Abstract
In women with previous gestational diabetes (pGDM), the risk of developing Type 2 diabetes is greatly increased, to the point that GDM represents an early stage in the natural history of Type 2 diabetes. In addition, in the years following the index pregnancy, women with pGDM exhibit an increased cardiovascular risk profile and an increased incidence of cardiovascular disease. This paper will review current knowledge on the metabolic modifications that occur in normal pregnancy, underlining the mechanism responsible for GDM, the link between these alterations and the associated long-term maternal complications. In women with pGDM, accurate follow-up and prevention strategies (e.g., weight control and regular physical exercise) are needed to reduce the subsequent development of overt diabetes and other metabolic abnormalities related to cardiovascular disease. Therefore, our paper will provide arguments in favor of performing follow-up programs aimed at modifying risk factors involved in the pathogenesis of Type 2 diabetes and cardiovascular disease.
Keywords
Gestational diabetes mellitus (GDM), defined as “glucose intolerance of any degree with onset or first recognition during pregnancy,” is a common complication in pregnancy, occurring in 4- –7% of pregnant women; it represents 90% of all cases of diabetes mellitus that are diagnosed during pregnancy [1].
Incidence rates of GDM are increasing in all ethnic groups, reflecting the increased prevalence of obesity and Type 2 diabetes mellitus (T2DM) within the general population. Both obesity and a family history of T2DM represent important risk factors for the development of GDM [2–4].
In the short term, GDM is associated with an increased risk of adverse obstetrical outcomes, particularly those related to fetal overgrowth, which include macrosomia, shoulder dystocia, birth injury, prematurity and an increased Caesarean section rate [5].
Despite normal glucose tolerance in the imme-diate postpartum, women with GDM are at high risk for subsequent development of metabolic diseases. Development of T2DM, for instance, is much greater among women with previous GDM (pGDM). These women also tend to display features of metabolic syndrome (MS) such as hypertension, dyslipidemia and microalbuminuria, which increase the risk for atherogenic insult [6]. Women with pGDM should, therefore, be enrolled in follow-up programs designed to ensure continuous surveillance with the aim of providing effective prevention of T2DM and cardiovascular diseases (CVD).
This review examines the available evidence with regard to the associations between GDM, T2DM and CVD. We also describe possible strategies to prevent these abnormalities in high-risk women, with a focus on lifestyle modification.
Glucose homeostasis in pregnancy
In order to better evaluate the potential longterm implications of GDM, we first considered the physiology of glucose homeostasis in pregnancy, underlining those modifications that are more likely to lead to the development of GDM.
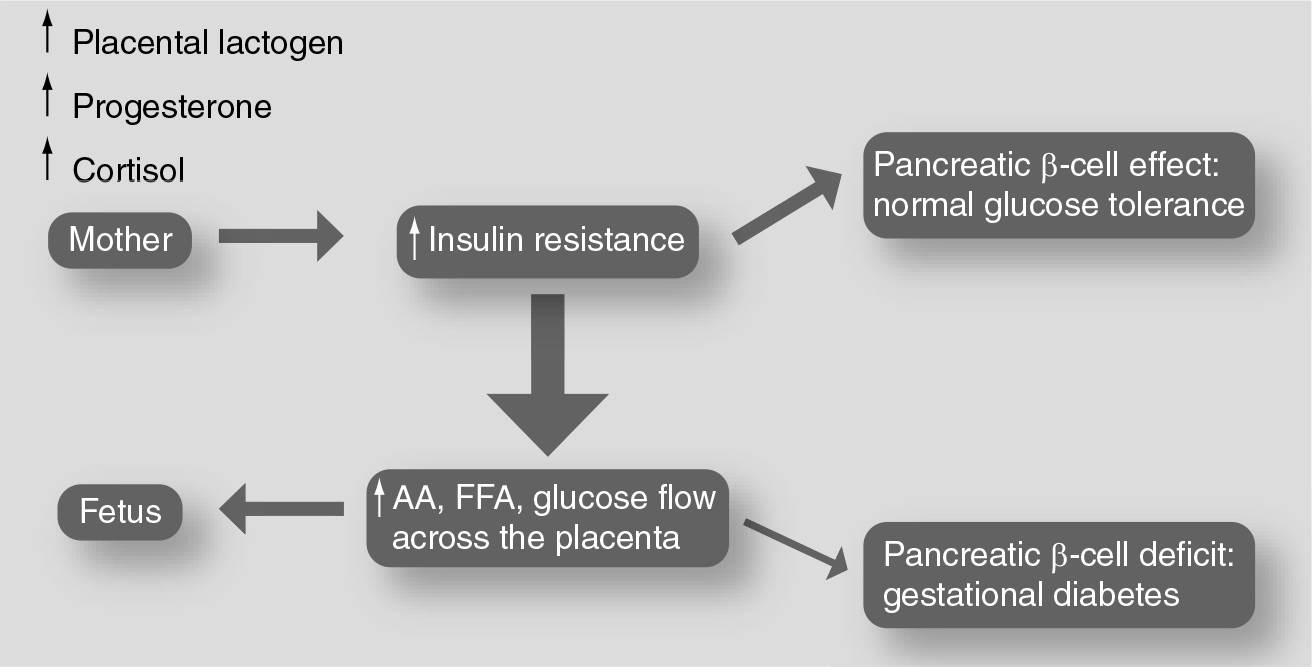
