Abstract
Although breast cancer is increasing in incidence, mortality from the disease is decreasing due to better treatment options. As the population ages, more women are being diagnosed with breast cancer at increasingly older ages. Tumors in elderly women tend to be more indolent than those in younger women, and physicians are sometimes reluctant to treat this cohort of patients simply because of their age. It is important for both primary care physicians and oncologists to be aware of breast cancer characteristics and treatments in the elderly in order to better serve this segment of the population. This review will discuss the benefits of both adjuvant chemotherapy and hormonal therapy in women with early stage breast cancer, with specific emphasis on the special attributes of elderly patients that should be considered when devising treatment plans.
Breast cancer is the most commonly diagnosed cancer in women. An estimated 211,240 new cases of invasive breast cancer will have been diagnosed in women in the USA in 2005 [101]. The incidence of breast cancer increases with age and, due to the aging of the population, over half of all breast cancers are now diagnosed in women over the age of 65 years [1]. Although the incidence of breast cancer has been increasing, mortality has been declining, suggesting a benefit from earlier detection and improved treatments [2]. However, breast cancer is still the second leading cause of cancer mortality in women and, of deaths due to breast cancer, 68% are in women over the age of 60 years [102].
In all women, regardless of age at the time of diagnosis, approximately two-thirds of cancers are localized to the breast, a quarter to a third have spread to regional lymph nodes and the remainder to distant sites [102]. Patients with localized and regional disease are treated either with breast-conservation therapy combined with radiation therapy or with modified radical mastectomy, followed by adjuvant systemic therapy to reduce the risk of both local recurrence and distant metastasis.
The definition of ‘elderly’ varies widely, although many consider people over the age of 65 years to be elderly. As the average age of the population in the USA increases, so does the number of people over the age of 65 years with breast cancer. The median life expectancy of elderly women is relatively long, approximately 15.7 years for a healthy 70-year-old and 9.4 years for a healthy 80-year-old [103].
Survival after breast cancer is age dependent, being worse in the very young (<35 years) and in older women (>60 years) [3]. Thus, for older women with few comorbid conditions but a high risk of breast cancer progression, such as women with estrogen receptor (ER)-negative, lymph node-positive disease, the risk of mortality from breast cancer outweighs the risk of mortality from other causes [4]. In women over the age of 75 years, breast cancer is the cause of death in 29–34% of patients [5].
In this article, an overview of adjuvant systemic therapy in breast cancer will be presented, the currently available literature regarding the use of hormonal therapy and chemotherapy in patients over the age of 65 years will be discussed, and recommendations regarding the treatment of this expanding patient population will be provided.
Adjuvant systemic therapy
Despite complete surgical resection and, if indicated, radiation therapy of early stage breast cancers, both local and systemic recurrences frequently occur. Adjuvant systemic therapy is therefore indicated for the treatment of the majority of patients with early stage breast cancer, as substantial data demonstrate that it decreases mortality due to breast cancer [6]. This therapy consists of chemotherapy and/or hormonal therapy, and treatment decisions are based on a series of classically accepted prognostic and predictive factors [7] (Box 1).
Most of what is known about adjuvant systemic therapy, as well as prognostic and predictive factors used to identify patients most likely to benefit from it, is derived from studies of women younger than 65 years of age. Therefore, with a few exceptions, it is instructive to review the current state-of-the-art for each area of knowledge, and then to either discuss what is known about elderly patients with breast cancer or to extrapolate to them.
Classically accepted prognostic and predictive factors for breast cancer.
– Large tumor size
– Lymph node involvement
– High tumor grade
– Age (bimodal, see text)
– High urokinase plasminogen activator and plasminogen activator inhibitor type 1 antigen level
– Estrogen and/or progesterone receptor expression
– HER2/neu overexpression
At the time of initial diagnosis, prognostic factors help to predict long-term clinical outcome and are used to determine if a patient has sufficiently high risk of recurrence that she might benefit from adjuvant systemic therapy, if effective strategies are available. Factors that predict worse outcome in breast cancer include large tumor size, positive lymph nodes, high histologic grade and, in many parts of Europe, elevated expression of urokinase plasminogen activator (uPA) and plasminogen activator inhibitor type 1 (PAI-1) [8].
Simultaneous analysis of multiple gene expression has also been used to improve on currently available prognostic factors [9]. One method analyzes paraffin-embedded tumor tissue using real-time reverse transcriptase polymerase chain reaction assays to quantify the expression of multiple genes. This methodology underlies a study by the National Surgical Adjuvant Breast and Bowel Project (NSABP), using the Oncotype DX™ (Genomic Health) assay, which appears to accurately quantify the likelihood of recurrence in patients with ER-positive disease without lymph node involvement, thereby providing additional prognostic information to help guide treatment decisions [9,10]. Tissue samples from two previously conducted NSABP trials, B-20 and B-14, were used to develop and validate, respectively, the Oncotype DX assay [9,10]. It is important to point out that most of the tumor samples used for the development of this assay were from younger women, as the B-14 trial specifically excluded women aged over 70 years and only 27% of women in the B-20 trial were aged 60 years or older [11,12].
