Abstract
The treatment and survival outcome of acute leukemia in women is generally similar to that of men. However, acute leukemia in women poses additional challenges in clinical practice. In addition to important precautions during therapy, such as prevention of abnormal uterine bleeding in premenopausal women and therapy during pregnancy, women who are survivors of acute leukemia face unique and potentially long-term health-related problems. In this review, we address the aforementioned issues, as well as the various health and psychosocial challenges faced by women who survive childhood leukemia during their path to adulthood. Finally, we address the issue of therapy-related acute leukemia in the category of women who are survivors of breast and ovarian cancer.
Keywords
The global burden of leukemia
Leukemias, both in their acute and chronic forms, comprise approximately 3% of all new cancer cases worldwide, with incidence patterns that are relatively consistent across geographic regions [1]. As with most malignant diseases, the incidence rates for all types of leukemia are slightly higher among males (57%) compared with females [2].
An overview of acute leukemias
Acute leukemia (AL) is a malignant clonal proliferation of immature hematopoietic cells. AL blast cells are nonfunctioning cells that rapidly accumulate in the bone marrow, ultimately replacing their functioning counterparts and resulting in many of the manifestations of the disease. Based on their origin, ALs are classified into two main categories: acute myeloid leukemia (AML) and acute lymphoblastic leukemia (ALL).
The ratio of incidence of ALL to AML varies according to age. AL represents a third of all cancers diagnosed in children aged 0–14 years, with ALL occurring five-times more frequently than AML [2–5]. This pattern is reversed in adults and in the elderly where AML accounts for approximately 80% of cases [6].
Treatment of AL is a complex process that aims at curing the disease and involves intensive chemotherapy protocols administered in two phases, known as induction and postremission therapy. Remission induction in AML is typically achieved using a chemotherapy regimen that includes an anthracycline, such as daunorubicin, administered for 3 days in combination with cytarabine for 7 days [7]. In acute promyelocytic leukemia (APL), a subtype of AML with t(15;17)(q22;q12), specific therapy in the form of all-trans retinoic acid (ATRA) or arsenic trioxide (ATO) are added to the conventional anthracycline-containing regimens [8,9].
Induction therapy in childhood ALL consists of weekly vincristine for 3–4 weeks and L-aspar-aginase for 6–12 doses, as well as daily corticosteroids. An anthracycline is added in high-risk patients. Treatment protocols for adult ALL are developed from childhood ALL regimens and consist of at least one glucocorticoid, vincristine and an anthracycline [10].
An integral part of ALL treatment is CNS prophylaxis. This is traditionally accomplished using a combination of intrathecal chemotherapy and craniospinal irradiation (CSI). Some studies have reported that this combination carries a higher risk of brain toxicities, especially affecting females, in addition to the increased risk for endocrinopathy and delayed fertility; this finding has prompted many centers to assume a more risk-adapted approach in an attempt to minimize or omit CSI [11–14].
The primary aim of postremission therapy of AL (i.e., consolidation and intensification) is to eradicate clinically undetectable diseases, otherwise known as minimal residual disease. Options for postremission therapy include additional courses of chemotherapy (consolidation or maintenance chemotherapy) or hematopoietic stem cell transplantation (HSCT). The choice depends upon the risk of relapse as determined by the presence of unfavorable tumor characteristics (e.g., unfavorable karytopes), as well as the expected morbidity and mortality as determined by age and the presence of comorbidities. HSCT and intensive chemotherapy are also options in the salvage of relapsed disease.
Survival in acute leukemia
Historically, survival rates in children with AL have markedly improved as a result of advances in treatment over the past 3 decades. In childhood ALL, 5-and 10-year survival rates were 88 and 84%, respectively, for those diagnosed from 2000 to 2004, compared with a survival of below 5% in the early 1960s [15,16]. Although the survival of children with AML has also improved, the 5-year survival rates have only increased by 40% [15,16]. By contrast, the 5-year survival rates for adults with ALL and AML do not presently exceed 40% [17,18].
Despite this success in the treatment of childhood leukemia, survivors of this disease face many health and psychosocial challenges during their path to adulthood, owing to the possible long-term consequences of the curative therapy. Survival rates in women of all age groups are similar to those of men, as represented in Figure 1.
