Abstract
Uterine papillary serous carcinoma (UPSC) and uterine clear cell carcinoma (UCCC) represent two rare subtypes that have an increased risk of recurrence and worse overall survival compared with the more common endometrioid endometrial cancers. Meaningful data in the form of prospective randomized trials is lacking for both advanced and early-stage UPSC and UCCC. Data extrapolated from prospective trials in advanced endometrioid endometrial cancer and retrospective trials on early-stage UPSC suggest that adjuvant platinum and taxane-based chemotherapy may provide a survival benefit for these patients. Future trials specifically examining UPSC and UCCC are needed to elucidate the optimal treatment regimen for these patients. Given the current data, the option of chemotherapy should be considered in treatment-planning discussions for all patients with UPSC and UCCC.
While the prognosis for most patients with endometrial cancer is favorable, two histologic subtypes exist for which prognosis is particularly poor. The most common histologic subtype is endometrioid endometrial cancer (EEC), which represents over 50% of all histologic types of endometrial cancer [1]. Uterine papillary serous carcinoma (UPSC), however, represents 10% of endometrial cancer cases and accounts for a disproportionate 35% of all endometrial cancer-related deaths [2–4]. The rarer histologic subtype, uterine clear cell carcinoma (UCCC), accounts for 1–6% of endometrial cancer diagnoses [5–6]. UPSC and UCCC are classified as type II endometrial cancers and lack the obesity and hormonal risk factors associated with the more common endometrioid subtype. Patients with UPSC and UCCC present at an older age and often with well-developed metastatic disease, which accounts, in part, for their worse prognosis [7–8]. However, even in stage I cancers, UPSC and UCCC have been shown to have a worse overall survival (OS) rate than EEC (50–80 vs 80–90%) [3,9–11]. Studies have confirmed that UPSC and UCCC are genetically distinct from each other and from type I EEC. Microarray gene-expression studies have established different gene expression profiles in UPSC, EEC and UCCC [12–13]. The recurrence patterns and incidence are different for UPSC and UCCC compared with EEC. Early-stage UPSC and UCCC tend to recur more frequently and at distant sites compared with EEC, which tends to recur locally, therefore limiting the curative intent of single modality radiotherapy in UPSC and UCCC.
The development of evidence-based management strategies for UPSC and UCCC has been hampered by the lack of prospective data due to the relative rarity of these tumors. This article will discuss the limited prospective data available, as well as the retrospective data examining the use of adjuvant chemotherapy in advanced- and early-stage UPSC and UCCC.
Adjuvant chemotherapy: advanced-stage disease
Historically, prospective chemotherapy trials in endometrial cancer have focused on the advanced-stage and recurrent disease. The Gynecologic Oncology Group (GOG) has reported the results of five Phase III randomized trials of chemotherapeutic agents in advanced or recurrent disease [14–18]. In the most recent trial, the addition of paclitaxel to cisplatin and doxorubicin following surgery and radiation did not improve progression-free survival (PFS) [17]. UPSC was represented in 13% in both arms, while UCCC represented only 4.5% of patients. A subset analysis revealed a nonsignificant trend towards improved survival with the addition of paclitaxel in patients with UPSC and no difference in survival in patients with UCCC. In the other four trials, UPSC represented 18% and UCCC represented 4% of the study population; however, no subset analyses were performed [14–,16,18]. The low numbers of patients with UPSC and UCCC in these prospective trials highlight the difficulty in obtaining meaningful data on how to manage patients with these rare histologies. Large, prospective trials of patients with UPSC and UCCC will probably not occur and we are left extrapolating data from overall results of trials that mostly include patients with EEC who may represent a different biological entity with different response patterns. That being said, McMeekin et al. analyzed the data from the four earlier GOG trials [14–,16,18] and determined that response rate was not related to histologic subtype [1]. The overall response rate in these four trials was 32% for UCCC and 44% for both EEC and UPSC; this difference was nonsignificant. These data would seem to suggest that results from EEC patients could be used to guide treatment strategies in patients with UPSC and UCCC. However, in these four trials, the hazard ratio for death was 1.2 for UPSC (p = 0.03) and 1.5 for UCCC (p = 0.01) and the hazard ratio for progression was 1.52 for UCCC (p = 0.009) relative to EEC, illustrating the worse overall prognosis for these patients compared with patients with EEC given the same chemotherapy regimen.