In contrast to prognostic factors, predictive factors help to predict the likelihood of a response to a given treatment [13]. Patients with tumors that express ER and/or progesterone receptors are likely to benefit from endocrine therapies, whereas patients with hormone receptor (HR)-negative tumors are not [6]. Similarly, patients with tumors that amplify and/or overexpress the human epidermal growth factor receptor type 2 (HER2)/neu receptor, a member of the epidermal growth factor receptor family, may benefit from treatment with the antibody trastuzumab, which specifically targets that receptor [14]. There is no a priori reason to suspect that these factors differ in their predictive abilities in older versus younger women.
Taken together, prognostic and predictive factors can be used to calculate the absolute reduction in odds of annual recurrence. For example, if a patient has a 40% risk of recurrence after 10–15 years, based on prognostic factors, and adjuvant therapy will likely decrease her annual odds of recurrence by approximately a third, then the adjuvant therapy will have decreased her absolute risk of recurrence by 10–12%. In general, adjuvant chemotherapy is recommended if the estimated absolute risk reduction is at least 5%; it is strongly considered if the reduction is 3–5%; and some patients are willing to endure the risks of chemotherapy for the possibility of as little as 1% absolute benefit [15]. Using available data, multiple calculators have been developed to assist oncologists in determining a patient's risk of recurrence based on these factors, and one of the most commonly used is called Adjuvant! Online [16,17].
The National Comprehensive Cancer Network (NCCN) has established guidelines for the adjuvant treatment of women with invasive breast cancer [18]. Adjuvant chemotherapy is recommended for patients with lymph node involvement at the time of diagnosis, as well as for patients with high-risk, node-negative disease. They recommend that all patients with HR-positive breast cancer and a tumor size of more than 0.5 cm receive adjuvant hormonal therapy. However, the evidence used in both NCCN and Adjuvant! Online was gathered from prospective clinical trials, which rarely include women over the age of 70 years. Thus, neither has sufficient data to make reliable recommendations regarding chemotherapy in this age group.
Adjuvant hormonal therapy
Endogenous estrogen is a primary growth factor for HR-positive breast cancer and endocrine manipulation is therefore used to minimize this stimulus. More than a century ago, it was recognized that ovariectomy could result in regression of advanced breast cancer [19]. Today, multiple methods are employed to limit endogenous estrogen, including ER modulators such as tamoxifen, ablation of estrogen production by aromatase inhibitors (AIs) and, in premenopausal women, chemical or surgical ovarian ablation (Box 2).
One method of limiting the systemic effect of estrogen is through ER modulation, and tamoxifen has been the primary adjuvant hormonal therapy used in the USA for more than 20 years [20]. Tamoxifen is a selective ER modulator (SERM) that acts as an antiestrogen in breast tissue, thereby decreasing the risk of both recurrence of breast cancer and incidence of second primary tumors. Both pre- and postmenopausal women with HR-positive invasive breast cancer benefit from treatment with tamoxifen, which is generally well tolerated and has an excellent long-term safety profile [6]. Surprisingly, tamoxifen acts as an estrogen agonist in other tissues, such as bone, liver and uterus (Table 1). Its estrogenic effects on bone result in a decreased risk of osteoporosis. Its estrogenic effects in the liver and uterus are associated with an increased risk of thrombosis (deep vein thrombosis [DVT] and possibly stroke) and endometrial cancer in women who have not undergone prior hysterectomy, respectively [21–24]. In studies of tamoxifen as a preventive agent, these agonistic secondary effects of tamoxifen are most evident in postmenopausal women. Specifically, the risk of thrombogenesis, especially related to cerebrovascular accident, may be particularly high in older women who are already at risk for the complication [21–24]. An additional side effect of treatment with tamoxifen is an increased risk of cataract formation. In a study of over 13,000 women who received either tamoxifen or a placebo, tamoxifen use was found to be associated with an increased risk of cataract formation and cataract surgery, especially in the older population [25].
Comparison of the side effects of tamoxifen and aromatase inhibitors.
Substantial progress has been made in the use of hormone therapy for breast cancer in the past few years. Although ovarian estrogen production has ceased, postmenopausal women produce estradiol and estrone by converting adrenal gland-derived steroid precursors via aromatization in peripheral tissues. Recent data from prospective, randomized clinical trials have supported the use of a newer class of medications that inhibit this conversion, the AIs, as first-line adjuvant hormonal therapy in postmenopausal women with HR-positive breast cancer [26]. Anastrozole and letrozole are examples of nonsteroidal AIs, which are competitive inhibitors of the aromatase enzyme, whereas exemestane is a steroidal AI that irreversibly inactivates aromatase.