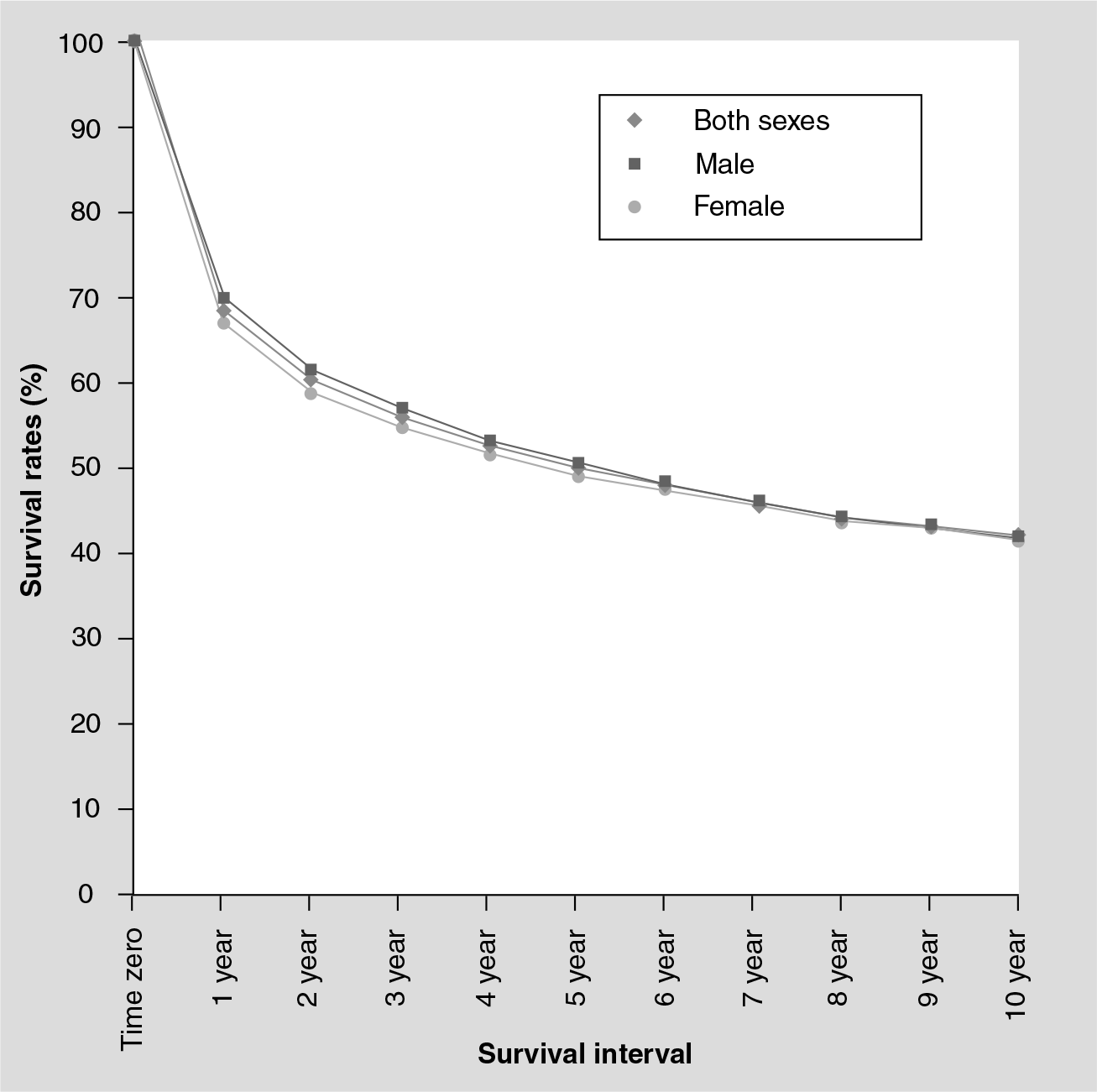
Survival rates for leukemia among women and men are similar, regardless of age group or race, based on SEER data from 1988-2005.
Special issues related to the treatment of acute leukemia in women
The National Cancer Institute's Surveillance, Epidemiology and End Results (SEER) review estimated that there would be 8300 women newly diagnosed with AL in the USA during 2009 [19]. When diagnosed in women, AL poses several unique challenges in medical practice.