Pregnancy is characterized by a complex endocrine-metabolic adaptation process including impaired insulin sensitivity, increased β-cell response, moderately increased blood glucose levels (particularly following the ingestion of a meal), and changes in the levels of circulating free fatty acids, triglycerides, cholesterol and phospholipids [7]. These changes do not reflect a pathological condition; rather, they represent a necessary and indispensable adaptation to meet the energy demands of the fetus and to prepare the maternal organism for delivery and lactation. In fact, the insulin resistance developing in pregnancy is likely to be a physiological event favoring glucose supply to the fetus. The reduced insulin-mediated utilization of glucose switches the maternal energy metabolism from metabolizing carbohydrates to lipid substrates (i.e., free fatty acids), redirecting carbohydrates toward the fetal tissues [8].
The insulin resistance that develops during pregnancy is similar to that observed in T2DM, with impaired insulin action mainly as a result of postreceptor alterations involving glucose transport and intracellular metabolism in insulin-sensitive tissues [9]. The degree of insulin resistance seems to be influenced by obesity and inheritance. In fact, in obese women (BMI > 30; or weight > 150% of ideal body weight) the incidence of GDM is 1.4- to 20-fold higher than that in normal weight subjects [10].
Despite insulin resistance, glucose homeostasis is maintained in normal pregnancies by a concomitant compensatory increase in insulin secretion. The intolerance to carbohydrates develops as soon as β-cell secretion is no longer sufficient to compensate for insulin resistance (Figure 1) [11].
Endocrine–metabolic adaptation to meet the energy demand of the fetus.
Although the specific mechanism(s) of the alteration of insulin secretion and action remain uncertain, a substantial contribution is made by endocrine modifications that accompany pregnancy. Changes in β-cell function occur in parallel with the development of the feto–placental unit and the local production of hormones, such as estrogens, progesterone, cortisol, human chorionic somatotropin, placental lactogen, prolactin and growth hormone (Table 1). These hormones have been shown to induce insulin resistance both
In women with GDM, the impaired insulin-mediated glucose utilization and the inadequate increase in first-phase insulin secretion are the initial alterations of glucose homeostasis that cause the plasma glucose excursion after meal ingestion and postoral glucose tolerance test (OGTT) hyperglicemia. The loss of early insulin release in these women is likely to contribute to the glucose intolerance and postprandial hyperglycemia, and is considered to be a marker of glucose homeostasis impairment. Xiang
GDM: a risk for T2DM
Although glucose tolerance returns to normal levels after delivery in the majority of women with GDM, this condition represents an early stage in the natural history of T2DM to many clinicians [15,16]. The strength of the association between GDM and T2DM, and the knowledge that both conditions have many of the same risk factors (e.g., family history for diabetes, overweight, increased age and ethnic group), suggest a common genetic background for GDM and T2DM. In accordance with this hypothesis, it has been reported that women with pGDM more frequently display some alleles associated with the high risk of T2DM [17,18].
The transition time from GDM to overt T2DM can be shortened as a function of the number of pregnancies. According to Peters
GDM is diagnosed before the 24th week of pregnancy (5-year risk: 80%);
Fasting glucose levels before pregnancy are higher;
A defect in insulin secretion is apparent;
Insulin therapy is indicated during pregnancy;
Obesity precedes pregnancy (risk; 50–75%);
A positive family history of diabetes mellitus exists;
Bodyweight increases excessively in the postpartum;
Ethnicity is non-Caucasian.
