Abstract
Addressing the sexual sequelae of chronic disease and its treatment is now accepted as a fundamental part of healthcare. Most of the sexual effects of chronic disease are negative, and ongoing illness continues to modulate a woman's sexual self-image, energy and interest in sexual activity, as well as her ability to respond to sexual stimuli with pleasurable sensations, excitement, orgasm and freedom from pain with genital stimulation or intercourse. Nevertheless, for many women with chronic illness, sexuality remains extremely important despite the commonly associated fatigue and acquired sexual dysfunctions; sexual resilience can be substantial. Following recovery from cancer surgery, chemotherapy and radiation, prognosis can be excellent and a return to full health can often be expected, and yet, there may have been devastating changes to sexual function owing to the cancer treatment. Women with metastatic disease may still treasure sexual intimacy. Assessment and management of sexual dysfunction is therefore necessary in all women with chronic illness or past or present cancer.
Chronic medical illness can interfere with sexual function directly and indirectly. Disease-related interruption of the neurovascular pathways and hormonal status may not be the major determinant of women's sexual satisfaction: depression is the major factor influencing sexual function in women with a chronic illness including end-stage renal disease (ESRD) [1], multiple sclerosis [2], diabetes [3] and after bone marrow transplantation [4]. The direct and indirect effects of chronic disease on women's sexual function are shown in Table 1. Table 2 outlines the direct and indirect effects of cancer on sexual function.
Factors involved in sexual dysfunction associated with chronic disease.
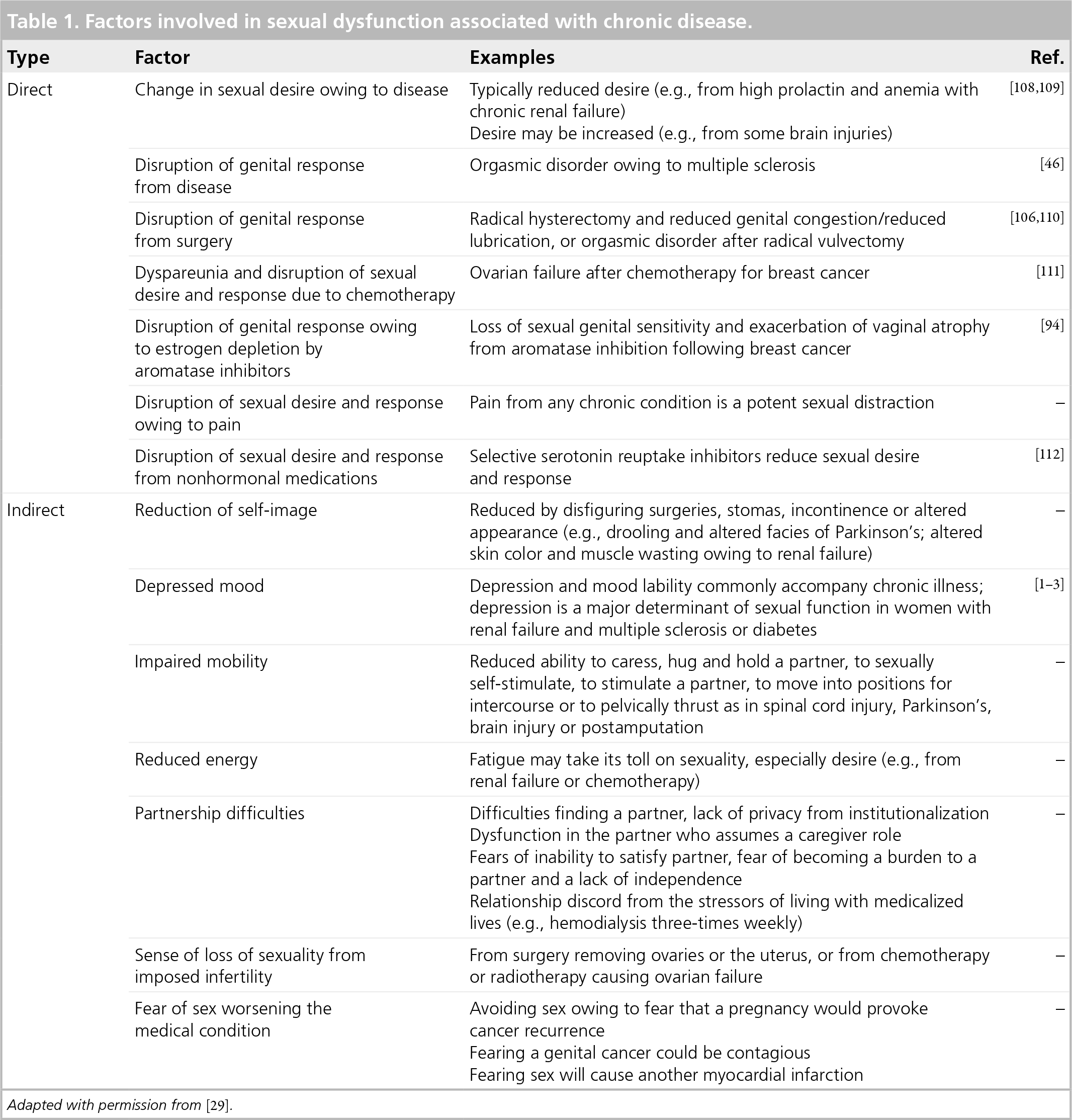
Adapted with permission from [29].
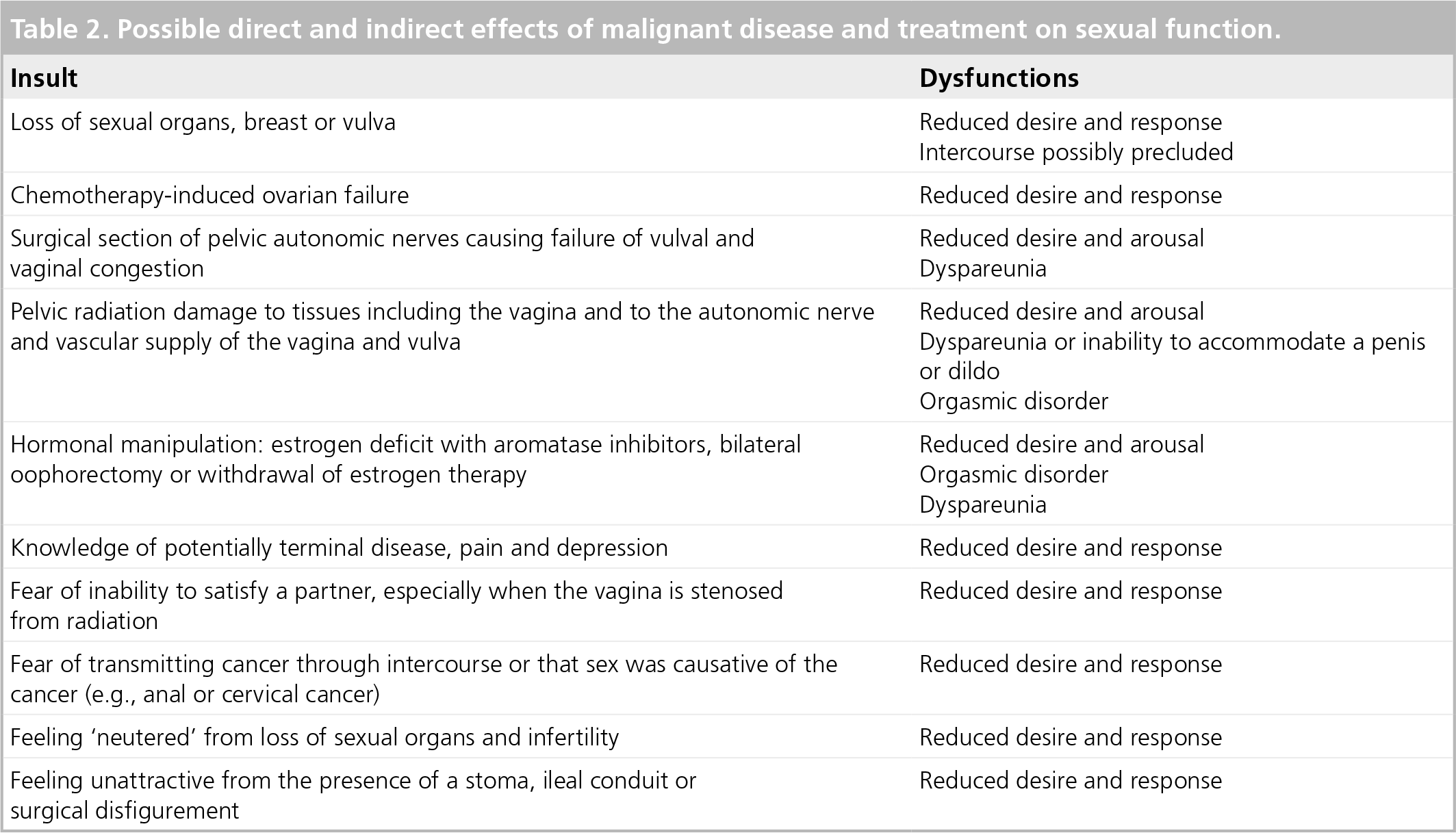
Possible direct and indirect effects of malignant disease and treatment on sexual function.