Multiple large, randomized trials comparing AIs and tamoxifen have demonstrated the effectiveness of AIs in the adjuvant setting for postmenopausal women with HR-positive breast cancer [27–32]. In one trial, patients were randomly assigned to anastrozole versus tamoxifen for initial adjuvant hormonal therapy, and women treated with anastrozole had prolongation of disease-free survival (DFS) as well as reduced distant metastases and contralateral breast cancers when compared with those treated with tamoxifen [29]. In a second trial, women were randomly assigned to exemestane after 2–3 years of tamoxifen therapy or to complete 5 years of tamoxifen therapy. Again, a significant reduction in breast cancer-related events was noted in women who were switched to the AI [27]. In a third trial that evaluated women treated with letrozole versus placebo after completion of 5 years of tamoxifen therapy, a significant improvement in DFS for all patients randomized to letrozole was observed, as was a statistically significant improvement in overall survival (OS) in the subset of women with lymph-node involvement treated with the AI [28].
AIs function by blocking the production of estrogen, rather than by acting on the ER, and therefore their side-effect profiles differ from that of tamoxifen (Table 1). Long-term safety data are not yet available, but it is known that osteoporosis and fracture risk are increased with these medications, and that a high percentage of women develop musculoskeletal symptoms [26]. In one study, fractures were reported in 11% of patients treated with anastrozole, compared with 7.7% of patients treated with tamoxifen [29]. Effects on lipids and cardiovascular complications are still being evaluated. AIs are now being routinely used for adjuvant therapy of postmenopausal women with ER-positive breast cancer, although additional studies are still underway to determine the appropriate timing of these medications in relation to tamoxifen [26].
Adjuvant hormone therapy in the elderly
Given the proven survival benefits and relatively favorable toxicity profiles, almost all elderly women with HR-positive disease should be treated with adjuvant hormonal therapy unless they have very poor performance status or are expected to have significantly decreased survival from an illness other than breast cancer. Tamoxifen has been shown to be beneficial in all age groups, including women aged over 70 years [6]. It improves bone density, which is advantageous in the older population, although as described above, it is associated with an increased risk of thromboembolic disease and endometrial cancer.
Endocrine agents used in adjuvant systemic therapy of postmenopausal patients with breast cancer.
– Tamoxifen
– Anastrozole
– Exemestane
– Letrozole
Several recent trials demonstrate that AIs appear to be superior to tamoxifen with regards to efficacy, although limited data specific to the elderly population are available for the AIs. The median age of enrolled patients in the three largest trials ranged from 62–64 years, and only a quarter of women in the MA17 trial were aged 70 years or older [27,28,33]. In the initial reporting of results from the Anastrozole or Tamoxifen Alone or in Combination (ATAC) trial after a median follow-up of 33 months, subgroup analysis suggested that anastrozole is at least as effective as tamoxifen in women aged 65 years and over [34]. Therefore, for an elderly woman without osteoporosis, an AI is likely the better initial choice for adjuvant treatment of breast cancer, especially if she is at a high risk of recurrence.
In a study of over 200,000 randomly selected women over the age of 50 years, osteopenia was present in 40% and osteoporosis in 7%, and the presence of osteoporosis was associated with a fourfold increase in fracture risk, which can lead to significant morbidity and mortality [35]. The additional increased incidence of osteoporosis and fracture due to treatment with AIs is of concern, especially in an elderly population. Therefore, patients with pre-existing osteoporosis and without a contraindication to tamoxifen should probably be treated with tamoxifen instead of an AI. Pretreatment and serial bone-density measurements are useful for the management of patients treated with AIs, with consideration of the addition of bisphosphonates if osteopenia or osteoporosis develop or worsen. Postmenopausal women, with few exceptions, should be urged to take daily calcium and vitamin D supplementation [36], especially if receiving an AI, although there are no data yet available to demonstrate decreased rates of bone loss with calcium supplementation.
Adjuvant chemotherapy
Evidence from the most recent overview analysis, performed by the Early Breast Cancer Trialists' Group (EBCTG) of clinical trials of adjuvant therapy in breast cancer, demonstrated a significant reduction in breast cancer deaths in women under the age of 70 years treated with chemotherapy, which was greatest if it included an anthracycline [6]. Since so few prospective, randomized clinical trials have included women over the age of 70 years, insufficient data are available to draw conclusions for this age group. However, evaluation of recurrence and mortality rates in women treated with chemotherapy demonstrated decreasing proportional reduction in the effect of chemotherapy with increasing age (Figure 1) [6]. Definitive explanations for this effect are lacking. However, it is probable that chemotherapy is less effective in women whose tumors are rich in ERs, and both the proportion of ER-positive tumors and absolute amount of ER expression increases with age [5,37–39].
