associated with the shortest survival and relapse-free survival rate. Interestingly, the basal-like subtype represented a high frequency of mutations in the TP53 gene, which predicted poor prognosis and was associated with poor response to systemic therapy [21]. Comparing multiple gene microarray datasets Sørlie et al. concluded that breast cancer subtypes were associated with significant differences, strongly supporting the theory that many of the subtypes represent biologically distinct disease entities. By including a group of tumors from BRCA1 carriers in the analysis, it has been found that this genotype predisposed to the basal tumor subtype [22].
Kapp et al., using centroid analysis, a novel method that can be applied to any microarray, confirmed the most consistently identifiable subtypes of breast cancer tumor tissue microarrays (ESR1+/CERBB2−, ESR1−/CERBB2− and CERBB2+) and showed that these subtypes significantly predict OS and distant metastasis [23]. Nielsen et al. immunohistochemically examined a panel of 21 basal-like tumors, which was determined using GEPs and subsequently reported that a panel of four antibodies (ER, HER1, HER2 and CK5/6) could accurately identify basal-like tumors and showed high specificity [12].
In general, all of the above studies classify breast cancer into four main groups: two of them are ER+ and include luminal A (ER+, progesterone receptor [PR]+ and HER2−) and luminal B (ER+, PR+ and HER2+) subtypes and two are ER−, HER2 overexpressing (ER2−, PR−, HER+) and basal-like (ER-/low, PR-/low, HER2-/low). Originally there was also a normal breast-like group (ER−, PR−, other nonepithelial cells, adipose cells) but this was probably due to inclusion of non malignant tissue. The profile of the subtypes is outlined in
Immunohistochemical subtyping of breast cancer.

+: Positive; -: Negative; ER: Estrogen receptor; PR: Progesterone receptor.
Stem cells
Several studies support the existence of stem/progenitor cells in mammary glands. Genetic and epigenetic changes in normal stem cells result in the development of cancer stem cells [28]. Disruption of the stem cell ability for self-renewal and differentiation may lead to carcinogenesis and the genomic alterations giving different cancer phenotypes [29]. In experimental models involving murine mammary cells, two groups of cells were identified, called ‘side population’ and ‘label retaining’. Although undifferentiated, when cultured, they grew as typical differentiated epithelial cells. The transplantation of these cells into clear mammary fat gave rise to epithelial ductal and lobular structures [30,31].
Similarly, in a study with human cells, a distinct population of cells was identified that expressed CK5, which is responsible for regeneration of the human adult breast epithelium. These cells differentiated towards glandular epithelial or myoepithelial CK5-negative passing through cells expressing either CK5/CK8/18/19 or CK5/smooth muscle α-actin (SMA)+ intermediates. Given that the vast majority of breast cancers demonstrate the glandular epithelial immunophenotype, it is likely that the neoplastic cells derive from a late stage of the glandular epithelial differentiation pathway [32]. In another study using cell surface markers and immunomagnetic sorting from primary cultures of reduction mammoplasties, a mucin-negative/epithelial-specific antigen-positive epithelial cell population within the luminal epithelial lineage was isolated with the ability to self-regenerate into myoepithelial cells and form terminal duct lobular units in the human breast [33].
Al-Hajj et al., using a model in which human breast cancer cells were grown in immunocompromised mice, found that only a minority of breast cancer cells had the ability to form new tumors and identified the tumorigenic from the nontumorigenic cancer cells based on cell surface marker expression. The tumorigenic CD44+CD24-/low lineage− population differentiated, proliferated and generated new tumors [34]. The identification of breast cancer stem cells in vivo and the reproduction in vitro offers the ability to investigate their origin, the molecular alterations that affect them and their interaction with the microenvironment. This knowledge will elucidate critical points for developing new therapeutic strategies and improving diagnosis and prognosis for breast cancer patients.
Defining triple-negative/basal-like breast cancers
Basal-like breast cancer, as defined by microarrays, may demonstrate either low expression of hormonal receptors and HER2 protein or often a clear hormonal and HER2− profile (triple negative). The overlap observed between the triple-negative and basal-like phenotype opened a new debate that often leads to confusion.
Molecular profile
Basal-like breast cancers originate from the outer cell layer of the ductal and lobular structures of the glandular breast tissue, adjacent to the basal membrane. These cells are myoepithelial [35] with features of both epithelial and smooth muscle cells and express markers specific to smooth muscle, including myofilament proteins, actin and myosin heavy chain. Additionally, myoepithelial cells are referred to as basal cells because they usually express high molecular basal CKs (CK5, CK14, CK17) [19,36].
While the original molecular subclassification of basal-like status provide a myoepithelial cell origin, most basal-like cancers express luminal-type CKs (i.e., CK8/18, CK19) [37] and in addition only a few basal-like cancers express typical myoepithelial markers (such as actin and p63) [38]. Rakha et al. reported that breast cancers with basal and myoepithelial phenotypes are distinct groups of tumors sharing some common morphological features and having a poor prognosis. Moreover, the study concluded that basal rather than the myoepithelial phenotype shows a reduced disease-free interval [38]. It is unclear whether basal-like tumors originate from differentiated myoepithelial cells, or involve cells that co-express both basal and luminal CKs or are composed of two distinct lineages of epithelial cells [39]. Current research on stem cell biology indicates that basal-like tumors may derive from mammary stem cells, identifying similarities between uncommitted cells with self-renewal potential and basal-like cancers [40].
Triple-negative breast cancer (TNBC) is defined by ER, PR and HER2 negativity in clinical assays for these biomarkers [41]. On the other hand, basal-like molecular phenotype is determined by an intrinsic gene set identified by using mRNA gene expression profiling [3]. Although the frequent ER, PR and HER2 negativity in BLBC was used as an indicator for identifying this subtype from the TNBC group, it is apparent that not all BLBCs express a triple-negative phenotype and conversely not all TNBCs bear a basal-like GEP [12,41]. According to the ‘intrinsic gene list’, 15 % of BLBCs classified express positivity in at least one of ER, PR or HER2 status and, conversely, not all TNBCs show basal-like characteristics as defined by microarray analysis [12]. Moreover, it has been found by microarray analysis that TNBC also includes a part of the normal breast-like subtype [3,24,42]. It is evident that TNBC is a heterogeneous group of tumors with BLBC comprising only a subset. Similarly, BLBC shows significant heterogeneity in microarray analysis and at least four distinct subsets have emerged according to the expression of different gene modules identified by a novel algorithm and clustering method [43,44].
The application of RNA expression profiling to accurately distinguish TNBC and BLBC revealed that most TNBCs fall in the BLBC subtype according to the intrinsic gene expression patterns [12,45]. These genes include:
Structural elements of basal epithelial cells: CK5, CK17, P-cadherin, vimentin, metalloprotein IX, activating transcription factor 3 and fatty acid binding protein 7;
Interaction with the extracellular matrix: laminin, fascin, α6β4 integrin, collagen type XVII α-1 and metalloproteinase 14 [3,24,46,47];
Carcinogenesis: via signaling pathways including MEK, ERK, PI3K, AKT kinases and NF-κB, and via signal transduction including EGFR, c-kit, caveolin 1 and 2, c-fos, c-jun and hepatocyte growth factor [46–50];
Proliferation: MCM2, STK6, cyclin E1, CDC2, MAD2L1, BUB1 and topoisomerase IIa;
Heat-shock proteins (hsps): αβ-crystallin and hsp27 [51];
Four distinct clusters of genes have been identified and classified according to the characteristic gene modules in ER− basal-like tumors: cell cycle and cell proliferation genes (CC+), immune response genes (IR+), cell cycle and immune response genes (CC+/IR+) and extracellular matrix genes (ECM+). The above findings confirm that the heterogeneity of ER− basal breast cancer is related to the variability in expression levels of complement and immune response pathway genes [52].
In microarray-based comparative genomic hybridization studies TNBC and BLBC are associated with greater genetic complexity as they demonstrate the highest frequency of DNA losses and gains compared with other subtypes; this is suggestive of distinct mechanisms of genomic instability that underlie their pathogenesis [53,54]. It has also been reported that TNBCs and BLBCs show low-level gains and deletions more often and high-level gains/amplifications less frequently, indicating that this pattern of genomic changes may be due to a defect in the double-strand DNA break repair mechanism.
