Abstract
For decades, hormone-replacement therapy (HRT) was considered safe and was the first choice in prevention of postmenopausal osteoporosis induced by estrogen deficiency. Numerous experimental and epidemiological studies further supported a protective effect of exogenous female sex hormones on atherogenesis and coronary heart disease (CHD) in women after the menopause. However, the fact that these promising results were not translated into lower incidences of CHD events in hormone-treated women compared with placebo in subsequent, large, randomized studies of healthy subjects as well as women with known CHD raised a very intense debate concerning the safety of HRT in terms of cardiovascular risk. A critical mass of data points toward a protective influence of HRT on cardiovascular disease end points in early postmenopausal women, but increased harm in elderly women, especially those with abdominal adiposity or metabolic syndrome. Once the quasi-hysterical reaction to the largest of the randomized studies (the Women's Health Initiative) has abated, a future strategy should be to concentrate on identifying those relatively few individuals who are not suitable for HRT, as HRT still remains the most thoroughly investigated pharmacological prevention strategy of osteoporosis.
Keywords
Osteoporosis is a bone disease characterized by microdeterioration of the bone architecture leading to an increased risk of fracture. This condition remains a serious disease as it affects millions of individuals worldwide. Estimates of the prevalence of osteoporosis in industrialized countries are that approximately 40% of postmenopausal women are affected at 60 years of age and as many as 70% of elderly women at 80 years of age suffer from manifest osteoporosis [1]. Moreover, the figures show increasing trends as a consequence of improved longevity. Postmenopausal osteoporosis is characterized by a reduction in bone mass due to increased osteoclastic bone resorption and simultaneous although less pronounced increased bone formation (when using biochemical markers of bone turnover) that occurs, especially in the first decade after the menopause, fuelled by a state of chronic estrogen deficiency. These biological processes will result in a net negative calcium balance. A cardinal feature of continuous bone loss is the occurrence of osteoporotic fractures (typically the wrist, the spine and the hip) that may cause considerable and even dramatic changes in the affected individual's health status.
Consequently, the burden of osteoporosis is substantial and serious in terms of morbidity as well as mortality. For example, in North America alone 10% of patients with a vertebral fracture die within the first year, and following hip fracture, as many as 30% die within a year, and another 50% never live independently again [2,3].
As postmenopausal bone loss is initiated and maintained by a chronic state of estrogen deficiency, one of the first logical approaches was to investigate the effect of exogenous estrogens on bone loss occurring after the menopause. For more than six decades, hormone-replacement therapy (HRT) has been used for the prevention of postmenopausal bone loss as it has been reported to significantly inhibit osteoclastic bone resorption. Despite the well-documented effects of estrogen replacement and HRT on bone mass in prevention of vertebral as well as nonvertebral fractures [4], it has been the nonskeletal effects of female sex steroids that have stimulated an intense debate in recent years. In this context, particular attention has been brought to the effect of estrogens on cardiovascular risk in postmenopausal women.
Fracture risk
Typically, bone mineral density (BMD) is measured using dual energy x-ray absorptiometry (DEXA) and the value is expressed in standard deviations (SD) of the mean value of premenopausal women, termed T-scores. As BMD values are normally distributed in pre- as well as post-menopausal women, it follows that the area under the curve for BMD in a given population from-2 SD through +2 SD covers approximately 95% of the population. The clinical implications of the T-score are that for every 1 SD that the BMD value of the spine is below the premenopausal mean value (T-score = 0), the risk of sustaining a vertebral fracture doubles in the individual [5].
Nevertheless, DEX A is not perfect at discriminating individuals who will subsequently sustain an osteoporotic fracture from those who will not. Applying the use of biochemical markers of bone turnover adds a quantitative and dynamic aspect to bone metabolism, reflecting resorption rate as well as formation rate. Furthermore, BMD measurements and assessment of bone turnover by biochemical markers were recently shown to be additive in predicting fracture risk [6]. Thus, by combining an array of verified independent risk fractures, today osteoporotic risk can be predicted with a higher degree of certainty in all individuals and particularly in those where BMD measurements may not alone indicate a significant relative risk.
Several risk factors are presently known for low bone mass and, thus, postmenopausal fracture risk, including belonging to an Asian or Caucasian race, family history of osteoporosis and modifiable risk factors, such as alcohol intake, smoking, certain diets and medications.