Prevention & treatment of abnormal uterine bleeding during chemotherapy
In premenopausal women receiving chemotherapy for the treatment of AL, special precautions should be taken in order to avoid abnormal uterine bleeding (mostly menorrhagia). This is particularly important in the early phases of treatment, as well as during conditioning regimens for myeloablative transplants when treatment protocols are more intensive.
Menorrhagia may result from thrombocytopenia induced by chemotherapy or as a manifestation of the disease process, as in the case of APL where there is a higher risk of disseminated intravascular coagulation with a life-threatening hemorrhagic diathesis. In this specific AL entity, supportive measures to counteract the coagulopathy should be instituted immediately after a diagnosis of APL is suspected and should consist of plasma, fibrinogen and platelet transfusions to maintain the fibrinogen concentration and platelet count above 100–150 mg/dl and 30–50 × 109/l, respectively.
Gonadotropin-releasing hormone (GnRH) agonists are recommended for preventive use before initiating chemotherapy [20]. Other preventive options include progestinonly pills (e.g., norethindrone acetate [21]) or the continuous administration of estrogen–progestin pills, with the added benefit of contraception when continued postchemotherapy [22–24].
Treatment of acute leukemia during pregnancy
Acute leukemia diagnosed during pregnancy is uncommon. The treatment approach is challenging and largely based on the trimester of pregnancy during which the disease is diagnosed. Decision-making should involve a hematologist, obstetrician and a neonatologist, in addition to the patient [25,26].
Pregnant women who are diagnosed with AL during their first trimester and who received anthracycline-containing regimens were reported to have rates of fetal death, malformation or spontaneous abortion of 4, 11 and 7%, respectively [27]. Therefore, the immediate termination of pregnancy is advisable in a hemodynamically stable patient, followed by conventional treatment of AL. Women who choose to continue pregnancy should receive conventional chemotherapy and be closely monitored.
For those diagnosed with AL during their second or third trimester, choice of treatment is controversial. Some reports provide evidence for the safe administration of full doses of chemotherapy [27–30] without an increased risk for either fetal abnormalities or the subsequent occurrence of leukemia during childhood. In one report, successful delivery was reported in 73% of 160 pregnant patients diagnosed with different types of cancer and treated with anthracyclines [27]. In conclusion, it appears that women in their second or third trimester can receive conventional chemotherapy doses but must be informed about the risks of fetal death, malformation and low birth rate [27]. When AL is indicated, induction of labor should be considered inbetween cycles and when the patient is hemodynamically stable.
For the various AL subtypes that may be diagnosed during pregnancy, additional precautions should be considered for related clinical manifestations and specific drugs. In APL, ATRA is contraindicated during the first trimester, and ATO is contraindicated throughout the entire period of pregnancy [17]. Women with APL who are pregnant in their first trimester and who choose to continue their pregnancy can receive chemotherapy alone. However, they should be informed that they are still at a high risk for chemotherapy-induced undesirable fetal outcomes. Bleeding tendency is a manifestation of the disease process in APL and is even less controlled when ATRA cannot be administered during the first trimester. Once remission is achieved in an otherwise normal pregnancy, ATRA can be administered during the second or third trimester [17]. Daunorubicin is the anthracyclin of choice in these patients, since it is less lipophilic and less transferable through the placenta than idarubicin and, therefore, theoretically less toxic to the fetus [31].
Patients who present with APL during their second and third trimesters can receive either chemotherapy and ATRA or ATRA alone and postpone chemotherapy until after delivery, an approach that is thought to be safer for both the mother and the fetus [31]. After delivery, breastfeeding is contraindicated during treatment with chemotherapy or ATO [17].
Long-term effects of exposure to chemotherapy in utero
Another area of major concern in patients with AL who are treated with chemotherapy during pregnancy is the long-term risk to the fetus. Information on the long-term effects of in utero exposure to chemotherapy is mainly obtained from retrospective series or case reports [30,32–38]. Although the available information is considered to be limited, it has been useful in suggesting a link between the risks of exposure to chemotherapy and the trimester of pregnancy, as well as the type of chemotherapy [30,32–38].