Some of the recurrent amplifications identified in these tumors include 12p13 [55] and 7p11.2, where the EGFR gene is involved; 7q31, affecting caveolin 1; and 6p21-p25, which harbors several candidate oncogenes such as DEK, E2F3, Notch4, Pim1 and CCND3 [56]. Furthermore, BLBC show a distinct comparative genomic hybridization profile [57,58] with higher rates of loss at chromosomes 16p, 17p, 19p and Xp, lower rates of loss at chromosomes 4q, 9q and 13q, and gains at 17q and 20q. BLBC is also associated with the lowest rate of loss of heterozygosity at 16q23-2 and 1p32-ter, and 100% lack of the locus at 5q11, where many checkpoint DNA repair and tumor suppressor genes are involved (i.e., MSH3, RAD17, APC, RAD50 and XRCC4) [59]. Interestingly, BLBC and TNBC are associated with sporadic mutations and/or p53 protein overexpression. Sorlie et al., using GEP, found p53 mutations in 82% of BLBCs compared with 13% in the luminal A subtype. Considering the genomic instability of BLBC, the lack of functioning p53 may be crucial in cell survival [13].
Despite the detailed information embedded in microarray analysis, this technology is not suitable for large population studies or routine clinical application because of the expense of arrays and the difficulty of having available a number of formalin-fixed, paraffin-embedded tissues to use for this purpose [60]. Basal CKs are currently regarded as one of the most reliable markers to classify BLBCs because of their overexpression in basal/myoepithelial cells of the normal breast. However, it is evident that not all CKs are expressed in BLBCs but only one or more of these. The most frequent CKs expressed in BLBCs are CK5/6 in up to 70% of cases and CK14 in up to 41.2%, and the least frequent is CK17. In addition, luminal CKs expressed by luminal epithelial cells such as CK7, CK8/18 and CK19 have been found to be present in up to 84% of BLBCs [37,61].
Several studies have evaluated CK5/6 [14,62,63], CK14 [57,58,64], a combination of CK5/6 and CK14 [61,63,65–68], CK5/6 and CK17 [69], and a combination of the three CKs [66,70–72] using variable staining patterns, resulting in contradictory results. In one of these studies a cut-off of 10% or greater basal CK staining was used to signify positivity [73,74], a cut-off of 1% was used in another study [75], whereas most studies do not explain how positivity was quantified [65,66]. Hence, it is unclear what percentage of positively stained cells should be present in a tumor to be determined as basal-like and whether different levels of positivity predict for specific behavior.
A plausible explanation for these contradictory results has been given by Rakha et al., who suggest that differences in the cohorts of patients, composition of the control group and the use of a single CK in basal-like defining led to the Will Rogers phenomenon; that is, the changes in the average values into two sets when an element moves from one set to the other [20,76]. Moreover, they insist that a single CK is not sufficient to identify all basal-like tumors, or in other terms the subgroup with the more aggressive clinical behavior. Therefore, CK5/6 is considered by many authors as the most precise marker for BLBC identification and is widely used by pathologists for diagnostic purposes.
Nielsen et al., in an IHC-based analysis in specimens previously defined as basal-like by DNA microarray, found that ER and HER2 negativity combined with CK5/6 and EGFR positivity define BLBCs more precisely, showing specificity of 100% and sensitivity of 76% [12]. In addition, in this study, both basal markers independently correlated with decreased survival. These criteria were consequently adopted by many other studies, either in combination with additional markers or by using only some of those mentioned above. Livasy et al., attempting to define the immunophenotype of BLBC further, applied a panel of IHC markers to luminal-like, HER2+ and basal-like cancers that were previously analyzed by DNA microarray. The immunoprofile best predicting the basal-like cancer genome was found to be vimentin, EGFR and CK5/6 positivity, and ER and HER2 negativity [37].
C-kit overexpression in combination with HER1 positivity and one basal CK in ER− and HER2− tumors have also been suggested to define BLBC. The presence of c-kit and HER1 in BLBC may represent a benefit for the patients because of the application of EGFR- and c-kit-targeted treatments [29].
Although BLBC has been identified by gene expression profiling and/or IHC methods as a distinct entity with special morphologic features, there is no generally accepted definition. Despite the large number of markers and myriad genes associated with the BLBC phenotype, there is no international consensus on the composition of a marker panel that could precisely define this subtype of triple-negative tumor.
Considerable interlaboratory variability, especially in relation to ER expression, has been documented showing a technical false-negative rate of between 30 and 60% [77]. This variability may be caused by differences in methodology; however, it is suggested any definition based on all negative results should be generally avoided.
In the last decade, the development of microarrays and gene expression analysis of human tumors elucidated the heterogeneity of breast cancer and the fact that distinct subtypes of breast cancer are completely different diseases. However, this technology cannot replace traditional clinicopathological markers in clinical practice and treatment decision-making. It has been shown to be rather complementary to traditional clinicopathological parameters [78]. On the contrary, currently there are no IHC markers that could efficiently detect all BLBCs and it is reported that approximately a third of BLBC is being missed by the use of various IHC markers [79]. To date, the most reliable and applicable definition in routine clinicopathological practice for BLBC is the ER−, HER2-/low, CK5/6+ and EGFR+ profile, which shows the highest degree of diagnostic specificity and sensitivity [12].
Pathological features of basal-like breast carcinoma
Breast cancers comprise a heterogenous group of tumors differing in morphology, molecular profile, phenotype, biological behavior, treatment and prognosis. Currently, pathologic features are generally used to determine the biological behavior of tumors in order for the management of the disease to be determined. Although these features are not specific to the different subtypes, many investigators have studied BLBCs on a histomorphological basis.
Large tumor size and high proliferative activity are common traits in BLBC. Livasy et al. reported a high mitotic rate in GEP-defined BLBC (average 45 mitoses per ten high power fields) [37] and subsequent studies confirmed this observation in BLBCs [38,63,75,80–84]. Most IHC studies evaluating tumors of different grades reported that basal phenotype correlates with larger tumor size [14,38,61,73,75,85] but some others reached different conclusions [63,65,86,87]. GEP studies did not find correlation between the molecular subclasses and tumor size for tumors less than 21 mm [24,47,88]. In a multivariate analysis tumor size was shown to be a statistically significant variable over the basal phenotype in association with disease-specific survival [75].
Central acellular zones and necrosis in BLBC specimens have been described in several IHC-based studies. Tsuda et al. reported 18 ductal carcinomas with myoepithelial differentiation that showed large, geographic central acellular zones with hyaline material, necrotic tissue and collagen [89]. Rakha et al. separated tumors showing basal and myoepithelial phenotype according to the markers they expressed and reported that one of the most characteristic features of the myoepithelial group was the presence of a central acellular zone [38]. One IHC study of grade III ductal cancers demonstrated that central scar was a more common feature in BLBC compared with other subtypes [82]. However, this finding may be artefactual because core biopsy is performed preoperatively, resulting in a central zone of fibrosis and scar formation in the final surgical specimen [79]. In the only GEP-based morphologic study none of the BLBCs displayed a central acellular zone but large zones of geographic necrosis were apparent in 75% of cases [37]. In addition, association between basal phenotype and tumor necrosis has been found in several other studies [38,82]. Other common features that have been described include the presence of spindle and/or sarcomatoid tumor cells, squamous metaplasia and/or squamous differentiation, metaplastic features/differentiation and high-grade nuclear features [12,37].
Specific architectural features have been observed in BLBC in both GEP- and IHC-based studies. Pushing noninfiltrative tumor borders and variable degrees of lymphoplasmacytic infiltration are more frequent in BLBCs than in nonbasal cancers [37]. Interestingly, Livasy et al. described a ‘ribbonlike’ architecture as a common finding in GEP-defined BLBCs [37,82,90]. Many BLBCs have the appearance of sheets of cells with minimal tubule formation; an ‘adenoid cystic’ change has also been described [38].