Typically, according to the WHO's guidelines, osteoporosis is present whenever the BMD T-score is below-2.5 SD of the premenopausal mean [7]. Depending on individual aspects that should always be taken into account by the patient's physician, this cut-off point will often constitute a guideline for pharmacological intervention. A history or the radiological identification of a nontraumatic low-energy fracture in combination with a T-score greater than-2.5 may, however, lead the physician to choose intervention in order to prevent new fractures from occurring. Presence of known risk factors or an increased bone turnover as assessed by biochemical markers may contribute to this decision. Many patients with manifest osteoporosis will probably require treatment for 10 years or more; however, safety and efficacy data of the majority of drugs available are based on studies of no longer than 3–5 years [8]. Some researchers believe that 4 years of treatment could be optimal for antiresorptive compounds such as bisphosphonates and raloxifene [9], but no consensus on duration of treatment exists as it is often based on an individual basis in practice according to the available results of published data.
Physiologic approach for prevention of osteoporosis: effect of HRT
Menopause is associated with an abrupt decline in the ovarian synthesis and secretion of sex steroids (primarily estrogens and progesterone) and this physiologic cessation in sex hormone production has been demonstrated to accelerate bone turnover with the net result of a negative calcium balance as bone resorption exceeds bone formation [10]. In line with this observation, estrogen-alone replacement therapy (ERT) or combined estrogen plus progestin (also referred to as HRT) have been convincingly demonstrated to significantly inhibit the menopause-induced accelerated bone turnover, evaluated by biochemical markers of bone resorption (e.g., serum C-terminal peptide of Type I collagen [CTX-I]) and formation (e.g., serum bone alkaline phosphatase) [11], which in turn lead to an increase in BMD, the magnitude of which is dependent on the HRT formulation, the dose(s) used, and the duration of therapy. The majority of randomized, controlled, Phase II and III clinical studies of HRT that have been conducted in relation to osteoporosis prevention or treatment have had change in BMD as the primary efficacy parameter, and changes in biochemical markers of bone turnover as the secondary parameter. However, although demanding much larger numbers, more importantly, the decrease in biochemical markers of bone turnover and the subsequent increase in BMD in association with administration of exogenous sex hormones (ERT or HRT) has now been shown to convey protection against osteoporotic fractures in women after the menopause [4,12]. Thus, identifying postmenopausal women with low bone mass at risk of developing osteoporotic fractures is crucial in an adequate prevention program.
HRT & osteoarthritis
Accumulating evidence suggests that estrogens may even confer protection against cartilage degradation in humans [13]. Estrogen has direct effects on various cells in the human articular cartilage or synovium (e.g., chondrocytes) that may be mediated through genomic as well as nongenomic mechanisms as these cells have been found to express estrogen receptors. Moreover, estrogen may affect the joints through indirect mechanisms, including inhibition of subchondral bone turnover thereby protecting the joint from cartilage degeneration. These estrogen effects on the cartilage thus point towards a favorable influence of estrogen replacement in patients with osteoarthritis (OA). In support of this notion, data from the Women's Health Initiative (WHI) study indicated that women on estrogen replacement had less arthroplasty surgery due to OA than women in the placebo group [14]. However, in terms of exogenous estrogens, factors such as dose, route of administration and duration of treatment as well as local conditions in the joint are of importance.
HRT & CVD: the evidence
For more than five decades, abundant animal experimental and observational data have supported the notion that ERT and HRT provide significant protection against atherogenesis and coronary heart disease (CHD). This conviction was primarily based on the multiple effects on cardiovascular risk factors that were observed in connection with exogenous estrogen, which is generally considered to be beneficial on the vasculature. These observations were supported by molecular studies showing that estrogen is also capable of eliciting genomic as well as rapid nongenomic effects on various vascular target cells, including the endothelial cells (with increased production of nitric oxide as a result, causing vasodilation), the vascular smooth muscle cells (causing relaxation), and monocytes (inhibiting conversion into foam cells). In addition to its direct effects, estrogen has also been reported to exhibit indirect influences on cardiovascular risk through its antioxidative properties, as well as effects on lipid and lipoprotein metabolism, glucose/insulin metabolism, the coagulation/hemostasis system, and the autonomous nervous system [15]. Published studies undertaken at cellular, tissue and
Much research on the influence of estrogens has been undertaken in the area of lipids and lipoproteins over the last 25 years. A significant reduction of atherogenous lipids and lipoproteins has been repeatedly reported with HRT [16]. The size of effect, however, depends on the specific HRT regimen, the doses used, the route of administration and duration of treatment.