The largest reported series describing the long-term effects of chemotherapy exposure in utero is from pregnant women treated with chemotherapy for hematologic malignancies [33]. Women participating in this study were followed-up for a median of 18.7 years (range: 6–29 years). No cases of cancer or AL were observed in the cohort of 84 patients or in the 12 second-generation children who were also found to have normal cognitive performance and no congenital, neurological or psychological abnormalities [33,34,36].
Despite these encouraging findings, many comprehensive studies failed to provide any solid data to support a uniform agreement on guidelines for the management of pregnant cancer patients, especially when it comes to newer drugs [30,32,33,36–38].
Special issues in women that are survivors of acute leukemia
Reproductive function & puberty development in leukemia survivors
In survivors of AL, reproductive function and sexual activity are affected by age and type of therapy [39]. Reports on the impairment of reproductive function and fertility in females treated for childhood leukemia raised interest regarding its mechanism and possible approaches to minimize the risk. This impairment could be in the form of acute ovarian failure or premature ovarian failure (POF), which is also termed premature menopause. The loss of ovarian function that occurs during or shortly after the completion of cancer therapy is termed acute ovarian failure. By contrast, the loss of ovarian function that occurs years after completion of treatment and after a period of functioning gonads is termed POF or premature menopause. Consequently, women who survived childhood AL may fail to enter puberty and develop endocrinopathy, or have an acute temporary ovarian failure or enter a premature menopause [39].
Ovarian failure
Young women treated for AL have been reported to have increased levels of FSH as a surrogate to chemotherapy-induced ovarian damage (caused by alkylating agents), even though the ovaries of prepubertal children and adolescents are known to be less sensitive to chemotherapy when compared with adults [40]. Fortunately, the majority of young women have reversible damage and normalization of FSH levels is still expected to occur even after many years following completion of therapy. By contrast, females who need to receive allogeneic or autologous stem cell transplantation are at a very high risk for developing acute ovarian failure with myeloablative doses of chemotherapy and especially with agents such as busulfan, melphalan and thiotepa [41,42]. This finding, which has been confirmed in several independent studies, highlights the need for long-term endocrine follow-up in these patients [40–43].
Pubertal delay
Delayed puberty is defined as the absence or incomplete development of secondary sexual characteristics by the age at which 95% of children of that gender and culture have initiated sexual maturation. Pubertal interruption can occur as a result of POF or, indirectly, through hypothalamic–pituitary injury and gonadotropin deficiency induced by high doses of cranial radiation. Delayed onset or progression of puberty has been described during the first year of AL therapy [44]. Puberty changes include breast development, pubic hair growth and distribution and vaginal estrogenization; checks for these changes should be made every 6 months. Absent signs of expected pubertal growth from 1 year after the completion of cancer therapy, or by 13 years of age in girls, warrants the consultation of an endocrinologist [45–47]. Estrogen-replacement therapy may be required and should be considered in order to accelerate growth and induce sexual development [42,48].
Sexual activity
Sexual dysfunction is an area that is commonly overlooked in the management of AL and is a condition that women are more susceptible to than men. Patients receiving chemotherapy are known to exhibit a decline in sexual desire and sexual activity [49]. Therapy for AL involves a lengthy and intensive process that is associated with fatigue and probably has a significant psychological impact. Altered body image as a result of therapy-induced alopecia, weight loss and disease-related complications all affect the woman's self-perception of desirability and beauty. As a result, many women avoid sexual activity altogether, which in turn may have additional psychosocial implications.
One prospective study with case-matched controls evaluated sexual function for 5 years after myeloablative allogeneic HSCT [50]. Both male and female survivors were found to be lower than controls in terms of their rates of sexual activity and sexual function at 5-years post-therapy. Men improved from their 6-month nadir in sexual function by 2-years post-therapy (p = 0.02), whereas women did not improve by 5-years post-therapy (p = 0.17). At 5-years post-therapy, more women reported residual sexual problems (80% of female survivors vs 61% of controls; p = 0.11) compared with men (46% of male survivors vs 21% of controls; p = 0.05). Reasons for lack of sexual activity included no partner, lack of libido, fatigue, sexual problems and the partner not being interested [50].
Fertility problems & pregnancy outcome
A significant drop in female fertility following chemotherapy and CSI as a treatment for childhood leukemia has been demonstrated in different studies [51,52].