Multiple IHC studies have demonstrated that the most common histologic subtype of BLBC is invasive ductal carcinoma not otherwise specified (IDC-NOS). Rakha et al. found that IDC-NOS constituted 68% of the basal phenotype [38] and Kim et al. found that IDC-NOS comprised 86% of BLBC [61]. However, this histological subtype is the most common among all breast cancers and it is anticipated that the IDC-NOS would be the most predominant in most of their subclasses [91]. By contrast, invasive lobular carcinoma showed basal phenotype in only 7.69% of cases in one IHC-based analysis [92]. In another study, immunoreactivity for the basal CK5/6 and CK14 was noted in 5.4 and 7.4% of invasive lobular carcinoma, respectively [93].
It has been documented that BLBC correlates with medullary carcinoma, with many studies reporting a significant percentage of these histological types showing positivity for basal CKs [38,61,94]. In one study Tot found that 25% of the typical, 43% of the atypical and 20% of the metastatic medullary carcinomas showed CK5/6 positivity [95] and another recent study also reported that more than 50% of medullary carcinomas demonstrate a degree of basal and/or myoepithelial phenotype and immunoreactivity for P-cathedrin, CK14, SMA and S100 [94]. GEP experiments showed that in a group consisting of IDC-NOS and medullary carcinomas, 95% of medullary carcinomas clustered with the basal subclass of IDC-NOS. It was apparent that genes characteristic for medullary carcinoma, such as those associated with muscle development, smooth muscle contraction, actin cytoskeleton organization and biogenesis, were included in basal grade III IDC-NOS tumors [96]. Furthermore, pushing tumor border and tumor edge stromal lymphocytic infiltrations are medullary features that have been noted in GEP-defined BLBC [37].
Most carcinomas that show myoepithelial differentiation display immunoreactivity for basal CKs and these are myoepithelial carcinoma (spindle cell and poorly differentiated), adenoid cystic carcinoma and low-grade adenosquamous carcinoma [93,97,98]. Several authors have demonstrated that basal and/or myoepithelial markers are expressed in 30–75% of metaplastic carcinomas. According to the WHO classification, metaplastic carcinomas include: adenocarcinoma with spindle cell differentiation, adenosquamous carcinoma (including mucoepidermoid carcinoma) and carcinoma with mesenchymal elements (carcinoma with chondroid or osseous metaplasia and carcinosarcoma) [99]. Some of these rare breast tumors showed ER and HER2 negativity and EGFR positivity in different studies [100,101].
Two studies using at least one basal marker in the more uncommon carcinoma subtypes found basal phenotype in the following: invasive papillary (43%), mucinous (21%), mixed ductal and lobular (17.6%), tubulolobular (16.7%), tubular mixed (11.4%), invasive cribriform (7.7%) and pure tubular (4.2%) [38,61]. It has also been reported that ER and PR negativity, metaplastic features or basaloid morphology are frequent findings in cancers arising from microglandular adenosis [102].
The theory that the high-grade basal type of IDC-NOS arises from high-grade ductal carcinoma in situ (DCIS) is under investigation. It has been reported that a subset of DCIS identified as basal type demonstrate basal phenotype markers [69,70,103–106]. Dabbs et al. described the morphologic features of 13 triple-negative DCIS with basal phenotype that were involved in the surgical specimen of an invasive tumor. All DCIS consisted of solid, flat or micropapillary type, were situated peripheral to the main invasive tumor, were of high grade and showed comedo necrosis and dense lymphocytic infiltration [90]. In a study population Bryan et al. evaluated 66 cases of pure DCIS and found 6% of cases to express triple-negative basal phenotype, suggesting that there are in situ precursor lesions for TNBCs and BLBCs [70]. Livasy et al., studying 245 pure DCIS, found a prevalence of 8% in basal-like DCIS [106]. It is unknown whether basal-like DCIS progress to invasive cancers more rapidly than the other subtypes but it has been hypothesized that the high proliferation rate seen in TNBCs and BLBCs leads to rapid overgrowth of basal DCIS. This could be a plausible explanation for the little or nonexistence of DCIS in specimens with invasive BLBC.
Heritable BRCA1 mutation breast tumors have been demonstrated to correlate with TNBCs and BLBCs. Approximately 70–80% of BRCA1 tumors diagnosed before the age of 50 years are triple negative and have molecular, morphological and IHC similarities with basal-like phenotype as determined by IHC and GEP arrays [14,107]. They are frequently of high grade, express basal CKs, EGFR, cyclin E, and present TP53 mutations, high mitotic index, metaplastic characteristics [108] and give rise to early visceral metastases [71]. Both basal-like and the majority of BRCA1 cluster within the cell cycle and immune response subclass (CC+/IR+) of ER− breast cancers showing similar biological pathways [109]. Moreover, loss of X-chromosome inactivation has been identified frequently in both basal-like and BRCA1 breast tumors [110].
Research is increasingly directed to sporadic BLBC, occurring in women without germline BRCA1 mutations [108]. An epigenetic dysfunction in the BRCA1 pathway has been hypothesized to facilitate sporadic BLBC, and BRCA1 gene promoter methylation has been implicated in over 60% of medullary and metaplastic breast cancers of basal-like phenotype [111]. Furthermore, an alternative epigenetic mechanism of BRCA1 inactivation has been reported: the upregulation of ID4. ID4 is a helix–loop–helix protein that has been shown to act as a BRCA1 gene silencer expressed in high-grade basal-like carcinomas of ductal/not otherwise specified morphology [112]. Notably, BRCA1 is known to be involved in the normal development of mammary gland and the regulation of ER expression [113]. It has also been been suggested to play a role the regulation of mammary progenitor cell fate and the lack of BRCA1 may inhibit differentiation into ER+ luminal cells so that stem cells progress to TNBC and BLBC development [114].
Risk factors
Several studies suggest that traditional risk factors such as age and race have different impacts in BLBCs and TNBCs versus luminal breast cancers defined by immunophenotyping [115–120]. The population-based Carolina Breast Cancer Study (CBCS) reported that 26% of African–American women had BLBC compared with 16% of non-African–American women, and that the excess of BLBC occurred more frequently in premenopausal African–American women compared with postmenopausal (27.2 vs 16%, respectively) [117]. It remains uncertain whether these racial differences are related to genetic or environmental factors or a combination of both.
Basal-like breast cancer appears to relate to young age, high parity, lack of breastfeeding, taking medications to suppress lactation, early onset of menarche [117], young age at first birth and abdominal adiposity [116,121] in contrast to the luminal variety, which is associated with nulliparity and later age of first birth [121]. Millikan et al. proposed that encouraging women to extend breastfeeding and reducing abdominal adiposity by diet could prevent approximately 68% of basal-like cancers [116]. Although lifestyle factors such as diet poor in vitamin D [122] and decreased physical activity [123] seem to increase the risk for ER− breast cancers, Bardia et al. do not find significant association between physical activity and ER− cancers [124]. Socioeconomic status as seen in lower levels of mammographic screening, late stage of survival and poor prognosis were also associated with BLBCs [60]. Moreover, it has been reported that BLBCs present more frequently as interval rather than screen-detected cancers. A Norwegian study also found that basal-like phenotype was significantly increased compared with the other types in the interval group, suggesting that specific pathways, determined early in the life of the cancer, may determine the rapid growth and aggressiveness of interval cancers [83]. It is possible that there are interactions between biology, socioeconomics and race, and future research in racially diverse populations will be needed to disentangle these factors and address disparities in breast cancer [120].
Mammographic prognostic indicators
Mammographic-specific features have been reported as prognostic indicators in small screening-detected tumors in terms of early detection and appropriate management [125]. Screen-detected breast tumors with basal phenotype have different mammographic appearance than the nonbasal tumors. BLBCs are more likely to exhibit an ill-defined mass and comedo calcification, indicating high grade and poor prognosis. By contrast, nonbasal tumors exhibit mammographic spiculation or stellate-like findings, predicting good outcomes. However, it is unknown why BRCA1-mutated tumors that particularly frequently express basal-like phenotype rarely exhibit comedo calcification or spiculation on mammograms [125].