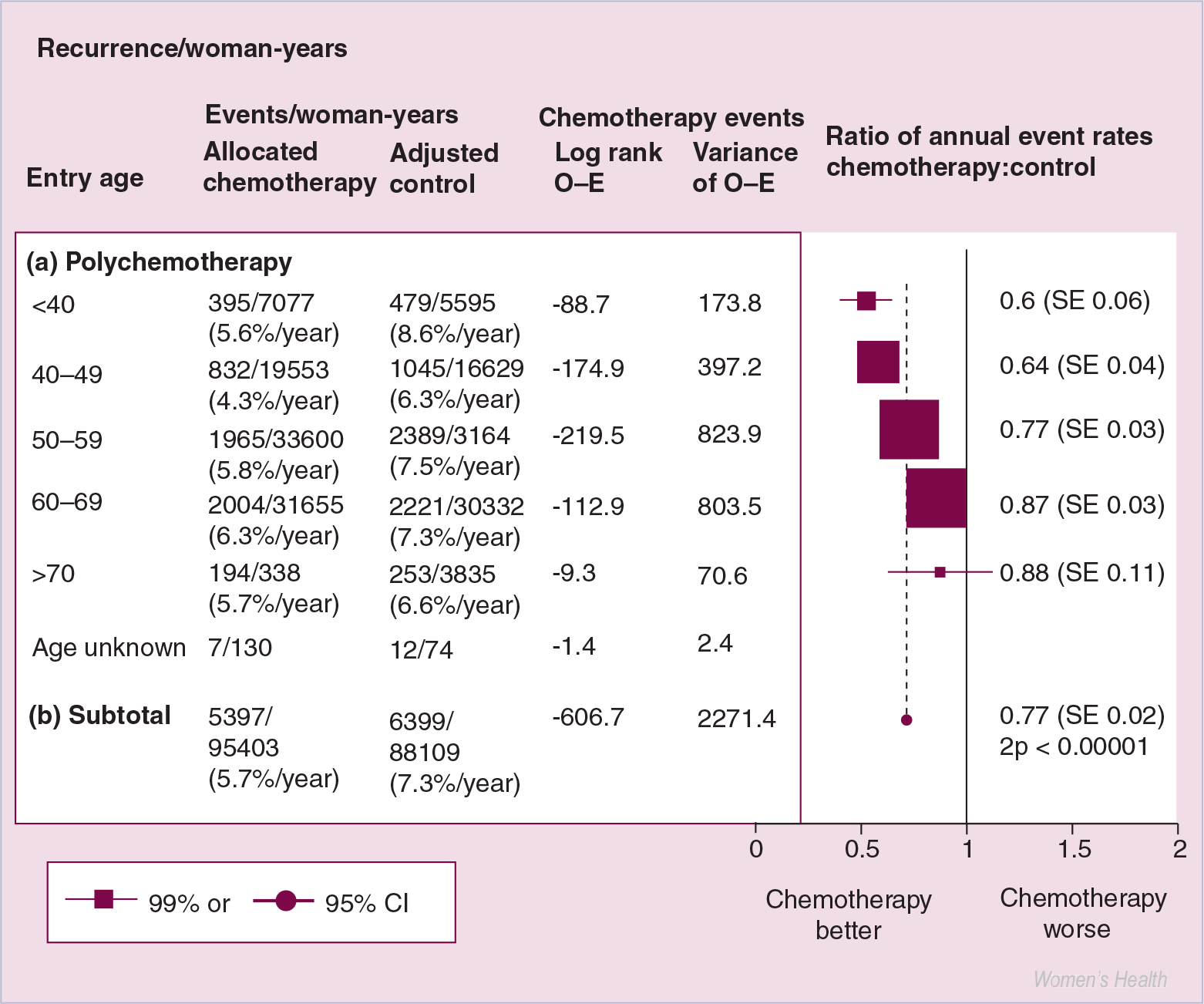
Decreasing effects of chemotherapy on recurrence rate with increasing age.
More recent studies have demonstrated the utility of the addition of taxanes to anthracycline-based chemotherapy, and now many women in the USA are treated with some combination and sequence of anthracycline, alkylating agent and taxane, especially if they are at a higher risk of recurrence [40]. In addition, results from a study evaluating the benefit of increasing the dose density of chemotherapy, by administering it every 2 weeks rather than every 3 weeks, demonstrate a survival benefit for the dose-dense regimen [41].
As noted above, trastuzumab is a humanized monoclonal antibody that specifically targets HER2/neu, a tyrosine kinase receptor that is overexpressed in 20–30% of breast cancers [14,42]. Overexpression of HER2/neu was found to be associated with a more aggressive form of the disease. In patients with metastatic breast cancer that overexpresses HER2/neu, both single-agent trastuzumab and trastuzumab in combination with chemotherapy were demonstrated to produce durable responses [14,42]. However, cardiac toxicity was a significant adverse event, especially in combination with anthracyclines and, to a lesser extent, paclitaxel [14,42].
More recently, results from three trials of adjuvant trastuzumab have been reported [43,44]. Taken together, these very early results suggest that adjuvant trastuzumab decreases the risk of recurrence by almost 50% and mortality by approximately 30%. In a combined analysis of two trials conducted in North America, the absolute increase in DFS was 8% at 3 years, and the increase in OS was 2% at 3 years. However, as with previously discussed prospective, randomized clinical trials, few of the enrolled subjects were older than 65 years of age, and subset analyses are lacking.