Prevalence of the new onset of sexual dysfunction in women with chronic disease or cancer
Unfortunately, studies regarding sexual dysfunction in women with chronic disease or cancer are limited. Assessment is usually via validated questionnaires focusing on the traditional components of sexual response (e.g., sexual fantasies, sexual thoughts, anticipatory desire of sexual activity with a partner, arousal – often equated to genital lubrication – and orgasm). These aspects of the sexual experience are not necessarily the most important for women, as will hopefully be recognized in the 2012 revisions of the American Psychiatric Association's Diagnostic and Statistical Manual 5th revision (DSM-V) definitions of women's sexual disorders [5,6], which will lead to the development of different validated questionnaires. While some surveys include the item of sexual satisfaction, important aspects of sexuality that emerge in chronic illness, and have been detected by qualitative research, are generally not assessed. These include a woman's altered sense of self, the need to adapt to and accept changes in her body and the realization that part of sexual intimacy involves meeting the needs of others [7]. A further factor is the need for a woman to be able to communicate her changed sexual needs and the degree to which she can meet the needs of her partner. Moreover, in a recent review of sexuality at the end of life, the authors were unable to find any articles addressing the types of sexual intimacies that patients desire or experience in their last few weeks of life, nor how such intimacies are encouraged or facilitated by healthcare providers, nor any publications regarding patient or partner's thoughts concerning sexuality at the end of their lives [8]. Acknowledging the above caveats, some conclusions can be made. While some 30% of the general female population report recently acquired sexual difficulties and approximately 10% report long-lasting problems that cause them distress [9], in some medical conditions (e.g., in ESRD), sexual dysfunction seems almost inevitable, given that the prevalence of low desire may reach 100% [10]. However, in other chronic diseases, including diabetes, some but not all studies have found little increase in sexual dysfunction compared with controls [11]. Studies commenting on the prevalence of sexual dysfunction within individual medical disorders will be outlined in later sections of this review. The disorders and cancers chosen for discussion are those that are both common and for which there are, albeit frequently few, data regarding associated sexual dysfunction.
Overview of assessment of women's sexual function
Understanding the evidence-based conceptualization of women's sexual responses in health allows clinicians to understand the changes brought about by chronic disease. The current model of sexual response is circular and depicts the phases of sexual response as overlapping and of a variable order. This is in keeping with data confirming that the majority of women in long-term relationships begin sexual experiences without (at that moment) sensing any sexual desire [5]. This is also in accordance with the known common association between disorders of desire, arousal, lubrication and orgasm [5].
The model demonstrated in Figure 1 indicates that many factors motivate women to initiate or agree to sexual activity – some 235 distinct reasons were recently identified [12]. Sexual stimulation is requried to move from this state of sexual neutrality and for the individual woman, this may need to be verbal, auditory, visual or physical. Nongenital and genital nonpenetrative stimuli are usually required but it is not only the stimuli themselves that is important but the context, interpersonal and environmental, is crucial. Simple environmental concerns such as privacy and not being too late at night when the woman is tired, as well as freedom from sexually transmitted diseases and freedom from unintended pregnancies may all be relevant. Empirical studies have shown the greater role of internal daily stress regarding the relationship, rather than external stresses from work, family or finances, in influencing a woman's sexual response and particularly her sexual desire [13]. The ability of these stimuli to arouse the woman will depend on their appraisal in her mind. A number of psychological factors can frequently interfere, including distractions, an inability to focus, fear of being sexually substandard, fear of a negative outcome such as pain and fear of not being able to please her partner, as well as some biological factors – importantly fatigue, depression, pain and medications. Any negative outcomes, such as genital pain from a lack of estrogen or pelvic radiation or gynecological surgery, will lessen the motivation for sex in the future. Similarly, emotionally negative outcomes will impede further motivation. Many women begin a sexual experience mainly to increase emotional intimacy, but if this does not occur during and after the sexual experience together, their motivation lessens.

Human sex response cycle.
FIGURE 2 illustrates changes to the response cycle when a healthy adaptation to chronic illness occurs. The woman's reasons for sex continue and her need of intimacy may increase. Innate desire boosting these motivations may well be absent. Provided that she is not depressed and pain is well controlled, she remains willing to attend to sexual stimulation that might arouse her. Sexual experiences may need to be planned and different types of stimuli used.

Healthy adaptation of sexual response to chronic illness.
However, healthy adaptation may not occur. Figure 3 shows the possibility of illness distancing the partners, the role of the caregiver reducing the eroticism of their interaction and thus lessening the woman's willingness to receive sexual stimulation. The latter may no longer be effective owing, in part, to nerve damage or distractions of pain or a lowered self-image. Minimal arousal follows and no desire for more intense sexual stimulation is triggered. Ultimately, there may be pain from a lack of physiological congestion as well as any tissue damage.

Sexual dysfunction resulting from chronic illness.
Details of assessment of the women complaining of sexual problems within the context of chronic illness: the interview
In order to fully assess a woman's sexual function and the couple's degree of adaptation to chronic illness, whenever possible, both partners are interviewed separately and together. The clinician conducting the interview may or may not be the one providing medical care for the chronic illness – he or she may be part of an interdisciplinary team. However, the treating physician or oncologist must screen for sexual dysfunction in order to make the appropriate referral to the team member. The assessment is comprehensive and yet respectful (Box 1). It involves addressing the woman's motivation to be sexual, the adequacy of the sexual stimulation and the environment, her ability to stay focused, her sexual self-view, her mood and her freedom from medications that are known to negatively affect sexual function. Her subjective arousal, ability to trigger desire to continue the experience for reasons of sexual pleasure (as well as her original reasons for becoming sexually engaged), genital sensations and orgasms are addressed. The partner's sexual response is also clarified. The sexual outcome for both partners is assessed in terms of enjoyment, satisfaction and freedom from pain. It is necessary to assess the partners' sexual lives before the onset of disease and how each partner has reacted to the changes. Sexual dysfunction associated with illness may be compounding longer-standing sexual difficulties. Therefore, it is often necessary to assess the woman's developmental history, focusing on her relationships with her parents or caregivers, losses, abuse or neglect, as well as themes regarding sexuality in her childhood home(s). Difficulties with trust, abandonment, control or expressing emotions may all have effects on subsequent sexuality. B
Details of assessment: the physical examination
Box 5 outlines the necessary components of physical examination – the general examination, the focused examination and the neurological genital examination, as well as the examination required when dyspareunia is present. The physical examination can be therapeutic and should not be rushed. Information and reassurance can be given. It may be the first time the woman has viewed her genitalia since her surgery, radiation or chemotherapy; inviting her to look with a mirror may allow some acceptance of the changes and hope that she can again have pleasure from her, albeit altered, body.
Definitions of sexual dysfunctions
Evidence-based definitions of sexual disorders recommended by the DSM-V committee will be published in 2012 [5,6]. Interim revisions of existing definitions were published by an international consensus committee organized by the American Urological Association (AUA) Foundation in 2003 [14]. Major themes that are expected to occur in the 2012 definitions are the importance of subjective arousal, the overlap of desire and arousal, and the normality of sometimes or always only experiencing desire once the sexual experience has begun. In the interim, and for the purposes of diagnosing dysfunction in a woman with chronic illness or cancer, the flow chart shown in FIGURE 4 may be helpful. First, it should be determined whether the concerns reflect a disorder or normal changes associated with relationship duration – a desire discrepancy between partners can masquerade as a disorder in the individual with lower desire. The three most common disorders are loss of arousability leading to complaints of low desire, loss of subjective arousal and pleasure, inability to experience orgasm despite high arousal and painful sex – usually dyspareunia.

Overview of sexual difficulties of women with chronic illness and cancer.