Basal-like versus triple negative
Triple-negative tumors usually have histopathological [126], clinical and demographic features resembling BLBCs [127], but despite the extensive overlap they are not synonymous [20]. Both types are ER−, PR− and HER2−, express HER1/EGFR in up to 66% of cases [127], behave aggressively and often present as interval cancers. In the main they are high-grade invasive ductal, of no special type, metaplastic carcinomas or medullary cancers and they are associated with BRCA1 mutation. Both types are more prevalent in African–American premenopausal women, do not successfully respond to the adjuvant therapy, more often metastasize to brain and lungs, and have poor prognosis. However, depending on the expression of at least one of ER− or HER2− status in conventional formalin-fixed, paraffin-embedded specimens, the difference between the two types ranged from 15 to 54% of basal-like cancers [20,127]. In gene expression arrays, 85% of triple-negative tumors were defined as basal-like according to their ER− and HER2− profile, and by IHC analysis 46–90% of triple-negative tumors expressed the basal CKs and HER1/EGFR markers [126–128]. BLBCs are associated with more aggressive behavior and worse prognosis. The specific molecular features make this subtype suitable for treatment with targeted therapy.
Prognosis
Triple-negative breast cancer/BLBC is associated with aggressive clinical behavior, visceral metastasis, decreased survival and high mortality. Recurrence and death rates are higher for TNBC/BLBC mainly in the first 3 years. Moreover, patients with residual disease after neoadjuvant chemotherapy have significantly worse OS compare with the non-TNBC/BLBC subtypes. If a pathologic complete response (CR) is achieved, patients with TNBC/BLBC have similar survival with non-TNBC [117]. A population-based, case–control study by Carey et al. showed that breast cancer-specific survival (BCSS) was shortest for HER2+/ER− and basal-like subtypes [27].
Locoregional relapse
Currently there are only limited data on the relationship between breast cancer subtype and the risk of local recurrence. Although the presence of TNBC and BLBC has been extensively evaluated for prognostic significance at the time of first diagnosis, the data for the prognosis after recurrence are limited. It is unclear if breast-conserving therapy is a safe therapeutic option for BLBC but one study showed the same rate of locoregional relapse compared with other subgroups [129].
A database of 482 patients with breast cancer managed by conservative surgery followed by radiation was studied by Haffty et al., aiming to determine the prognostic significance of TNBCs with respect to locoregional relapse and distant metastasis. At 5 years, although the triple-negative cohort had a poorer distant metastasis-free rate, no significant difference in local control between the triple-negative and other subtypes (83 vs 83%, respectively) was noted [129].
Freedman et al. confirmed the above, but additionally observed that HER2 overexpression had the highest rate of distance metastasis and therefore the lowest recurrence-free survival rate. This finding may be due to the fact that in the study period trastuzumab (Herceptin®; Roche) was not administrated routinely and was offered only to a few patients [130]. Additionally, Dragun et al., in a multivariate analysis of 793 patients with breast cancer that had undergone lumpectomy and subsequent radiation, assessed that both the HER2+ and basal subtypes were associated with an increased risk of local recurrence and exhibit a statistically significant increase in distance metastasis. The HER2+ overexpressing group exhibited slightly poorer outcomes but no patients received adjuvant Herceptin.
Triple-negative breast cancer patients not given radiotherapy after total mastectomy had a significantly higher locoregional recurrence rate compared with those who had undergone conservative surgery combined with radiation therapy, but in part this may have been due to larger tumor size [131]. In a prospective randomized trial, Kyndi et al. studied the effect of postmastectomy radiotherapy (PMRT) in locoregional recurrence in terms of isolated first event, distant metastases and OS among breast cancer subtypes. No significant OS improvement after PMRT was found among patients with an a priori poor prognosis (HER2+ and triple negative) and in particular the HER2+ subtypes. Furthermore, significantly smaller improvements in locoregional recurrence after PMRT were found for the triple-negative and HER2+ subtypes compared with the luminal group [132]. Dawson does not suggest that a more radical approach to local or axillary surgery is required, although distinct mechanisms of metastatic spread may be important in these tumors [39].
It is notable that some patients with the basal subtype have BRCA1 mutations. This appears to be the only contraindication for BCT as the multifocal and multicentric distribution in TNBC/BLBC is an uncommon feature.
Another study by Dent et al. in patients treated either by mastectomy or by BCT reported that both TNBCs and non-TNBCs had similar local recurrence rates. However, there was a significantly shorter mean time to local recurrence among the triple-negative patients (2.8 vs 4.2 years; p = 0.02) [133].
Parikh et al. evaluated the prognostic significance of basal-like breast carcinoma at the time of ipsilateral breast tumor recurrence after conservative surgery and radiation treatment [134]. They found that basal-like phenotype was associated with decreased median time to recurrence (<3 years) and despite administration of chemotherapy at the time of recurrence, the 5-year metastasis-free survival and 5-year OS were significantly poorer in BL/triple-negative tumors than the non-triple-negative tumors. The clinical, pathological and molecular features of BLBC are shown in
Features of triple-negative/basal-like breast cancers.
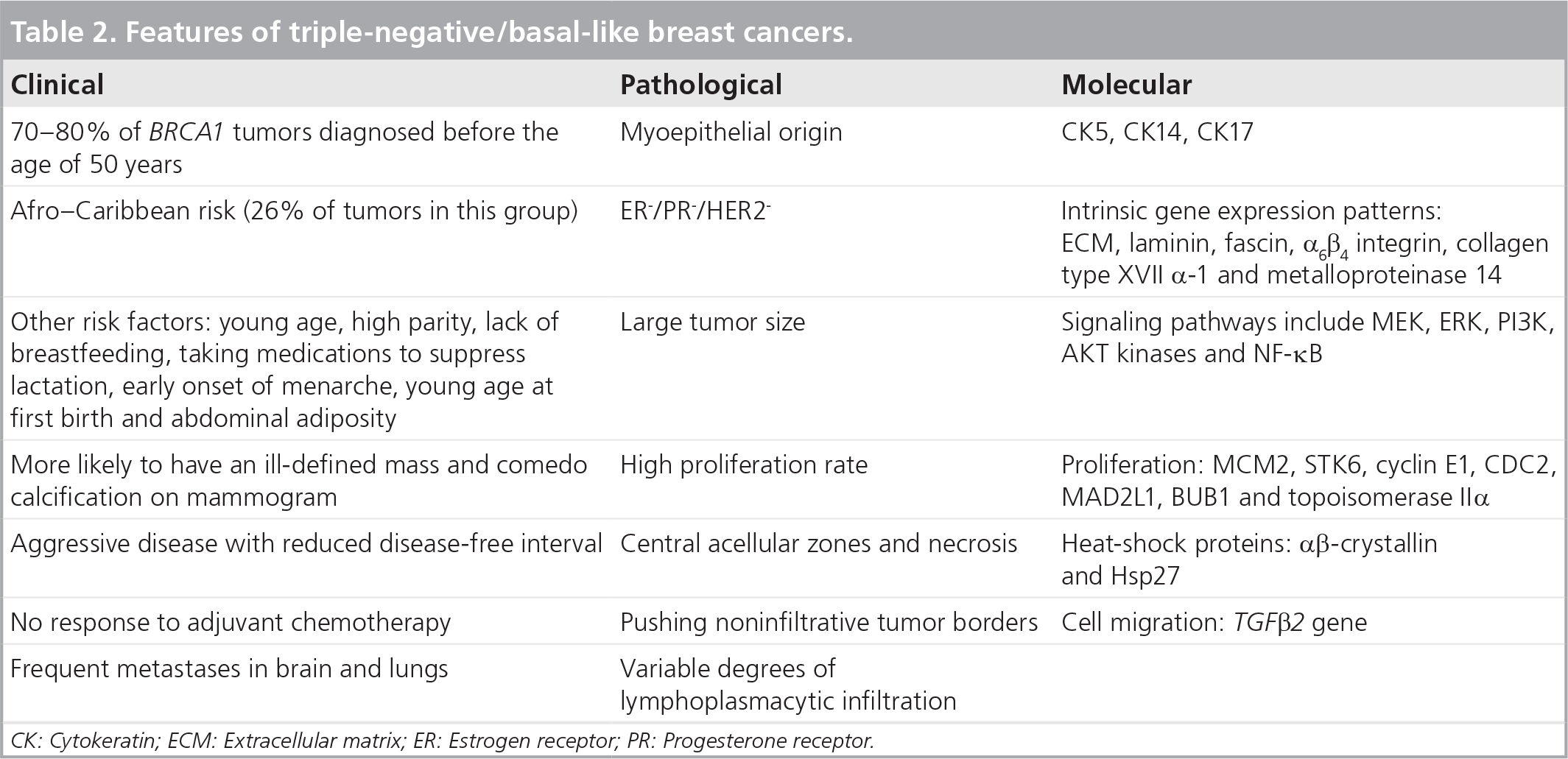
CK: Cytokeratin; ECM: Extracellular matrix; ER: Estrogen receptor; PR: Progesterone receptor.