In summary, substantial progress has been made in adjuvant systemic therapy of early breast cancer, with dramatic reductions in mortality in the Western world. However, the studies discussed above have been conducted almost exclusively in women under the age of 65 years.
Adjuvant chemotherapy in the elderly
As noted, although hundreds of thousands of women have enrolled in prospective, randomized, controlled trials over the last three decades, hardly any were over the age of 65 years [6]. Only a few studies have specifically addressed adjuvant systemic therapy in the elderly [38,45–48].
For example, investigators from the Cancer and Adult Leukemia Group B (CALGB) retrospectively compared adjuvant chemotherapy administered to lymph node-positive women younger than 65 versus those older than 65 years of age [48]. Only 8% of the 6487 women in the included trials were over the age of 64 years. In this group, multiple variables, including smaller tumor size, fewer positive lymph nodes, and chemotherapy and tamoxifen use, all correlated with longer DFS and OS. Age did not have a significant impact on DFS rates, although, as with the older population-based studies, OS was decreased in women aged 65 years and older due to nonbreast cancer deaths. However, it is likely that the elderly women enrolled in these trials represent a highly selected group and are not representative of the general elderly population.
In both the NSABP B-20 trial and the International Breast Cancer Study Group (IBCSG) trial VII, lymph node-negative, primarily ER-positive women were randomly assigned to tamoxifen alone versus tamoxifen plus cyclophosphamide, methotrexate and 5-fluorouracil (CMF) [38,45]. In both trials, a benefit was demonstrated for younger women. However, in a subset analysis of older patients, the addition of chemotherapy resulted in no improvement in recurrence-free survival, DFS or OS. A randomized study from France, specifically evaluating women aged over 65 years with operable, node-positive, primarily ER-positive breast cancer, was conducted to compare epirubicin plus tamoxifen versus tamoxifen alone [46]. Approximately 40% of patients were aged 70 years or older at time of enrollment. DFS and OS at 6 years were similar between the two groups, although the risk of relapse was increased in patients treated with tamoxifen alone.
In ER-negative women, however, chemotherapy may be beneficial regardless of age. NSABP trials B-13, B-18 and B-23 evaluated node- and ER-negative breast cancer patients. In these trials, all women benefited equally with regard to reduction in recurrence-free survival and OS, including those aged 60 years and older versus those younger than 60 years of age [47]. Compared with women aged over 60 years and treated with surgery alone, ER-negative women treated with surgery followed by adjuvant doxorubicin and cyclophosphamide (AC) had an absolute benefit in recurrence-free survival of 5% and in OS of 7%. The benefit from the addition of chemotherapy in older women is significant, but less than the benefit demonstrated for younger women.
Taken together, the results of these trials suggest that the benefits of chemotherapy in the older population are modest, at best, if HR status is not considered. However, in the trials of women with HR-positive disease, no clear benefit has been observed from the addition of chemotherapy, even in patients with lymph-node involvement. In studies of women with HR-negative disease, a benefit for chemotherapy in addition to surgery has been suggested, but the improvements in outcome were not as great as those seen in younger women [6]. More clinical trials of elderly women are clearly indicated in order to determine in which subpopulations of elderly women the benefits of chemotherapy are likely to outweigh the risks.
Special considerations in the elderly
The preceding discussion demonstrates that precious limited prospective, randomized clinical-trial data are available to permit evidence-based clinical decision-making regarding adjuvant systemic therapy in the elderly with breast cancer. Nonetheless, this disease causes substantial morbidity and mortality in this age group. Unfortunately, extrapolation from prospective, randomized clinical-trial results in younger women is complicated. Two considerations distinguish elderly women from younger patients: cancer biology, and comorbid conditions and host factors that may affect and/or alter pharmacology (Box 3).
Breast cancer biology in the elderly
Breast cancers in the elderly population are biologically different from those that develop in younger patients [5,49,50]. In general, older women have more early stage cancers that express more favorable characteristics. These include lower histologic grade, higher frequency of HR expression and lower S-phase fractions compared with younger women [50,51]. In addition, tumors from older women are less likely to express HER-2/neu [5].
Of these factors, ER expression is the most important consideration regarding the effects of adjuvant systemic therapy according to age. In the Surveillance, Epidemiology and End Results (SEER) database, ER expression increased with increasing age [5]. In women aged 55–64 years, 68% of tumors expressed ERs, whereas in women over the age of 85 years, 84% of tumors expressed ERs. Among patients with ER-positive tumors and node-negative disease, there was a higher ER concentration with advancing age [38].
Special considerations in the elderly.
– Higher percentage of tumors express estrogen and/or progesterone receptors
– Decreased expression of HER2/neu
– More favorable biologic characteristics
– More likely to have comorbidities
– More likely to have reduced hepatic and renal function
– Polypharmacy leading to drug interactions
– Patient and physician perceptions result in decreased treatment
Decreased treatment of older patients
Numerous patient factors, other than comorbidities, are also a factor when treating older patients. Physiologic changes occur in adults as they age, including alterations in drug metabolism and decreases in renal and hepatic function. Multiple social factors also become relevant, including poor social support and financial concerns. Another primary focus of older cancer patients is maintaining quality of life (QoL).