The differences of diagnostic tests, of criteria for GDM, of the ethnic groups and of the length of follow-up, do not allow one to assess the true prevalence of T2DM in women with pGDM, nor the rate of conversion from GDM to T2DM.
In the classic studies by O'Sullivan performed 30 years ago, diabetes was diagnosed in 36% of women 22–28 years after pregnancy with GDM [22]. In 2002, Kim and colleagues conducted a systematic literature review of articles published between 1965 and 2001 [23]. A total of 28 follow-up studies, performed between 6 weeks to 28 years postpartum were examined, and the cumulative incidence of diabetes ranged from 2.6% to over 70%. The authors reported that after adjustment for various lengths of follow-up and testing rates, the cumulative incidence of T2DM increased in the first 5 years after delivery and reached a plateau after 10 years.
More recently, in a meta-analysis performed by Cheung and colleagues from six controlled follow-up studies, the overall relative risk for developing diabetes after GDM was calculated to be 6.0 [24]. The authors, estimating that 10–31% of parous women with diabetes have had GDM, underlined the population health significance of GDM.
Our population, including 160 women with pGDM, underwent an OGTT 1–3 years after delivery; five (3.81%) had diabetes and 31 (19.37%) had impaired glucose regulation (i.e., impaired glucose tolerance and/or impaired fasting glucose) [25]. Prepregnancy BMI, fasting and 120-min glucose levels were independent contributors of postpartum diabetes or impaired glucose regulation. Notably, at postpartum follow-up, all women with normal pregnancy were normotolerant and all pGDM women who were found to be normo-tolerant had significantly higher fasting and post-challenge plasma glucose levels compared with control women.
More recently, Bellamy and colleagues performed a new meta-analysis of cohort studies in which women who had developed T2DM after GDM were followed-up between 1965 and 2009 [26]. Their analysis, including 20 studies of 675,455 women and 10,859 cases of T2DM, reported that women with pGDM have at least a 7.5-times increased risk of developing T2DM in the future compared with those with normoglycemic pregnancy. Moreover, the authors reported that within 5 years of a pregnancy complicated by gestational diabetes, women had a relative risk of 4.69, which more than doubled to 9.34 in those who were examined more than 5 years postpartum.
Antepartum predictors of diabetes risk after gestational diabetes mellitus.
Ethnic group
Family history of maternal Type 2 diabetes
BMI at diagnosis of gestational diabetes mellitus
Diagnosis early in pregnancy
Impaired β-cell function
Magnitude of hyperglycemia
Insulin therapy during pregnancy
Increased body weight during pregnancy
GDM: a risk for cardiovascular disease
In addition to T2DM, women with pGDM also tend to display features of the MS, such as hypertension, dyslipidemia and microalbuminuria [6], and they may present an increased cardiovascular risk factor profile (Figure 2) [27,28]. In this context they show elevated total cholesterol, LDL-cholesterol and triglyceride concentrations and low HDL-cholesterol [29,30].

Gestational diabetes mellitus: a risk for Type 2 diabetes and for cardiovascular disease later in life.
Moreover, other nontraditional cardio-metabolic risk factors, such as higher levels of plasminogen activator type 1, acute-phase inflammatory biomarkers (e.g., leukocyte count, fibrinogen, C-reactive protein and sialic acid) and low circulating levels of adiponectin have been found in women with pGDM [31–34]. As a consequence, MS prevalence after a GDM- complicated pregnancy is significantly higher than in women with normal pregnancy, in just the first 10 years following the index pregnancy (Figure 3) [35–37]. In a previous study, we have demonstrated that the rate of MS was 9% in women with pGDM and 1% in the control group in a cohort of women with pGDM 16 months after delivery [34]. Based on this observation, it appears rational to propose that pGDM should be considered a manifestation of this syndrome.