Distant metastasis
It is well established that TNBCs/BLBCs are associated with higher rates of distant metastasis and specific patterns of distant relapse, with greater likelihood of visceral metastasis to the brain and lungs, and lower incidence to axillary nodes, liver and bones [117,135]. The incidence of lymph node metastasis at time of diagnosis in TNBC is controversial. Dent et al. found a higher rate of lymph node metastasis compared with controls [133], while other studies showed inverse results [73].
Many studies have shown that patients with TNBC have shorter time to develop brain metastasis from the initial diagnosis, shorter survival after the development of metastasis and poorer OS compared with the other subtypes of breast cancer [136–138]. Venkitaraman et al. proposed routine screening of the brain and prophylactic cranial irradiation in patients with TNBC to prevent brain metastasis [137]. Several reasons have been implicated in the higher propensity of brain metastasis in TNBC. These include the cyclooxygenase COX2, the EGFR expression and the a2,6-sialytransferase that mediate cancer cells’ passage through the blood–brain barrier [139]. In particular, the impermeability of the blood–brain barrier to chemotherapy and the high mitotic rate combined with EGFR expression explain the tendency of BLBC to metastasize to the brain [137].
In their pivotal study, Perou et al. found that overall patterns of gene expression were as similar between metastasis and primary tumor as they were in repeated samplings of the same primary tumor. This suggests that the molecular program of a primary tumor is retained in its metastases. This may explain the poorer survival of BLBCs after distant metastasis compared with the other subtypes. Ding et al. carried out genomic analysis on the primary BLBC and a brain metastasis and found that there were two new mutations in the metastasis and a deletion not present in the primary tumor, suggesting origin from a minority of cells in the primary tumor [140].
Tsuda et al., using Cox's multivariate analysis, showed that myoepithelial phenotype combined with large, central acellular zones in invasive ductal carcinomas were an indicator of high risk of brain and lung metastases and of death by cancer, independent of nodal status and tumor size [135]. Examination for large, central acellular zones and myoepithelial immunophenotype in high-grade invasive ductal carcinomas appears helpful in predicting patient prognosis and preferential metastatic sites of the tumors. McSherry et al. have suggested that individually invading cells from poorly differentiated cancers may have a better chance of establishing viable metastases than collectively migrating cell clumps from well-differentiated tumors [141].
Differences in the timing of relapse
Recurrence risk is highest the first 5 years after diagnosis and relatively few systemic recurrences occur after this period. For TNBC distance metastasis peaked at approximately 3 years after diagnosis. By contrast, the recurrence risk
for non-TNBC remains constant over time [133]. Dent et al. studied a cohort of 1601 patients with breast cancer with a median follow-up of 8.1 years. In total, 180 out of 1601 patients (11.2%) had TNBC. Compared with other women with breast cancer, those with TNBC had an increased likelihood of distant recurrence and death within 5 years of diagnosis but not thereafter. The pattern of recurrence was also qualitatively different; among the triple-negative group, the risk of distant recurrence peaked at approximately 3 years and declined rapidly thereafter. Among the ‘other’ group, the recurrence risk seemed to be constant throughout the period of follow-up. TNBC seems to be cured if it remains recurrence free for the first 5 years after diagnosis.
Patients with TNBC expressing a basal phenotype have a significantly shorter disease-free survival compared with those without [73]. Additionally, they appear to have a significantly shorter survival following metastasis than the non-TNBC/BLBC controls [74,133]. Fulford et al. specifically demonstrated that basal-like tumors differ in their biological behavior from other tumors, showing a distinct pattern of metastatic spread. Compared with other grade III tumors, basal-like tumors appear to have a relatively good long-term survival but survival after metastases is poor [74]. They recognized that basal CK14 expression in breast cancer is associated with ER− status and axillary nodes negative for metastasis, but with more frequent lung and brain metastases and poorer prognosis than other carcinomas. Moreover, two subgroups of BP tumors were identified: one with early relapse and more aggressive clinical course and another that despite the expected poor prognosis does not relapse.
Similarly, Cheang et al. insisted on the superior prognostic value of basal markers when compared with relying on the triple-negative status alone. This study compared the prognostic significance of BCSS between three- and five-biomarker surrogate panels to define intrinsic breast cancer subtypes. Among 3744 interpretable cases, 17% were basal with the triple-negative definition showing 10-year BCSS of 67%. Using the five-marker definition, 9% were basal with a 10-year BCSS of 62%. Among triple-negative patients treated with adjuvant anthracycline-based chemotherapy, the group with basal markers demonstrated significantly worse outcome. This indicates that the addition of EGFR, and CK5/6 in triple-negative cancers, provides a more specific definition of BLBC that better predicts breast cancer survival [126].
It has been reported that basal CKs are independent markers of poor prognosis after adjustment for variables such as age, tumor grade, tumor size and lymph nodes status [38,67,72,93,126]. Rakha et al. emphasized the strong predictive value of basal against the myoepithelial phenotype with the basal type being associated with reduced OS and disease-free interval [38]. In another study of 1872 patients with invasive breast carcinomas and long-term follow-up the clinical significance of BP was assessed. It was found that grade III BLBC had highly significant prognostic impact followed by tumor size, while the other variables were nonsignificant. Furthermore, patients with BLBC appear to have better response to chemotherapy than nonbasal tumors. Another study has similarly recorded that low-grade histological subtypes of BLBC were consistently associated with improved prognosis [142].
Other studies show that the prognostic significance of the basal subtype disappears when evaluated alongside other risk variables such as ER negativity, HER2 positivity and grade III status [61,143]. Kim et al., in contrast to previous findings from western countries [61], reported that the HER2/neu status is the most important prognostic factor of breast cancers. Moreover, Jumppanen et al., analyzing 375 tumor specimens, found that even if basal CK expression predicts early recurrence among nonselected tumors, the clinical outcome of basal tumors is similar to nonbasal ER− cancers [143].
Finally, it should be noted that there are some basal-like subgroups with specific features and molecular phenotypes that show a favorable prognosis, such as those with myoepithelial differentiation including myoepithelial carcinoma [93,97,98], adenoid cystic carcinoma, low-grade adenosquamous carcinoma and metaplastic carcinomas. It is apparent that BLBC do comprise either a single entity or a biologically uniform group of breast cancers. Significant variables in immunoprofiles, molecular phenotypes, grades and clinical behavior may determine the potential schedule for the appropriate treatment. Some studies gave contrary results because different criteria used for BLBC definition, different treatment protocols in the same group or different patients grouped together undergoing different treatment protocols may lead to an incorrect interpretation of the studies. Well-designed studies and large prospective clinical trials are necessary to provide conclusive evidence for the prognostic impact of BLBCs in association with the traditional clinicopathological grouping.
Current therapy & ongoing trials
Chemotherapy
Basal-like breast cancer as a subgroup of triple-negative cancer lacks the expression of all three receptors (ER, PR and HER2) and for this reason cannot benefit from hormone therapy or anti-HER2-targeted agents. At present no specific tailored therapy is available and chemotherapy with anthracyclines and/or taxanes remains the only systemic treatment option [20].
Rouzier et al. first described the high response rate of basal-like and ERB2+ to chemotherapy after distinguishing breast cancer subtypes through gene expression profiling or IHC. It has been shown that different molecular subtypes of breast cancer respond differently to preoperative chemotherapy.
An important observation was that the basal-like and cerbB2+ subgroups were associated with the highest rates of pathological CR, 45 and 45% (95% CI: 23–68), respectively, whereas the luminal tumors had a pathologic CR rate of 6% (95% CI: 1–21). Molecular class was not independent of conventional clinicopathological predictors of response such as ER status and nuclear grade. Moreover, none of the 61 genes associated with pathologic CR in the basal-like group were associated with pathologic CR in the cerbB2+ group, indicating that the molecular mechanisms of sensitivity to chemotherapy differ between these two ER− subtypes.
