Abstract
Systematic reviews of randomized, controlled trials have shown that use of hormone replacement therapy in women increases the risk of total cardiovascular disease (venous thromboembolism, stroke and coronary heart disease; there may also be an increased risk of peripheral arterial disease). The relative increase in risk is similar to that for use of combined oral contraceptives, but the absolute increase in risk is higher owing to the higher age of hormone replacement therapy users. The increased risk appears confined to current users, increases with age and obesity, and may differ with type of preparation. Transdermal hormone replacement therapy may carry a lower risk of venous thromboembolism compared with oral hormone replacement therapy. The mechanism for the increased risk is probably activated blood coagulation. The risk of venous thromboembolism is higher in women with thrombophilias; however, the value of screening for thrombophilias is not established. Further research is required to establish the risks of cardiovascular (and other) diseases in different groups of women for different types of hormone replacement therapy, including an individual participant meta-analysis of reported randomized, controlled trials.
What is the overall risk of cardiovascular disease in women using HRT compared with nonusers?
While it is commonly believed by both doctors and their patients that coronary heart disease (CHD) accounts for most cardiovascular disease (CVD) events in middle-aged women, Table 1 shows that CHD accounts for less than half of CVD events in those who do not use hormone replacement therapy (HRT) or other hormonal preparations. This is also observed in recent whole-population studies of CVD events [1]. In addition to stroke and peripheral arterial disease (PAD), venous thromboembolism (VTE) is common. Two-thirds of cases of VTE present as deep-vein thrombosis (DVT) and a third as pulmonary embolism (PE).
Relative risks and estimated absolute risks of cardiovascular events in users of hormone replacement therapy over 5 years, compared with nonusers.

From [2].
From [4]. CHD: Coronary heart disease; CI: Confidence interval; CVD: Cardiovascular disease; f.o.d.: Fatal or disabling; HRT: Hormone replacement therapy; MI: Myocardial infarction; VTE: Venous thromboembolism.
By 2004, systematic reviews of randomized, controlled trials (RCTs) (which provide the least biased estimates of risks and benefits) had established that use of HRT increased the risk of VTE and stroke, and was not protective against CHD [2–4]. The relative risk of VTE in HRT users is approximately 2.0 (95% confidence interval [CI]: ~1.5–3.0) [2]. The relative risk of stroke is 1.23 (95% CI: 1.06–1.44), and of fatal or disabling stroke is 1.56 (95% CI: 1.11–2.20) [4]. This increased risk was confined to ischemic stroke and was not apparent for hemorrhagic stroke [4]. These increased risks appear confined to current users of HRT.
The absolute increases in risk of CVD events can be estimated from these overall relative risks, and from the expected rates in nonusers of HRT [5,6] (
For women aged 50–59 years not using HRT, 13/1000 (1.3%) will experience a CVD event over 5 years. In HRT users of this age, the absolute risk increases by approximately 30% to 17/1000 (1.7%), comprising two additional cases of DVT, one of PE, and one of stroke (with a 50% chance that it will be fatal or disabling). For women aged 60–69 years not using HRT, 33/1000 (3.3%) will experience a CVD event over 5 years. In HRT users of this age, the absolute risk increases by approximately 30% to 43/1000 (4.3%), comprising five additional cases of DVT, three of PE and two of stroke (one of which will be fatal or disabling) (
The number needed to harm from a CVD event by prescribing HRT for 5 years is therefore 250 for women aged 50–59 years, and 100 for women aged 60–69 years. To such harms should be added the increased risks of endometrial and ovarian cancer (approximately 6 cases over 5 years for unopposed estrogen HRT), the risk of breast cancer (approximately 6 cases over 5 years for estrogen–progestin HRT), and the increased risk of dementia [5,7]. Benefits of HRT include on-treatment relief of menopausal symptoms, on-treatment reductions in bone loss and risk of fractures, and a reduced risk of colon cancer in users of combined HRT [5,7].
Meta-analyses of RCTs have shown that HRT does not increase mortality, whether cardiovascular, cancer or total [8,9]. However, doctors need to inform their patients considering HRT regarding the cardiovascular, cancer and other risks of HRT preparations. This review updates a previous review of CVD risk [2], and focuses on CHD and PAD (final reports on these were recently published from the Women's Health Initiative [WHI] trial of unopposed estrogen [10,11]), mechanisms for increased cardiovascular risk, risks of different types of HRT, and risk factors for CVD in HRT users.