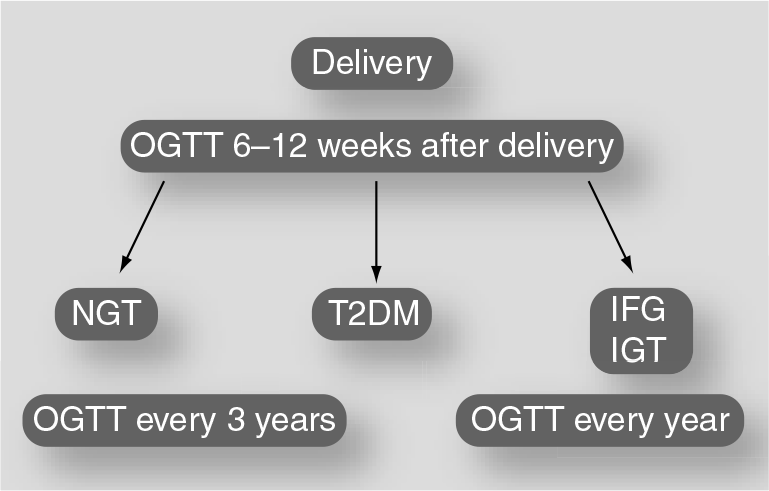
Follow-up program for the prevention of T2DM in women with previous gestational diabetes mellitus.
The observation that the association between subclinical inflammation and MS occurs more frequently in women with pGDM than in women with normal pregnancy makes plausible the theory that these women may be exposed to a more atherogenic insult [38]. In fact, studies performed in these women have demonstrated increased vascular dysfunction, including impaired endothelium-dependent vasodilatation, increased vessel stiffness, early abnormalities in diastolic function and impaired cardiac autonomic function [33,39–41]. In this context, we observed that relatively young women, just 2 years after index pregnancy, are at risk for the early onset of subclinical atherosclerosis, as indicated by increased carotid artery intima-media thickness (Table 2) [42]. Therefore, pregnancy offers an important opportunity for assessing the risk of cardiovascular and metabolic disease, and it could be considered as an event with the potential to unveil a pre-existing susceptibility that is higher in women who previously developed GDM.
Vascular function after gestational diabetes mellitus.

GDM: Gestational diabetes mellitus.
Recently, this anticipated association between pGDM and subsequent CVD has been confirmed by Shah
The potential role of overt T2DM in determining the vascular risk associated with pGDM remains unclear. However, considering the low period of time generally required for the development of CVD in T2DM, it seems unlikely that diabetes precedes the onset of CVD in women with pGDM. T2DM and CVD probably develop in parallel in this patient group.
Prevention of T2DM & CVD
The American Diabetes Association recommends that women with GDM should be reclassified at 6 weeks after the index pregnancy in order to detect persistent glucose abnormalities [45]. For those who maintain impaired fasting glucose or glucose intolerance after the pregnancy, annual OGTTs should be performed. Furthermore, women with pGDM who give normotolerant results at the first evaluation after delivery should have an OGTT with regular intervals, beginning 1 year after pregnancy. Markers of MS, such as abdominal circumferences, blood pressure and lipid profile may be investigated in addition to the OGTT (Figure 3).
Although follow-up recommendations for women with pGDM are well defined, there are numerous citations in the literature documenting a lack of follow-up care during the postpartum period for GDM [46–48]. A lack of primary care survillance in a relatively young, mobile population that underestimates their risk of T2DM, and the difficulties associated with adherence to diet and exercise in women who are busy providing care for young children, do not easily allow for a high rate of postpartum follow-up that remain only approximately 50% in this population [49].
It is now well documented in different populations, including Americans [50], Finnish [51] and Asians [52] with prediabetes, that lifestyle changes with increased physical activity, weight loss and a healthy diet can reduce the risk of progressing to T2DM. In addition, pharmacological intervention (e.g., with metformin [50]) has been found to reduce progression to T2DM. Although a common characteristic of these studies is that the subjects were seen intensively over a long period of time, it seems rational to consider similar interventions in women with pGDM. Women with self-reported pGDM, who form a subset of the population who enrolled in the Diabetes Prevention Program, experienced a 55% reduction in the development of T2DM with the lifestyle intervention when compared with those in the placebo group [53]. Considering that this study was limited by the self-reporting of pGDM and the fact that diagnosis of GDM was not validated, these results should not be considered conclusive. Other studies on this topic are not available. Therefore, in order to determine the potential benefit of lifestyle modification and in order to evaluate the most effective way to achieve it, postpartum studies of healthy diet and exercise plans should be performed in women with pGDM.