Typically, oral estrogen replacement lowers total cholesterol levels and LDL-cholesterol, and raises HDL-cholesterol, thus counteracting the changes in the lipoprotein profile that occur in connection with the menopause. In addition, estrogen replacement has consistently been found to significantly decrease serum lipoprotein (a), an independent cardiovascular risk factor [17]. Collectively, these effects point towards a protection against atherogenesis and ultimately CHD events. However, a simultaneous estrogen-induced increase in triglycerides, especially when using conjugated equine estrogens (CEE) [18], is considered less desirable as it may accelerate atherogenesis. However, transdermal addition of 17β-estradiol is known to have a neutral effect on triglycerides. Addition of a progestin to estrogen as commonly required in postmenopausal women with an intact uterus may further modify these estrogenic effects on lipid and lipoprotein metabolism [19]. As for the estrogen component, the progestogenic effect also depends on dose, regimen used (continuous or sequential), and perhaps most importantly, on the nature of the progestin [20,21]. Progestins that are used clinically vary considerably in chemical property, exerting varying degrees of mineralocorticoid, androgenic or glucocorticoid properties [22]. In general terms, the favorable influence of estrogens is somewhat attenuated by addition of a progestin, albeit wide differences are present.
Data from large epidemiological studies have consistently corroborated the experimental data indicating that women who use HRT after the menopause have a significantly lower incidence of fatal as well as nonfatal CHD. For example, following more than 85,000 women (pre- and post-menopausal) for 15 years in the Nurses' Health Study demonstrated that among the postmenopausal women, current use of HRT reduced the relative risk of CHD by 19–32% compared with non-users (after adjustment for age) [23]. However, these prospective epidemiological results were challenged a decade ago by the first results from a large randomized placebo-controlled study in postmenopausal women with diagnosed CHD participating in the Heart and Estrogen Replacement Study (HERS) [24]. To the surprise of most specialists and common people alike, the HERS study found signs of early harm (during the first year) in terms of increased CHD followed by later protection (during the last 3 years). Initial data from another and even larger randomized placebo-controlled study–the WHI, which was designed to last for 7 years – suggested cardiovascular harm (i.e., increased risk of fatal and nonfatal CHD events) in women without overt clinical symptoms of CHD at baseline [4]. However, the WHI study was prematurely stopped when it was indicated that 5.2 years of HRT, but not ERT, promotes cardiovascular harm in postmenopausal women [4]. Nevertheless, a substudy consisting of hysterectomized women within the WHI study who were randomized to CEE only or placebo showed no statistically significant difference in coronary events between groups [25]. Owing to these surprising and ‘negative’ results, there still remains much confusion among laypeople and doctors alike as to the benefits and risks of using HRT for the control of menopausal symptoms or prevention of longterm consequences of the menopause, such as osteoporosis. Interestingly, the WHI authors recently released a new publication that superseded the original 2002 publication [26]. Here it was stated that there was in fact no statistically significant difference between HRT- and placebo-treated women when the years since menopause were taken into account.
HRT & breast cancer
The data available on the effect of HRT on breast cancer risk are conflicting. The large WHI study for combined estrogen plus progestin reported a borderline statistically significant increase in the risk of invasive breast cancer of 26% (38 vs 30 per 10,000 person-years) in the HRT group compared with placebo [4]. Results from another large study of 5692 late postmenopausal women (Women's International Study of long Duration Oestrogen after Menopause [WISDOM] study) reported no statistically significant difference in number of breast cancer cases between women randomized to HRT or placebo after 12 months (the study was prematurely stopped during ongoing recruitment after the publication of WHI data in 2002) [27]. Combining the WISDOM data with the WHI data has, however, shown that after 1 year of treatment, the hormone-treated women had a statistically significant lower incidence of breast cancer than placebo; however, this was reversed after 4 years of treatment, with a statistically significant trend over time for increased risk of breast cancer [28]. Subsequent analysis of the WHI study revealed that cumulative rates of invasive breast cancer in HRT-treated women increased more rapidly than in the placebo-treated ones after 3 years in those who had previously been using HRT, and after 5 years in those who had never used HRT before. Interestingly, the data for the WHI substudy (estrogen only users vs placebo) showed a statistically significant decreased incidence of invasive breast cancer in ERT-treated women compared with placebo [20], suggesting that the progestin (e.g., medroxyprogesterone acetate [MPA]) may play a role in the development of malignant breast neoplasia. A recent systematic review of the literature using risk models for estimating breast cancer risk among women taking HRT in prospective studies found that estradiol was associated with a 2—2.9-fold increased relative risk of developing breast cancer, comparing the highest with the lowest quintile of estradiol (p < 0.01) [29]. However, it remains unknown whether the increased risk of breast cancer using HRT is because of a
Timing of HRT in relation to menopause
Since the publication of the WHI data in 2002, considerable controversy exists in the scientific community towards the harm/benefit ratio of postmenopausal HRT. It has been argued that the women participating in the WHI study, who were almost 15 years after the menopause at baseline and of which the majority were overweight, were in fact not those who would choose HRT in ‘real life’ because of their lack of menopausal symptoms [4]. Furthermore, the use of MPA as the progestational agent was criticized, despite being the most widely used in the world. Nevertheless, today the general belief is that HRT – regardless of regimen, duration and the women taking it – is associated with an increased risk of CHD and stroke in all post-menopausal women.