In a retrospective cohort study, the detrimental effect of CSI on fertility rates has been documented in women treated for childhood ALL [51]. CSI at any dose was significantly associated with lowered fertility rates; however, the timing of CSI was found to be crucial. CSI within 2 years before or after the onset of menarche resulted in fertility reduction to only approximately a quarter of that of controls. It is worth noting that female survivors treated after menarche had their first pregnancy at an older age, possibly owing to delayed resumption of fertility after the initial decline [51].
In another population-based prospective cohort study of fertility and offspring of female leukemia survivors, a nearly normal reproductive function was observed, except in patients who had received prophylactic CNS radiotherapy, who were found to have a lower firstbirth rate [52]. Fortunately, there was neither an increase in the incidence of birth defects nor in the incidence of childhood malignancies compared with the general population in both studies [51,52].
Although different studies provide evidence for possible structural damage to the ovaries, changes in gonadotropin levels and a delay in menarche and fertility in survivors of childhood leukemia [40,53–59], the recovery and resumption of normal reproductive function is expected in the majority of patients [60]. Moreover, in some centers, cranial radiotherapy has been replaced with extended intrathecal chemotherapy in most CNS prophylaxis protocols – a modification that allows for fertility rates similar to those expected in the general population [14,61]. Promising methods in female fertility preservation include the cryopreservation of embryos and oocyte or ovarian tissue. However, the future role of these methods is yet to be defined [62–64].
Cognitive functions
Decline in cognitive functioning is a concerning and common late effect following AL. This is particularly common among children with ALL younger than 6 years of age who are treated with high-dose cranial radiotherapy (>24 Gy) and/or intrathecal chemotherapy [65,66]. Studies have demonstrated variable and conflicting results regarding the effect of childhood leukemia treatment protocols on CNS development in combination with different treatments; these studies have suggested that CNS development may be affected by the treatment protocol used and by gender [11–13,65–67].
Although a decline in cognitive functioning has been observed in leukemia patients treated with chemotherapy and cranial irradiation [66], it has also been observed in females treated at a young age with chemotherapy alone [65]. Moreover, this decline was found to be more significant in females treated at younger ages compared with females treated at older ages [65].
In one study of 150 ALL survivors followed for up to 10 years and evaluated for cognitive functioning, 83% had minimal or no cognitive dysfunction, 14% had a mild form and 3% had a severe form of cognitive dysfunction. However, in this study, gender has not been found to have a statistically significant impact (although females scored slightly poorer than males in verbal IQ) [11].
By contrast, another study demonstrated similar neurophysiologic performance for children with leukemia who were treated only with intrathecal and systemic chemotherapy compared with control siblings (except for poorer fine-motor functioning in children with leukemia) at a mean follow-up of 4.5 years (range: 4–12.3 years). Interestingly, in this study, sex was a significant confounding variable, with girls scoring higher in neurophysiologic performance compared with boys [67].
As a result, current ALL treatment protocols support the use of a risk-adapted approach, limiting cranial irradiation to high-risk patients and patients who relapse, in order to minimize the incidence and severity of cognitive impairment [11–13,67]. In turn, this led to a reduction, but not the disappearance, of late neuropsychological sequelae. Moreover, known risk factors, such as young age at ALL diagnosis and female gender, were reproducible in chemotherapyonly treated patients [12]. Therefore, long-term follow-up and screening for neurocognitive deficits is necessary for ALL survivors in order to improve their long-term outcomes.
Secondary neoplasm & breast cancer as a long-term complication of treatment for acute leukemia
As described in various studies, breast cancer as a secondary malignancy only occurs infrequently in female survivors of childhood leukemia [68,69]. Moreover, with modern treatment protocols, even fewer cases of secondary cancers are observed in survivors of childhood leukemia [68,69]. In a large cohort of 8831 children diagnosed with ALL, the cumulative incidence of a secondary solid nonhematopoietic malignancy was 0.82% (95% CI: 0.5–1.1%) at 10-years and 1.55% (95% CI: 0.9-2.2%) at 15-years post-diagnosis. The median time to the development of secondary solid malignancies was 7.1 years (range: 1.1–15.8 years). The most frequent secondary tumor after leukemia was brain cancer, while breast cancer was the least common. Female sex, radiation therapy and disease relapse were independent factors for an increased risk for a secondary solid malignancy. This study also suggested a trend towards a lower incidence of secondary neoplasms (mainly hematopoietic ones, owing to a short follow-up duration) among children diagnosed with ALL after 1983 [68].
