Abstract
Each year, approximately 780,000 strokes occur in the USA, representing a major public health problem. Stroke is also the leading cause of disability in the USA [1]. Gender differences in relation to stroke incidence, mortality and outcomes are being increasingly recognized. A systematic review of epidemiology studies from 19 countries and five continents reported an age-adjusted incidence rate ratio of 1.33 (95% CI: 1.30–1.37), which means that, worldwide, stroke incidence is 33% higher in men than in women [2]. In addition, women have their first strokes an average of 4.3 years later than men [2]. A recent analysis of the Framingham Heart Study cohort reported a higher incidence of stroke in men of all age groups except for men aged 85–94 years. The study also reported that women had an incidence of 21.6 out of 1000 person-years, whereas men had an incidence of 15.5 out of 1000 person-years [3]. Women also live longer and represent the majority of stroke deaths. There has been a general decline in stroke mortality in men, but not in women, for reasons that are unclear at present [1]. In addition, despite the lower likelihood of having a stroke, women appear to have worse outcomes than men, as implicated by scales of activities of daily living (ADLs) [3–5], quality of life [6] and disability [3,4,7–10]. Some studies have also found that women have more severe strokes at onset compared with men [4,5]. The systematic review of studies worldwide reported that stroke severity measured with the NIH Stroke Scale (NIHSS), which ranges from 0 (for least severe) to 42 (for most severe), is only slightly higher in women (NIHSS = 10) than for men (NIHSS = 8.2; p = 0.06) [2]. This is important because initial stroke severity is the single most significant predictor of most long-term stroke outcomes, including ADLs, handicap and disability. Older age has been associated with poorer outcomes after stroke [4], and could help to explain the disparity for outcomes in women, even if the initial stroke severity was the same as a man's.
Other sex differences include the observation that women have worse prestroke disability and more comorbidities than men [5,8,9]. The Stroke Hormones and Outcomes in Women (SHOW) study was designed to document outcomes in women with stroke from the initial stroke presentation to long-term outcomes, accounting for hormone-therapy (HT) use and baseline comorbidities at the time of stroke. In a prior analysis of this cohort, we demonstrated that overall comorbidities at baseline (measured with the Charlson Index), as well as specific comorbidities (e.g., coronary heart disease [CHD] and diabetes), predicted disability at 90 days or more after stroke [11]. This analysis highlighted the importance of CHD as a factor related to stroke outcome other than mortality, whereas other studies reported diabetes, prior stroke [12], atrial fibrillation and decreased level of consciousness [13] as predictors of stroke outcome at 90 days.
Considering the potential for poor outcomes in women, stroke prevention is of the utmost importance. HT was thought to be protective against heart disease in an early meta-analysis of observational studies, along with a neutral benefit for stroke [14]. HT has since been tested in randomized, controlled trials for its efficacy for preventing stroke [15] and heart disease [16], but there was no protection in those randomized to estrogen formulations. Furthermore, in the Women's Health Initiative (WHI) trial of apparently healthy women, HT (whether conjugated equine estrogen alone or combined with medroxyprogesterone) increased the risk of stroke by approximately 40% in HT users [17]. There are multiple potential explanations for the disparate results from the observational and the randomized trials, but one of the most likely is the healthy-user effect; that is to say, women who use estrogens are healthier than nonusers. For example, a comparison of perimenopausal estrogen-replacement users with nonusers showed distinct differences between these groups of women [18]. Estrogen users were more educated, had higher HDL-cholesterol levels, were more active, more likely to consume alcohol and had lower blood pressures, weight and fasting insulin compared with nonusers [18]. A second healthy-user effect relates to factors associated with stopping estrogen and an increased mortality associated with the morbidities that lead to discontinuation. In a large study of all-cause and cause-specific mortality in women followed for breast cancer, current estrogen users had a lower all-cause mortality rate than never users [19]. However, women who had recently discontinued estrogens had a higher all-cause mortality rate compared with never users. The ‘healthy estrogen-user survivor effect’ is a potential explanation for the low mortality rate among current users of estrogen. However, when estrogen is stopped, it signifies the diagnosis of a serious diseases; this will lead to a decrease in survival, potentially leading to an overestimation of the benefits of estrogens in current users that have not yet had a serious event [19]. Considering that some women may still be using HT at the time of stroke (currently for only limited indications), it is useful to understand the longer-term outcomes in these women, especially when most women with stroke would be encouraged to discontinue HT shortly after stroke onset.