The overall improvement in PFS and OS seen with the addition of paclitaxel to doxorubicin and cisplatin (TAP) in GOG 177 [14], has led many clinicians to view this regimen as the ‘gold standard’ for patients with advanced endometrial cancer. However, given the increased toxicity of the TAP regimen and the relative low toxicity of the carboplatin and paclitaxel doublet, several investigators have examined the use of the carboplatin and paclitaxel doublet (CT) in advanced endometrial cancer, specifically in patients with UPSC and UCCC. Several retrospective studies have reported response rates between 60 and 89% with CT in advanced UPSC and UCCC [19–22]. Hoskins et al. conducted a Phase II trial of carboplatin and paclitaxel in women with advanced or recurring endometrial cancer. Patients with advanced UPSC had a response rate of 60% [23]. The GOG has recently completed recruitment of patients in a prospective Phase III trial comparing the TAP regimen with carboplatin and paclitaxel in advanced or recurrent endometrial cancers, including patients with UPSC and UCCC (GOG 209). Although the number of patients with UPSC and UCCC included in this trial will probably be too small for a statistically meaningful subset analysis, the results will hopefully provide an insight into the optimal treatment regimen for these patients.
It seems reasonable that all patients with advanced-stage UPSC and UCCC should receive some form of adjuvant chemotherapy despite the lack of specific prospective data. The controversy lies with which regimen will produce the best outcomes. Based on GOG 177, it seems reasonable to treat these patients with the TAP regimen knowing that they may experience significant toxicity, including some treatment-related deaths. That being said, the patients with measurable disease in GOG 177 derived the most benefit from the TAP regimen and it may be reasonable to limit this regimen to only those patients. For patients with significant medical comorbidities, carboplatin and paclitaxel is a reasonable alternative based on Phase II and retrospective data. The results of GOG 209 will hopefully shed light on this issue. Conclusive data on the optimal adjuvant chemotherapy regimen for these patients will probably remain elusive. The concept of neoadjuvant chemotherapy in patients with UPSC combined with interval debulking surgery has been mentioned in the literature and represents an interesting alternative to upfront surgery followed by chemotherapy. However, this topic is beyond the scope of this article.
Adjuvant chemotherapy in early-stage disease
Most studies examining adjuvant therapy in early-stage endometrial cancer have focused on identifying those patients with an increased risk of recurrence. Factors such as deep myometrial invasion, lymph-vascular space invasion, grade 3 disease, and advanced age all signify an increased risk of recurrence and decreased OS in patients with EEC compared with EEC patients without these factors [24–25]. However, patients with early-stage UPSC and UCCC have an increased risk of metastasis and recurrence even when these factors are absent [26–28]. Approximately 60% of patients with tumors clinically confined to the uterus will be upstaged after surgical staging [26,29–30] and 36% of patients with the disease clinically confined to a uterine polyp will have evidence of extrauterine disease after surgical staging [31]. Lymph node metastasis will be present in over 30% of patients with no myometrial invasion, further highlighting the importance of comprehensive surgical staging, which includes omentectomy and peritoneal biopsies in addition to hysterectomy, bilateral salpingo-ophorectomy, pelvic and paraaortic lymph node dissection and pelvic washings [26]. In addition, these patients often recur in adjuvant radiation fields, severely limiting the effect of salvage therapy. While adjuvant treatment of early-stage EEC has focused on local disease in the pelvis, adjuvant treatment of UPSC and UCCC has targeted increasingly larger regions and has incorporated systemic therapy.