Decreased recommendation
In clinical practice, elderly patients may not receive the same or as much adjuvant systemic therapy as younger women. Elderly patients are more likely to have comorbidities and decreased performance status than younger women, and therefore adjuvant systemic therapy may not be feasible or appropriate. In a longitudinal, observational study of 936 women aged 40–84 years with breast cancer, higher rates of comorbidity conferred a higher mortality rate compared with women without comorbid conditions, regardless of age or cancer stage at diagnosis [52]. There was a 20-fold higher rate of mortality from causes other than breast cancer in women with three or more comorbid conditions. In a retrospective single-institution study of women over the age of 75 years with breast cancer, researchers demonstrated an association between a higher number of comorbidities and decreased treatment [53]. Although 24% of patients had node-positive disease, only 5% of patients over the age of 75 years received adjuvant chemotherapy. Likewise, only 79% of patients over the age of 80 years received tamoxifen, although 94% had ER-positive disease. Other studies have shown that chronologic age, as opposed to overall health status, correlates with decreased treatment with chemotherapy, regardless of comorbidity score [54].
Perceptions
In addition to the patient factors discussed above, another reason for decreased adherence to treatment guidelines is the perception of the patient and/or the physician regarding treatment. Newly diagnosed women with breast cancer were studied to determine social factors that are central to treatment decisions [55]. Patients who were older, unmarried, and who had less interaction with their breast cancer physicians were less likely to opt for more aggressive initial therapy. Although a majority of women were interested in minimizing risk of recurrence, QoL and financial factors were also significant considerations.
Decreased tolerability
Drug delivery & pharmacokinetics in the elderly
Even if standard adjuvant systemic therapy is offered, patients may not be able to tolerate it. Physiologic changes that occur with aging can affect the effectiveness and toxicity of hormonal therapy and chemotherapy in the older population by altering pharmacokinetics and bioavailability [56,57]. Changes in the gastrointestinal system can affect the absorption of oral medications. Decreases in total body water and increases in total body fat cause changes in the volume of distribution, thereby leading to altered peak concentrations and half-lifes of drugs. Hypoalbuminemia can result in an increased risk of toxicity of protein-bound drugs. Many chemotherapy agents are metabolized via the cytochrome P (CYP)450 system, and hepatic drug-metabolizing enzymes, especially of the CYP450 system, have 30% less activity in the elderly [57]. Similarly, decreases in glomerular filtration rate result in decreased clearance of renally excreted drugs. In addition, older patients are more likely to take other medications to manage comorbidities, and this polypharmacy increases the risk of drug–drug interactions.
Few studies have been performed specifically to evaluate the pharmacokinetics of chemotherapy medications in the elderly. A detailed analysis of pharmacologic considerations for the majority of commonly used antineoplastics in the elderly is beyond the scope of this article; however, the interested reader is referred to recent comprehensive reviews [58,59].
Tolerability of chemotherapy in the elderly
Given the increased likelihood of impaired metabolism of many chemotherapy agents in elderly patients [58], several studies have been conducted evaluating the tolerability of different adjuvant chemotherapy regimens in this group of patients. In the IBCSG Trial VII, women over the age of 65 years treated with CMF had increased rates of grade 3 toxicities, hematologic toxicity and mucosal toxicity, and were more likely to receive dose reductions in comparison to younger women [45]. Despite this increase in toxicity, however, a QoL assessment failed to demonstrate a significant difference between the age groups.
These same investigators have reported that treatment-related deaths in patients treated with the combination of CMF and tamoxifen in multiple IBCSG trials are higher in the elderly [60]. Of the 545 women over the age of 65 years, 1.28% had treatment-related deaths, mostly from toxic effects, sepsis or cerebrovascular accidents. In comparison, there were only 0.16% treatment-related deaths in the 6381 women under the age of 65 years.
In contrast, in the French trial discussed above, which specifically involved women over the age of 65 years, 96.9% of patients treated with epirubicin received all six cycles and 96.5% of patients received all three doses per cycle [46]. The chemotherapy was well tolerated, with grade 3 nausea and vomiting occurred in only 4.6% of patients. Eight cases (0.5%) of decreased left ejection fraction occurred in the 160 patients who received the anthracycline epirubicin, and none resulted in longstanding cardiac symptoms.
Specific chemotherapy complications in the elderly
Rate of hospitalization
Given the unclear benefit of chemotherapy in the elderly population, it is important to weigh the risks of chemotherapy and to understand if there are ways to predict those risks. In a study using the SEER database, 9% of breast cancer patients aged 65 years and older treated with chemotherapy were hospitalized for neutropenia, fever, thrombocytopenia or other adverse effects, whereas of those who did not receive chemotherapy, only 0.5% of patients in that age group required hospital admission [61]. Risk of admission increased with increasing comorbidity score, use of an anthracycline and tumor stage. Among this elderly population, there was no increased rate of hospitalization with advancing age. However, the study did not include patients under the age of 65 years, therefore the comparative hospitalization rate of younger patients treated with chemotherapy is unknown.