The aims of the SHOW study were to determine whether women using HT at the time of an ischemic stroke would have differences in initial stroke severity compared with women who were not using these drugs. In addition, the impact of HT and other comorbidities on disability and ADL scales were examind over 2 years of follow-up in order to assess whether the healthy-user survivor effect is valid in women with stroke.
Patients & methods
The methods of the SHOW study have been previously published [11]. The inclusion criteria for this study were women over the age of 18 years having an acute ischemic stroke within 24 h of hospital admission. Women admitted to Duke Hospital (Durham, NC, USA) were consecutively screened for eligibility and approached for participation. The study protocol was approved by the Duke University Medical Center Institutional Review Board, and all participants consented to participate. Ischemic stroke diagnosis was verified by clinical symptoms and routine neuroimaging (i.e., CT or MRI scan). Women with transient ischemic attack (symptoms resolving within 24 h of onset), intracerebral or subarachnoid hemorrhage, other intracranial abnormalities (e.g., subdural hematoma and brain tumor), severe dementia, life expectancy of less than 6 months or a pre-existing condition resulting in a handicap were excluded.
Hormone-therapy use (including current and prior use, type, dose, duration and reason for use), all other admission medications, risk factors (e.g., diabetes, hypertension, hypercholesterolemia, prior stroke/transient ischemic attack, CHD, congestive heart failure [CHF], atrial fibrillation, peripheral vascular disease, venous thrombosis and cigarette smoking) and family history of vascular disease were recorded at the time of enrollment. Medical comorbidities were assessed using the modified Charlson Index, which excluded stroke and hemiplegia. Scores were assigned by the study principal investigator (Cheryl Bushnell) from medical records [20,21]. Parity and number of miscarriages were documented. Current height and weight were collected for BMI calculation. Amount of exercise (frequency/week), illicit drug use, alcohol use (drinks/week), primary-care use (visits/year) and socioeconomic details, such as education years, type of health insurance and social support at home, were also ascertained.
During the acute stroke hospitalization, details of the acute stroke treatment (e.g., thrombolytics and heparin), discharge-prevention therapy (e.g., antithrombotics, lipid-lowering and specific classes of antihypertensive therapies) were documented. Diagnostic test results were assessed to determine the stroke subtype according to the Trial of Org 10172 in Acute Stroke Treatment (TOAST) criteria, which categorizes patients into small vessel or large vessel atherosclerotic, cardioembolic, other or unknown subtypes [22]. NIHSS score is a scale that measures neurologic impairment and has been widely used in acute stroke clinical trials [23,24]. The scores range from 0 to 40, with lower scores reflecting lesser impairment. The NIHSS was obtained by certified study personnel at enrollment and at 90 days or more in those with in-person follow-up.
The modified Rankin Score (mRS) [25], the Stroke Impact Scale-16 (SIS-16) [26] and the Barthel Index (BI) were obtained from subjects 90 days or more after stroke by personal or telephone interview. The mRS is a scale of disability that ranges from 0 to 5 (
Modified Rankin scores and description of each grade of disability.

The mRS and BI scores were obtained by telephone interview at 6-month intervals for a minimum of 2 years among survivors. Additional information collected at the 6-month interviews included a screen for stroke symptoms, current medications (including HT), new hospitalizations, vascular diagnoses or risk factors since the prior contact. If the subject could not be contacted by telephone, the in-patient and outpatient medical records were reviewed to determine the date the patient was last known to be alive, as well as changes to medications and medical history.
