Abstract
Contrary to the exaggerated risks associated with HRT that developed after the initial press reports held by the Women's Health Initiative (WHI) writing group, the recent approach to hormone therapy is more balanced and evidence based. A review of over 40 years of scientific studies demonstrates that estrogen is a medication that can decrease mortality, cardiovascular disease, osteoporosis fracture, urogenital atrophy and dementia. When timing of administration, dose of therapy and route of administration are considered, estrogen is associated with low risks and substantial benefits. The decision of whether or not to take HRT for either short symptom relief or for long-term therapy, should be based on an accurate risk–benefit analysis. Adjusting the dose of therapy and considering a transdermal approach, particularly in high-risk patients, are important considerations.
Keywords
After the release of the Women's Health Initiative (WHI) press reports in the USA in 2002, the use of HRT among women around the world declined abruptly. Since then, a better understanding of risks and benefits of hormone therapy has clarified the WHI findings and led to a more balanced approach to counseling. One of the most important findings of the WHI is that the age of initiation of therapy has profound effects on the risk and benefit profile. Currently there are very important ongoing studies designed to further characterize the timing hypothesis that simply says that timing of initiation is critical to understanding the therapeutic actions of estrogen [1,101]. These data are not expected to be known for several years. However, even without these results, careful review of over 40 years of studies demonstrates several important points. Estrogen is a medication that can decrease mortality, decrease cardiovascular disease, decrease osteoporosis fracture, urogenital atrophy and dementia. Strategies to minimize risk include initiation of therapy within 10 years of menopause or under 60 years of age, using low-dose estrogen, adding a low-dose progestin in women with a uterus, and use of transdermal therapy in women with risk factors for cardiovascular disease.
Strategies to minimize risk
Many women may want the benefits associated with hormone therapy including protection from cardiovascular disease, osteoporosis and fracture, urogenital atrophy, skin atrophy and dementia. Minimizing the risks of taking hormone therapy for these women is an important consideration.
Timing hormone therapy: when started in women under 60 years or within 10 years of their menopause, is not associated with an increased risk of heart disease and many studies show that estrogen therapy actually protects the coronary vessels. Continuing therapy, at least through 65 years of age, appears to continue this protection [2].
Minimizing the dose: using the lowest effective dose for menopausal symptoms lowers the risk of side effects and bleeding problems. Low-dose therapy is also associated with beneficial effects on bone metabolism and vaginal tissue.
Delivery method may be important: estrogen can be administered by patch, gel, mist, vaginal cream, vaginal suppository or vaginal rings. Use of these non-oral delivery methods minimizes the effect of estrogen on hepatic proteins. The non-oral delivery methods are recommended for women with suspected or known cardiovascular disease, clotting abnormalities, thromboembolic history, pronounced obesity, prolonged hypertension or diabetes, advanced age or prolonged immobilization. Minor differences among these transdermal estrogen preparations or among the many oral estrogen preparations is not the focus of this article.
Adding a progestin: for women who have not had a hysterectomy, addition of a progestin to estrogen therapy is necessary to protect the endometrium from overstimulation. One of the advantages of using a low-dose estrogen product is that it allows for minimizing the dose of the progestin. The combination products on the market contain balanced levels of estrogen and progestin and generally minimize uterine bleeding. Some clinicians and patients prefer to select separate estrogen and progestin products. There are a large variety of progestins that can be used in this manner and differences in the progestins may offer clinical advantages. In general, these differences are minimal when a low-dose option is used and are not the focus of this article.
Consider high risk factors: women with a personal history of breast cancer or active liver disease are generally advised to avoid hormone therapy. Those with heart disease, a history of blood clots, advanced age, longstanding diabetes, longstanding hypertension or those with prolonged immobilization should fully evaluate a risk–benefit profile before initiating hormone therapy and consider low-dose transdermal therapy.