The results from Bhatia et al. were confirmed in another population-based study with more precise data from 13 well-established cancer registries [69]. In this study, the risk of secondary cancer after childhood leukemia was assessed in a total of 12,731 patients with leukemia with an average follow-up of 6.5 years. Brain tumors were again the most common secondary malignancies and breast cancer was the least common [69].
Therapy-related myelodysplasia & acute myeloid leukemia Breast cancer survivors & therapy-related myelodysplasia & acute myeloid leukemia
Breast cancer is the most commonly diagnosed cancer in women, accounting for 26% of new cancer cases [70]. In recent years, progress in early diagnosis and management has contributed to a remarkable improvement in disease outcomes, with the 5-year survival rate now approaching 90% [70]. However, survivors of breast cancer are at an increased for risk of therapy-related myelodysplasia and AML (t-MDS/t-AML) [71–73].
The 2008 WHO classification system recognized t-AML, t-MDS and t-MDS myeloproliferative neoplasms as therapy-related myeloid neoplasms (t-MNs) [74]. Patients with t-MNs are known to be more resistant to chemotherapy and have a shorter median survival time compared with patients with de novo AML, MDS or MDS/myeloproliferative neoplasm [73].
Breast cancer is by far the most common primary solid tumor reported in patients with t-MN [75,76]. Breast cancer patients treated with adjuvant therapy are at a twofold increased risk of t-AML/t-MDS compared with controls. Alkylating agents and topoisomerase II inhibitors have well-established leukemogenic effects. Radiotherapy alone or in combination with chemotherapy, as well as granulocyte colony-stimulating factor, have all been linked to the risk of developing t-AML/t-MDS [73,77–86].
A long latency period of 5–7 years after exposure to alkylating agents is expected before the development of t-MNs [87–90]. Two-thirds of these patients will first develop myelodysplasia with complex karyotype abnormalities and monosomies (-5 or −7), which are characteristics associated with an unfavorable prognosis. By contrast, patients exposed to topoisomerase II inhibitors have a shorter latency period of 1–3 years before developing t-AML, and they rarely pass through a phase of MDS. These patients typically have favorable and intermediate cytogenetic characteristics, such as t(8;21) or t(15;17) and t(9;11), respectively [91–97].
By contrast, the incidence of therapy-related APL (t-APL) is emerging as one of the most frequent secondary cancers that can develop after the treatment of breast cancer [75,76,98,99] – a phenomenon that is thought to be related to the increased use of topoisomerase II inhibitors in breast cancer treatment [75,98,99]. Recent laboratory studies have demonstrated that mitoxantrone (a toposiomerase II inhibitor) results in the formation of the t(15;17) chromosome translocation that is characteristic of APL and results from the fusion of the retinoic acid receptor a gene on chromosome 17 with the promyelocytic leukemia gene on chromosome 15 [100]. Moreover, DNA breakpoints induced by this drug have been discovered to be tightly clustered in a small DNA region within the promyelocytic leukemia gene (termed the ‘hotspot’) [100,101].
Ovarian cancer survivors & therapy-related myelodysplasia & acute myeloid leukemia
Ovarian cancer is the second most common gynecologic malignancy [102]. Improvement in survival has been achieved over the past 2 decades with changes in the standard of care in newly diagnosed ovarian cancer patients [103–107].
Alkylating agents, in particular platinum compounds (cisplatin and carboplatin), are the cornerstone of treatment in ovarian cancer [108,109]. The occurrence of t-MDS/t-AML as a late sequale of ovarian cancer treatment has been increasingly reported and has been linked to alkylating agents [62,107,110–114]. The reported incidence of secondary leukemia in ovarian cancer patients treated with different alkylating agents was 0.24-1.15%, with a relative risk (RR) of 12–67 [115–118].