Statistical analysis
The primary outcome of interest in the SHOW study was initial stroke severity measured with the NIHSS, and the aim was to compare HT users with nonusers. We estimated that 70 women in each group would provide 80% power (β = 0.2) with an α-value of 0.05 to determine a difference in NIHSS of 4 points. The target enrollment was therefore 75 HT users and 75 nonusers. Nonparametric tests (i.e., Wilcoxon rank sum) were performed in order to assess differences in NIHSS between HT users and nonusers, and in prespecified subgroups of subjects with factors potentially affecting stroke severity. Univariately significant factors associated with HT use and NIHSS were entered into a linear regression model (using ranked NIHSS as the outcome) and the stepwise method (retaining variables with p < 0.1) was used to determine which factors were independently associated with initial NIHSS.
Secondary outcomes in this cohort were those collected at intervals between 90 days and 2 years, and included mRS, BI and SIS-16 (obtained only at 90 days). Median mRS obtained at the last follow-up contact were compared between prespecified or previously significant subgroups, such as HT use, Charlson comorbidity index, history of atrial fibrillation, coronary artery disease and diabetes. Median SIS-16 scores were also analyzed in a similar fashion. BI was dichotomized into those with a good outcome (BI score of 95 or 100) versus those with a poor outcome (BI < 95). Variables with a p-value of less than 0.05 were entered into a logistic regression model adjusted for initial NIHSS and age. The stepwise method was used to determine the final model results as described above.
Results
From September 2001 to February 2005, 142 women consecutively admitted for ischemic stroke met the inclusion criteria and were approached regarding participation. Nine women refused, leaving 133 (94%) who were enrolled. The flow of enrollment and follow-up are shown in
Baseline characteristics based on hormone-therapy status at time of stroke (including NIH Stroke Scale).

HT: Hormone therapy; IQR: Interquartile range.

Enrollment and follow-up in the Stroke Hormones and Outcomes in Women (SHOW) study.
Patterns of HT use
A total of 93 women were nonusers and 40 (30%) were using HT at the time of stroke (herein referred to as baseline-HT users). Of those not currently using HT, 38% were former HT users, 57% were never users and 5% were unknown. The types of HT used at baseline included: conjugated equine estrogens (n = 15), conjugated equine estrogens/medroxyprogesterone (n = 5), estradiol (n = 4), oral contraceptives (n = 7), raloxifene (n = 3), transdermal estradiol (n = 1) and other (n = 5). The median duration of HT use was 9.5 years prior to the stroke (range: 0.5–32 years). The reasons for self-reported use in the 40 HT users at baseline were hysterectomy in 29%, menopausal symptoms in 33%, osteoporosis in 10% and other uses in 24%.
Hormone-therapy users were more likely to be White, leaner, have insurance other than Medicare or Medicaid, and have lower levels of family support than nonusers (
Of the women who provided self-reported age at menopause (n = 79), the median age in those without hysterectomy (48 years; range: 36–63 years) was significantly older than those with hysterectomy (45 years; range: 21–56 years; p = 0.016). Hysterectomy status was obtained from the subject's self-report and/or verification in the admission note. Of the 49 (37%) women whose status was posthysterectomy, 14 (29%) had at least one ovary remaining, and for 11 (22%), the ovariectomy status was unknown.
Discharge to home following the stroke hospitalization was similar between HT users (60%) and nonusers (61%). After 90 days, 110 (83%) women could be contacted regarding HT use. At this interview, 12 (11%) were continuing to use HT, 16 (15%) were no longer using HT and 82 (74%) remained off HT.
Initial stroke severity
The median initial NIHSS was 4.0 (range: 0–22). There was no difference in initial NIHSS scores between HT users (median: 3.5; range: 1–21) and nonusers (median: 4.0; range: 1–22; Wilcoxon p = 0.4;
Factors associated with initial stroke severity in women with acute ischemic stroke.