Accurate evaluation of the WHI: risks & benefits
Clinical practice was abruptly changed after data from the WHI were released to the media in June 2002. Rather than a first presentation to the medical profession to allow wide peer review, the results were first presented to a media conference in a manner that allowed a media-driven frenzy and an exaggerated public view of the risks associated with hormone therapy.
The WHI results were published in July 2002 showing small increases (often borderline or not statistically significant) in the risk of cardiovascular disease, breast cancer and stroke in postmenopausal women taking combination estrogen and progestin therapy compared with untreated controls [3]. These results were in stark contrast to the vast majority of earlier studies that reported large decreases in overall death rate, cardiovascular disease, osteoporosis, bone fractures, dementia, colon cancer and a host of other diseases in postmenopausal women taking hormone therapy compared with nontreated age-matched women [4–14]. The public has yet to understand the limitations of the study or to appreciate the study reported benefits associated with hormone therapy including a decreased hip fracture rate, decreased overall fracture rate and decreased colorectal cancer risk associated with combination estrogen plus progestin therapy
Women's Health Initiative reported risks and benefits of estrogen, estrogen plus progestin and combined studies.
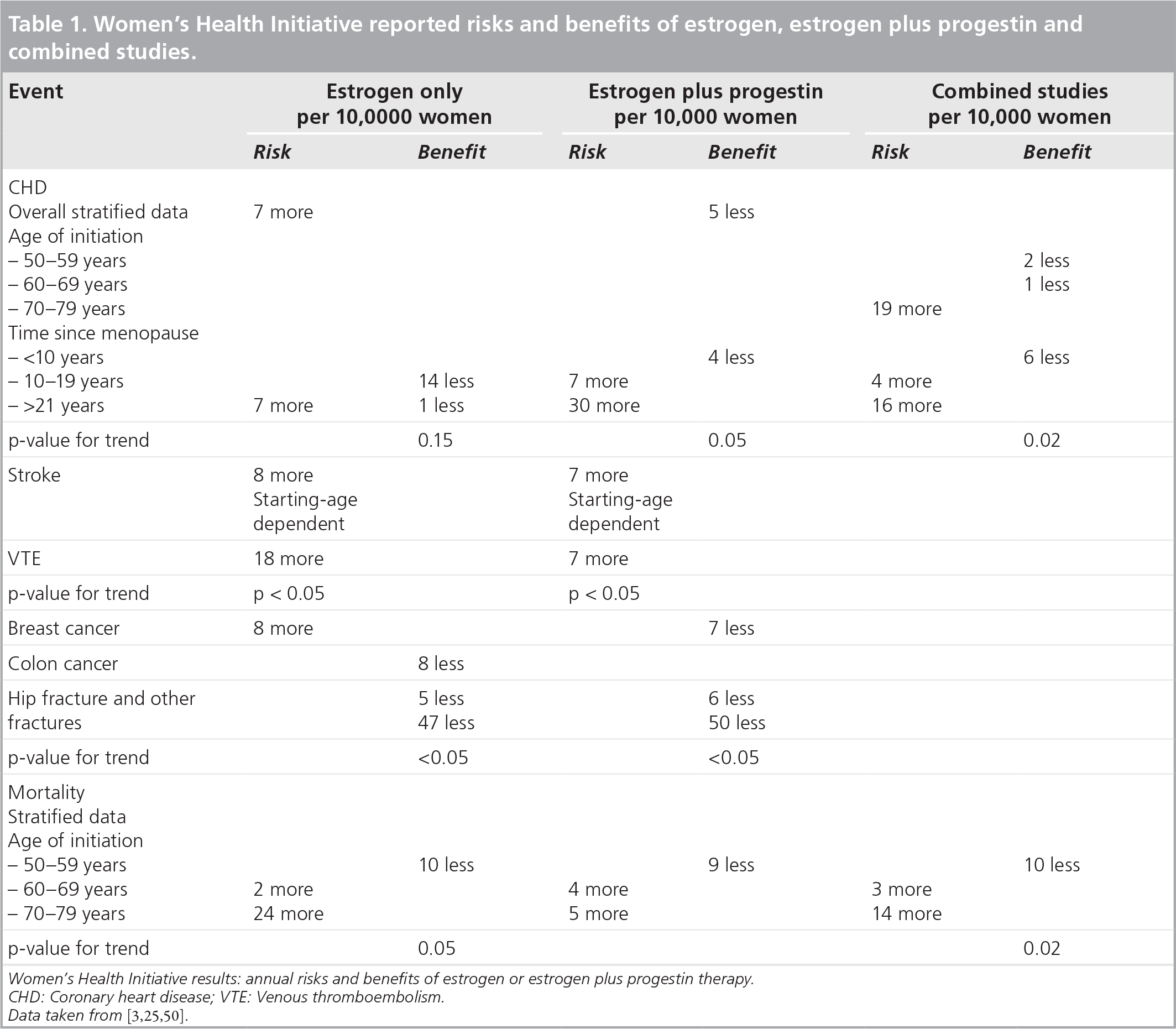
Despite the wealth of evidence contrary to this single study and the fact that the paper was criticized heavily for a number of important reasons as shown in
Criticisms of the Women's Health Initiative.