Coronary heart disease & peripheral arterial disease
Women have a lower risk of CHD compared with men of similar age, and it has been suggested that this may reflect a protective effect of estrogen in the arteries of premenopausal women. It was therefore hypothesized that estrogen ‘replacement’ with HRT would continue such ‘premenopausal protection’, and animal studies suggested that estrogens might reduce the progression of early atherosclerosis [7]. Observational studies also suggested that women using HRT had a lower risk of CHD [7,12]. However, over the past 10 years epidemiological, animal, clinical and (above all) RCT studies have challenged these hypotheses and conclusions [2,7]:
Gender differences in CHD risk may not necessarily reflect estrogenic mechanisms in women, but rather androgenic mechanisms in men, such as postpubertal reductions in ‘protective’ high-density lipoprotein cholesterol, and increases in hematocrit and blood viscosity [2,7];
There is no change in the gender difference in incident CHD around the menopause, hence the ‘postmenopausal increase’ in risk of CHD in women is a myth [2];
Oral HRT preparations are not ‘replacement of natural estrogen’. Similar to COCs, they have ‘first-pass’ effects on plasma protein synthesis in the liver, which increase activation of blood coagulation and possibly inflammation. Hence, they are prothrombotic and proinflammatory drugs which, like COCs, increase the risk of venous and arterial thrombosis [2];
Both animal studies and human angiographic trials have shown that HRT does not reduce the progression of later atherosclerotic lesions, which are frequently present in women considering HRT [2,7];
Observational studies are systematically biased, leading to an erroneous estimate of reduced CHD risk. In particular, such studies have incomplete capture of early CHD events [2,7].
It has taken a long time to establish, perform, and report long-term RCTs (which minimize these biases in observational studies) and to establish in meta-analyses of these trials that HRT increases the risk of VTE [2] and stroke [4], and does not protect against CHD [3].
In 2004, a provisional meta-analysis of all RCTs of HRT observed that the relative risk of CHD was 1.10 (95% CI: 0.95–1.27) [3]. Table 2 summarizes these trials and their results. The two largest primary prevention trials were performed in the USA: the WHI trial of oral, opposed HRT (conjugated equine estrogens [CEE] plus medroxyprogesterone acetate [MPA]) in nonhysterectomized women [13–15], and the WHI trial of oral, unopposed estrogen HRT (CEE) in hysterectomized women [10,11,16]. The latter trial was stopped due to the increased risk of stroke [16], and its final results for CHD and PAD were published only recently [10,11]. Few data are available from a meta-analysis of trials reported before 1997 [17], or from the Women's International Study on long-Duration Oestrogen after Menopause (WISDOM) study, which was stopped in 2002 after publication of the WHI results. The largest secondary prevention trial was the Heart and Estrogen/Progestin Replacement Study (HERS), which was also performed in the USA and tested oral opposed HRT (CEE plus MPA) [18]. The results of other secondary prevention trials are summarized in Table 2 [19–22].
Randomized, controlled clinical trials of hormone replacement therapy for prevention of cardiovascular disease.