Overview of therapy for sexual dysfunction in women
Legitimizing and clarifying the women's sexual concern is therapeutic in itself. The woman's own sexual response cycle is outlined and guides the therapy. FIGURE 5 demonstrates the main modalities of treatment. Standard therapies include cognitive–behavioral therapy, sex therapy and psychoeducation. Sex therapy includes sensate focus exercises, whereby each partner is encouraged to take turns giving and receiving sensual, and later sexual, touches, caresses and kisses. Initially, genital areas and breasts are off-limits. The idea of any goal or expectation is put aside for these ‘homework assignments’. Usually, each session lasts 15–20 min and two or preferably three sessions should occur each week for 3–6 weeks. The couple, together with the clinician, decide as to when the breasts and genital areas are included. Ultimately, the act of intercourse (vaginal penetration with a dildo) may be included. For some women with chronic disease or cancer, intercourse may not be possible and encouragement is given to expand the sexual menu for more erotic, varied and exciting nonpenetrative activities.
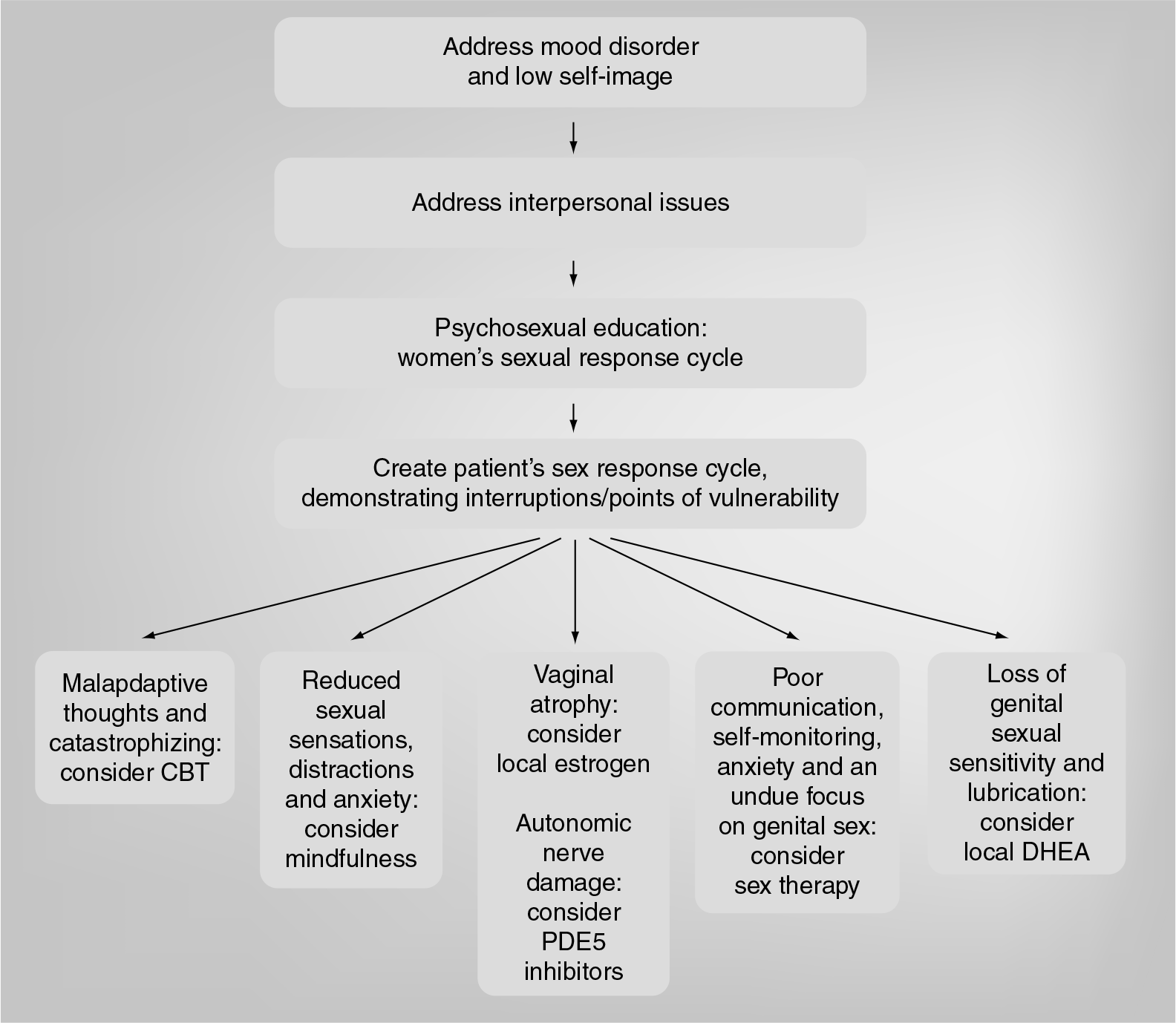
Overview of management of women's sexual dysfunction.
Assessment of sexual dysfunction associated with chronic illness.
Review past medical and psychiatric history
Review current medical status: consider respiratory, cardiac, mobility and continence requirements for sexual activity including intercourse, self-stimulation and orgasm
Review current medications
List the sexual dysfunctions, their duration and whether the dysfunction is also present with self-stimulation or with other partners
Clarify relationship status and quality
Review the environment for sex: home/institution/‘medicalization’ (e.g., hemodialysis machines, respirators and lack of independence in daily living)
Review any chronic pain
Assess current mood
Assess the consequences of illness on sexual self-image (concerns regarding attractiveness and physical appearance)
Review dysfunctions in detail: ask what the sexual difficulties are:
– Clarify the motivations for sex including ‘desire/drive’ and the desire to satisfy the partner
– Identify reasons for avoiding sex
– Clarify subjective arousal/excitement and pleasure
– Clarify genital congestion and lubrication
– Review the variety and usefulness of sexual stimuli and sexual context
– Assess the couple's sexual communication
– Enquire about distracting thoughts or negative emotions during sex
– Determine if wanted orgasms are possible, very delayed, nonintense or painful
– Review if intercourse is possible
– Assess dyspareunia: introital, depth, how constant, exacerbation from partner's ejaculation fluid, postcoital burning or postcoital dysuria
– Assess any sexual dysfunction of the partner
Clarify sexual response pre-illness: any dysfunction, how rewarding, how important was sex and any desire discrepancy between partners
Review the impact of medications on desire and response
Review the treatment for sexual dysfunction to date
Complete a full physical examination, including a genital examination; this is usually necessary because of the medical condition and is particularly important for neurological illness and whenever there is dyspareunia or pain with arousal
Laboratory investigations – especially necessary when needed to monitor anemia, high prolactin levels and thyroid replacement; estrogen levels are usually assessed by the history and the genital/pelvic examination
Adapted with permission from [29].
Questions used to assess the woman's sexual function.
Can you explain the sexual difficulties to me? Give some options (e.g., are there fewer physical sensations; is there less pleasure or little excitement and arousal; perhaps there is no interest or even too much desire; is the difficulty that you would like to experience orgasm but it is not possible; is there pain with sexual stimulation or pain with penetration?). Discuss intercourse or dildo dependant on orientation.
For how long have these problems been present? Are they constant or intermittent?
How was your sexual life before the illness?
How is your mood? (Formally screen for depression.)
Tell me about your current relationship (including any abuse) and about your partner's sexual function.
Briefly, can you tell me a little bit about your childhood? Was the home dysfunctional or functional? Would you describe your childhood as happy or not? Were there any major traumas or losses?
Any abuse?
What are your medical conditions right now and what are your current medications?
Seeing partners is usually very helpful. What would you think about my seeing your partner with you and on his/her own?
New to western medicine is the recent addition of mindfulness, which has already shown promise in the therapy of sexual dysfunction in medically healthy women [15] and in those with pelvic cancer [16,17]. Mindfulness is an eastern practice with roots in Buddhist meditation, which focuses on the present moment and non-judgmental awareness. Randomized waiting-list control studies are in progress.
Cognitive–behavioral therapy can assist the woman to change her often catastrophic selfview imposed by the illness. Sadly, some women view themselves as sexually substandard, feeling that they do not ‘deserve’ reasonable treatment in a relationship and will therefore even stay in abusive relationships. Other exaggerated or catastrophic thoughts that are amenable to cognitive therapy include: ‘sex is only for well women’, ‘no longer fertile, I am no longer sexually attractive’, as well as ‘if intercourse is impossible, then no one will want me’.