Basal-like and cerbB2+ subtypes of breast cancer proved more sensitive to paclitaxel (taxane) followed by doxorubicin-containing preoperative chemotherapy than the luminal and normal-like cancers [144]. p53 mutation, a characteristic marker in TNBC/BLBC patients, is implicated in inverse results concerning chemosensitivity. For this type of tumor there are reports of resistance to anthracyclines [145] but response to taxanes [146].
In a recent study of anthracycline-based neoadjuvant chemotherapy, involving 107 patients treated with doxorubicin/cyclophosphamide, basal-like tumors demonstrated the highest pathologic CR (85%) compared with HER2+ (70%) and luminal (7%) tumors [27]. However, disease-free survival and OS were significantly worse in basal-like and HER2+ subtypes. Liedtke et al. compared response to neoadjuvant anthracycline/taxane-based chemotherapy and survival between patients with TNBC and non-TNBC [147]. Patients with TNBC had significantly higher pathologic CR rates (22 vs 11%; p = 0.034), but decreased 3-year progression-free survival (PFS; p < 0.0001) and 3-year OS (p < 0.0001). It is important to note that prognosis for TNBC patients who experience a pathologic CR after neoadjuvant chemotherapy was similar to the other subtypes [144]. By contrast, the TNBCs that failed to achieve pathologic CR had a poor prognosis [27,147].
Currently, in patients who have undergone neoadjuvant chemotherapy with docetaxel, adriamycin and cyclophosphamide within the GEPARTRIO trial, a trend to more tumor relapses has been reported in basal-like (30.0%) compared with the cerbB2+ (0%), normal-like (28.6%) and luminal (13.6%) tumors [64].
The biological similarities between BRCA1 mutation carriers and basal-like cancers point to therapeutic agents targeting BRCA1 pathway dysfunction. However, BRCA1-mutated tumors appear resistant to common chemotherapy drugs and less sensitive to taxanes [148]. Taking into account the results of randomized clinical trials on the role of taxanes in breast cancer, further studies concerning the use of taxanes in BLBC and TNBC are required. Platinum salts are under investigation in patients with advanced TNBC and a Phase II neoadjuvant trial has reported that the regimen is well tolerated and can be as effective as other multiagent therapies [127].
The genetic instability of basal-like cancers may potentially result in resistance to chemotherapy. Epothilones, a novel group of cytotoxic agents, have demonstrated that they are able to overcome the mechanism of drug resistance and are suggested in metastatic disease treatment of TNBC [149]. Of these, ixabepilone is at the most advanced stage of clinical development and is indicated for the treatment of metastatic and locally advanced breast cancer after nonresponse to anthracycline agents and taxanes. It has been found that ixabepilone also demonstrates a significant antitumor activity in patients with TNBC. In the neoadjuvant setting, in particular, this agent achieves a comparable pathologic CR and overall response rate among TNBC and non-TNBC patients [150]. A Phase II study (NCT00633464) is now underway to evaluate the overall response rate, PFS, time to respond and duration of response of therapy with ixabepilone alone or in combination with cetuximab in TNBC patients with metastatic disease [201].
Patients with triple-negative and basal-like tumors have a poor prognosis, especially those with local recurrence or metastatic disease. Development of novel molecular targets and advanced chemotherapeutic regimens is urgently needed to improve disease management of this vulnerable group of patients.
New molecular agents
Based on the information provided above it is apparent that no single systemic therapy is suitable for all TNBCs. Although BLBCs show significant response to anthracycline, taxane and platinum salts in the neoadjuvant/adjuvant setting, many patients do not respond to this therapy and they still have poor prognosis in comparison to the other molecular subgroups [27]. Several promising molecular agents for targeted therapy are under investigation
New molecular treatments.
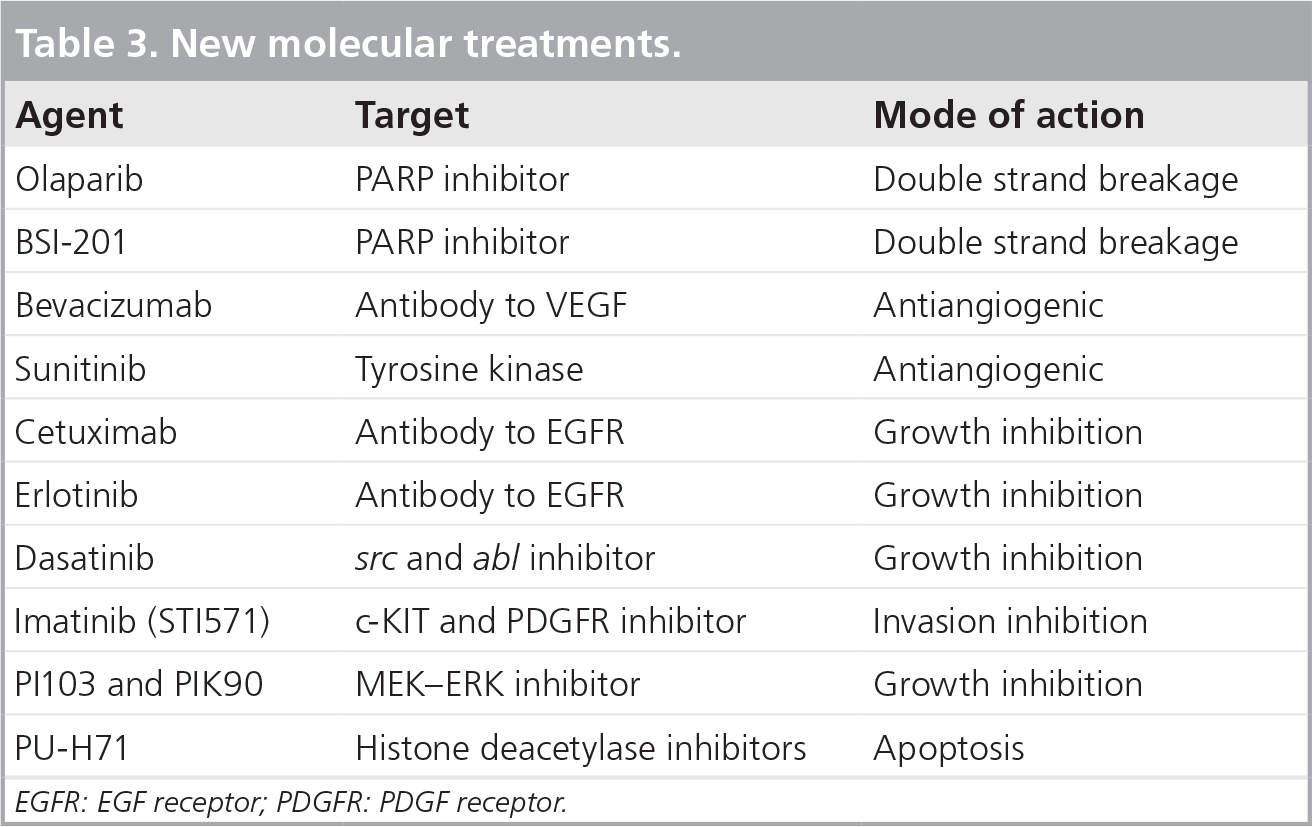
EGFR: EGF receptor; PDGFR: PDGF receptor.
Similarities between BLBC and BRCA1 mutation carriers direct interest to BRCA1 genotype-specific sensitivity and resistance to currently used chemotherapy drugs. It is known that BRCA1 mutation carriers lack the ability of homologous recombination DNA repair that leads to genetic instability in mitotic breakdown and finally cell death. This approach led to the higher evaluation of poly(ADP-ribose) polymerase (PARP) inhibitors. These are a group of pharmacological inhibitors of the enzyme PARP. In tumors with BRCA1 or BRCA2 mutations, drugs that inhibit PARP1 result in multiple double strand breaks, and because of the BRCA1 or BRCA2 deficiency these cannot be efficiently repaired, leading to cell death. Normal cells do not replicate their DNA as frequently as cancer cells, and still have homologous repair ability, allowing them to survive the inhibition of PARP [151].
