Abstract
After the Ibandronate Osteoporosis Vertebral Fracture Trial in North America and Europe (BONE) study had demonstrated the strong vertebral and nonvertebral antifracture efficacy of daily and intermittent oral ibandronate, the Monthly Oral Ibandronate In Ladies (MOBILE) study gave evidence for an increased efficacy on the bone mineral density (BMD) of higher intermittent oral ibandronate doses (150 mg monthly) compared with 2.5 mg daily. The BONE study also observed nonvertebral antifracture efficacy in patients with a high risk for fractures (mean femoral neck T score of −3.0 or less). A recently published meta-analysis assessing the nonvertebral antifracture efficacy corresponding to the annual cumulative exposure (ACE) of ibandronate demonstrated a significantly better antifracture efficacy of higher compared with lower doses of ibandronate. The Dosing Intravenous Administration (DIVA) study demonstrated evidence for the high efficacy and good tolerability of intravenous ibandronate delivered by quarterly injections. This review summarizes the efficacy and tolerability data of ibandronate concerning monthly oral treatment as well as quarterly injection therapy.
Keywords
Osteoporosis, which is characterized by low bone mass and bone microarchitectural deterioration, is a common cause of fractures in postmenopausal women. Approximately half of the osteoporotic fractures are vertebral and result in height loss, chronic pain and restrictions of the activities of daily living [1]. Nonvertebral fractures frequently observed in osteoporotic patients affect the hip, the wrist and other regions.
Nitrogen-containing bisphosphonates together with vitamin D and calcium are currently the mainstay of the pharmacologic treatment of osteoporosis. Bisphosphonates such as alendronate, ibandronate, risedronate and zoledronate inhibit enzymes in the mevalonate pathway, especially farnesyl pyrophosphate synthase (FPPS). This leads to inhibition in recruitment, differentiation and function of osteoclasts, thereby increasing bone mineral density (BMD) and reducing fracture risk [2].
Ibandronate is a very potent nitrogen-containing bisphosphonate. It is the only bisphosphonate available for intravenous and oral use. Oral ibandronate has demonstrated the numerically highest vertebral antifracture efficacy of all oral bisphosphonates in the pivotal Ibandronate Osteoporosis Vertebral Fracture Trial in North America and Europe (BONE) study [3]. In this study, 3-year ibandronate treatment (2.5 mg/day) was demonstrated to reduce incident vertebral fracture by 62% compared with placebo in postmenopausal women. The BONE study was conducted to prove vertebral – not nonvertebral – antifracture efficacy. However, a post hoc analysis in a group of patients with a high risk for fractures (femoral neck T score of −3.0 or less) showed a significant risk reduction of 69% for nonvertebral fractures.
Owing to stringent intake guidelines (fasting before and after application, upright posture after application) with all oral bisphosphonates, patient adherence to daily treatment is poor. The intake guidelines are necessary to maximize absorption and to reduce gastrointestinal side effects [4]. Therefore, the dosing frequency of oral bisphosphonate treatment was reduced to improve adherence and convenience for the patient. The only bisphosphonate available for the more convenient once-monthly oral application is ibandronate.
Once-monthly oral treatment with ibandronate 150 mg was shown to result in higher and superior BMD increases compared with daily oral treatment with ibandronate 2.5 mg [5]. Furthermore, intravenous ibandronate delivered by quarterly injections was associated with BMD increases similar to ibandronate 150 mg once-monthly [6].
Evidence for therapeutic efficacy should be demonstrated by meta-analyses in addition to randomized controlled studies. A Canadian meta-analysis compared the nonvertebral antifracture efficacy of different ibandronate doses using annual cumulative exposure (ACE) [7]. The analysis delivered strong evidence for the nonvertebral antifracture efficacy of ibandronate.
This article will present an up-to-date overview regarding ibandronate clinical data concerning monthly oral treatment as well as quarterly injection therapy. Nonvertebral antifracture efficacy will be a second important focus.
Oral treatment
The BONE study
Ibandronate licensing was originally based on the results of the BONE study, which reported striking evidence for the vertebral antifracture efficacy of this potent bisphosphonate [3]. This randomized, controlled, double-blind study compared two different ibandronate dosing regimes with placebo in 2946 postmenopausal women. Study patients were required to have a lumbar spine T score of less than or equal to −2.0 in at least one vertebra (L1–L4) and one to four prevalent vertebral fractures (T4–L4). The participants were randomized 1:1:1 to daily oral ibandronate (2.5 mg), intermittent ibandronate (20 mg every other day for 12 doses every 3 months) or placebo.
During the 3-year treatment period, the rate of incident vertebral fractures diagnosed using annual lateral radiographs was 9.6% (placebo) 4.7% (daily oral ibandronate) and 4.9% (intermittent ibandronate). The relative vertebral fracture risk compared with placebo was reduced by 62% (daily oral ibandronate; p = 0.0001;

Reduction of the 3-year incidence of vertebral fracture by daily ibandronate treatment compared with placebo (BONE study).