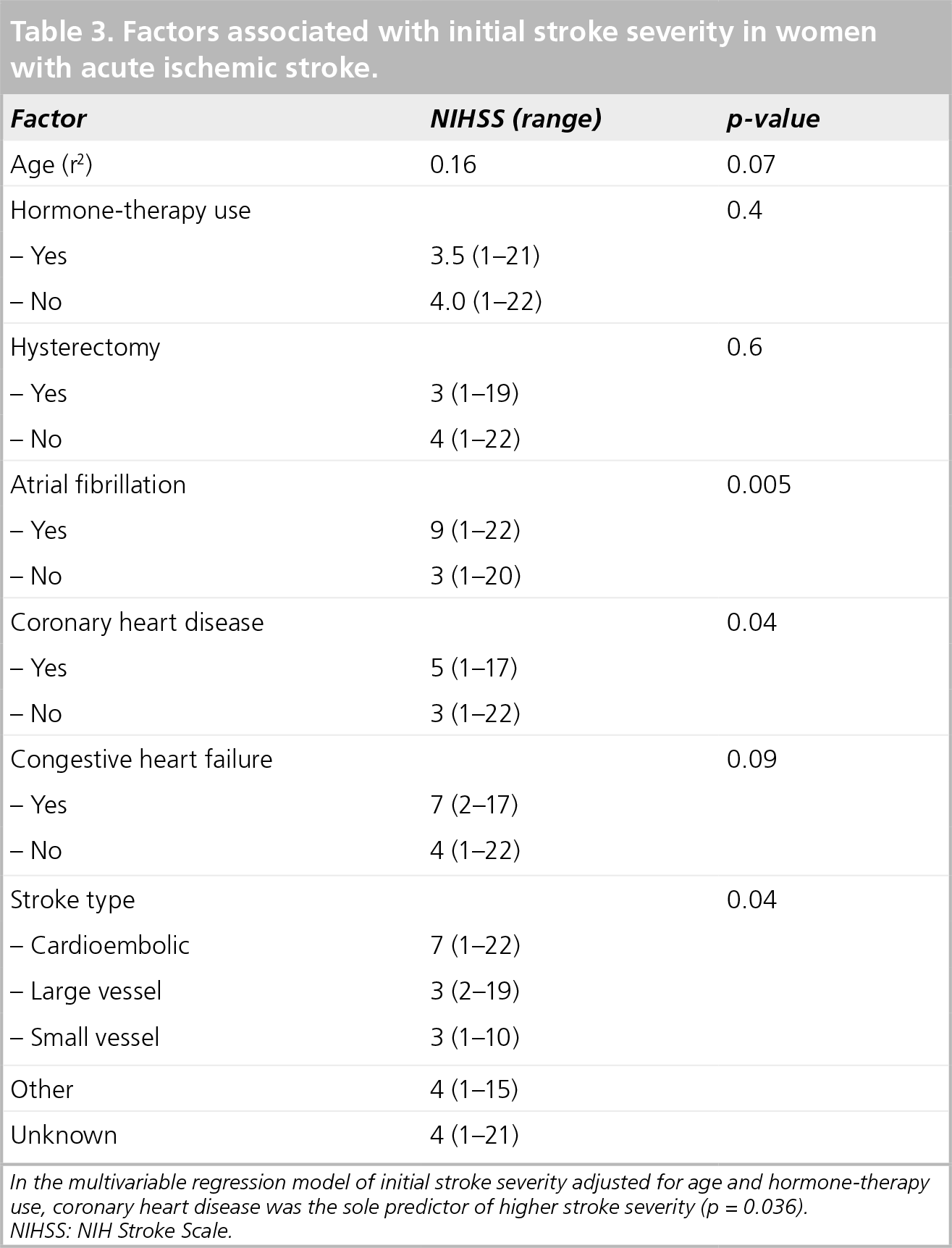
In the multivariable regression model of initial stroke severity adjusted for age and hormone-therapy use, coronary heart disease was the sole predictor of higher stroke severity (p = 0.036).
NIHSS: NIH Stroke Scale.
90-day outcomes
At total of three women (2%) died during hospitalization, six (4.5%) died prior to 90-day follow-up, five (3.8%) withdrew and 14 (10.5%) were lost to follow-up. The median time from enrollment to follow-up was 105 days (interquartile range [IQR]: 91–151 days). The majority of women alive at 90 days or more follow-up were living at home (n = 107 [95%]).
The stroke severity at 90 days was obtained for 100 women (median NIHSS score: 1; range: 0–18). The NIHSS at 90 days was similar in HT users (n = 27; median NIHSS score: 1; range: 1–11) at the time of stroke versus nonusers (n = 73; median NIHSS score: 1; range: 1–18;
Stroke severity and functional status in survivors categorized by baseline hormone-therapy use.