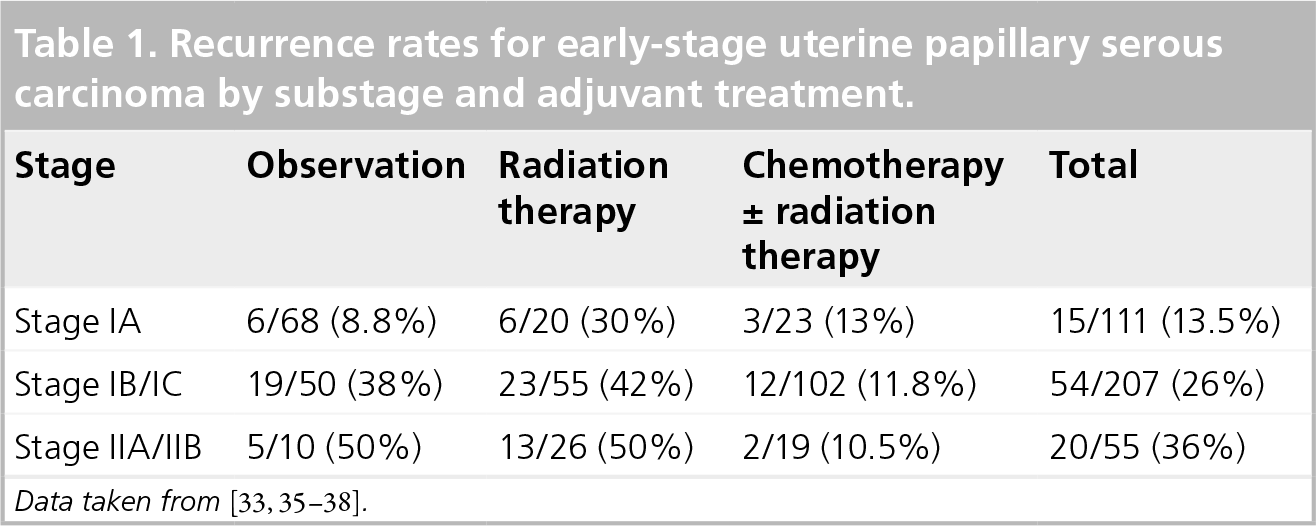
Nevertheless, we are then left with the question: should all patients with early-stage UPSC and UCCC receive adjuvant chemotherapy? As with advanced-stage disease, there are no homogeneous prospective randomized trials that can answer this question. However, several large retrospective studies have proved informative. Dietrich et al. reported data on 21 patients with surgical stage I UPSC treated with carboplatin and paclitaxel [32]. With an average follow-up of 41 months, no recurrences were observed. In a study of 83 patients with surgically staged UPSC, Havrilesky et al. reported a 9% overall recurrence rate in patients with stage IA cancers. However, only 5% of stage IA UPSC who underwent observation recurred versus 20% of stage IA UPSC patients who received adjuvant therapy, suggesting that adjuvant therapy may have little benefit in stage IA UPSC [33]. Thomas et al. reported retrospective data on 15 stage IA UPSC patients who underwent systematic pelvic and paraaortic lymph node dissection [34]. None of the stage IA patients recurred regardless of adjuvant therapy (11 out of 15 underwent observation alone). None of the stage IB/IC patients treated with adjuvant chemotherapy in this study experienced a recurrence compared with 25% of stage IB/IC patients not treated with adjuvant chemotherapy. Kelly et al. examined 72 patients with stage I UPSC treated with adjuvant platinum-based chemotherapy with or without vaginal radiation versus observation [35]. No recurrences were noted in 12 stage IA patients without evidence of residual disease in the hysterectomy specimen regardless of adjuvant therapy or in seven stage IA patients with evidence of residual uterine disease treated with adjuvant chemotherapy. However, 43% of stage IA patients with residual uterine disease recurred who underwent observation alone. Stage migration may account for the high recurrence rate in stage IA cancers in this study, secondary to a lack of mandated systematic lymph node dissections. One of the 22 patients (4%) with stage IB/IC UPSC recurred following adjuvant chemotherapy, whereas 14 out of 18 patients (78%) with stage IB/IC UPSC who underwent observation alone experienced a recurrence. OS was also significantly improved with adjuvant chemotherapy (p = 0.05). The largest retrospective study of patients with surgical stage I UPSC examined at outcomes in 142 patients who either had no further treatment, radiation therapy (RT) alone, or carboplatin and paclitaxel ± RT [36]. The recurrence rate in stage IA patients was 12% and was not associated with adjuvant therapy. However, PFS was significantly improved in stage IB/IC patients who received adjuvant chemotherapy over those that did not (28 vs 11%; p = 0.001). A trend toward improved cancer-specific survival was also noted (p = 0.08). In an analysis of adjuvant chemotherapy in 55 patients with surgical stage II UPCS, this same group reported an improvement in recurrence rate (p = 0.013) and PFS (p = 0.028) in patients who received adjuvant platinum-based chemotherapy compared with those who did not [37]. The effect of adjuvant chemotherapy on OS was not significant in patients with stage II UPSC (p = 0.115). The same group has recently updated their findings in early-stage UPSC patients with similar results [38]. Multivariate analysis of 151 patients with stage I and 55 patients with stage II UPSC, demonstrated that only adjuvant chemotherapy improved PFS (p = 0.006). Interestingly, their combined analysis of stage I and stage II UPSC revealed that adjuvant chemotherapy, in addition to patient age and depth of myometrial invasion, was significantly associated with OS (p = 0.02), while survival was not improved with the use of adjuvant chemotherapy in stage I or stage II UPSC when analyzed separately.