A series of well-conducted experimental studies in ovarienctomized and fat-fed cynomolgus monkeys and rabbits (the two species that have been most utilized for experimental atherogenesis) have shown virtually similar effects of exogenous female sex hormones on atherosclerosis. In general, when estrogens are introduced after ovariectomy in animals that have been fed a normal, not fat-rich diet before becoming menopausal, but a fat-rich diet thereafter (in combination with estrogen), there was a 70% inhibition of coronary artery atherosclerosis. However, if instead of normal diet the animals were fed a fat-rich diet while being premenopausal and this diet continued after ovariectomy where it was given in combination with estrogen administration; then there was a 50% reduction in coronary artery atherosclerosis. Interestingly however, when the cholesterol-rich diet was given before as well as after ovariectomy, but estrogen replacement was postponed to a time occurring several months after surgery and then introduced in combination with a normal diet for the remainder of the study, then the effect of estrogen replacement on coronary artery atherogenesis was negligible [31–34]. Thus, collectively, these data suggest that timing of HRT is crucial in terms of the degree of atherosclerosis prevention obtained: when introduced early after becoming menopausal, HRT has a pronounced inhibitory effect on atherosclerosis, but this effect diminishes as the time elapses between menopause transition and initiating the hormone replacement. In support of this notion, late introduction of HRT in elderly postmenopausal women, especially in the presence of cardiovascular risk factors, is currently believed to augment destabilization of prevalent atherosclerotic plaques through increased inflammatory responses [35] and activation of matrix metalloproteinases (MMPs) within the plaque [36].
Extrapolating the monkey data from Clarkson's group to the human situation, suggesting an approximate 3:1 ratio of monkey to human years, this would imply that 6–7 years after the menopause, the beneficial effects of HRT on atherogenesis abate. This would account for the limited influence on coronary events in the WHI study participants more than 10 years after menopause [26].
These important observations may help to explain, at least in part, the results of the large randomized placebo-controlled WHI and HERS studies that failed to demonstrate any overall beneficial effect of HRT on CHD events in these late postmenopausal women [4,24]. Interestingly, a recent more detailed analysis of the WHI data revealed that when split into decades after the menopause, for women who were 0–10 years since menopause, the hazard ratio (HR) for CHD was 0.76 (95% CI: 0.501.16), in women 10–19 years since menopause the HR was 1.10 (95% CI: 0.84-1.45); and in those with 20 or more years since menopause, the HR was 1.28 (95% CI: 1.03–1.58; p for trend = 0.02) [26]. However, HRT was found to increase the risk of stroke in this study (HR: 1.32; 95% CI: 1.12–1.56).
Who are candidates for postmenopausal HRT & who are not?
Recent evidence suggests that the potential harm of HRT is linked to a certain body phenotype or metabolic representation with respect to body fat distribution. Another important issue relates to the progestin itself. Beneficial effects of HRT have been demonstrated on glucose metabolism by improving insulin sensitivity in nonobese postmenopausal women [16], thus inhibiting the tendency towards more abdominal adiposity and higher circulating concentrations of atherogenic lipoproteins that occur after the menopause. However, in women with abdominal adiposity or metabolic syndrome, the concentration of free estradiol and of IL-6 (a marker of low-grade inflammation) are notoriously elevated. Consequently, treatment with HRT will increase the concentration of free estradiol even further, in addition, some progestins appear to also lower adiponectin concentrations, both of which will nurture atherogenesis and eventually the risk of cardiovascular events in these subjects [37]. Hence, harm may arise from inappropriate use of HRT in postmenopausal women with an intrinsically elevated risk due to their phenotype. In addition, high doses of HRT may further promote thromboembolic events as a result of increased coagulation activation and adverse vascular remodeling, suggesting that some women may not benefit from HRT after the menopause.