Several randomized clinical trials have specifically studied diabetes prevention with pharmacological intervention in women with pGDM. Buchanan
Future perspective
Several opportunities exist for the prevention of T2DM and CVD in women with pGDM, such as education regarding risk awareness, implementation of a healthy lifestyle and pharmacological intervention.
Although GDM is a well-established risk factor for T2DM, many women could be unaware of this increased risk. Interventions are clearly needed in order to increase awareness and acceptance of the personal risk of developing T2DM in women with pGDM. Moreover, considering that not only women with GDM, but also those with mild glucose intolerance in pregnancy are at risk of CVD [57], public education campaigns could be performed for all women with any degree of glucose disturbance during pregnancy.
Postpartum studies of healthy diet and exercise plans should now be performed in women with pGDM in order to determine their potential benefits and to evaluate the most effective way to achieve lifestyle modification in this population. Moreover, considering that breastfeeding is associated with a reduced incidence of T2DM among both women with pGDM and women in the general population [58], the promotion of a combination of breastfeeding, diet and physical activity could reduce maternal T2DM risk, and might be particularly important in women with pGDM.
Executive summary
Gestational diabetes mellitus (GDM) is a common complication in pregnancy, occurring in 4–7% of pregnant women.
GDM develops as soon as β-cell secretion is no longer sufficient to compensate for physiological insulin resistance, which becomes evident in the second half of pregnancy.
Obesity and a positive family history of Type 2 diabetes (T2DM) are the most significant risk factors for the development of GDM.
Although after delivery glucose tolerance returns to normal in the majority of women with GDM, for many clinicians GDM represents an early stage in the natural history of T2DM.
Factors that contribute to evolution toward overt T2DM are: an early diagnosis of GDM, fasting glucose levels before pregnancy, insulin therapy, prepregnancy obesity, positive family history of diabetes mellitus and excessive weight gain.
Cumulative incidence of T2DM increases in the first 5 years after delivery and reaches a plateau after 10 years.
Women with previous GDM (pGDM) tend to display features of the metabolic syndrome (MS), such as hypertension, dyslipidemia and microalbuminuria, and they may present an enhanced cardiovascular risk factor profile.
MS prevalence after a GDM-complicated pregnancy is significantly higher than in women with normal pregnancy.
Impaired endothelium-dependent vasodilatation, increased vessel stiffness, early abnormalities in diastolic function and impaired cardiac autonomic function are reported in women with pGDM at a relatively young age.
The hazard ratio for cardiovascular disease (CVD) events in pGDM women is reported to be 1.71.
GDM must be reclassified at 6 weeks after the index pregnancy to detect persistent glucose abnormalities; women with pGDM should have an oral glucose tolerance test (OGTT) with regular intervals, beginning 1 year after pregnancy. Markers of the MS, blood pressure and lipid profile may be investigated in addition to the OGTT.
To reduce the risk of progressing to diabetes and other metabolic abnormalities, lifestyle changes with increased physical activity, weight loss and a healthy diet must be recommended to these women.
Several randomized clinical trials, using drugs that ameliorate insulin action, have specifically studied diabetes prevention with a pharmacologic intervention in women with pGDM.
Interventions are needed in order to increase awareness and acceptance of the personal risk for the development of T2DM in women with pGDM. Public education campaigns could help to increase patient risk-awareness.
Additional pharmacological studies are still needed to evaluate the cost-effectiveness of the prevention of both T2DM and CVD.
Long-term, continuous programs specifically addressed to women with pGDM could be performed in order to permit them to regularly check glucose tolerance, lipid profile, blood pressure and other parameters aimed at improving women's health.
Several randomized clinical trials using drugs that improve insulin sensitivity have specifically studied T2DM prevention with pharmacological intervention in women with pGDM [54–56]. Although the results of these studies are promising, it should be noted that these medications are not currently approved for use in T2DM prevention. Additional studies are still needed to evaluate the cost-effectiveness of these medications for the prevention of both T2DM and CVD, alone or in combination with new medications.
Furthermore, all women with pGDM should be offered regular checkups of their glucose tolerance, lipid profile, weight and blood pressure.
In order to implement these measures, it is necessary to offer a long-term, continuous programs for these women. Based on the available evidence, a major aim during the next decade will be to implement such programs that might have an important effect on women's health.
Footnotes