The study was conducted mostly in elderly, largely asymptomatic, obese women, many with cardiovascular disease, and not an appropriate population to extrapolate its results to normal users of HRT who initiate therapy around menopause
The preparation/dose of HRT used was not appropriate for at least 70% of the population included in the trial
The study had an unacceptably high dropout in rates, high unblinding rate, less than adequate compliance (only approximately half or study subjects were on therapy at the end of trial)
Risks of treatment exaggerated by a media frenzy
The dismissal of estrogen as a long-term beneficial treatment for postmenopausal women was not universally accepted. The beneficial effects of estrogen on bone and urogenital health remained important reasons to continue estrogen therapy beyond the early years of menopause. And even more importantly, it was not easy to dismiss the numerous studies demonstrating significant protection from cardiovascular disease and dementia and lowered risks of mortality associated with long-term estrogen therapy in women. Detailed analysis of older studies and the WHI data revealed important findings that helped to explain why the studies had such disparate results [15–17]. A major factor affecting the risk–benefit profile, that was appreciated before is the age of initiation of estrogen therapy. Estrogen therapy if started within 10 years of the menopause or generally between 50 and 60 years minimizes the risk of therapy
Understanding menopause & the consequences of hypoestrogenism
The transition from reproductive status to menopausal status is a result of a substantial reduction in hormone production by the ovaries. Generally this is not a sudden or abrupt transition but occurs over a period of years. The word menopause literally means cessation of monthly menses and it is a natural consequence of aging. There is a wide age range in which menopause can occur but for most it occurs in the late 40s or early 50s. For many women, the symptoms accompanying the menopausal transition and early menopausal years can significantly disrupt their daily activities and their sense of well-being. The loss of estrogen has profound effects on a variety of tissues including urogenital, skeletal, vascular, skin, soft tissue, cardiovascular and CNS
Overall risks of estrogen therapy: looking at the WHI & other studies
The original studies published by the WHI study group identified increased risk for a number of diseases that have, in later publications, been substantially changed or clarified. Presentation of the risks as defined by the original WHI data are included in this section. A summary of the risks and benefits of hormone therapy from both the early and later WHI publications is shown in
Reported effects of short- and long-term estrogen deficiency.