A large case-control study of more than 28,0 women with invasive ovarian cancer demonstrated a significant association between the use of alkylating agents (mainly melphalan and platinum compounds) and the occurrence of secondary leukemia within an average of 4-years postdiagnosis. The calculated RR of secondary leukemia was 6.5 and 8.1 with platinum-based therapy alone and in combination with radiation therapy, respectively. The study also demonstrated a significant association between the cumulative dose of cisplatin and the risk of secondary leukemia. The RR was 1.9, 2.1, 4.1 and 7.6 with cumulative doses of less than 500 mg, 500–749 mg, 750–999 mg and 1000 mg or greater, respectively (p < 0.001). t-APL was also reported in women treated for ovarian cancer. In two different studies, tumors of the female reproductive system, including ovarian cancer, comprised 71 and 85% of primary solid tumors of females diagnosed with t-APL [76,110].
The heterogenicity of treatment protocols used in older studies, and the fact that most of these regimens consisted of agents that are not used in current practice, makes these data difficult to utilize when making conclusions concerning the actual risk of t-MDS/t-AML in ovarian cancer patients. Secondary leukemia after ovarian cancer treatment with current standard protocols continues to be reported [119–121]. However, only a few case reports exist regarding secondary leukemia occurring after the use of current standard protocols (carboplatin and paclitaxel) [119,122–124]; therefore, larger studies and longer follow-up of patients are both warranted.
Management of t-AML is based on risk stratification by karyotype. Patients with favorable karyotypes are offered standard induction therapy followed by high-dose cytarabine consolidation rather than allogeneic hematopoietic cell transplantation, which should be considered for patients with intermediate karyotypes who have a human leukocyte antigen-matched donor. Unfavorable karyotypes do poorly with all available treatment options and are candidates for clinical trials and allogeneic HSCT, while t-APL carries a good prognosis and is treated in a similar fashion as de novo APL, with a conventional anthracycline-containing chemotherapy regimen combined with ATRA. However, taking into account cardiac toxicity and prior anthracycline exposure, some drug or dose modifications may be necessary in these patients, particularly in the postinduction phase [17,76,98,113,125–130].
Conclusion
A considerable amount of data is available regarding the effects of therapy on the health of women with AL, such as premature menopause and its consequences, delayed fertility and secondary malignancies. However, a better understanding of the mechanisms behind these issues, as well as the best preventive and therapeutic approaches, has yet to be established. Furthermore, there is a clear need for the development of a central registry specifically responsible for the long-term follow-up of AL survivors and their children and, in particular, those at higher risk owing to chemotherapy exposure in utero.
Therapy-induced AML is an often fatal long-term complication in some women who have received therapy for breast cancer. The underlying molecular mechanisms, as well as the interaction between therapy and genetic predisposition and the choice of the best targeted therapeutic approach, are all areas of ongoing research.
Future perspective
Risk-adapted therapy for the treatment of leukemia is evolving rapidly and personalized treatment will be possible in the coming years. Improvements in genomic technology have made understanding the underlying molecular mechanisms of leukemogensis possible, as well as enabling predictions regarding responses to treatment to be made. Moreover, defining markers in order to monitor the disease, which occur in a very low levels within leukemic cells (minimal residual disease), is an extensive area of research that will change the management of leukemia in the near future. Hopefully, with risk-adapted therapy and the availability of more successful targeted therapy, optimum individualized treatment will be achievable.
Executive summary
The treatment and survival outcome of acute leukemia (AL) in women is generally similar to that of men. However, women face certain unique challenges.
Special precautions should be taken in premenopausal women receiving chemotherapy for the treatment of AL in order to avoid abnormal uterine bleeding.
The treatment approach for AL diagnosed during pregnancy is challenging and largely based on the trimester of pregnancy during which the disease is diagnosed.
Reproductive functioning, sexual activity and cognitive functioning are affected by age at AL diagnosis and the type of therapy that is employed.
With modern treatment protocols, fewer cases of secondary cancers – in particular breast cancer – are seen in survivors of childhood leukemia.
Breast cancer is by far the most common primary solid tumor reported in patients with therapy-related myeloid neoplasms. Therapy-related acute myeloid leukemia has also been reported in ovarian cancer patients treated with alkylating agents.
There is a clear need for the development of a central registry specifically responsible for the long-term follow-up of AL, breast and ovarian cancer survivors.
A better understanding of the underlying molecular mechanisms and novel targeted approaches to therapy are areas of ongoing research in AL.
Footnotes
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