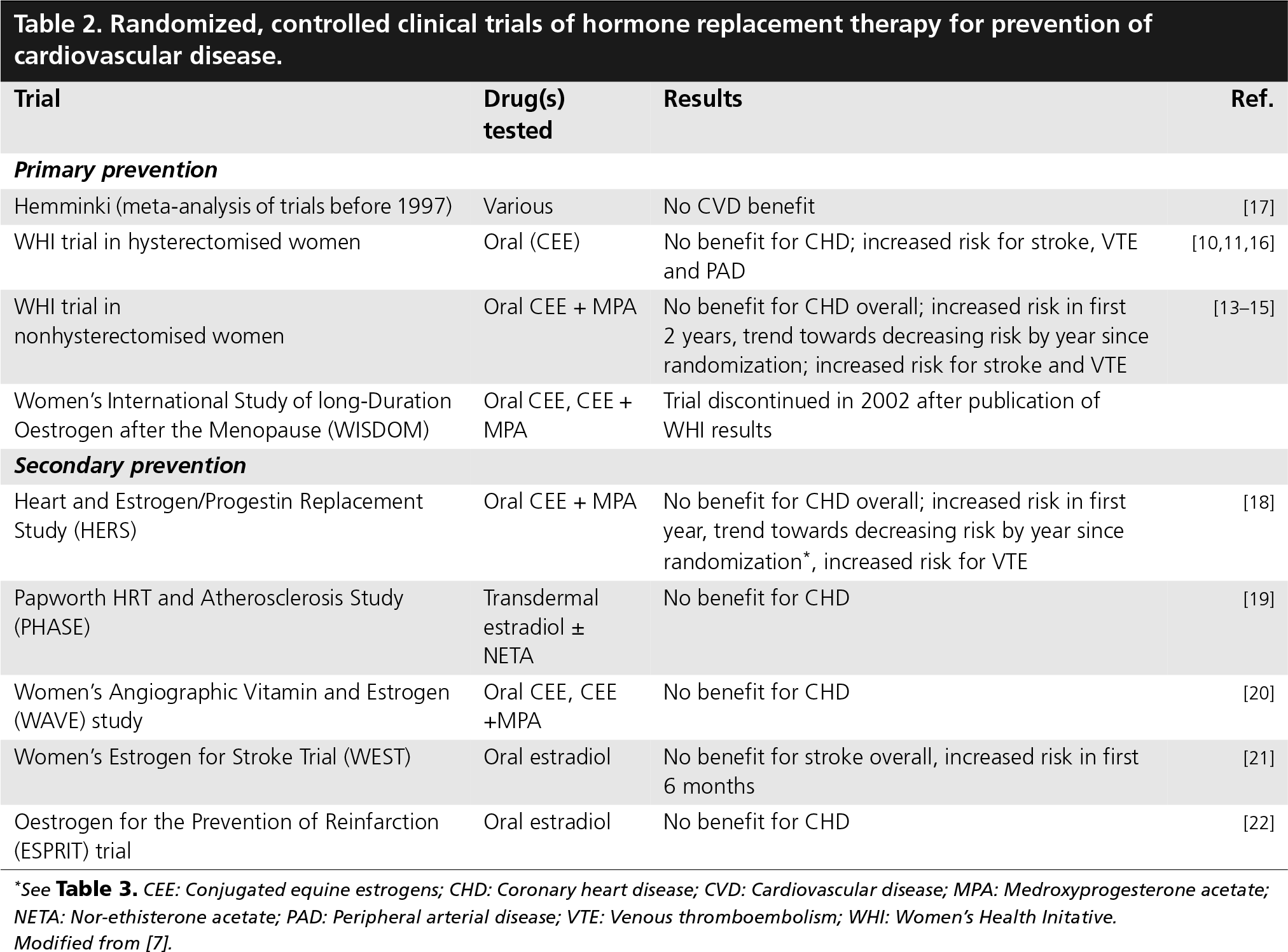
See
Summary of cardiovascular risks in the WHI and HERS randomized, controlled trials of conjugated equine estrogens with or without medroxyprogesterone acetate.