Hot flushes
Night sweats
Headaches
Rapid heart beat/palpitations
Itching
Dryness
Bleeding
Infection/discharge
Loss of elasticity
Shrinking
Urinary frequency Incontinence Urgency Increased UTIs
Bone loss Osteoporosis Hip fracture Vertebral fracture Back pain Height loss Joint pain Immobility
Decreased collagen content and elasticity
Thinning
Wrinkling
Redistribution of fat (from hips and thighs to abdomen)
Mood disturbance
Depression
Fatigue
Irritability
Insomnia
Dyspareunia Decreased libido
Accelerated atherosclerosis Cardiovascular disease
Dementia Memory loss Parkinson's Macular degeneration
Breast pain Atrophy
In the original (estrogen plus progestin) WHI study, in the overall study population, women treated with estrogen and progestin had a significant increased risk of blood clots (≥18 per 10,000 women/year) and an increased risk of stroke (≥8 per 10,000/year). Treatment with estrogen plus progestin also resulted in a higher risk of heart disease (≥7 per 10,000/year) and breast cancer (≥8 per 10,000/year)
In the original (estrogen only) WHI study, there was an increased risks of blood clots (+7 per 10,000/year) and stroke (+12 per 10,000/year), no increased risk cardiovascular disease, or ovarian cancer. The use of estrogen only resulted in a lower breast cancer risk (-6 per 10,000/year) that continued to be lower (statistically significant) in a 10-year follow-up study
WHI substudy
In a separate substudy of the WHI Memory Study (WHIMS) women aged 65 years and older (average age of initiation 71 years of age) reported the combination hormones had a modestly increased risk of developing dementia, compared with those taking placebo (45 per 10,000 vs 22 per 10,000) [21]. These findings again clarify the importance of early initiation of therapy and the increased risk (early harm) that can occur when starting hormone therapy in women over 65 years of age.
WHI: follow-up studies
In an 11-year follow-up study, the death rate in participants who received estrogen plus progestin for 5 years was 2.6 per 10,000 versus 1.3 per 10,000 per year in the group that took placebo (hazard risk [HR]: 1.96; 95% CI: 1.00–4.04; p = .049). A total of 24 % of the breast cancer patients who took HRT had tumors that spread to the lymph nodes, compared with just 16% of women taking placebos [22].
Million Women Study
The Million Women Study reported an increased risk of breast cancer in women currently using HRT. Women on current combination hormone therapy were at twofold increased risk of developing breast cancer, and current users of estrogen-only therapy had a 1.3-fold risk. The effects were similar for all doses, patterns of use, delivery methods and types of estrogen and progestogen [23].
Slovenia study
In a case–control study carried out in Slovenia, hormone therapy was associated with a reduced risk of breast cancer. A total of 784 cases and 709 controls aged 50–69 years were enrolled in the study. The reduced risk was highest in women on estrogen-only replacement therapy (odds ratio [OR]: 0.51; 95% CI: 0.30–0.87). Longer duration of HRT use did not result in a significant change in risk (1–5 years of HRT use: OR: 0.44; 95% CI: 0.26–0.73; >5 years of HRT use: OR: 0.51; 95% CI: 0.30–0.87). Risk factors for breast cancer identified included obesity, smoking and any first-degree relative with breast or ovarian cancer
WHI: reduced risks–benefits associated with estrogen or estrogen plus progestin therapy
The WHI reported a reduced risk of colon cancer in women on combination therapy; this equates to six women per 10,000 women [3]. In the WHI study group that received estrogen only, estrogen replacement therapy did not effect the risk of colorectal cancer
The WHI reported reductions of bone fractures (45 total fractures and five hip fractures per 10,000 women/year on combined HRT and six fewer hip and spine fractures and overall 56 fewer osteoporotic-related fractures in the estrogen alone arm), reduction in breast cancer (six less per 10,000/year) and reduced cardiovascular disease risk (five less per 10,000/year) [3,18,19]. The overall number of deaths was reduced by six (49 in the estrogen only arm and 54 on placebo)
The overall (reduced) risk for developing osteoporotic fracture in the combination therapy group, or (reduced) risk for osteoporotic fracture, breast cancer, and coronary heart disease (CHD) in women on estrogen alone in the WHI ranges between −0.45 and −0.67%.