The pathophysiologic explanation behind this contrasting influence of central and peripheral fat tissue on atherogenic risk factors and on atherogenesis may be explained by decreased plasma adiponectin concentrations, increased serum-free estradiol concentrations, lower insulin sensitivity, and a chronic inflammation present in abdominally obese women compared with peripherally obese women. Consequently, abdominally obese postmenopausal women probably should avoid hormone therapy, and the relative contraindication is further stressed in the presence of metabolic syndrome and advancing age [37].
Support for a difference in response to HRT depending on a person's metabolic representation in terms of body fat distribution came from a recent analysis of the results from the Women's Angiographic Vitamin and Estrogen (WAVE) study by Howard and colleagues [38], which pointed out some interesting differences in the progression of CHD during 2.8 years of HRT depending on whether participants had a normal or an abnormal glucose tolerance test at baseline, the latter being a metabolic condition closely associated with central (abdominal) adiposity. Although in subjects with abnormal glucose tolerance test levels there was no increase in C-reactive protein in the HRT-treated women and a decrease in placebo-treated women (p < 0.05), and even a favorable change in serum lipoproteins in HRT-treated compared with placebo-treated women (p < 0.05), women with abnormal glucose tolerance had accelerated atherosclerotic progression that was further enhanced by HRT.
Progestational component: does it have any cardiovascular impact?
We have recently investigated the influence of the progestational component used on the circulating levels of adiponectin, an adipokine with insulin-sensitizing, anti-inflammatory and anti-atherogenic properties [39]. We found that at least some progestins (e.g., drospirenone) exert an inhibitory effect on circulating adiponectin concentrations, which could be a plausible mechanism of action underlying the adverse cardiovascular response, especially if exposing women who have low serum levels of adiponectin (i.e., women that are centrally obese). By contrast, when nonobese women take HRT for a period of 2–3 years after the menopause, both all-cause and cardiovascular mortality is significantly reduced compared with nonusers [40].
To investigate the impact of postmenopausal HRT on human atherogenesis and all-cause and CHD mortality, we invited women who had previously been screened for or randomized to various double-blind, placebo-controlled HRT studies of 2–3 years' duration. At this follow-up visit, which occurred approximately 9.8 years after the initial contact, 1284 of the original 1458 postmenopausal women for whom baseline data were complete were alive while 174 had died during the follow-up period. HRT used in the previous clinical studies consisted of oral regimens (estradiol or estradiol valerate combined with one of various progestins). The main finding of this Prospective Epidemiological Risk Factor (PERF) study was that all-cause mortality among women assigned to 2–3 years of HRT was reduced by 30% compared with women who were not assigned to hormones (HR: 0.70; 95% CI: 0.50–0.97) even after adjusting for age and body mass index (BMI). Similarly, cardiovascular mortality was reduced by 46% (HR: 0.54; 95% CI: 0.29–0.98) among women who had previously taken HRT compared with those who had not. Severity of lumbar aortic calcification – a surrogate measure of atherosclerosis – was significantly less in hormone-treated compared with nonhormone-treated women (by 9.7%; p < 0.0001) having adjusted for age and smoking. Thus, our data indicate that even a relatively few years of HRT after the menopause may retard atherogenesis and positively affect cardiovascular mortality [40].
Why did our results differ from those reported by the WHI authors? There may be several reasons, but some important issues may in fact relate to differences in mean age (63.2 vs 55.8 years for WHI and PERF, respectively), in BMI (28.5 vs 25.3 kg/m2 for WHI and PERF, respectively), and in the HRT regimen used (WHI, CEE plus MPA as commonly used in the USA vs PERF, estradiol plus norethisterone acetate or levonorgestrel as commonly used in Europe). In addition, differences in study design may account for the contrasting outcome. As a result of the ‘negative’ WHI outcome – even in the light of a follow-up report stating that the women in the WHI cohort who sustained the CHD events in the HRT and the placebo groups were also those who at baseline had subclinical low-grade inflammation, a risk marker of subsequent CHD events [41], as well as the recently published WHI breakdown into years since menopause [26], it is unlikely that a similar, massive, randomized, placebo-controlled HRT trial on CHD mortality and morbidity of similar proportions as the WHI study but performed in a ‘young’ (i.e., early postmenopausal) cohort of (clinically as well as biochemically) healthy women will ever be undertaken again. The considerations for such a trial also would be numerous, and include BMI range and metabolic phenotype, type of HRT (oral vs transdermal), type and dose of progestin(s), duration of treatment, age groups, presence of known risk factors for osteoporosis and exclusion of familiar disposition for gynecological cancers. Nevertheless, a smaller yet pertinent randomized study has been designed and is currently ongoing in order to address these relevant clinical issues. The Kronos Early Estrogen Prevention Study (KEEPS) is a 5-year randomized study that will investigate the effectiveness of low-dose oral estrogen and transdermal estradiol in preventing progression of coronary artery atherosclerosis in recently postmenopausal women [42]. However, it should be kept in mind that the study is not comparing equivalent doses of estrogen as the equivalence is based on the effects on urinary calcium excretion [42], which is likely to limit the relevance of the results. Furthermore, the end point in this study is also limited to surrogate markers rather than fatal and nonfatal coronary events. Nevertheless, time will show whether the ‘years since menopause’ hypothesis can be confirmed by the KEEPS study.