Medical therapies include local or systemic estrogen for dyspareunia and reduced lubrication from a lack of estrogen, and the occasional use of investigational phosphodiesterase type 5 inhibitors for a neurogenic genital congestion deficit. More recently, the investigational use of vaginal dehydroepiandrosterone (DHEA) has been trialed for postmenopausal vaginal atrophy. The off-label prescription of systemic testosterone for low desire is frequent among some clinicians treating women with and without chronic disease; however, this remains without sound scientific basis – randomized controlled trials recruited postmenopausal women who reported distressing low desire since their menopause and who, at baseline, experienced two to three sexually satisfying events per month on average. These sexually satisfying events increased to approximately five per month with the active drug and to four per month with placebo [18–21]. It is by no means certain whether these women reflect the patients seen clinically, whose lack of desire is associated with consistently unsatisfying sexual experiences. Sexual function does not correlate with any measures of testosterone activity to date (i.e., there is no evidence of an endocrine deficiency being related to sexual dysfunction). Nevertheless, intracrine production of testosterone and estrogen from adrenal precursors (the sole source of estrogen following menopause and the major source of testosterone) does markedly decline with age, but a recent study found no difference in total androgen activity (ovarian plus intracrine), as measured using androgen metabolites, between women with and without disorders of desire and arousal [22]. Research is urgently needed to clarify exactly which women might safely benefit from supplemental testosterone based on a hormonal deficit and sexual improvement with supplementation. Given the current dilemma regarding testosterone treatment to otherwise healthy women, any role for the same treatment in women with chronic illness is unclear.
Questions used to assess partner's sexual function
What is your take on the current difficulties you two are experiencing?
Please tell me about your own sexual function. Do you have any difficulties with desire or interest, pleasure or arousal (erections or ejaculations if male), orgasm or pain?
Have your past sexual experiences been positive or negative? Was there any abuse?
How would you assess your current relationship?
How is your health currently, including your mood? (Screen for depression as appropriate.) What are your current medications?
Questions for the assessment of sexual pain.
Where does it hurt? How would you describe the pain?
For heterosexual women: is the pain experienced with penile contact to the opening of your vagina, once the penis is partially in, with full entry, after some thrusting, after deep thrusting, whenever there is penile movement, with the partner's ejaculation or after withdrawal, and for how long or with subsequent micturition? Ask similar questions for a dildo or female partner's fingers.
Do you find your body is tensing when your partner is attempting, or you are attempting, to insert his penis? What are your thoughts and feelings at this time?
How long does the pain last? Does touching cause pain? Does it hurt when you ride your bicycle or when you wear tight clothes? Do other forms of penetration hurt (e.g., tampons or fingers)?
Pelvic floor muscle tension
– Do you recognise the feeling of pelvic floor muscle tension during sexual contact?
– Do you recognise the feeling of pelvic floor muscle tension in other (nonsexual) situations?
Do you feel subjectively excited when you attempt intercourse? Does your vagina become sufficiently moist?
What do you do when you experience pain during sexual contact? Do you still continue to include intercourse or attempts at intercourse, or do you use other ways to make love instead? If so, are you both clear that intercourse will not be attempted?
What consequences does the pain have on the rest of your sexual relationship?
What tests have been done/diagnoses given? What treatment have you received?
Do you have other pain syndromes, such as temporomandibular pain or irritable bowel syndrome?
Adapted with permission from [114].
When deterioration of sexual function coincides with beginning a medication, it may be possible to change the medication (Table 3). When this is not possible, the patient is advised that at least part of the problem is the required medication, and adjustments can be suggested. The context might be made more erotic, more intense sexual stimulation may be provided and specific goals can be removed; for example, that intercourse or orgasm must necessarily occur.
Medications associated with sexual dysfunction in women.

GnRH: Gonadotrophin-releasing hormone; SHBG: Sex hormone-binding globulin; SSRI: Selective serotonin reuptake inhibitor.
Individual medical disorders & cancer
Cardiovascular disease
Congestive cardiac failure
Data on the sexual function of women with heart failure are few. One recent study of 24 women with stable New York Heart Association (NYHA) classes I–III confirmed sexual dysfunction in a majority of the women (87%) and half of them reported that sexual problems predated the onset of the symptoms of heart failure [23]. A total of 40% also confirmed that sex remained an important part of their lives and their dysfunction reduced their quality of life. Enquiry from their physicians regarding possible sexual problems had been absent for most of the women.
Coronary artery disease
The link between coronary artery disease and genital sexual dysfunction (erectile dysfunction in men) has not been confirmed in women. However, most patients reduce the frequency of sexual activity after myocardial infarction owing to fear of further cardiac damage [24]. Patients can be advised that the risk is low and short-lasting and that cardiac vascular symptoms are very unlikely to occur if no symptoms arise during exercise testing to six metabolic equivalents [25]. Comorbid depression is common and is the most likely cause of low sexual desire and arousability.
Hypertension
The larger epidemiological studies have not confirmed an increase in sexual dysfunction in older women with hypertension [26]. One small study of premenopausal women with mild hypertension, irrespective of treatment, demonstrated more lubrication difficulties and fewer orgasms than age-matched controls [27]. Minimal study of optimal medication for women's sexual function has been conducted, but avoiding β-blockers would remove the theoretical chance of decreasing desire and avoiding thiazides would prevent reduction in genital blood supply. A randomized trial of the angiotensin II AT1-receptor antagonist valsartan and the selective β1-blocker atenolol in hypertensive postmenopausal women showed some increase in sexual desire in women receiving valsartan and some loss of desire in those receiving atenolol [28].
End-stage renal disease
Sexual dysfunction is extremely common in women with ESRD. There is a strong link between ESRD and comorbid depression [1], but also with other comorbid conditions that can interfere with sexual response, including diabetes, hypertension and coronary artery disease. Moreover, nonsexual symptoms can impede sexual activity and motivation, including pain from bones and joints owing to osteodystrophy, fatigue, anorexia, nausea, stomatitis, pruritis and malnutrition. Some studies confirmed an absence of sexual desire in up to 100% of women with ESRD and in those undergoing hemodialysis and possibly in as many as 80% post-transplantation [29]. Many factors are involved, including anemia and estrogen deficiency with vaginal atrophy and hyperprolactinemia, as well as chronic pain, psychosexual and interpersonal issues and repeatedly negative outcomes of sexual interaction. Some 40% of younger women with ESRD are totally amenorrheic – less than 10% have regular menstrual periods. Premature menopause is common. Young adolescent women have major difficulties forming sexual relationships – puberty may have been delayed and their lives are medicalized, limiting social interactions. Recombinant human erythropoietin therapy is guided by hemoglobin levels but may improve sexual function indirectly by increasing energy. There is no evidence that drugs that reduce prolactin also ameliorates sexual dysfunction.
Rheumatic disease
Pain, debility, reduced mobility and the changes to genital tissues found in some rheumatic disorders as well as comorbid depression can all reduce women's sexual responses. Interestingly, not all studies have confirmed an increased prevalence of sexual dysfunction in women with rheumatic disease: one study in which patients had different types of rheumatic disease involved long-term couples where one partner did or did not have rheumatic disease and these were compared; no differences in sexual satisfaction were found. However, the degree of functional impairment was demonstrated to be related to sexual dissatisfaction in both partners [30]. Specifically, the movement needed to participate in intercourse may be precluded or too painful, and sexual caressing and stimulation of self and partner may not be possible. Especially for young women, the altered mobility can impair their sexual self-image, causing hesitancy in starting a sexual relationship. Hip replacement in rheumatoid arthritis was found to improve sexual function to previous levels in 50% of cases [31]. Specifically in the context of rheumatoid arthritis, pain, age and depression have been demonstrated to be the important modulators of women's sexual function [32].
Physical examination.