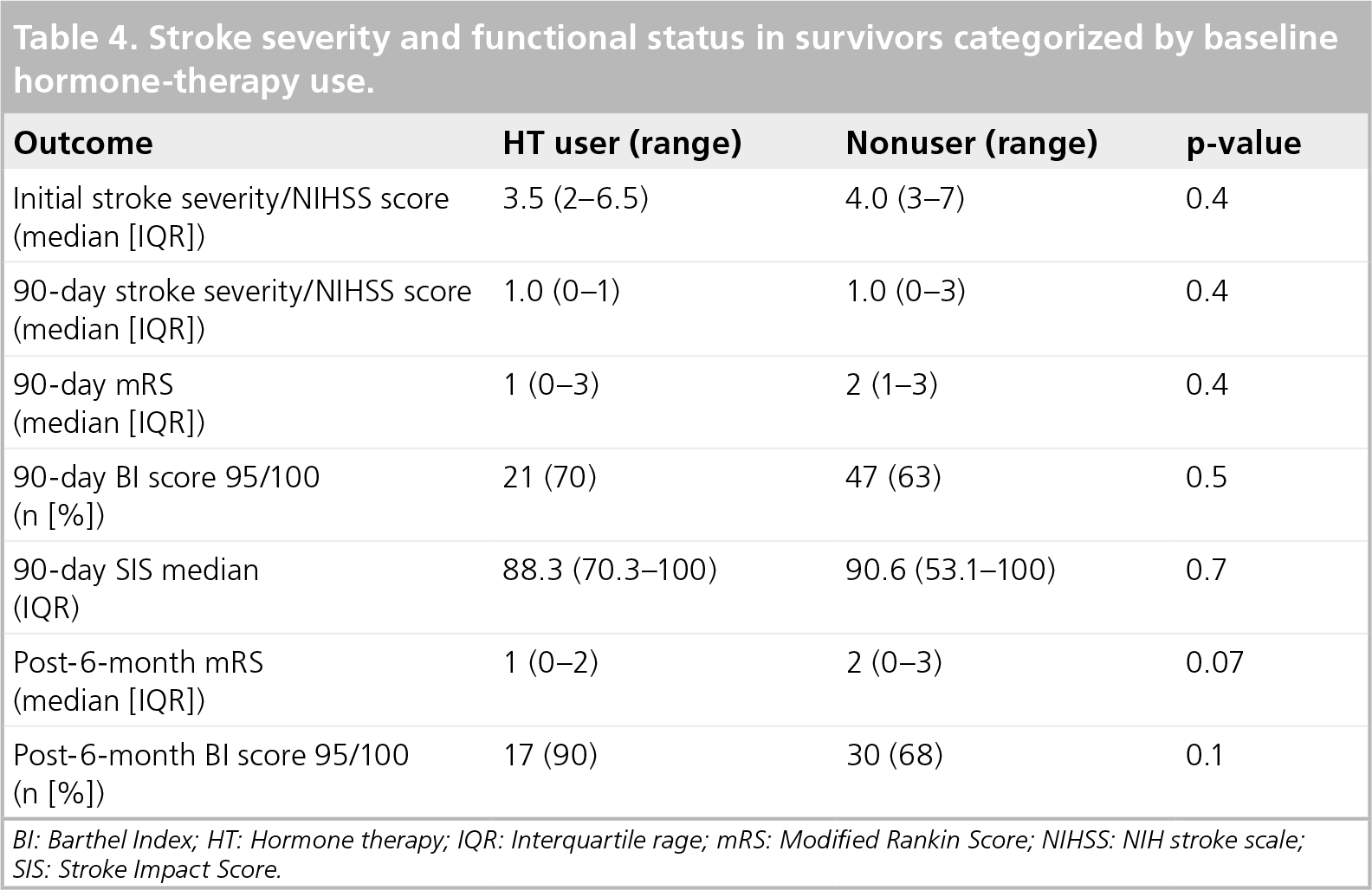
BI: Barthel Index; HT: Hormone therapy; IQR: Interquartile rage; mRS: Modified Rankin Score; NIHSS: NIH stroke scale
SIS: Stroke Impact Score.