Progestins, administered in combination with estrogen primarily to avoid endometrial hyperplasia and cancer, vary considerably in their physiological and metabolic effects [20–22]. Endothelial synthesis of nitric oxide (NO) is a strong vasodilator and is crucial in preventing endothelial dysfunction and inhibition of atherogenesis [43]. Evidence supports a concern for some, but not all, progestins, in particular for MPA. In contrast to progesterone, which appears to have a neutral influence on the anti-atherosclerotic effect of estrogen [44], MPA has a negative effect [45], including vasocontraction rather than dilation and promoting acceleration of plaque formation and thrombus formation – effects that collectively block the cardioprotective effects of estrogen [45,46]. By contrast, other progestins, such as dydrogesterone, seem to have opposite effects on NO production than MPA [47], such as a favorable influence on atherogenesis and lipoprotein metabolism, but more data are needed to determine the effect of this and other progestins on hard coronary end points and cardiovascular mortality.
Based on the data presently available, an essential question to ask is: can we identify the women least likely to benefit from HRT after the menopause? Accumulating evidence suggests that early initiation of HRT (i.e., in the menopausal transition), is crucial for full benefit of the therapy, and that delaying this onset of HRT in the years following the menopause markedly reduces the cardioprotective influences [48]. Hence, the benefit/harm ratio of HRT may vary from being primarily beneficial when started in the early menopause towards largely harmful if begun in the late menopause. However, regardless of time of HRT onset, body fat distribution
HRT & fracture prevention
Results from the large randomized WHI study also included a significant protection of fractures (vertebral and non-vertebral) in postmenopausal women [4]. Interestingly, the subjects of this study were not women typically at risk of developing osteoporosis as most were overweight and 63 years of age at baseline, and furthermore they were not selected for the study to have low bone mass. Yet even in this cohort of elderly women, HRT prevented osteoporotic fractures.
Currently, HRT is no longer prescribed as the treatment of choice for osteoporosis prevention mainly owing to the effect of the initial WHI publication (2002) and other safety concerns associated with long-term HRT use, such as breast cancer [51]. Nevertheless, HRT remains most frequently used for the control of menopausal symptoms impairing a woman's quality of life and less frequently nowadays for prevention of long-term consequences of estrogen deficiency (e.g., osteoporosis). Thus, a majority of women with menopausal symptoms will use HRT only for a few years around the menopause to alleviate these symptoms [52]. Therefore, we found it was interesting to raise the question of long-term prevention of osteoporosis in women who had taken HRT for a period of 2–3 years after the menopause (at various years after the menopause). Data from the large PERF cohort of 347 postmenopausal women, followed 5, 10 and 15 years after stopping HRT, revealed that long-term prevention can be obtained by relatively few years of HRT in the early years after the menopause or even later (Figure 1) [12]. After stopping HRT following 2–3 years of treatment, the decline in bone mass was heterogeneous, thus women with a more accelerated bone loss were also at a higher risk of sustaining a fracture. Therefore, it is crucial to identify those individuals with an increased rate of bone loss after cessation of HRT in order to consider alternative treatment modalities for future prevention of osteoporosis. The data by Bagger and colleagues may have far-reaching socioeconomic consequences as the results clearly indicate that use of HRT for even relatively few years after menopause may have a long-term impact on bone health for as long as 10 years. Hence, timely prevention of osteoporotic fractures by simple and short-term means can translate into considerable and longterm benefits, even in woman demonstrating accelerated postmenopausal bone loss and, thus, increased risk of osteoporosis.