The general examination can show difficulties with mobility or shortness of breath, the degree of tremor or muscle weakness limiting sexual activity and highlight concerns regarding hygiene, stomas and continence
The focused examination will depend on the patient's history (e.g., it may be necessary to examine painful scars or the focus may well be on the cardiovascular system when the ability and safety to engage sexually is in question)
A neurological genital examination may be necessary; for example, in multiple sclerosis or spinal cord injury. Sensory modalities can be examined using a light touch (Q-tip), cold (lubricant), vibration (tuning fork) and pain (broken Q-tip). The voluntary contraction of muscles around the vagina and around the anus may be tested. When clitoral sensation is absent and/or perivaginal/perianal muscle contractions are absent, orgasm is unlikely. For women with spinal cord injury, test the sensation in the T-10 to L-1 areas, as intact sensation here indicates the potential to have psychogenic lubrication
The genital examination can evaluate changes from radiation, surgery, chronic skin disease or hormonal deficit. Genital pain with sex necessitates a detailed examination; for example, for vulval disease involving introitus (e.g., pallor, friability, loss of elasticity and moisture of vulval atrophy, loss of elasticity in systemic sclerosis, graft-versus-host disease, incidental vulvar conditions such as lichen sclerosis, provoked vestibulodynia – test for allodynia of introital margin using moistened Q-tip). Inability to tighten and relax perivaginal muscles should be tested for and is often associated with hypertonicity of pelvic muscles and mid-vaginal dyspareunia. Abnormal vaginal discharge associated with burning dyspareunia will be examined
An internal examination of the following can evaluate the causes of deep dyspareunia: vaginal size and elasticity after radiation or surgery, pelvic muscle tone, pelvic floor prolapse associated with urinary incontinence and related sexual dysfunction, presence of tenderness, ‘trigger points’ on palpating deep levator ani owing to underlying hypertonicity, the presence of nodules and/or tenderness in the culdesac or vaginal fornix along uterosacral ligaments and retroverted fixed uterus; tenderness and palpating of the posterior bladder wall from the anterior vaginal wall suggests bladder pathology
In patients attending a pain clinic for chronic musculoskeletal pain, sexual dysfunction was found to be common and more frequently present in those with greater disability, those with depression and, interestingly, those with a shorter pain duration [33]. The authors suggested that the results support a ‘response shift’ model of adaptation to illness that is influenced by personality but can be further modulated by the use of various behavioral, cognitive and affective processes, including changing expectations and priorities. It is suggested that women who have lived with pain for a shorter period of time have had limited opportunities to make this accommodation.
Sjögren's syndrome can be associated with vaginal dryness, a loss of elasticity and dyspareunia [34]. Systemic sclerosis gives rise to similar symptoms and even ulcerations [35]. These symptoms are difficult to manage and when local lubricants, moisturizers, polymers that attract water (e.g., vaginal moisturizer) and, if necessary, local estrogen are insufficient, support and encouragement of nonpenetrative sexual activity is necessary.
Neurological disease
Neurological disease can interrupt the sexual response system at the level of the brain, spinal cord and all peripheral nerves. Moreover, it can alter sexual motivation, reduce sexual selfimage and desire and interfere with communication regarding sexual needs. Its presence can increase vulnerability to sexual exploitation or coercion. Usually, neurological disease leads to sexual deficit but women with Parkinson's disease may experience unwanted hypersexuality when given dopaminergic agonists. In addition, severe trauma to the prefrontal lobes or damage to the amygdyla in Klüver–Bucy syndrome can lead to a disinhibited hypersexuality. However, it is important to note that sometimes the neurological deficits are ultimately accepted and accommodated, such that women report more intimate sexuality compared with preillness; this has been shown after spinal cord injury [36].
Traumatic brain injury & cerebral vascular accidents
Depression is the most sensitive single predictor of sexual outcome after brain injury [37], and at least 50% of patients are diagnosed with depression following moderate and severe brain injury [38]. The prevalence of sexual dysfunction owing to traumatic brain injury itself, especially to the deeply located limbic structures or to their connections, is difficult to document given the usual extensive bodily injuries other than those of the brain. No clear relationship has been confirmed between the location of brain injury or duration of coma and the degree of sexual impairment. Most studies lack input from sexual partners, thereby probably lowering the true prevalence of dysfunction, especially when frontal lobe damage has occurred that can distort the sense of reality. Brain damage may also lead to impaired insight and cognition that interferes with social interaction; sexual relationships may be difficult to begin and maintain.
There can be considerable risks for women whose brain damage leads to hypersexuality given their increased risk of abuse, coercion and acquisition of sexually transmitted diseases and unwanted pregnancies.
Parkinson's disease
Sexual dysfunction is common in women with Parkinson's disease with various studies reporting its prevalence to be between 30 and 88% [39]. Difficulties with arousal, lubrication and orgasm, as well as impaired sexual satisfaction and desire, are reported. Despite the involvement of autonomic, limbic and somatomotor systems, it has been found that it is the attitudes regarding their sexuality and disease that influence women's sexual functioning more than biomedical factors [40]. Moreover, studies have suggested that healthy female partners of patients with Parkinson's disease are more sexually dissatisfied than women suffering with Parkinson's disease [41]. Reduced genital sensitivity is common in women diagnosed with multiple system atrophy [42]. The syndrome, recently coined as ‘hedonistic homeostatic dysregulation’, which signifies sexual exhibitionism, multiple sexual liaisons and excessive use of sex phone lines and sex shops, appears to be far more common in men than women, and it is attributed to therapy using dopamine agonists in conjunction with L-dopa. Other features include pathological gambling, excessive shopping and aggression [43]. Risk factors for this syndrome include a history of mood disorders, alcohol intake, personality traits of novelty seeking and an early onset of Parkinson's disease [44]. It is important to note that sexual compulsions such as these can be completely resolved by discontinuing the dopamine agonists while continuing L-dopa therapy [45].
Multiple sclerosis
Sexual dysfunction is common in women with multiple sclerosis. One study reported that even when symptoms had only been present for an average of 2.7 years, sexual dysfunction was present in 35% of women compared with 20% of healthy controls [46]. Eventually, some 62% of women complain of loss of genital sensation [47]. Contributing factors include fatigue, spasticity, pain and incontinence. Limited studies suggest that phosphodiesterase inhibitors might improve vaginal lubrication [48] but not other aspects of sexual function. Agents such as baclofen, tizanidine, and botulinum toxin or sclerosing agents may reduce the spasticity that can interrupt sexual function.
Spinal cord injury & cauda equina injury
Although sexual response may be markedly interrupted, sexual desire is typically retained. In cases of complete upper motor neuron damage, orgasms are usually lost. However, even when lesions are complete and at any level of the spinal cord, women may experience orgasm from cervical vibrostimulation. It is not clear how this is mediated, although there is possibly a neural supply to the cervix that travels separately in the vagus nerve [49]. When the injury involves segments below L2, the T10–L2 sympathetic outflow from the spinal cord is spared and this is thought to underlie the retained ability for vaginal congestion and lubrication when there are mental sexual stimuli. Lower lesions involving the S2, 3 and 4 nerve roots will preclude any reflex lubrication as well as any physical sexual sensations from genital stimulation.
Cauda equina injuries interrupt the innervation of the genitalia from the autonomic nerves, which have left the lower end of the spinal cord to ‘hitchhike’ to the S2, 3 and 4 nerve roots. In addition, they cause a complete loss of genital sensation together with a loss of voluntary control of the bladder and bowel. The distress from this can be immense – the women typically note how they ‘don't appear to look unwell or have any injury’ but have a truly devastating loss.
Neuroplasticity may underlie the changes that occur following injury whereby areas of the body, including the neck, shoulders and earlobes, can become highly sexually arousing when stimulated. This can occur when injuries have precluded any activity or sensation below the lesion level.
Sildenafil has been demonstrated to produce a minor increase in vaginal lubrication in spinal cord-injured women [50].
Sexual activity is not without risk for some women with spinal cord injury. Autonomic dysreflexia (acute hypertension from unopposed sympathetic nervous activity) can occur with orgasm in women with lesions above neurological level T6. More commonly reported in men [51], this dangerous consequence is managed with the prophylactic use of prazocin or captopril, and the acute situation is managed with calcium channel blockers.
Epilepsy
Epilepsy may be associated with hyposexuality, especially when the focus is in the temporal lobe. Less commonly, epilepsy provokes involuntary sexual gestures during the seizures. Moreover, a seizure can be preceded by an erotic aura. Of the antiseizure drugs, there is some evidence that lamotrigine has the lowest profile of negative sexual side effects [52]. However, most of the data are on men and are related to the lowering of bioavailable testosterone owing to increased serum sex hormone-binding globulin, brought about by older antiseizure drugs including phenytoin, barbiturates and carbamazepine (but not oxcarbamazepine).
Diabetes
Although most studies suggest that rates of low sexual desire are similar in women with and without diabetes, generally, difficulties with lubrication are found to be as twice as common in women with diabetes and some, but not all, studies show increased levels of dyspareunia, orgasmic difficulties and sexual dissatisfaction in diabetic women [3]. Somewhat different findings were reported in a recent study that differentiated the generalized blunting of desire and all phases of response in diabetic women over 50 years of age from the apparently unaffected sexual responses of younger women who only showed increased problems with low desire, when compared with control women [53]. An even more recent large study of women enrolled in the long-term Epidemology of Diabetes, Interventions and Complications (EDIC) study, albeit without comparison with control women, confirmed dysfunction in 35% of the unknown percentage of women who were sexually active. Low desire was present in more than half of women and problems with orgasm, arousal and lubrication occurred in 40–50%, with pain reported in 21% of women [54]. In a multivariate analysis, only depression and marital status predicted sexual dysfunction. In general, studies are somewhat limited in that many do not separate premenopausal from postmenopausal women, nor do they control for estrogen status. The majority of studies have clearly identified the strong link between dysfunction and comorbid depression. Diabetic control and length of disease do not generally appear to correlate with the incidence of sexual dysfunction in women, in contrast to the evidence for men.