The median mRS after 90 days was 1 (range: 0–4) for the 105 women with this outcome. There was no difference in 90-day mRS scores between HT users (median: 1; IQR: 0–3) and nonusers (median: 2; IQR: 1–3; p = 0.4;
The median BI score at 90 days was 100 (range: 20–100). For the dichotomized scores (≤95 vs >95), 70% of HT users had a BI score greater than or equal to 95, versus 63% of nonusers (p = 0.5). After adjustment for initial stroke severity, increasing age by decade (OR: 1.99; 95% CI: 1.28–3.10) and Charlson Index score greater than or equal to 2 (OR: 3.42; 95% CI: 1.10–10.64) were associated with lower BI scores.
The median SIS-16 score at 90 days was 90.6 (IQR: 56.3–100), and there was no difference between baseline-HT users (median: 88.3; IQR: 70.3–100) and nonusers (90.6; IQR: 53.1–100; p = 0.7;
Mortality
The median total follow-up time for the cohort was 743 days (IQR: 467–919 days). A total of 15 women (11%) died during study follow-up, three during hospitalization and the remainder died after discharge. The likelihood of survival to 3.25 years in this cohort was 77.6% (95% CI: 59.6–88.3%). There were eight women with recurrent stroke during follow-up, three of whom suffered both recurrent stroke and death.

Kaplan–Meier survival curves based on hormone-therapy status (log rank p = 0.8).

Kaplan–Meier survival curves based on age less than or greater than 65 years at the time of stroke (log rank p = 0.03).
Long-term functional status
The median time to the final assessment of functional status was 642 days (IQR: 118–808 days). A total of 67 (50%) of the original cohort had mRS scores that were obtained 6 months or more after hospital discharge. The most common mRS was 0, indicating no disability. The median mRS was 1 (IQR: 0–2) for baseline-HT users and 2 (IQR: 0–3) for nonusers (p = 0.07). After adjustment for age and initial NIHSS, the multivariable linear regression analysis demonstrated that history of CHD was the independent predictor of long-term disability measured with mRS (p = 0.025; model r2 = 0.453).
A total of 62 women had BI scores performed after a minimum of 6 months of follow-up. The final median BI was 100 (IQR: 90–100). Baseline-HT users had a significantly higher BI (median: 100; IQR: 100–100) than nonusers (median 100; IQR: 85–100; p = 0.05). However, when the BI scores were dichotomized, 89% of HT users had good outcomes (score of 95 or 100), whereas the scores from 68% of nonusers did not reach significance (p = 0.1). In the multivariable logistic regression model, only age (OR: 1.056 per year; 95% CI: 1.002–1.112) and initial NIHSS (OR: 1.572 per point; 95% CI: 1.250–1.977) were independent predictors of BI scores less than 95 (i.e., poor outcome).
Discussion
The results of the SHOW study provide detailed documentation of outcomes of women with stroke categorized by HT use at the time of stroke and offered a unique opportunity to examine the healthy-user effect. This is likely to be the only study of its kind for two reasons. First, the WHI trial demonstrated an increased risk of stroke for women using HT even though they were apparently healthy at the time of randomization [17], and this led to a dramatic decline in HT prescriptions soon afterward [31]. In fact, the lack of HT users admitted with stroke limited our enrollment to just over 50% of the original target sample size. In addition, clinical trials with multiple outcomes, such as the WHI trial, did not focus on stroke-related outcomes beyond discharge [17]. Therefore, the SHOW study fills gaps in the knowledge regarding the clinical issues specific to women longitudinally from the time of stroke through the assessment of long-term functional status, as well as addressing the healthy-user effect in women who have suffered a stroke.