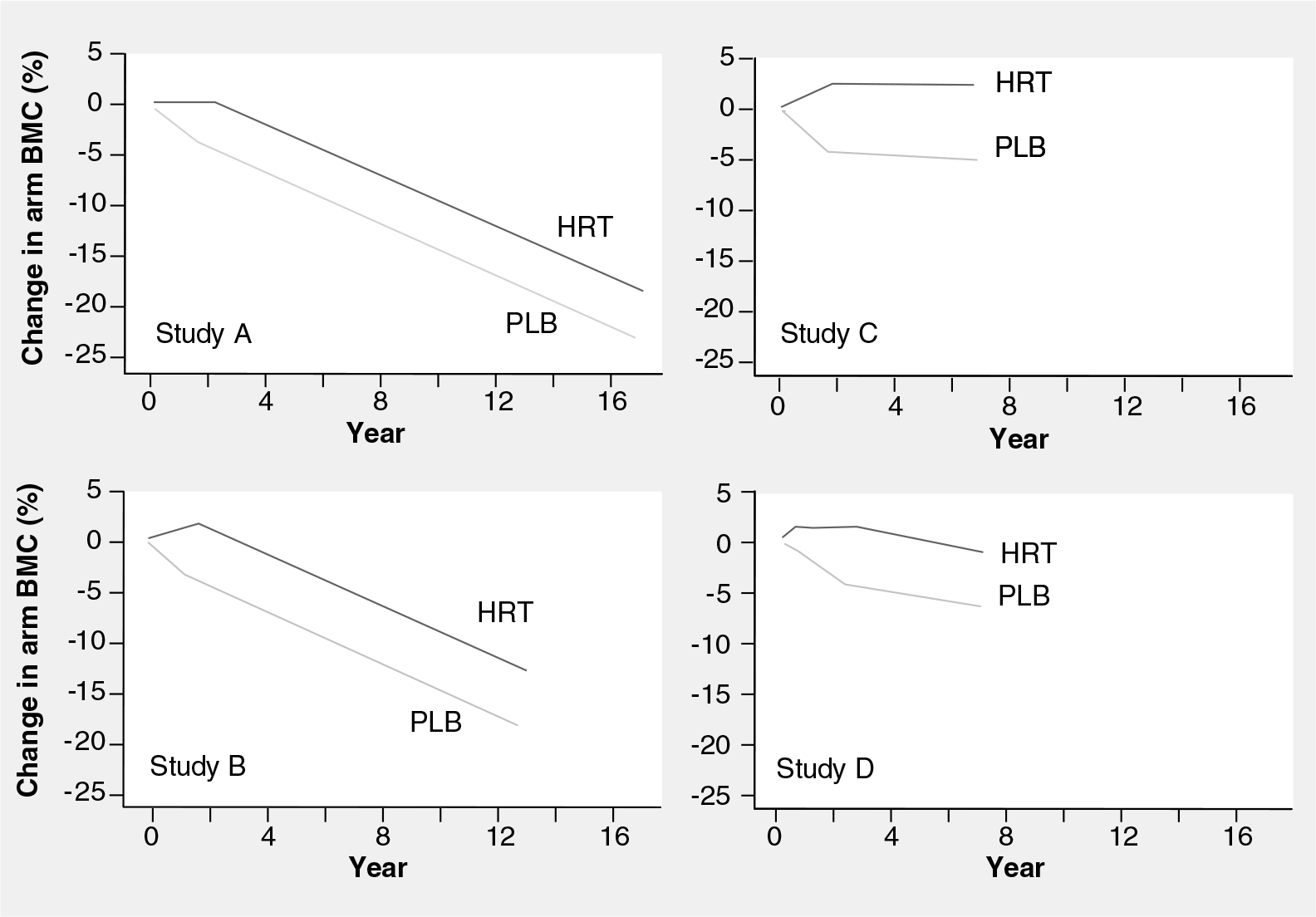
Longitudinal changes in distal forearm bone mineral content from baseline during randomization to 2–3 years of hormone-replacement therapy or placebo, and a number of years later at a follow-up determination.
Conclusion
In conclusion, observational studies consistently found that women who choose to use HRT had a reduction in mortality and CVD incidence relative to women who did not use HRT. Randomized, controlled trials have demonstrated that initiation of HRT in elderly women (>60 years of age) who are more than 10 years since menopause is potentially associated with more risk than benefit. In addition, randomized, controlled trials have confirmed observational studies indicating the safety and benefit of HRT in younger (< 60 years of age) and recently menopausal women (<10 years since menopause). Thus, there seems to be a rationale for a caveat concerning initiation in elderly women. For nonobese women less than 60 years of age and with no history of gynecological cancers, HRT still appears to be a logical and safe therapy for the prevention of CVD and osteoporosis.