In terms of any specific therapy, when sexual dysfunction is in the context of diabetes, one small study demonstrated improved genital congestion with sildenafil [55]. Generally, systemic estrogen is avoided and topical estrogen can be used with a vaginal ring, in a tablet or as a cream.
In men, insulin resistance and increased adiposity are associated with a hypoandrogenic state, but in women, glucose intolerance and insulin resistance are associated with hyperandrogenism. Sex hormone globulin levels are lowered by Type 2 diabetes and postmenopausal women with diabetes have been demonstrated to have higher levels of free estradiol than controls. Estrogen therapy has been demonstrated to decrease androgen levels and improve glucose control [56]. A polymorphism in a repeated sequence of the androgen receptor gene has been reported to modulate the activity of the receptor. However, the molecular structure of the androgen receptor in women with diabetes has not been studied. In women with polycystic ovary syndrome, those with smaller numbers of the repeated sequence in the receptor gene demonstrated a close association between increased testosterone levels and increased insulin resistance. As the length of the repeated sequence decreased, the association became weaker [56]. Animal studies suggest that estrogen is an important regulator of pancreatic function. In addition, studies suggest that estrogen serves to reduce oxidative stress, and an increase in oxidative stress is considered to be one of the main mechanisms responsible for the diabetic pathology. Animal work has also identified reduced clitoral blood flow and reduced clitoral cavernosal smooth muscle content in diabetic animals and there is one small clinical study of women with diabetes suggesting decreased sensation in the clitoris, labia and vagina as well as extragenital sites in diabetic compared with nondiabetic women [57]. This would support the concept that in some women, diabetes-induced peripheral, vascular and neurological dysfunction may impair their genital response. Animal studies also suggest that estrogen can improve vaginal structure and function in a persistently diabetic animal. Human research is urgently needed to investigate the apparent protective effect of estrogen on both the diabetes itself as well as on the health of women's genital tissue.
Metabolic syndrome
Recent studies have shown metabolic syndrome to be associated with increased sexual dysfunction independent of associated diabetes and obesity [58]. This is more marked in pre-compared with post-menopausal women [59]. Whether or not improved management of metabolic syndrome improves sexual function has not been studied.
Polycystic ovarian syndrome
Sexual satisfaction has been demonstrated to be reduced in women with polycystic ovarian syndrome compared with controls in some but not all studies. Data are limited and the presumption is that lower sexual satisfaction is related to obesity and cosmetic androgen-related effects of hirsutism and acne. Whether antiandrogen treatment improves sexual function is unclear; one small case study showed desire to increase in six women and decrease in 13 women [60]. One further small study showed metformin to improve sexual function in women with polycystic ovarian syndrome [61].
Psychiatric disease
Depression
Low desire is almost universal in women suffering from major depression [62] but arousal, lubrication and orgasm may also be impaired [63,64]. Case reports of increased libido and hypersexuality associated with depression are infrequent and may have been due to an unrecognized onset of hypomania. Selective serotonin reuptake inhibitors (SSRIs) and serotonin norepinephrine reuptake inhibitors (SNRIs) are associated with a new onset of sexual dysfunction in some 40% of women [65]. Bupropion, which has dopaminergic and nor-adrenergic effects without any changes in serotonin activity, is associated with a low rate of sexual dysfunction, differing significantly from SSRIs and SNRIs [65]. Mirtazapine has been associated with less sexual dysfunction, as confirmed in a recent meta-analysis [66]. It is possible that total daily doses of antidepressants in the upper half of the normal dosing range are more commonly associated with sexual dysfunction than doses in the lower half, except perhaps for fluoxetine, bupropion and mirtazapine [67]. A newer SNRI, duloxetine, may also lessen sexual function but to a smaller extent than an SSRI [68,69].
Monoamine oxidase inhibitors have also been demonstrated to be associated with sexual dysfunction. However, recently, transdermal administration of selegeline did not appear to induce sexual dysfunction. Both placebo and selegeline were associated with minimal onset of new dysfunction in one study [70].
The older tricyclic and heterocyclic antidepressants can negatively impact sexual functioning. Although rarely used for depression, amitriptyline is frequently prescribed for chronic pain – sexual functioning in this population is not well studied. There are occasional reports of clitoral priapism from trazodone.
Available in Europe, agomelatine (an agonist of melatonin receptors MT1 and MT2, and antagonist at 5HT2C) has been shown to cause a very low degree of dysfunction, which is comparable to placebo and significantly less than paroxetine or venlafaxine [71,72].
Anxiety disorders
Anxiety disorders are associated with an increased prevalence of sexual dysfunction, especially orgasmic or arousal disorders; obsessive compulsive disorder is particularly associated with orgasmic difficulties [73]. There may be an association between women with both obsessive compulsive disorder and orgasmic disorder and past sexual abuse [74]. Although studies are limited, anxiety and hypervigilance have been noted in women with persistent genital arousal disorder (a condition of distressing genital congestion and feelings of impending orgasm that are unremitting, unwanted and minimally lessened by experiencing an orgasm). Pharmacological treatments of anxiety disorders involve administering typically higher doses of SSRIs or SNRIs, the sexual side effects of which have been outlined. Buspirone, a serotonin 1A partial agonist used for the treatment of generalized anxiety disorder, negatively affects sexual function only rarely [75].
Psychotic illness
Changes in sexual function are common just prior to the full-blown psychosis in women with schizophrenia. In one study, some 40% of women with schizophrenia reported never having been in a sexual relationship [76] but nevertheless may be sexual alone.
Antipsychotics can further lower sexual desire in 50–73% of women [77]. Traditional antipsychotics and risperidone reduce dopamine and increase prolactin levels as well as blocking α-muscarinic receptors. Second-generation antipsychotics do not raise prolactin levels and may have fewer sexual side effects [78] but other mechanisms might still cause dysfunction [79]. Reducing prolactin with dopaminergic drugs does not improve sexual dysfunction [80]. Despite the occurrence of antipsychotic-induced sexual dysfunction, the increased ability for emotional regulation from effective antipsychotic therapy seems to benefit sexual function [81]. Managing antipsychotic-associated sexual dysfunction is extremely important given the risk of non-compliance and subsequent distress to patients and their families. There is a lack of control data to support the use of antidotes such as amantadine or cabergoline, but substitution may be helpful –switching to olanzapine, quetiapine, aripiprazol or ziprasidone is suggested [82–84].
Breast cancer
Difficulties in sexual function following breast cancer treatment are the most likely causes of distress to persist beyond 1 year after diagnosis [85]. A predictive model for sexual outcome in breast cancer patients has evolved from two independent groups of women with a past history of breast cancer [86]. An absence of vaginal dryness, presence of emotional wellbeing, a positive body image, a high quality of the interpersonal relationship and a lack of sexual problems in the partners proved to be the most important predictors of sexual health.
Studies of the prevalence of sexual dysfunction subsequent to the diagnosis and management of breast cancer range from 30 to 100%. Sexual self-image can be negatively impacted (Table 3) and fear of recurrence may cause hesitancy to start a new relationship – the need to disclose the medical details and fear of rejection may act as deterrents. The stress and sadness of possible imposed infertility may further lessen self-image and sexual desire. However, many studies conclude that the major factors negatively influencing sexual response are also treatment-related. Chemotherapy may bring about permanent premature ovarian failure, often leading to dyspareunia owing to estrogen deficiency-related vulval vaginal atrophy. This in turn can reduce arousability and the likelihood of orgasm and can lessen sexual motivation. There will also be a loss of ovarian testosterone, DHEA and androstenedione, but sexual function has not yet been shown to correlate with any of these hormonal levels. A small study of women with past breast cancer and a complex endocrine status owing to ongoing estrogen therapy found that, whereas relationship factors predicted desire, a history of chemotherapy predicted disorders of arousal, lubrication, orgasm and the presence of sexual pain [87]. However, there was no relationship between sexual function and androgen levels, including androgen metabolites. The latter reflects total androgen activity (i.e., intracrine testosterone from adrenal precursor hormones, such as DHEA sulphate and androstenedione, as well as ovarian testosterone). The authors noted that the experience of chemotherapy appeared to disrupt sexual function by nonendocrine mechanisms. Notably, weight gain from chemotherapy can be very troublesome to a woman's sexual self-image and, in one series, only 10% of women reported a return to their precancer diagnosis weight [88].