Factors that affect the risk–benefit profile of hormone therapy
When evaluating the WHI or any study in regards to the risk–benefit profile for hormone therapy, there are important factors
The timing hypothesis simply says that timing of initiation is critical to understanding the therapeutic actions of estrogen. This theory is supported by large numbers and varieties of studies that consistently demonstrate an optimum therapeutic window for initiation of estrogen therapy [15–17]. The risks are increased if hormone therapy is started after 60 years or after more than 10 years since menopause
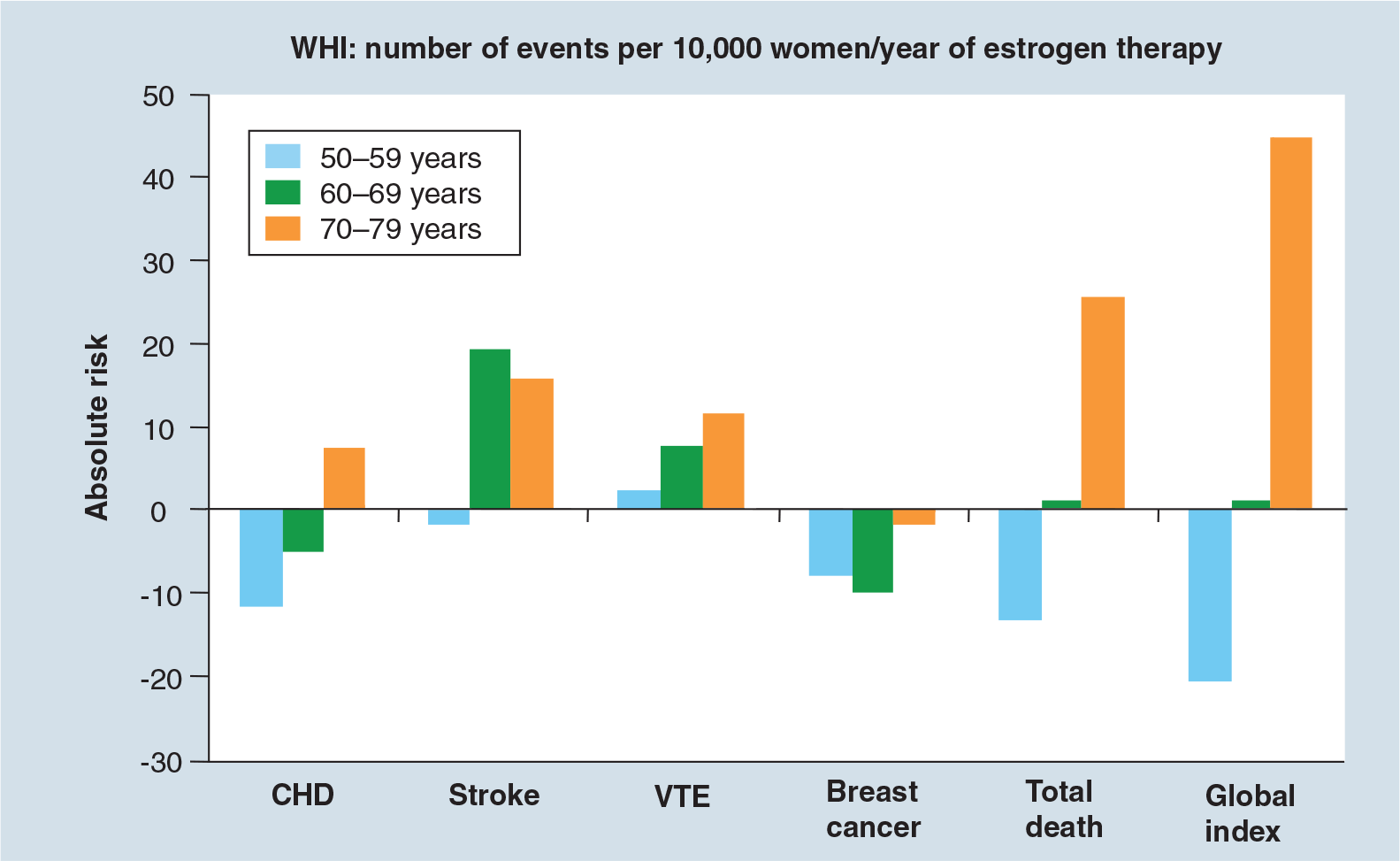
Overall risk and benefit are profoundly affected by age of initiation of therapy.