The current practice of assessing risk during hospitalization in women with stroke would now routinely include the recommendation to discontinue HT. The WHI randomized trial primary results were published 1 year into enrollment of the SHOW study. While 30% of women continued on HT at 90 days or more of follow-up, there were only two women (5%) who reported HT use during long-term follow-up. Since several women in the HT group were lost during follow-up, the actual number of women using HT after stroke is still uncertain. However, assessing the cases with data demonstrates that the decreasing use of HT over time was consistent with the general temporal trends to discontinue this drug in women with evidence of vascular disease. By contrast, a retrospective database analysis of HT users who suffered a stroke from 1997 to 1999 and were admitted to the same hospital as those enrolled in the SHOW study reported that 82% of the women were maintained on HT at discharge [32]. For this cohort, we were able to study one facet of the healthy-user survival effect, which is the change in health status (i.e., having a stroke) leading to discontinuation of HT. In addition, we did not identify a significant increase in mortality immediately after discontinuation (
Another important result from the SHOW study is that HT use at baseline did not significantly impact initial stroke severity (
Stroke is associated with a 30-day mortality rate of approximately 10%. In addition, of those who die from stroke, over 60% are women [1]. In the Women Estrogen Stroke Trial (WEST) of HT for secondary prevention, the post hoc analysis suggested that women randomized to HT had a higher early mortality [15]. This would also be the concern in the SHOW study for HT users. There was no difference in mortality based on baseline HT status (n = 4 vs n = 11 for nonusers; see
Functional status in women with stroke
Stroke severity, or neurological impairment, is an important outcome, especially because the initial stroke severity is the primary predictor of most longer-term outcomes of interest. However, it is also important to understand how women are able to function despite the impairment. Therefore, functional independence after stroke, measured with scales of disability or ability to perform ADLs, are just as important as stroke severity. The SHOW study collected outcomes at most of the major landmarks of stroke recovery, which included the initial severity, severity at 90 days or more (when 90% of the neurological recovery has occurred) and functional status at 90 days, as well as at 6-month intervals up to 2 years. After adjusting for initial stroke severity, the predictors of 90-day disability with the mRS scores (previously published) included CHD and diabetes, as well as increased comorbidities measured with the Charlson Index [11]. The predictors of worse 90-day independence with ADLs (measured with the BI) and physical functioning domain of the SIS-16 score both included age and increasing comorbidities, measured with the Charlson Index. The SIS-16 was performed because it includes specific tasks that reflect upper extremity as well as lower extremity functions. In addition, it is more sensitive than the BI, which is often skewed towards higher scores and thereby limited by a ceiling effect [26]. The finding that women have less functional independence following stroke than men has been reported in multiple studies [4,5]. However, very few studies have offered explanations for this disparity besides older age in women. Our results suggest that, in addition to age and initial stroke severity, overall comorbidities as well as specific comorbidities, such as CHD and diabetes, also need to be considered when assessing functional status following stroke.
The predictors of functional status at 6 months or more poststroke revealed some minor differences when compared with the 90-day results. For instance, the long-term disability measured with mRS was associated with a history of CHD, but not diabetes or comorbidities, as with the 90-day assessment. Therefore, it appears that long-term disability continues to be influenced by the presence of heart disease at the time of the stroke, which could impair physical mobility owing to unstable or exertional angina, or the interval development of congestive heart failure. In addition, the only predictor of long-term independence with ADLs (BI) was age. It may be that the effect of comorbidities is reduced with time as these women compensate for stroke deficits or have increased support.
HT use at baseline & the healthy-user effect
One of the major disparities between observational studies versus randomized trials of HT use can be at least partially explained by the healthy-user effect. In other words, women using HT in observational studies tended to be healthier than women who were not using these drugs [18,34]. Similarly, women with risk factors, such as hypertension, obesity and diabetes, may be discouraged from using HT by providers. Randomized trials, such as the WHI trial, reduced this bias and therefore demonstrated an increased risk of heart disease and stroke events that were not predicted by the observational data. Consistent with this healthy-user bias is the fact that the cohort of women enrolled in the SHOW study who were using HT at baseline were leaner and less likely to have hypertension than nonusers (
Strengths & limitations
The strengths of this study are the prospective and detailed characteristics and outcomes relevant to women with stroke. This has been an under-studied field, but it is now moving to the forefront of clinical medicine because of the increasing recognition of sex differences related to stroke and many other conditions. The outcomes that were obtained are standardized and have been used routinely for stroke patients in many clinical trials, including the National Institute of Neurological Diseases and Stroke tissue Plasminogen Activator trial [24]. The proportion of patients lost to follow-up was approximately 15%, which is acceptable for a stroke observational study.