In terms of body composition and in particular body fat distribution, there is emerging evidence suggesting that obese women (especially abdominally obese women) or women with metabolic syndrome receiving HRT probably have an increased risk of CVD events compared with women without this metabolic representation [37,50].
Thus, in elderly women or women with abdominal obesity and possibly also with metabolic syndrome [50], preventive pharmacological strategies other than HRT need to be considered, of which many are on the market and still more are likely to be available within the next 5 years. The progestational component of HRT may play a significant role in the relative risk of CVD events
Future perspective
The WHI publications caused an abrupt and pronounced decline in the sales of HRT across the globe, regardless of type, regimen, doses and the women that this treatment was intended for. Some 7 years later, after a massive criticism and a heated debate about the WHI study design and especially the results, leaving many menopausal women untreated or at least not treated with HRT owing to a generalized fear among many physicians to prescribe estrogens regardless of the obvious indications, slowly there appears to be basis for a new consensus of how and when to use HRT based on a more detailed picture of the benefit/harm ratio in postmenopausal women. Presently, a history of breast cancer or other gynecological cancers is usually regarded as a relative contraindication of estrogen use in early postmenopausal women. Likewise, in elderly women with a history of symptomatic CHD, considering a prescription of estrogens or HRT today happens only after careful consideration of the pros and cons and is done with caution. Nevertheless, in early as well as late postmenopausal women, being abdominally obese (i.e., a high waist:hip ratio) or having signs of metabolic syndrome should probably also be regarded as relative contraindications of oral estrogen use, whereas parenteral estrogens seem to have a neutral effect [50]. For this purpose, it may be useful to measure the waist circumference (should not exceed 88 cm in women), which could be combined with a panel of parameters such as blood pressure, fasting triglycerides and blood sugar levels, sex hormone-binding globulin, and inflammatory markers. For women with an intact uterus, if HRT is regarded as relevant, ultra-low-dose ERT without addition of a progestin seems an interesting and promising option [53,54], but this particular therapy warrants further investigation. For hysterctomized women, ERT either as patch or oral treatment, is a useful option, but the metabolic differences among the different progestins should also be taken into account. The intention, dose and regimen of HRT must be individualized, based on the principle of choosing the lowest appropriate dose in relation to the severity of symptoms and the time and menopause age. Estrogen-like compounds, such as tibolone or selective estrogen-receptor modulators, still need to be further validated in terms of their long-term cardiovascular benefit/harm ratio in postmenopausal women at high risk of osteoporosis.
Executive summary
Hormone-replacement therapy (HRT) has previously been extensively used for alleviation of menopausal symptoms and for long-term prevention of osteoporosis.
More than 50 years of abundant experimental and epidemiological studies have consistently shown that HRT prevents atherogenesis and the risk of coronary heart disease is less in women using HRT after the menopause than women who do not use it.
Initial published results from the large randomized and placebo-controlled Women's Health Initiative (WHI) study (2002) did not confirm a benefit of HRT in terms of cardiovascular outcomes, in fact there was an increased risk of myocardial infarction in women randomized to HRT compared with placebo (p < 0.05).
A subsequent report from the WHI group (2007) has, however, confirmed a recent hypothesis that women close to the menopause benefit from HRT, but as the time distance to menopause increases so does the cardiovascular risk associated with HRT (due to plaque instability and low-grade inflammation in elderly women).
Specific phenotypes (abdominal adiposity and metabolic syndrome) seem to be caveats for use of HRT. Certain progestins may also be associated with great cardiovascular risk when given in combination with estrogen.
Despite this more detailed understanding of HRT in relation to years since menopause, this initial WHI report directly caused an abrupt, pronounced and global drop in sales of HRT regimens alike. This is slowly changing again with a more detailed understanding of the importance of timing of the start of HRT.
Estrogen-like compounds such as tibolone and selective estrogen-receptor modulators are currently being developed and investigated in lieu of HRT; however, long-term data on cardiovascular risk are sparse. Unopposed, ultra-low-dose estrogen may provide a useful alternative, but further studies are warranted in terms of cardiovascular risk assessment. HRT probably has no role in existing coronary heart disease.
Footnotes