Another important factor to consider in an overall risk–benefit assessment is the dose of estrogen. There is substantial evidence that while low-dose estrogen is effective for menopausal symptoms [27] and for the prevention of bone loss [28] and other specific diseases associated with the low menopausal levels of estrogen, lower doses are associated with fewer side effects [27] and lowered risks.
Use of transdermal estrogen therapy adds a specific additional safety benefit as it avoids the hepatic ‘first pass’ effect thus avoiding changes in clotting factors and sex hormone-binding globulin. Multiple studies show no increased risk of blood clots and venous thromboembolism, even in high-risk postmenopausal women when transdermal estrogen is used [29,30].
Risks versus benefits of hormone therapy as defined by Women's Heath Initiative.
If the major risks of hormone therapy as reported in the Women's Health Initiative (WHI) are combined, the total number of affected women/year in the primary studies was 41/10,000 in the combination arm and 19/10,000 in the estrogen only arm. This is a very low number and confirms the low risk associated with hormone therapy. The overall increased risk of developing breast cancer, blood clots, stroke or coronary heart disease associated with estrogen or estrogen plus progestin therapy in the WHI is between 0.19 and 0.41%. These risks appear to be lower if hormone therapy is started in women within 10 years of their menopause or by 59 years of age, the risk of cardiovascular disease drops substantially. Use of low-dose therapy and transdermal therapy reduces the risks of venous thromboembolic problems
The benefits of hormone therapy are substantial when therapy is started within 10 years of menopause or by 59 years of age
Important factors to lower the risks and side effects of hormone therapy.
Age of initiation: ideally therapy begins within 10 years of menopause or by 60 years of age
Low dose: use of low-dose estrogen with low-dose progestin when appropriate
Route of administration: transdermal administration has reduced risk of blood clotting (venous thromboembolism risk) compared with oral administration
Consideration of progestin: side-effect profile of various progestins may play a clinical role in selecting the optimum treatment regimen. The newer progestin drosperinone (derivative of spironolactone) maybe a good consideration in hypertensive women
Benefits of estrogen treatment if started within a therapeutic window
As with the risk profile, the age of initiation plays a large part in maximizing the benefit profile. There is a therapeutic window where estrogen therapy is optimal. After many years of estrogen deficiency, the tissue changes may be advanced and the benefit of estrogen may be dramatically reduced. The primary beneficial action of estrogen is in prevention, that is protecting healthy tissue
Cardiovascular protection
A total of 40 years of trials support the cardiovascular protection of estrogen [2,4–13,31–37]. A few of the studies are highlighted below:
The Nurses Health Study followed over 70,000 women for over 30 years. They reported that the 0.3 mg dose of conjugated estrogen therapy was associated with a significant reduction in both major coronary disease 0.58 (95% CI: 0.37–0.92) and stroke 0.43 (95% CI: 0.22–0.83) [5,8];
Both arms of the WHI reported a major reduction in new cases of diabetes during the 7-year trial. This is an important morbidity that was inexplicably omitted from the arbitrary and invalidated ‘global index’ used by the WHI authors [3,18];
The WHI Coronary-Artery Calcification (CAC) studied 1064 women, randomized on the estrogen only arm of the WHI between age 50–59 years, with computed tomography of the heart. After 7.4 years on the study, the women who had taken estrogen had a significantly lower calcium score (30–40% reduction) compared with those taking placebo. (83.1 vs 123.1; p = 0.02). Women adherent to therapy or at least 5 years had a 64% reduction in score (p = 0.001) [2]. An accompanying editorial wrote “The results of WHI-CAC are clear and striking: women randomly assigned to receive estrogen had significantly less coronary-artery calcification than women randomly assigned to receive placebo” [38];
In a randomized study of 222 postmenopausal women 45 years of age or older without preexisting cardiovascular disease but with low-density lipoprotein cholesterol levels ≥130 mg/dl, the average rate of progression of subclinical atherosclerosis was lower in those taking estradiol compared with controls. (-0.0017 mm/year vs 0.0036 mm/year); the difference between average progression rates was 0.0053 mm/year (95% CI: 0.0001–0.0105 mm/year; p = 0.046). In women not on lipid-lowering medication (n = 77), the difference between average rates of progression between placebo versus estradiol was even greater at 0.0147 mm/year (95% CI: 0.0055–0.0240; p = 0.002) [9];
In a meta-analysis of 23 randomized controlled trials, women starting HRT <10 years after menopause or <60 years of age had significantly lowered risk of cardiovascular disease
Data from 23 trials, with 39,049 participants followed for 191,340 patient-years, concluded that hormone therapy reduced the risk of CHD events in younger postmenopausal women (OR: 0.68; 95% C I: 0.48–0.96). For older women, hormone therapy increased the risk the first year (OR: 1.47; 95% CI: 1.12–1.92), then reduced events after 2 years (OR: 0.79; 95% CI: 0.67–0.93) [37].