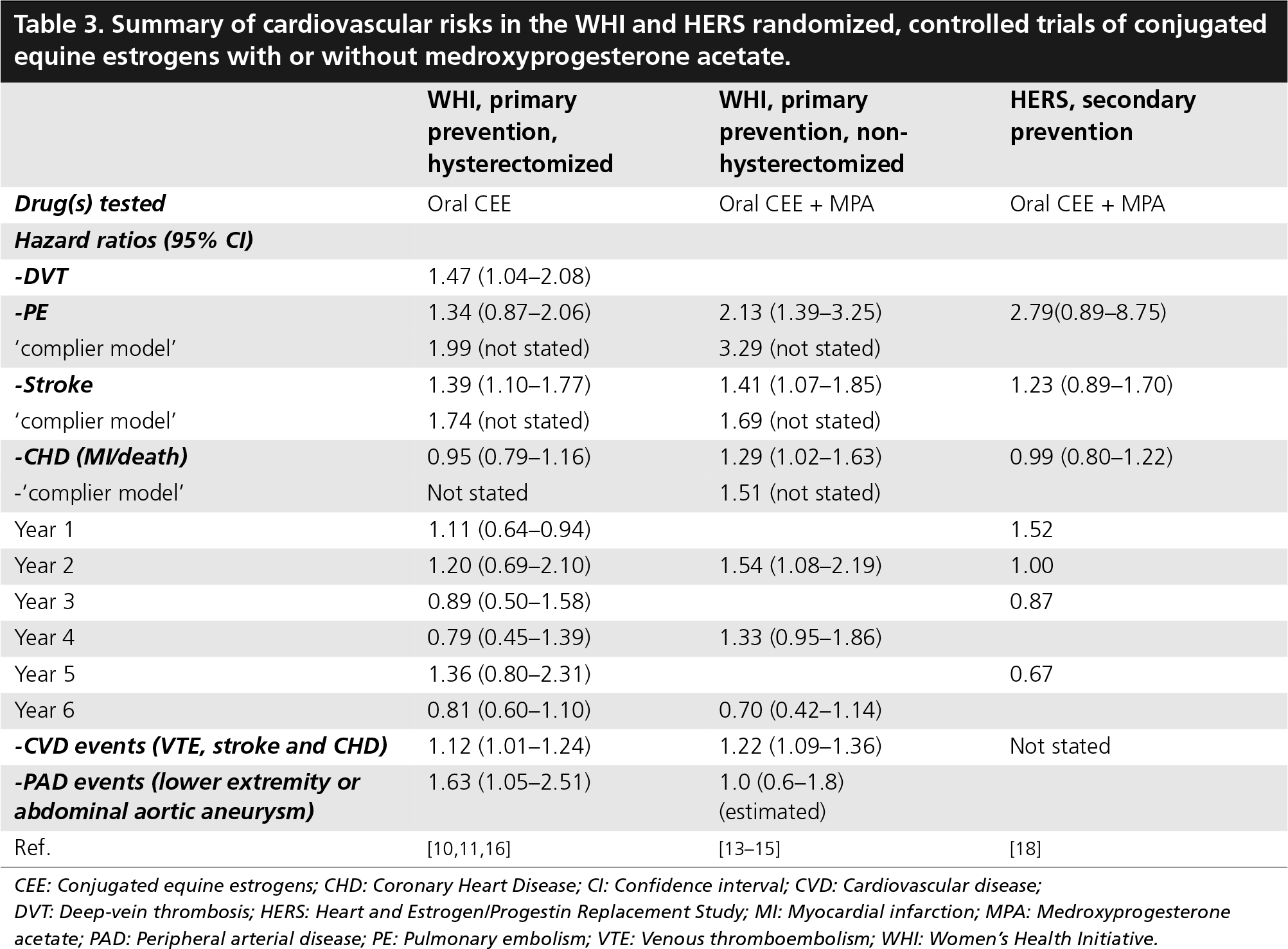
CEE: Conjugated equine estrogens; CHD: Coronary Heart Disease; CI: Confidence interval; CVD: Cardiovascular disease
DVT: Deep-vein thrombosis; HERS: Heart and Estrogen/Progestin Replacement Study; MI: Myocardial infarction; MPA: Medroxyprogesterone acetate; PAD: Peripheral arterial disease; PE: Pulmonary embolism; VTE: Venous thromboembolism; WHI: Women's Health Initiative.
Table 3 summarizes results from the WHI and HERS trials. For VTE, stroke and CHD, these more recent results remain consistent with those of meta-analyses of all RCTs [2–4,8]. In both of the WHI trials, the risk of total CVD events was significantly increased, with substantial overlap between their 95% CIs; hence the increased cardiovascular risk appears similar for unopposed and opposed HRT. In the two WHI trials, the risks of PE and stroke were higher in a ‘complier’ model, which incorporated adherence to allocated treatment. For CHD events, a significant increase in risk (hazard ratio: 1.29; 95% CI: 1.02–1.63; 1.51 in a ‘complier model’) was reported in the WHI trial of combined CEE and MPA [13,14]. However, the WHI trial of CEE did not report a ‘complier model’ for CHD. It should be noted that in the final follow-up of the other two trials the upper 95% CIs for the hazard ratio were 1.16 and 1.22 respectively [10,18]; and in the provisional meta-analysis of all RCTs this was 1.27 [3]. Hence, from current data, an overall 27% increase in relative risk of CHD in HRT users in randomized trials cannot be excluded. Conversely, the lower 95% CI for the hazard ratio in the meta-analysis was 0.95 [3], hence, from current data, an overall 5% decrease in risk of CHD in HRT users cannot be excluded. Nevertheless, there are convincing data suggesting that total cardiovascular risk increases with both unopposed and opposed HRT.