Anthracycline-induced congestive heart failure
Anthracyclines are associated with a dramatic, cumulative dose-dependent risk of congestive heart failure [62]. Given the frequency of this condition in elderly patients in general, one must be concerned about anthracycline use in older patients. A retrospective analysis of breast cancer patients treated with doxorubicin-containing regimens at a single institution was conducted, in which almost 17% of patients were aged 65 years or older [63]. All patients had good performance status and baseline cardiac function. These older patients tolerated the chemotherapy as well as a cohort aged between 50–65 years, with no worse long-term morbidity due to cardiotoxicity. These data suggest that, with careful selection, anthracyclines can be used safely in this population.
Taxane-induced neuropathy
Peripheral neuropathy is a common complication of taxane chemotherapy [64]. The neuropathy symptoms generally worsen with subsequent cycles, but are frequently reversible. In one study, 9% of patients with metastatic breast cancer developed grade 3 neuropathy after treatment with paclitaxel [65]. Of the enrolled patients, 34% were over the age of 65 years, and subgroup analysis demonstrated a similar toxicity profile for both young and elderly patients. However, patients with comorbidities, such as diabetes, are likely to have peripheral neuropathy at baseline and may have more difficulty tolerating taxane therapy than the more healthy patients who are typically enrolled in clinical trials.
Risk of febrile neutropenia & growth factor support in the elderly
A retrospective study of febrile neutropenia in elderly breast cancer patients treated with chemotherapy correlated poor performance status and presence of bone marrow metastasis, but not age, with higher rates of neutropenic fever [66]. Although age alone may not predispose elderly patients to higher rates of febrile neutropenia, a greater incidence of serious medical complications develop in older, compared with younger, patients with febrile neutropenia [67,68].
Growth factors such as granulocyte colony-stimulating factor (G-CSF) and erythropoietin are endogenous factors that help to stimulate the production of hematopoietic precursors in the bone marrow. Recombinant G-CSFs, such as filgrastim and pegfilgrastim, stimulate increased bone marrow production of neutrophils. Routine use of G-CSF is indicated for patients receiving moderate-intensity chemotherapy, although most breast cancer regimens are less intense [69,70]. In addition, growth factor support with G-CSF is indicated to allow for administration of dose-dense chemotherapy, such as AC administered every 2 weeks.
No studies have been published to date recommending the prophylactic use of growth-factor support to decrease the risk of neutropenic fever specifically in elderly patients with breast cancer. In the most recent guidelines from the American Society of Clinical Oncology (ASCO) in 2000, being elderly is not specifically mentioned as a reason for giving colony stimulating factors (CSFs) as front-line therapy with nonmyelosuppressive chemotherapy. Use of CSFs is recommended if the patient has specific risk factors for developing fever and neutropenia, or infection. CSF use has been more extensively studied in elderly patients with non-Hodgkin's lymphoma. In that setting, CSFs are sometimes used to allow completion of the chemotherapy regimen, but have not been demonstrated to improve clinical outcomes, such as survival [70].
A recent evidence-based guideline for the use of CSFs in elderly cancer patients recommended that G-CSF should be used with the initial cycle of chemotherapy in patients aged 70 years or older with breast cancer who experience severe neutropenia [68]. This recommendation is similar to the recommendations for younger patients. QoL also improved with the use of CSFs in elderly patients receiving moderate-intensity chemotherapy [68].
Anemia
Anemia is a common problem for patients with breast cancer, and it contributes to decreased QoL and perhaps even survival [71]. ASCO and American Society for Hematology (ASH) guidelines recommend the use of recombinant human erythropoietin in cancer patients with hemoglobin levels less than 10 mg/dl, and in selected patients with hemoglobin levels between 10–12 mg/dl [72]. In a trial evaluating recombinant human erythropoietin in breast cancer patients receiving adjuvant therapy, improvement in QoL was seen, and outcome was not assessed [73].
One study evaluating prophylactic erythropoietin to prevent anemia in breast cancer patients receiving first-line chemotherapy for metastatic disease was stopped early due to increased mortality in the erythropoietin arm [74]. Similar studies have not been conducted in the adjuvant breast cancer treatment setting. Incidentally, similar results have been reported in a study of chemotherapy with and without erythropoietin in head and neck cancer patients [75].
The ASCO/ASH guidelines recommend considering the use of erythropoietin in elderly patients with limited cardiovascular reserve if their hemoglobin levels are less than 12 mg/dl [72]. No studies have specifically examined the use of erythropoietin in elderly patients with breast cancer. At the present time, there is no clear evidence that erythropoietin is detrimental, especially in women with established, symptomatic anemia. Consistent with the guidelines, it is reasonable to consider its use in elderly patients with hemoglobin levels lower than 12 g/dl who are receiving chemotherapy, especially if they are symptomatic.