A Phase I clinical trial used olaparib, an oral PARP inhibitor, and indicated an effective response in BRCA1 and BRCA2 in advanced and pretreated breast, ovarian and prostate cancer [152]. Two recently reported Phase II studies investigated the effect of PARP inhibitors in breast cancers and showed a notable toxicity but confirmed the high response. In a multicenter study Tutt et al. investigated the use of olaparib in chemotherapy-pretreated BRCA1/2 mutation carriers, showing promising results with an increase of the PFS [153]. Another Phase II study compared carboplatin and gemcitabine with or without the PARP inhibitor BSI-201 in TNBC, resulting in a significant increase in OS. The Phase III study is currently open, but the pharmacological effects of BSI-201 on PARP are yet to be explored [154].
Increased understanding of the important role of angiogenesis in breast cancer biology has led to the development of different antiangiogenic therapies. Application of antiangiogenic agents such as monoclonal antibodies against the VEGF receptor (bevacizumab) and multikinase VEGF receptor inhibitors is another option for targeted therapy [155]. Bevacizumab, a humanized monoclonal antibody against VEGF receptor is the most developed agent, being the first approved angiogenesis inhibitor for use in metastatic breast cancer. A recent study showed that TNBCs have higher intratumoral VEGF levels compared with non-TNBCs [156]. Three randomized Phase III studies showed that the combination of bevacizumab with chemotherapy increased overall response rate and prolonged PFS compared with chemotherapy alone in a first-line setting. In an open-label, randomized, Phase III trial (E2100), Miller et al. compared the efficacy and safety of paclitaxel alone with that of paclitaxel plus bevacizumab, as initial treatment for metastatic breast cancer including the TNBC subgroup [157]. The combination of paclitaxel plus bevacizumab prolonged PFS, but not OS, as compared with paclitaxel alone.
Another Phase III study showed that 15 mg/kg of bevacizumab every 3 weeks significantly increased PFS when combined with docetaxel compared with docetaxel plus placebo for the first-line treatment of human EGFR2-negative metastatic breast cancer [158]. In both trials patients with TNBC experienced similar PFS benefit with the addition of bevacizumab compared with ER+/PR+ patients. The RiBBON I trial confirmed the above findings and RiBBON II evaluated the combination of bevacizumab with chemotherapy in metastatic breast cancer in the second-line setting with similar findings [159]. In addition, bevacizumab in combination with cisplatin showed encouraging results in a reported Phase II study [160]. The role of bevacizumab is currently being studied in the adjuvant setting. A two-arm Phase III study, Bevacizumab Adjuvant Therapy in Triple Negative Breast Cancer (BEATRICE), randomizes patients to receive either standard chemotherapy (anthracycline ± taxane or taxane alone) or standard chemotherapy in combination with 1 year of bevacizumab [153].
The multikinase inhibitors sorafenib, sunitinib and pazopanib have also shown promising early results and are being further evaluated [155]. Sunitinib is an oral, multitargeted tyrosine kinase inhibitor that blocks VEGF receptor, PDGF receptor (PDGFR), stem cell factor receptor, and colony-stimulating factor-1 receptor. A Phase II, open-label, multicenter study evaluated sunitinib monotherapy in patients with metastatic breast cancer previously treated with an anthracycline and a taxane and showed significant response in a subset analysis of triple-negative tumors [161].
Triple-negative breast cancer and BLBC overexpress EGFR in up to 60% of cases. In particular, EGFR appears to play an important role in growth and proliferation for BLBC cell lines [162]. EGFR amplification is present in up to 25% of metaplastic cancers, a subgroup that is associated with the triple-negative/basal-like phenotype [163]. EGFR signaling responds to anti-EGFR-targeted therapies in other types of cancer, either by using EGFR-directed antibodies or inhibitors of receptor phosphorylation [164]. Cetuximab, a chimeric monoclonal antibody, is currently under evaluation in metastatic triple-negative disease either alone, with carboplatin added in progression, or as cetuximab and carboplatin throughout [165]. Initial results of a Phase II randomized trial (TBCRC 001) demonstrated a possible benefit in the cetuximab arm [165] compared with the cetuximab plus carboplatin arm. Another anti-EGFR agent, erlotinib, is currently being evaluated in the neoadjuvant setting in TNBC in a Phase II clinical trial in combination with chemotherapy [154].
Dasatinib is an orally active small molecule (tyrosine kinase inhibitor) of both the src and abl proteins. Huang et al., investigating the potential role of dasatinib in human breast cancer cell lines, observed a highly significant relationship between breast cancer subtype and sensitivity to dasatinib [166,167]. In particular, basal-type and postepithelial-to-mesenchymal-transition breast cancer cell lines demonstrated greater growth inhibition by dasatinib.
Using baseline gene expression profiling of a panel of 23 breast cancer cell lines, Huang et al. identified genomic signatures highly correlated with in vitro sensitivity to dasatinib [166]. The dasatinib-sensitive signature subgroup of breast tumors included a specifically ‘basal’ breast cancer phenotype. These results led to the concept that dasatinib may represent a valuable treatment option in this difficult-to-treat population and a Phase II clinical trial is currently underway to determine the activity of dasatinib in these patients with advanced disease. However, a Phase I trial of dasatinib and capecitabine in an unselected cohort of breast cancer patients yielded discouraging results [168].
KIT (CD117) is a transmembrane tyrosine kinase receptor that is expressed in different levels in breast cancer. STI571 (imatinib mesylate) inhibits c-KIT and PDGFRa tyrosine kinase [169]. Some KIT-overexpressing solid tumors have responded favorably to STI571. In a study by Roussidis et al. the activity of STI571 on the growth and invasiveness of three human epithelial breast cancer cell lines of low (MCF-7) and high (ZR-75-1 and MDA-MB-231) invasive potential was examined. A significant inhibitory effect on the invasion of the highly invasive breast cancer cell lines was found that was cytostatic rather than cytotoxic [170].
A Phase II trial evaluating the safety and efficacy of imatinib in patients with metastatic breast cancer showed that there is no evidence of clinical benefit. Imatinib therapy at doses of 800 mg/day is associated with significant toxicity in patients with heavily pretreated metastatic breast cancer and is not indicated as monotherapy in these unselected patients [171]. Further studies on the efficacy of KIT/PDGFRa inhibition are necessary.
Recently, specific inhibitors of MEK–ERK that sufficiently inhibit the oncogenic RAF–MEK–ERK pathway have been developed. Basal-type breast cancer cells are particularly susceptible to growth inhibition by small-molecule MEK inhibitors and consequent activation of the PI3K pathway in response to MEK inhibition leads to limited efficacy. Targeting inhibition of MEK and PI3K pathways produces synergistic effects, which has implications for the design of future clinical trials of MEK inhibitors in BLBC [172].
The oncogenic PI3K pathway has been studied in 13 in vitro BLBCs, and was compared with a control series of 11 hormonal receptor-negative- and grade III-matched HER2+ carcinomas defined by immunohistochemistry and gene expression arrays. The PI3K pathway is activated and upregulated in BLBC as shown by a significantly increased activation of the downstream targets AKT and mTOR. Furthermore, BLBCs expressed significantly lower levels of the tumor suppressor ‘phosphatase and tensin homolog’ (PTEN) and PTEN levels were significantly negatively correlated with AKT activity in that cancer subtype. Reduction in PTEN DNA copy numbers were observed specifically in BLBCs. Both PI3K and mTOR inhibitors led to basal-like cell growth arrest with subsequent apoptosis after PI3K inhibition. While these findings point to the PTEN-dependent activated AKT signaling pathway as a potential therapeutic target for the management of patients with poor-prognosis BLBCs [173], other studies have shown that PIK3CA and PTEN mutations are uncommon in BLBC [174]. Since PI3K may promote cancer through both AKT-dependent and AKT-independent mechanisms and differential PI3K/PDK1 signaling, this could lead to new approaches in cancers harboring PIK3CA mutations [175].
Histone deacetylase inhibitors may play a role in breast cancer therapy, in combination with a DNA repair targeted agent [176]. PU-H71, a hsp90 inhibitor, has shown efficacy in a TNBC xenograft [177]. Interestingly, TNF-related apoptosis [178] demonstrated efficient inhibition of triple-negative cell lines with mesenchymal features in vitro [179].