While the above data refer to the total risk in CHD over trial duration, an early increase in risk (‘starter effect’) was shown for CHD in the WHI trial of combined CEE and MPA, and in the HERS trial of combined CEE and MPA, but not in the WHI trial of unopposed CEE (
Hazard ratios (95% confidence intervals) for cardiovascular disease events in the Women's Health Initiative trial of conjugated equine estrogens and medroxyprogesterone acetate, as a function of years from randomization.

Cox regression analyses stratified on age, presence or absence of prior coronary event, and randomly assigned diet modification group, and adjusted for previous coronary artery bypass grafting or percutaneous transluminal coronary angioplasty
Cox regression analyses stratified on baseline age in 5-year intervals (50–54, 55–59, 60–64, 65–69, 70–74 and 75–79). Note that a hazard ratio below one would not necessarily imply risk reduction in this time period because high-risk hormone replacement therapy users have been differentially included from follow-up by the risk elevation at earlier times from randomization. Reproduced with permission from [53].
The final report on CHD of the WHI trial of unopposed CEE reported a large number of subanalyses: four primary outcomes and nine secondary outcomes were reported, followed by further analyses of eight outcomes by three baseline age groups (50–59, 60–69 and 70–79 years) [10]. Prominence in this report (and in its abstract and discussion) was given to lower hazard ratios for some outcomes in the age-group 50–59 years. However, extreme caution is required due to the large number of subanalyses in this report, resulting in a high probability of false-positive (or -negative) findings, as recently pointed out in a statistical commentary on another randomized trial (adding the antiplatelet agent clopidogrel to routine use of aspirin in prevention of CVD) [23].
The final report on CHD of the WHI trial of unopposed CEE also compared its results for CHD (in the discussion part of this paper) to those of the WHI trial of CEE and MPA [14], adjusting for differences in baseline variables between these two trials [10]. The authors reported that the hazard ratio for CHD was lower for CEE compared with CEE plus MPA (p = 0.03) and concluded that this “difference in CHD risk in the two trials is unlikely to be due to chance or to these baseline risk characteristics”. This further, data-derived subanalysis likewise requires cautious interpretation, especially as the results are not presented in detail.
The final report on CHD of the WHI trial of unopposed CEE also did not update, highlight or discuss its previous report that the risk of total cardiovascular disease (VTE, stroke and CHD) was increased (hazard ratio: 1.12; 95% CI: 1.01–1.24) in the HRT arm of the study (
To clarify the increased relative risks of all CVD events (and other major events, e.g., cancers, dementia) in HRT users (
How does HRT increase the risk of cardiovascular events?
Venous thromboembolism is a multicausal disease, resulting from interactions between multiple genetic and environmental variables, in which blood coagulation and its activation play a central role [26]. Like COCs, oral HRT increases activation of blood coagulation by altering hepatic synthesis of coagulation factors and coagulation inhibitors [2]. Increased synthesis of coagulation factors VII and IX [2] may be relevant, because elevated plasma levels of these factors have been associated with increased risk of VTE in some epidemiological studies [27]. Decreased synthesis of coagulation inhibitors (antithrombin, protein S, protein C and tissue factor pathway inhibitor) [2] may also be relevant, because decreased plasma levels of these inhibitors have also been associated with increased risk of VTE in some epidemiological studies [27]. Increased levels of coagulation activation markers (e.g., fibrin D-dimer) may also be relevant, because increased plasma levels have also been associated with risk of VTE in epidemiological studies [27].