However, it is important to recognize the limitations. The median stroke severity was mild (NIHSS of 4); therefore, the cohort was perhaps not representative of most hospitalized stroke patients. This is an inherent issue with enrollment in trials investigating acute stroke, since severe strokes and poor outlook for survival will influence the patients who are approached for participation, as well as the likelihood that patients or their legal representatives will decline participation. However, we consecutively screened the women admitted with stroke who met the specific inclusion/exclusion criteria, making the best attempt possible to avoid any selection bias. Another explanation for the milder stroke severity is that the NIHSS was performed 24 h after onset, and may not represent the actual severity at onset, which could have been higher. As mentioned previously, the decreasing number of women using HT during enrollment limited the ability to enroll subjects in the HT group, resulting in a smaller-than-desired sample size and thereby limiting the analyses in certain subgroups. The number of women who could not be contacted for 90-day follow-up assessments was 15%, and these missing data could have biased the results of the outcomes. An additional 29% of the cohort was lost to late follow-up, limiting the statistical power even further. Therefore, these results must be considered exploratory, and further research is needed to validate these findings.
Conclusion
In a cohort of women with stroke followed for stroke severity and longitudinal functional status, there were no differences in any of the outcomes of interest based on HT status at the time of the stroke. The mortality rate of 11% and the significant impact of age at the time of stroke were similar to other observational studies of stroke subjects. Although there was clear evidence of a healthy-user effect in women using HT at the time of stroke, the healthy estrogen-user survivor effect did not apply because there were no differences in the long-term survival or outcomes based on HT status. The design of future studies should compare men and women in order to determine whether the clinical factors associated with stroke outcomes in women identified in this cohort are equally important in men, or are unique to women.
Future perspective
At present, it almost appears that the door has been shut on the field of HT and stroke in women owing to the overwhelming negative influence of the results from the WHI trial. However, there are many questions that have arisen, and these have opened many avenues of investigation. These include determination of the ‘window of opportunity’ for women who are perimenopausal or recently postmenopausal, and whether HT provides a cerebrovascular benefit to certain women during this time. With additional research to answer this question, physicians will be able to develop risk strata for women in order to select those who would or would not benefit from HT during the critical time period surrounding menopause. Finally, considering the intense interest surrounding gender differences in stroke, the design of future outcome studies and clinical trials will prospectively compare predictors of outcomes in men and women to determine if clinical factors (such as CHD) are equally important in men, or are unique to women.
Ethical conduct of research
The author states that they have obtained appropriate institutional review board approval or have followed the principles outlined in the Declaration of Helsinki for all human or animal experimental investigations. In addition, for investigations involving human subjects, informed consent has been obtained from the participants involved.
Executive summary
Women admitted to hospital with stroke were categorized by hormone-therapy (HT) use at the time of stroke, in order to determine the healthy-user effect for characteristics, as well as stroke severity and outcomes.
HT users were more likely to be White, lessly likely to have hypertension and had a leaner body mass compared with nonusers.
There was no difference in initial stroke severity or any of the longer-term measurements of disability, independence of activities of daily living or mortality between HT users and nonusers.
At 90-days follow-up, only 12 of 40 HT users at baseline were still on HT, but at 6 months there were only two women using HT.
Age over 65 years was the single most important predictor of mortality in this cohort.
The only predictor of worse long-term disability (measured with the modified Rankin score) was coronary heart disease at baseline.
The only predictor of long-term independence with activities of daily living (measured with the Barthel Index) was decreasing age.
Although there was clear evidence of a healthy-user effect in women using HT at the time of stroke, the healthy estrogen-user survivor effect did not apply because there were no differences in the long-term survival or outcomes based on HT status and discontinuation after stroke.
Footnotes
This study was supported by grant number NIH K23NS041929 from the National Institute of Neurological Disorders and Stroke (NINDS) and by a National Stroke Association Research Fellowship Award. The author has no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
No writing assistance was utilized in the production of this manuscript.