Cardiovascular heart disease events associated with HRT in older and younger women.

Meta-analysis of 34 randomized trials; 191,340 woman-years. OR: Odds ratio. Data taken from [33].
Prevention of dementia & Alzheimer's disease
A therapeutic window for protection from dementia and Alzheimer's disease is reported in a large number of studies [39–45]. As with cardiovascular disease, estrogen therapy initiated within 10 years of the menopause or in women <60 years of age has a higher benefit profile. Starting hormone therapy after 60–65 years needs to be done with caution. Use of a very low dose of hormone therapy and consideration of a transdermal administration is recommended.
In a prospective study in incident dementia among 1357 men and 1889 women (mean age around 73–74 years), the incidence among women increased after age 80 years and exceeded men of similar age. Women who used HRT had approximately a 50% reduction in risk (HR: 0.59; 95% CI: 0.36–0.96). Risk varied with years of use of hormones. Prior HRT use is associated with reduced risk of Alzheimer's disease, but there was no benefit with current HRT use unless such use had exceeded 10 years [39].
Benefits associated with hormone therapy.
Protection from bone loss and osteoporotic fracture
HRT is effective in fracture prevention [3,17,18] in a population unscreened for osteoporosis. This is in contrast to the other therapies used for osteoporosis that have been shown to be effective in populations that have been selected for high risk of fracture
Protection from urogenital atrophy, menopausal symptoms and hot flashes (estrogen is gold standard)
Cardiovascular disease prevention
Prevention of dementia and Alzheimer's disease
Decreased mortality
Decreased arthritis, colon cancer, memory loss, fat redistribution, tooth loss, Parkinson's disease and collagen loss in skin/wrinkling
Reduced mortality
Misconceptions still exist that cardiovascular disease is not a real problem for women, although it is estimated that one in two women will eventually die of heart disease or stroke, compared with one in 25 who will eventually die of breast cancer. Cardiovascular disease, particularly CHD and stroke, remains the leading killer of women in the USA and most developed countries [46,47]. Evidence suggests that postmenopausal women on hormone therapy, if started within the therapeutic window, have a lowered mortality. This is largely thought to be due to estrogen's cardiovascular protection.
A 10 year follow-up of the Nurses' Health Study including 48,470 postmenopausal women, reported that the age-adjusted relative risk of mortality form all causes in women who used estrogen at any time was 0.81. For cardiovascular mortality it was 0.68. After adjustment for other risk factors, the relative risks remained significant. The age-adjusted relative risk for cardiovascular mortality was 0.52 for current estrogen users and 0.77 for former users [102].
In a study utilizing data from 30 trials with 26,708 participants, hormone therapy significantly reduced mortality in the younger age group (OR: 0.61; 95% CI: 0.39–0.95)
Meta-analysis of total mortality associated with HRT in younger versus older postmenopausal women.

Meta-analysis of 30 randomized trials; 119,118 women-years. OR: Odds ratio. Data taken from [37].