One measure of reduced coagulation inhibition is resistance to activated protein C (APC), a natural anticoagulant that limits fibrin thrombus formation, particularly in the venous circulation. APC resistance has been associated with increased risk of VTE in some epidemiological studies [27]. As in pregnancy and use of COCs (each of which is associated with an increased risk of VTE, stroke and early CHD), use of oral HRT is associated with APC resistance [28–34]. The mechanisms are unclear, but may include changes in protein S and tissue factor pathway inhibitor [32] or in coagulation factor VIII [34]. Because APC resistance has been associated with several other risk factors for VTE (genetic thrombophilias including the factor V Leiden and prothrombin
Consistent with these hypotheses, the risk of VTE in women using HRT is increased in women with thrombophilias such as high coagulation factor IX, low antithrombin, APC resistance and the factor V Leiden mutation (Figure 1) (
Studies showing supra-additive interactions between factor V Leiden, exogenous hormone use and risk of venous thromboembolism.

Reproduced with permission from [54].

Model showing increases in risk of venous thromboembolism with single and combined thrombophilias, with and without hormone replacement therapy use.
The prothrombotic effects of HRT may also be relevant to the increased risks of stroke and early CHD [2]. Recent meta-analyses of the factor V Leiden and prothrombin
Recent reports from the WHI study have generally shown no interaction of HRT use with plasma levels of blood lipids, or of inflammatory markers such as fibrinogen, coagulation factor VIII or C-reactive protein (CRP) [10,14–16]. While oral HRT increases circulating CRP levels, and the WHI study of unopposed CEE reported an interaction of CHD risk with CRP and HRT use of borderline statistical significance, the clinical significance of these markers in CVD remains to be established [27,47].
Risks of different types of HRT
As reviewed above, most data on the cardiovascular risks of HRT in RCTs come from the USA, where CEE and MPA were chosen in the WHI and HERS trials owing to their common use. Several European commentators have hypothesized that other estrogens and progestins, used more commonly in European countries, may carry a lower risk than CEE and MPA. However, results from other trials using estradiol alone have shown similar results to the WHI and HERS studies [2,4,19,21,22]. Moreover, some ‘European’ preparations have significant effects on activation of coagulation and inflammation [3,28–39].
Unopposed estrogen versus opposed estrogen–progestin preparations
In general, there appears no difference between unopposed and opposed estrogen preparations with regard to risk of VTE [39], stroke [4] or total CVD events (
Type of estrogen
In a recent case–control study from a USA population [49], current users of esterified estrogen had no increase in risk of VTE (odds ratio [OR]: 0.92; 95% CI: 0.69–1.22), in contrast to users of CEE (OR: 1.65; 95% CI: 1.24–2.19). Among all estrogen users, concomitant progestin use was associated with increased risk compared with use of unopposed estrogen (OR: 1.60; 95% CI: 1.13–2.26). Further studies are required to replicate these findings.
Dose of estrogen
Low-dose estrogen HRT preparations may have lesser effects on activation of coagulation and inflammation compared with higher-dose preparations [34]. Whether or not this results in a lower risk of CVD events remains to be proven.
Route of administration
Three case–control studies have reported on the risk of VTE in users of transdermal HRT compared with oral HRT. A pooled analysis of these studies reported an overall odds ratio for VTE of 1.25 (95% CI: 0.80–1.95) [2]. This possible lower risk may lead to consideration of their prescription to women at increased risk of VTE, such as those with thrombophilias [27]; however, it should be noted that the upper 95% CI (1.95) is similar to the mean estimated risk of VTE with oral HRT (approximately 2) [2].
Risk factors for CVD in HRT users
As discussed above, the baseline absolute risks of CVD increase with age, hence the absolute extra risks of CVD in HRT users also increase with age (
Obesity is increasingly recognized as a risk factor for both VTE and arterial disease. In the WHI trial of CEE and MPA, the hazard ratio for VTE was 3.8 in overweight women (body mass index [BMI] 25–30 kg/m2) and 5.6 in obese women (BMI>30 kg/m2) [51]. This interaction may reflect effects of both HRT and obesity on coagulation pathways, such as APC resistance [28,29].
As discussed previously, women with thrombophilias have a high risk of VTE during HRT use (Figure 1) (
Executive summary
Systematic reviews of randomized, controlled trials (RCTs) (which provide the least biased estimates of risks and benefits) have shown an increased risk of cardiovascular disease (CVD) events (venous thromboembolism [VTE], stroke, early coronary heart disease [CHD], and possibly peripheral arterial disease [PAD]) in women currently using hormone replacement therapy (HRT).