Several small retrospective studies have suggested the potential benefits of adjuvant platinum-based chemotherapy in patients with UCCC [39–41]. However, most of these studies include either heterogeneous histologic subtypes with very few UCCC, or very few patients treated with adjuvant chemotherapy. Thomas et al. reported data on 49 patients with stage I and II UCCC. In 22 definitively staged patients (>20 lymph nodes removed), of whom 50% received some form of adjuvant RT or platinum-based chemotherapy, only one recurrence (vaginal) was noted. In the 27 stage I and II patients with a suboptimal lymph node dissection, nine experienced a recurrence, including three distant failures. This study highlights the importance of complete surgical staging for UCCC, but offers little insight into the efficacy of adjuvant chemotherapy in early-stage UCCC [34]. Studies examining only UCCC patients are probably not feasible and we are, therefore, forced to extrapolate from studies including other high-grade endometrial subtypes.
Recurrence rates for early-stage uterine papillary serous carcinoma by substage and adjuvant treatment.
Progression-free survival and overall survival in early-stage uterine papillary serous carcinoma by adjuvant treatment.

CSS: Cancer-specific survival; C T: Chemotherapy; N/A: Not available; OBS: Observation; OS: Overall survival; PFS: Progression-free survival; RT: Radiation therapy.
Conclusion
Adjuvant chemotherapy is recommended for all patients with advanced-stage UPSC and UCCC. Pending the results of GOG 209, the regimen of choice at present is paclitaxel, cisplatin and doxorubicin, especially in women with measurable disease. The carboplatin and paclitaxel doublet represents a reasonable alternative and should be considered in patients with significant medical comorbidities and nonmeasurable disease. It is likely that UPSC and UCCC will continue to be included with EEC in large prospective chemotherapy trials in advanced endometrial cancer and definitive data on the optimal treatment regimen for these two histologic subtypes will probably remain elusive.
The decision to treat all patients with early-stage UPSC and UCCC is less straightforward. The risk of recurrence in patients with surgical stage IA UPSC is approximately 13% and seems to be unaffected by choice of adjuvant therapy, suggesting that these patients, especially those with no residual uterine disease and those who undergo systematic lymph node dissection, may be observed. However, the 13% risk of recurrence is not trivial and careful patient counseling should take place prior to making the decision to forego adjuvant chemotherapy. Two large retrospective trials have demonstrated decreased recurrence rates that resulted in improved OS in patients with early-stage UPSC [35,38]. Although the data is retrospective in nature, the results support the routine use of adjuvant chemotherapy in these patients. Perhaps the most compelling reason to advocate routine adjuvant chemotherapy in these patients is that most recurrences occur at distant sites and less than 15% are salvageable [37–38]. In this context, observation or radiation therapy alone seem unreasonable options despite the lack of randomized prospective data. The paucity of data, retrospective or otherwise, on early-stage UCCC makes treatment recommendations difficult. Given the lack of meaningful data on UCCC, and the similarities in the outcomes of UCCC and UPSC, the treatment recommendations for UPSC should guide the treatment of UCCC. Therefore, the improvement in recurrence rate and survival in stage IB–IIB UPSC combined with the noninsignificant risk of recurrence in stage IA UPSC compels these authors to recommend routine adjuvant platinum and taxane-based chemotherapy for all patients with early-stage UPSC and UCCC.
Future perspective
Currently, the GOG has completed accrual for a prospective randomized trial comparing TAP versus carboplatin and paclitaxel in advanced-stage endometrial cancer (GOG 209). Accrual is ongoing in a separate GOG trial examining adjuvant pelvic radiation versus adjuvant vaginal brachytherapy plus six cycles of carboplatin and paclitaxel in patients with early-stage high-risk endometrial cancer (GOG 249). Both trials include patients with UPSC and UCCC. The results of these two trials are eagerly awaited and may provide additional insight into the management of these rare histologic subtypes.
Executive summary
Uterine papillary serous carcinoma (UPSC) and uterine clear cell carcinoma (UCCC) represent aggressive endometrial histologic subtypes.
Data extrapolated from randomized trials on adjuvant chemotherapy in advanced-stage endometrioid endometrial cancer (EEC) suggest that patients with advanced-stage UPSC and UCCC should receive adjuvant chemotherapy, either paclitaxel, doxorubicin and cisplatin or carboplatin and paclitaxel.
Established risk factors for recurrence in early-stage EEC do not predict recurrence in early-stage UPSC and UCCC.
Early-stage UPSC and UCCC patients have a higher recurrence rate, recur more often at distant sites, and are often unsalvageable once they do recur, compared with patients with early-stage EEC.
Retrospective data suggests that adjuvant platinum and taxane-based chemotherapy may improve recurrence rates and survival in patients with early-stage UPSC.
Data detailing the management of early-stage UCCC is lacking; therefore, current recommendations for the management of early-stage UPSC may be applied to UCCC given their similarity regarding recurrence patterns and outcomes.
Footnotes
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