Comparison of overall mortality rates in first decade menopausal women with HRT compared with lipid-lowering medication and aspirin.

Summary for individualizing hormone therapy: start with an accurate analysis of the risks & benefits
Contrary to the 2002 media-driven hype surrounding the initial presentation of the WHI, there are new, better considered and much more reassuring consensus statements on HRT. The new consensus statements on HRT are based both on reanalyses of published data from the WHI and on other extensive studies that must be included in an accurate risk–benefits analyses of HRT. The North American Menopause Society, The American Menopause Society and the International Menopause Society now concur that the risk–benefit ratio is in favor of HRT when initiated near menopause.
Estrogen is a medication that is reported to decrease overall mortality, cardiovascular disease, decrease osteoporosis fracture, urogenital atrophy and dementia. When timing of administration, dose of therapy and route of administration are considered, estrogen is associated with low risks. The decision to take or to recommend low-dose estrogen therapy for either short or long term, should include an accurate risk–benefit analysis:
Estrogen/hormone therapy is FDA-approved for the prevention of bone loss and urogenital atrophy and is approved as a treatment of menopausal symptoms. Multiple other studies support the protective effect of estrogen therapy on cardiovascular disease, dementia and overall mortality. Data suggests that hormone therapy is singularly effective in preventing cardiovascular disease for women when therapy is started within 10 years of their menopause;
The risks associated with low-dose estrogen/hormone therapy are very low if started within 10 years of menopause or under 60 years of age;
Women with cardiovascular or thrombotic risk factors, those initiating therapy after age 60 or >10 years since menopause, or those with significant obesity should consider low-dose transdermal estrogen/hormone therapy;
Initiation of hormone therapy for treatment of menopausal symptoms in early menopause or perimenopause is associated with low risk. Decisions regarding long-term use of hormone therapy should be based on an accurate risk–benefit analysis.
Future perspective
Within the next 5 years, the results of both the Early Versus Late Intervention Trial With Estradiol (ELITE) and Kronos Early Estrogen Prevention Study (KEEPS) trial will be available [1,101]. These trials are designed to further clarify the cardiovascular protective effects of estrogen replacement when given to women in their early menopause years compared with those that have 10 years or more of menopause. If these trials demonstrate a protective effect of estrogen against atherosclerotic plaque formation, a very compelling argument for initiating estrogen therapy in the early menopause will be difficult to counter. Estrogen is already an established medication for preventing bone loss and treating menopausal symptoms in the early menopause. It is also expected that new low-dose estrogen only and combination products with new progestins will be introduced that will increase the choices for the large numbers of women entering the menopause. It is anticipated that within the next decade, healthcare providers and the general public will have a better and a more accurate understanding of the low risks associated with hormone therapy and that the accepted benefits associated with the initiation of hormone therapy for post-menopausal women within 10 years of their menopause will include protection from cardiovascular disease, prevention of bone loss, and effective treatment of menopausal symptoms.
Executive summary
A review of over 40 years of scientific studies demonstrates that estrogen is a medication that can decrease mortality, cardiovascular disease, osteoporosis fracture, urogenital atrophy, colon cancer and dementia.
When timing of administration, dose of therapy and route of administration are considered, estrogen is associated with low risks and substantial benefits.
Contrary to the exaggerated risks associated with HRT that developed after the initial press reports held by the WHI writing group, the recent approach to hormone therapy is more balanced and evidence based.
WHI has been heavily criticized as the study was conducted in mostly elderly, largely asymptomatic, obese, diseased women and this is not an appropriate population to extrapolate results to normal users of HRT who initiate therapy around menopause.
The decision on whether or not to take HRT for either short symptom relief or for long-term therapy, should be based on an accurate risk–benefit analysis.
The loss of estrogen has profound effects on a variety of tissues including urogenital, skeletal, vascular, skin, soft tissue, cardiovascular and the CNS.
Using a low dose of estrogen, adding a low dose of progestin in women with a uterus, and considering a transdermal approach in high-risk patients, are important considerations.
Footnotes
The author has no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