The relative increase in risk is similar to that for use of combined oral contraceptives (COCs), but the absolute increase in risk is higher due to the higher age of HRT users.
The absolute increase in risk increases with baseline age and with risk factors for CVD.
An individual participant meta-analysis of these trials should now be performed, to clarify individual and total risks of CVD (and other) events, and to clarify time trends, effects of risk factors, and effects of type of HRT and route of administration.
The relative risk of VTE in RCTs and observational studies of HRT users is approximately 2 (95% confidence interval (CI): approximately 1.5–3.0).
As with COC use, the risk is higher in the first year of treatment – the ‘starter effect’.
The ‘starter effect’ may be due to precipitation of thrombosis in women who are susceptible to the prothrombotic effects of HRT, such as those with thrombophilias.
Screening for thrombophilia may reduce these risks and be cost-effective; however, the risks and benefits of such screening require further evaluation.
Transdermal HRT may carry a lower risk of VTE compared with oral HRT; however, further studies are required.
In women undergoing surgery, HRT increases the risk of postoperative VTE, hence withdrawal of HRT prior to elective surgery and antithrombotic prophylaxis according to overall risk factors should be considered. VTE prophylaxis should be given routinely in emergency surgery.
The relative risk of stroke in RCTs of HRT use is 1.23 (95% CI: 1.06–1.44).
The relative risk of fatal or disabling stroke in HRT users is 1.56 (95% CI: 1.11–2.20).
The increased risk is confined to ischemic stroke, and is not apparent for hemorrhagic stroke.
There is an overall trend for a ‘starter effect’.
The overall relative risk of CHD in RCTs of HRT use is a nonsignificant increase (1.10; 95% CI: 0.95–1.27).
There is an overall trend for a ‘starter effect’.
Subanalyses in the final report of the Women's Health Initiative study of unopposed HRT suggest a lower risk of CHD events in women aged 50–59 years at baseline. However, extreme caution is required in interpretation owing to the large number of subanalyses in this report, resulting in a high probability of false positive or false negative findings.
Furthermore, the Women's Health Initiative study of unopposed HRT also reported an increased risk of total CVD (CHD, stroke, VTE, congestive heart failure) (hazard ratio: 1.12; 95% CI: 1.01–1.24), as well as an additional increased risk of PAD events (lower extremity event or abdominal aortic aneurysm; hazard ratio: 1.63; 95% CI: 1.05–2.51).
The most likely mechanism for increased CVD risk in HRT users is activation of blood coagulation (as in COC users), as shown by studies of blood coagulation factors, inhibitor pathways (e.g., resistance to activated protein C), and activation markers, and by increased risk in women with thrombophilias.
Most RCT data comes from studies of oral conjugated equine estrogens and medroxyprogesterone acetate; however, results from trials of other preparations (e.g., estradiol) have shown similar results.
Overall, there appears to be no difference between unopposed and opposed estrogen preparations for CVD risk.
Hospitalization for surgery, trauma or other illness is an important risk factor for VTE [101]. In the HERS study, the risk of VTE was increased five- to sixfold during the first 90 days after surgery or other hospitalization, and remained increased for at least 30 days after HRT was stopped [52]. Hence it has been recommended in an evidence-based clinical practice guideline that medical practitioners discuss the balance of risks and benefits with the patient when considering stopping hormones prior to elective surgery; consider specific antithrombotic prophylaxis according to overall risk factors; and give VTE prophylaxis routinely in emergency surgery [101].
Future perspective
The time is ripe for a collaborative meta-analysis of individual data in randomized trials, which would clarify the risks of different CVD (and other) events by time, type of preparation, and risk-factor subgroups (age, obesity, baseline CVD etc.). Pharmacological research in HRT (or in its alternatives such as selective estrogen-receptor modulators) will likely be directed at discovering new hormones with fewer prothrombotic effects on biomarkers (e.g., APC resistance, activation markers of coagulation and inflammation), investigation of lower doses of estrogen, and investigation of nonoral routes of administration. However, testing the cardiovascular safety of newer preparations will be a major challenge, in view of the low baseline CVD risk in women aged 50–54 years who are now the main users of HRT. Postmarketing surveillance of such newer preparations and ongoing analysis of major CVD (and other) events is important [2].