The estrogen deficiency of the genital tissues presents a clinical dilemma. Local estrogen is beneficial but there is currently insufficient safety evidence to recommend the various currently available minimally absorbed local estrogens. Zero systemic estrogen is desirable, particularly when a woman is on aromatase inhibitors; however, some oncologists will permit the use of local estrogen via a sialastic ring or via conjugated estrogen cream or vaginal estradiol pills. All three formulations are under investigation for efficacy at lower doses than are currently available. In addition, the use of investigational local vaginal DHEA in women using aromatase inhibitors (implying the safety of DHEA in these women who must receive no additional estrogen) is currently being studied in clinical trials. There is theoretical and animal evidence that local testosterone produced from the DHEA will remedy the vaginal atrophy, despite the fact that local estrogen would not be produced from the DHEA owing to the presence of the aromatase inhibitor. There is recent evidence of sexual benefit when DHEA is applied vaginally for estrogen deficiency-related vulval vaginal atrophy in otherwise healthy postmenopausal women [89]. Interestingly, there was no increase in systemic levels of estrogen, testosterone, DHEA or androgen metabolites – the therapy would appear to be truly local. Systemic estrogen is contraindicated in women with breast cancer and systemic testosterone is currently contraindicated as well, although there are theoretical reasons for both harm and benefit. Importantly, one small study of women with a past history of cancer, including breast cancer, did not report benefit from systemic testosterone that was prescribed for low sexual desire [90].
Regarding the changes in sexual self-image after partial or complete mastectomy, scientific reviews have concluded that reconstruction or choosing between lumpectomy versus mastectomy have minimal effects on sexual function [91]. The sexual consequences of the often painful and frequently distressing lymphedema of the arm have been minimally studied. Radiation can cause skin fibrosis and alter the appearance and sensitivity of the skin.
A temporary ‘medical menopause’ from gonadotrophin-releasing hormone agonist treatment is associated with reversible dysfunction [92]. Tamoxifen is considered to be sexually neutral [92].
Approximately 10% of women with breast cancer have a genetic predisposition for the development of ovarian cancer owing to BRCA gene mutations, and some of these women choose to undergo a risk-reducing bilateral salpingo-oophorectomy. Sexual outcome has received limited study, but a recent report showed a negative impact owing to dyspareunia and lowered body image [93]. There have also been limited studies of sexual function subsequent to prophylactic mastectomy of the breast unaffected by cancer.
Given that the aromatase inhibitors prevent the production of any estrogen, many women complain of moderate and severe dyspareunia, exacerbation of menopausal symptoms and, indirectly, the loss of sexual desire [94].
For younger women with breast cancer, a strategy for fertility preservation is considered along with the overall oncological treatment plan. The threat of infertility arises from a need for systemic chemotherapy and decisions to administer these treatments depend on the stage of diagnosis. Women with stage I disease and favorable tumor biology (estrogen receptor-positive, progesterone receptor (PR)-positive and HER-2-negative tumors) are typically treated with surgery, sometimes followed by antiestrogen therapy and radiation. Provisional data suggest that antiestrogen therapy can be delayed to allow for pregnancy once surgery and radiotherapy have been completed [95,96]. Neither pregnancy nor any attempt to retrieve eggs for IVF should occur during radiation therapy because there can be some internal scatter to the pelvis and ovaries. Women with tumors larger than 1 cm, metastases to regional lymph nodes or women with estrogen receptor/PR-negative tumors often require chemotherapy and face more serious threats to their fertility. Risk factors include the woman's baseline ovarian reserve and the need of an alkylating agent. Some options for fertility preservation are emerging, one of which is to delay treatment in order to undergo a cycle of hormone stimulation and oocyte retrieval. However, this may not be appropriate for women with hormonal-sensitive estrogen receptor/PR-positive tumors, since there are data that suggest that exogenous estrogen could promote such tumors. A further fertility preservation technique avoiding exposure to exogenous hormones is that of ovarian tissue retrieval. The oocytes can be aspirated from the ovary and cryopreserved. Alternatively, ovarian tissue strips can be preserved. These strips can be used for subsequent retransplantation, but reintroduction of cancer cells is a theoretical risk. An experimental procedure termed ‘in-follicle maturation’ involves obtaining the immature follicles from the cryopreserved ovarian tissue, maturing them in vitro and then using them in IVF procedures [97].
Gynecological cancer
Sexual confidence, desire and function can frequently be negatively affected by the diagnosis and management of gynecological cancer. Loss of sexual organs from surgery, loss of ovarian function and fertility from chemotherapy, interference with vulval and vaginal congestion from damaged autonomic nerves in radical hysterectomy and tissue damage from radiation – especially vaginal agglutination – as well as misgivings relating to the etiological role of past sexual activities may all contribute to the sexual changes. Another fear is that cancer may be transferred sexually. Epidemiological studies of sexual dysfunction after different gynecological cancers vary with regard to oncological stages and follow-up times, but recent studies suggest that when women are specifically asked, between 30 and 63% of women who undergo treatment for cervical cancer experience some sexual complaints [98]. Sexuality after ovarian cancer has been less well studied but one study of 232 women, of whom almost half were still receiving treatment, showed that of the 50% who had been sexually active in the past month, almost half reported little or no desire, 80% reported problems with vaginal dryness and 62% with dyspareunia [99].
Despite the strong possibility of interruption of sexual response from the treatment of gynecological cancer, many women show particular sexual resilience. A recent study of sexual morbidity in very long-term survivors of vaginal and cervical cancer, of whom most received a radical hysterectomy and partial or complete vaginectomy, showed that despite having significantly more sexual problems than control women, the rates of sexual activity and partnerships were similar in patients and controls [100].
Radical hysterectomy for cervical cancer has been noted to have persistent negative effects on sexual desire, whereas difficulty with lubrication may lessen over time, as noted in one study where the autonomic nerves in the cardinal and uterosacral ligaments were probably spared [98]. Nerve-sparing radical hysterectomy is in its infancy [101]; however, early data suggest that neurovascular preservation leads to improved sexual function [102,103]. A recent study of 30 women with early-stage cervical cancer receiving nerve-sparing radical hysterectomy type III, of whom 14 also received bilateral salpingo-oophorectomy, indicated minimal new sexual dysfunction at 6 months, using a visual analog score [104]. However, using a frequently used and validated questionnaire, other researchers found that similar radical hysterectomy (type III) was associated with deterioration in sexual function whether the approach was via laparoscopy or laparotomy [105]. Some research suggests that the more marked sexual dysfunction in women with cervical cancer compared with controls is seen in those with a history of sexual abuse [106]. Whereas an absence of satisfaction was reported by 20% of women with neither abuse nor cancer of the cervix and by 31% of women who had been sexually abused but did not have cancer – similar to the 28% of women with cancer of the cervix but not past abuse – it was of note that 45% of women with both abuse and a cancer history reported sexual dissatisfaction. Moreover, this dissatisfaction caused a marked decrease in wellbeing in 18% of women with neither abuse nor cancer but in 39% of women who had been abused but had no cancer, and similarly, a decrease in wellbeing was observed in 44% of women with both abuse and cancer history. The same study showed that dyspareunia was extremely rare in women without cancer of the cervix – dyspareunia was reported by 12% of those with cancer of the cervix alone but by 30% of those with cancer of the cervix and past abuse [106].
Management strategies include the provision of information and support and symptom management. A preliminary study of mindfulness training that was incorporated into a psychoeducational program for women with arousal disorder subsequent to gynecological cancer showed benefit [16,17]. Given that the majority of women who receive radiation are at risk for developing vaginal agglutination and stenosis within the first 3 months of therapy, vaginal insert treatment is extremely important and measures to increase compliance require urgent study. Local estrogen can be given in the form of a sialastic ring, estradiol cream or tablet. Theoretically, phosphodiesterase type 5 inhibitors might help women with partial autonomic nerve interruption owing to radical hysterectomy, but studies are lacking. The multidisciplinary approach to sexual rehabilitation within the context of ongoing chronic illness, as well as past or present cancer, may have excellent outcome, as suggested by a small pilot study [107].