Recently, it has been reported that FGF receptor (FGFR)2 is present in 5% of TNBCs and silencing this by RNA interference methods or by chemical inhibition could result in death of TNBC cells with FGFR2 amplification [180].
From this and other studies, FGFR2 amplification is suggested as a new target for therapeutic intervention in TNBC [154].
Claudin-1 is an integral protein component of tight junctions with the gene CLDN1 expressed in mammary epithelial cells but with low or undetectable levels of expression in breast cancers, suggesting that it may be a possible tumor-suppressor gene. The epithelial-to-mesenchymal transition produces cancer cells that are invasive, migratory and associate closely with the claudin-low and metaplastic malignancy [181]. Most claudin-low tumors are triple negative and resemble mammary epithelial stem cells [182]. This may represent yet another potential therapeutic target.
Executive summary

Most breast diseases originate from terminal ductolobular units lined by inner luminal and outer myoepithelial layers.
Gene expression arrays identify breast cancer subtypes of with different behavior.
There are four main subtypes: luminal A, luminal B, HER2-overexpressing and basal-like breast cancer (BLBC).
Cancers may arise from genetic/epigenetic disruption of stem/progenitor cells.
BLBC originates from the myoepithelial cell layer, which has both epithelial and smooth muscle cell features, usually expressing basal cytokeratins.
Triple-negative breast cancer (TNBC) is defined by estrogen receptor, progesterone receptor and HER2 negativity.
BLBC may not express triple-negative phenotype and conversely TNBC has no basal-like profile.
TNBC is heterogenous; BLBC is only a subset. RNA profiling indicate most TNBCs fall in the BLBC subtype.
In array-CGH studies TNBC and BLBC show a high frequency of DNA losses, low-level gains/deletions and/or p53 protein overexpression.
Basal cytokeratins are the most reliable markers to classify BLBCs but using the combination of CK5/6, CK14 and CK17 results in contradictory results.
The most reliable definition for BLBC in routine clinical practice is the ER−, HER2-/low, CK5/6 and EGFR+ phenotype.
These include large tumour size, high proliferation, central acellular zones/necrosis, squamous metaplasia, pushing noninfiltrative borders, lymphoplasmatic infiltration and adenoid cystic changes.
Most BLBCs are invasive ductal carcinoma not otherwise specified. A subset of ductal carcinoma in situ is BLBC.
BLBC correlates with medullary carcinoma.
70–80% of BRCA1 mutation breast cancers are TNBCs and BLBCs.
BLBC is associated with African–Caribbean race, young age and abdominal adiposity.
Mammographic features are ill-defined mass and comedo calcifications.
In routine practice ER or HER2 status of the two types differs from 15 to 54% of basal-like cancers.
In gene expression arrays, 85% of TN tumours are BLBC according to ER−/HER2− profile and by IHC analysis 46–90% of TNBC express basal cytokeratins and HER1/EGFR markers.
BLBC/TNBC is associated with aggressive clinical behavior, visceral metastasis, decreased survival and high mortality, and these are worse for BLBC.
Both have higher recurrence and death rates in the first 3 years compared with other breast cancer subtypes. Patients with residual disease after neoadjuvant chemotherapy have significantly worse overall survival. However, if pathologic complete response is achieved, survival is comparable with non-TNBC.
Locoregional relapse and distance metastases are more frequent in TNBC/BLBC tumors, indicating a need for more radical surgery.
Patients with TNBC have shorter time to develop brain metastasis from the initial diagnosis and shorter survival after this event.
Recurrence risk in TNBC is highest the first 5 years after diagnosis, whereas for non-TNBC risk remains constant over time.
Patients with TNBC and BLBC phenotype have a significantly shorter disease-free survival than those without.
Some basal-like subgroups with myoepithelial differentiation have a better prognosis.
BLBC cannot benefit from hormone therapy or anti-HER2-targeted agents. At present anthracyclines/taxane chemotherapy is the only option.
Basal-like and cerbB2+ subgroups have the highest rates of pathologic complete response after neoadjuvant chemotherapy and are more sensitive to paclitaxel followed by doxorubicin-containing therapy.
P53 mutation, a characteristic marker in TNBC/BLBC patients, indicates anthracycline resistance but taxane sensitivity.
Platinum salts are under investigation in patients with advanced TNBC.
Ixabepilone is indicated for the treatment of metastatic and locally advanced TNBC after nonresponse to anthracycline/taxanes.
Poly(ADP) ribose polymerase inhibitors show promising results in tumours with BRCA1 or BRCA2 mutations increasing progression-free and overall survival.
Several randomized Phase III studies showed that the combination of bevacizumab with chemotherapy increased overall response rate and prolonged PFS.
BEATRICE, a two-arm Phase III study randomizes patients to receive either standard chemotherapy (anthracycline ± taxane or taxane alone) or standard chemotherapy in combination with 1-year bevacizumab.
Cetuximab, a chimeric monoclonal antibody against EGFR is currently under evaluation in metastatic TNBC and similarly erlotinib in the neoadjuvant setting in triple-negative disease.
FGFR2 inhibitors and CLDN1 tumor-suppressor gene, may represent additional potential therapeutic targets.
An agreed definition of BLBC is needed for better clinical management.
Carefully designed prospective trials are required to confirm the activity of targeted therapies against BLBC/TNBC.
Better understanding of patterns of metastatic spread and relapse between different subtypes of breast cancer may enable targeted adjuvant therapy and surveillance decisions.
The similarities observed between TNBC/BLBC phenotypes and BRCA1 tumours suggest the use of triple negativity in combination with cytokeratin staining to select patients for genetic testing.
Most clinical trials use the triple-negative definition overlapping the basal-like cases, a fact that leads to inconsistent and uncertain findings. More carefully designed prospective studies are required to assess the biological behavior of basal-like tumors and the consequent response to specific therapy.
Conclusion & future perspective
Basal-like breast cancer is a distinct entity but also has heterogeneous morphologic, genetic and immunophenotypic features, resulting in variable behavior in terms of response to neoadjuvant/adjuvant therapy. An agreed realistic definition is needed for better precision regarding prognosis and clinical management. The activity of targeted therapies need confirmation through appropriately designed clinical trials. This effort should not only increase the opportunity to intervene against TNBC and BLBC but also enable a personalized approach to all patients with breast cancer.
It is reported that 5000 basal-like tumors may be missed every year in the UK because of discrepancies in diagnosis. Many subgroups of BLBCs may be unrecognized, leading to incorrect management and therapeutic failure [74]. Simpler methods to identify BLBCs and specific subgroups of the same category are required for tailoring management appropriately.
A better understanding of patterns of metastatic spread and relapse between different subtypes of breast cancer may influence adjuvant therapy and surveillance decisions and determine which investigations and therapies are appropriate when distant disease or relapse has been diagnosed. Conventional imaging is considered to be ineffective in detecting all metastatic disease and subclinical metastasis is frequently missed because of inappropriate imaging [183] or false-negative findings. Given that follow-up recommendations for patients with TNBCs and BLBCs do not differ from the other subtypes [184] and metastatic disease diagnosis is still based on history and physical examination, a new approach should be developed in these patients with increased risk or recurrence and distal metastasis.
Since DCIS constitutes approximately 20% of mammographic breast malignancies [185], the identification of the basal-like subtype may change the therapeutic approach in these patients and lead to a better prognosis.
The similarities observed between triple-negative and basal-like phenotypes and BRCA1 tumors suggest the use of triple negativity in combination with CK staining as a predictor for the selection of patients for BRCA1 mutation testing. The correlation in the function of BRCA1 breast stem cells between normal breast development and breast cancer deserve further consideration.
Recognition of differences in mammographic appearance between basal and nonbasal tumors is of great importance as BLBCs tend to appear as interval tumors. Knowledge of the specific features of the tumors can help not only in the identification of a malignancy but also in the potential management with more effective treatment. It is of particular interest that TNBCs comprise a wide family that includes not only the basal-like but also other subtypes such as claudin-low cancers, molecular apocrine tumors, interferon-rich and normal-like tumors. All these subtypes have distinct clinicopathological features and respond differently to systemic therapy giving rise to the necessity of the recognition of these subtypes that may require more specific therapeutic approaches.
Footnotes
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