Summary
Due to the few studies performed to date specifically evaluating chemotherapy use in the elderly, making broad statements regarding chemotherapy tolerability in the elderly can be difficult. Healthy older patients with no comorbidities, especially those without pre-existing heart disease, liver or kidney dysfunction, are more likely to tolerate chemotherapy, although side effects such as myelosuppression may still occur. Some of these side effects can be minimized with the use of supportive factors, as described in the previous section. However, patients with more comorbidities are at a higher risk of adverse outcomes from chemotherapy, and the risks may outweigh any benefits of chemotherapy in this population.
Future perspective
One of the primary barriers to developing guidelines for adjuvant treatment of older breast cancer patients is the lack of evidence derived from clinical trials [76]. Historically, enrollment has been poor, as can be seen in a large, retrospective analysis of cooperative group trials, where only 8% of patients were over the age of 64 years, and only 2% were over the age of 69 years [48]. An education campaign aimed at physicians, with the goal of increasing enrollment of elderly patients in cooperative group trials, was implemented, but was unfortunately unsuccessful [77].
Despite the difficulties accruing patients, a number of studies evaluating adjuvant chemotherapy in the older population are currently underway. The US Breast Intergroup is conducting a trial in women aged 65 years and older who have undergone surgical resection for early stage breast cancer, comparing either CMF or AC with oral capecitabine. In Europe, the Adjuvant CyTotoxic chemotherapy In Older womeN (ACTION) trial is comparing no chemotherapy with standard AC chemotherapy in women 70 years of age or older with HR-negative breast cancer [78]. It is hoped that successful completion of these trials, aimed specifically at elderly patients with breast cancer, will add to the sparse data set available for this age group.
Conclusion
Adjuvant systemic therapy for early stage breast cancer has been demonstrated to significantly reduce recurrence rates and improve OS in women of all ages. The choice of adjuvant therapy used is based on risk of recurrence of disease as well as an estimate of patient mortality from nonbreast cancer-related causes. For a majority of older women with HR-positive breast cancer, hormonal therapy with an AI or tamoxifen is appropriate, as these medications have few side effects and are unlikely to substantially affect QoL, while decreasing the risk of disease recurrence. In general, treatment with an AI is preferred, although for each individual patient, the risks of side effects from AIs versus tamoxifen must be carefully weighed.
In older, otherwise healthy women with ER-positive breast cancer who are at a high risk of recurrence, such as those with large tumors or lymph-node involvement at presentation, consideration should be given to treatment with adjuvant chemotherapy in addition to hormonal therapy. Similarly, otherwise healthy older women with HR-negative disease, who are thus unlikely to benefit from adjuvant hormonal therapy, should be offered treatment with adjuvant chemotherapy to reduce the risk of recurrence. This patient population should also be encouraged to participate in clinical trials.
Executive summary
This therapy has resulted in decreased mortality in breast cancer patients with early stage disease.
Patients with poor prognostic factors benefit more from adjuvant systemic therapy.
Adjuvant endocrine therapy, such as ovarian ablation, tamoxifen or an aromatase inhibitor (AI), is used in patients with hormone receptor (HR)-positive disease.
Adjuvant chemotherapy is used primarily in patients with a higher risk of recurrence or HR-negative disease.
Elderly women are more likely to have HR-positive disease, and therefore benefit from adjuvant hormonal therapy.
Tamoxifen is well tolerated and decreases the risk of osteoporosis, but causes an increased risk of thromboembolic disease and endometrial cancer and may cause a higher incidence of stroke in older women.
AIs are more effective than tamoxifen, although they cause osteoporosis and there are no long-term safety data available at present.
This therapy is indicated in almost all patients with HR-positive disease unless they have an extremely low risk of recurrence or low likelihood of survival due to nonbreast cancer-related comorbidities.
Elderly women with HR-positive disease may not derive as much benefit from chemotherapy as younger patients.
It is indicated in women with few comorbidities at a high risk of disease recurrence.
It is indicated in women with HR-negative disease who are otherwise healthy.
It is not indicated in women with lower risk of recurrence or with significant comorbidities.
Breast cancers in elderly patients are more likely to be less aggressive and to be HR-positive.
Elderly patients are more likely to have comorbidities, which can affect both tolerability of treatment and overall survival.
Elderly patients are more likely to have reduced hepatic and renal clearance of medications.
Growth factors such as granulocyte colony-stimulating factor can be used in elderly breast cancer patients receiving chemotherapy, especially to minimize the risk of febrile neutropenia.
Erythropoietin has been shown to improve quality of life in patients with chemotherapy-related anemia, although it may result in decreased survival in patients with mild, asymptomatic anemia.
Significant advances have been made in adjuvant systemic treatment of breast cancer in recent years. As the population continues to age and the proportion of older women with breast cancer continues to increase, it is becoming increasingly important to focus on this population in order to develop evidence-based guidelines for management of the disease.