Future perspective
As advances in surgical and medical treatments continue, the survival of women with chronic illness and cancer is likely to improve markedly and their quality of life, including their sexual life, will become even more important. An increased acceptance that sexual dysfunction matters to women with chronic illness has probably encouraged the increased teaching of sexual medicine to physicians in training as well as media focus on women's as well as men's sexual lives. Many factors, including the presence of HIV and AIDS, have made it necessary for women to be more vocal regarding their own sexual experience and needs, such that women themselves are less likely to allow their sexual lives to be overlooked by their healthcare providers.
Within the next 5–10 years, clarity regarding what constitutes a sexual disorder versus ‘changes within a broad spectrum of normal female sexual experience’ is expected. DSM-V definitions of disorders will be published in 2012 or 2013, and draft versions are currently available, albeit for a short period [201], in order to receive input from a wide audience. Provided that a broader perspective of what is within the range of normal is accepted, this will have a therapeutic effect on women sensing changes in their sexual experiences associated with acquiring chronic illness. Knowing that their response may still be ‘within normal limits’ will improve sexual self-image, motivation to find more erotic stimuli and a healthier emotional connection with their partner, benefiting sexual function.
Given the strong link between sexual dysfunction within chronic illness and depression, sexually neutral antidepressants are needed. Agomelatine, which is approved in Europe, appears to be one such molecule and others are likely to be developed in the next 5–10 years.
Our understanding of any hormonal etiology underlying the loss of sexual arousability may well improve. Specifically, postmenopausal and older women who are still willing to be sexual with their partners, to whom they are still attracted and with whom they were able to become aroused in the past, but now only experience unrewarding/unsatisfying sexual experiences, are potential candidates for hormonal-based dysfunction. Alterations in the sensitivity of androgen and estrogen receptors may prove to be linked to subtypes of sexual dysfunction. This may not immediately lead to obvious therapeutic interventions, but may prevent inappropriate hormonal supplementation. Development of selective estrogen receptor modulators and selective androgen receptor modulators that possess sexual benefit with minimal adverse effects are attractive possibilities.
Further research into the area of neurosteroids may well shift attention from the measurement, and perhaps even from the supplementation of systemic steroids to an understanding of which factors modulate the production and activity of neurosteroids. These include the sex steroids that are entirely manufactured within the brain, beginning with cholesterol and acting within the CNS. Adaptive changes occur in the brain in response to reductions in the serum levels of sex hormones that are associated with age and with menopause – in women, upregulation of steroidogenic enzymes and of sex receptors occurs. Research into whether this upregulation is variable and if it is linked to sexual function is needed. Whether systemic supplementation interferes with upregulation may also become clear.
Medications with variable antagonist/agonist profiles on serotonergic (± other) receptors might well receive approval for desire and arousal disorders in women with and without chronic illness during this timeline. However, the potential benefits of such medications will only be demonstrated if patients are carefully assessed and psychological factors including distractibility, as well as interpersonal factors, are addressed first. Complex as this is, combinations of psychological and medical therapy are likely to be needed.
Further study of the use of mindfulness will be published in the future – controlled studies are ongoing. The focus on the present moment rather than on any goal of the sexual encounter, as well as acceptance rather than judgment of that moment, may well prove key to the improvement of sexual responses of women with (as well as those without) chronic illness. There is currently considerable integration of this very old eastern skill within western medicine, including the management of mood disorders, attention deficit disorder and various chronic pain syndromes. The dualistic thinking of psychological versus biological is, albeit slowly, fading; medical students are now more likely to be taught that the mind and body are inseparable.
Executive summary
Despite the fact that chronic disease and cancer are all associated with an increased prevalence of sexual dysfunction, women's sexual resilience can be remarkable nevertheless.
Depression is strongly linked to sexual dysfunction in women living with chronic disease, such that the assessment and management of mood disorder is the initial focus in sexual rehabilitation.
Despite medical and surgical interruptions of sexual response, sexual dysfunction is usually multifactorial in etiology, such that assessment and management needs to be comprehensive. Usually, both direct and indirect mechanisms are operating: chemotherapy-induced ovarian failure may be compounded by pain, depression, low sexual self-image, fear of inability to satisfy the partner, guilt regarding the cause of cancer and even fear of transmitting the cancer sexually.
Discussion of women's sexual response and clarifying areas that are currently disturbed is the basis of both assessment and management.
Physical examination is usually necessary and an unhurried detailed examination with adequate explanation before and after can itself be therapeutic.
Standard therapies for sexual dysfunction include cognitive-behavioural therapy, sex therapy and psychoeducation. Mindfulness has recently been added to this and has been found to show early promise in women and in those with pelvic cancer.
Medical therapies include local or systemic estrogen for dyspareunia owing to a lack of estrogen. Investigational therapies include phosphodiesterase type 5 inhibitors for a neurogenic genital congestion deficit, local vaginal dehydroepiandrosterone for postmenopausal vaginal atrophy and systemic transdermal testosterone for low sexual desire. The latter, being systemic, remains of questionable safety in the long term.
Fear of further cardiac damage from sex is common in women with cardiac disease: they can be advised that the risk is low and short-lasting and that cardiac vascular symptoms are very unlikely to occur if no symptoms arise during exercise testing to 6 metabolic equivalents. Comorbid depression is common and the most probable cause of the presenting symptoms of low desire and arousability.
Sexual dysfunction is extremely common in women with end-stage renal disease. Many medical and psychological factors are involved. Studies of sexual benefit from the use of erythropoietin for the associated anemia, or of sexual benefit from transplantation, are extremely limited.
Pain, age and depression have been demonstrated to be important modulators of women's sexual function when rheumatic disease is present. Changes to genital tissue may compound sexual difficulties from loss of mobility.
Neurological disease usually leads to sexual deficit but there are some exceptions. Parkinson's disease treated with dopaminergic agonists can cause unwanted hypersexuality. There can be considerable risk to women whose brain damage has caused hypersexuality given the increased chance of abuse, coercion, acquisition of sexually transmitted diseases and unwanted pregnancies. Loss of genital sensation is common in established multiple sclerosis. In addition, it is of note that devastating sexual loss arises from cauda equina injuries, involving complete loss of genital sensation as well as loss of voluntary control of the bladder and bowel: these women appear to be healthy and not have obvious disability.
Most studies report increased sexual dysfunction in women with diabetes. In contrast to studies in men, there is no relationship between glucose control, duration of diabetes or its complications and sexual dysfunction.
Metabolic syndrome and polycystic ovarian syndrome have been demonstrated to be associated with an increased prevalence of dysfunction.
All aspects of sexual response can be decreased with depression: selective serotonin reuptake inhibitors and serotonin norepinephrine reuptake inhibitors are associated with the new onset of sexual dysfunctions in approximately 40% of women. Less sexual deficit is reported from bupropion and mirtazapine, and the monoamine oxidase inhibitor selegeline. Agomelatine, an agonist of melatonin receptors MT-I and MT-II and antagonist of 5HT2C receptors and sexually neutral, is available in Europe. The higher dose of selective serotonin reuptake inhibitors needed for anxiety disorders can be sexually detrimental, but buspirone, a partial agonist for serotonin 1A receptors, is rarely sexually negative. Antipsychotics can further lower sexual desire in women with schizophrenia. Traditional antipsychotics and risperidone reduce dopamine levels and increase prolactin, and while second-generation antipsychotics do not raise prolactin levels and may have fewer sexual side effects, other mechanisms can still cause dysfunction. Nevertheless, the increased stability and emotional regulation from effective antipsychotic therapy benefits sexual function. Sexual side effects of medications must be routinely inquired about, given the very real risk of poor compliance with therapy.
Difficulties in sexual function after breast cancer treatment may persist. The most important predictors of sexual health are an absence of vaginal dryness, presence of emotional wellbeing, positive body image, a high quality of interpersonal relationships and a lack of the partner's sexual dysfunction. Treatment with aromatase inhibitors can be particularly sexually damaging, given the total lack of estrogen production and the frequent preclusion of local estrogen. The safety and benefit of local dehydroepiandrosterone applied vaginally is under investigation. Fertility prevention is considered along with the overall oncological treatment plan.
Gynecological cancer can be sexually devastating, especially ovarian cancer, which has been minimally studied. Nerve-sparing surgeries for cervical cancer attempting to avoid the pelvic plexus in the cardinal ligaments may prove to limit sexual dysfunction. Some research suggests the more marked sexual dysfunction in women with cervical cancer is linked to past sexual abuse. Treatment includes the provision of information and support, chronic symptom management and psychosexual education with the possible addition of mindfulness, along with vaginal insert treatment and local estrogen to prevent agglutination and stenosis.
Footnotes
Acknowledgements
The author would like to thank Angela Wong for her secretarial assistance.
The author has no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