An analysis by Felsenberg et al., which retrospectively analyzed the prevention of new vertebral fractures of greater severity in the BONE study, confirmed the robust antifracture efficacy and the fast onset of action of oral ibandronate [8]. This analysis confirmed a pronounced, rapid onset and consistent efficacy of daily oral ibandronate during the whole 3-year study period on the incidence of moderate and severe vertebral fractures, which were reduced compared with placebo by 59.5% after 1 year (p = 0.014), 58.7% after 2 years (p = 0.0004) and 59.0% after 3 years (p < 0.0001).
The nonvertebral efficacy of ibandronate in the BONE study depended on the baseline femoral neck BMD, which is a predictor for higher fracture risk. Therefore, the results were considerably influenced by the relatively high mean femoral baseline BMD of the total study population being considerably higher than in studies investigating other bisphosphonates, such as the Fracture Intervention Trial 1 [9]. The BONE study was primarily conducted to show vertebral antifracture efficacy. Therefore, neither nonvertebral fracture risk nor patient numbers proved that risk was considered in the design.
However, detailed analyses of study results revealed a significant interaction between baseline BMD of the femoral neck and nonvertebral antifracture efficacy of ibandronate. In the subgroup of patients with a high risk of fracture (femoral neck baseline BMD T score ≤ −3.0; n = 375), daily oral ibandronate treatment resulted in a relative-risk reduction of 69% for nonvertebral fractures compared with placebo (ibandronate 2.5 mg, n = 123; ibandronate 20 mg, n = 128; p = 0.013).
In the BONE study, active treatment was further associated with a progressive significant increase in lumbar spine BMD (daily oral ibandronate: +6.5%; intermittent ibandronate: +5.7%; placebo: +1.3%) after 3 years. Both ibandronate regimes also resulted in hip BMD increase, normalization of bone turnover and reduced height loss.
Based on the BONE study results, it can be concluded that daily oral ibandronate shows a rapid onset and consistent vertebral antifracture efficacy during the 3-year treatment period. The prevention of nonvertebral fractures was highly effective in patients with a high risk of fracture. In addition to the vertebral antifracture efficacy of daily ibandronate, the BONE study demonstrated the efficacy of an intermittent ibandronate treatment schedule.
Bridging concept
For all oral bisphosphonates, antifracture efficacy data were originally provided by randomized controlled trials investigating daily oral bisphosphonate treatment compared with placebo. However, adherence to long-term daily oral bisphosphonate therapy is very poor owing to the difficult oral application schedules requiring a long fasting period before application and a further fasting period, as well as an upright position, after application.
Consequently, alternative oral treatment schedules requiring weekly (alendronate or risedronate) or monthly application (ibandronate) were developed for oral bisphosphonates. The weekly alendronate and risedronate doses correspond to seven daily doses whereas dosing of once-monthly ibandronate (150 mg) doubled the sum of 30 daily doses (30 × 2.5 mg = 75 mg).
The higher dose for once-monthly ibandronate was selected because a slight decline in effect size was observed in the preclinical development program, which was more pronounced with longer dose-free intervals [10]. Similar effect decreases, although not statistically significant, were also apparent in clinical trials of oral alendronate and risedronate after prolonging dosing intervals [11,12]. Evidence in favor of increased oral ibandronate doses when moving from daily to monthly intervals was originally provided by the Phase I pilot, double-blind, placebo-controlled Monthly Oral Pilot Study (MOPS) [13]. The MOPS trial, which was a dose-titration study, investigated the effect on bone turnover and tolerability of three once-monthly oral ibandronate doses (50, 100 and 150 mg). During the 3-month treatment period, monthly ibandronate treatment resulted in dose-dependent decreases of serum C-telopeptide of the α-chain of type I collagen (sCTX), with the 150-mg dose showing the most pronounced effect.
The Monthly Oral Ibandronate In Ladies (MOBILE) study design complied with the requirements of the European Medicines Agency (EMEA) for the licensing of intermittent bisphosphonate therapies [5]. To license intermittent treatment regimes, the EMEA requires evidence for the noninferiority on BMD of intermittent treatment schedules compared with daily treatment regimes investigated in Phase III trials. However, there is no need to deliver new placebo-controlled antifracture efficacy data. The assumption of a comparable antifracture efficacy of daily and intermittent bisphosphonate treatment regimes based on noninferior BMD data is called ‘bridging’.
The MOBILE study randomized 1609 patients to four different monthly ibandronate dosing schedules (50 + 50 mg, 100 mg and 150 mg) or ibandronate 2.5 mg daily [5]. In all four treatment groups, substantial increases of lumbar spine, total hip, femoral neck and trochanter BMD (measured by dual-energy x-ray absorptiometry) were observed. The three once-monthly ibandronate regimes were noninferior compared with ibandronate 2.5 mg daily. Ibandronate 150 mg/month was the most effective treatment regime (lumbar spine BMD +6.6 vs 5.0% [2.5 mg/day]; p < 0.001;
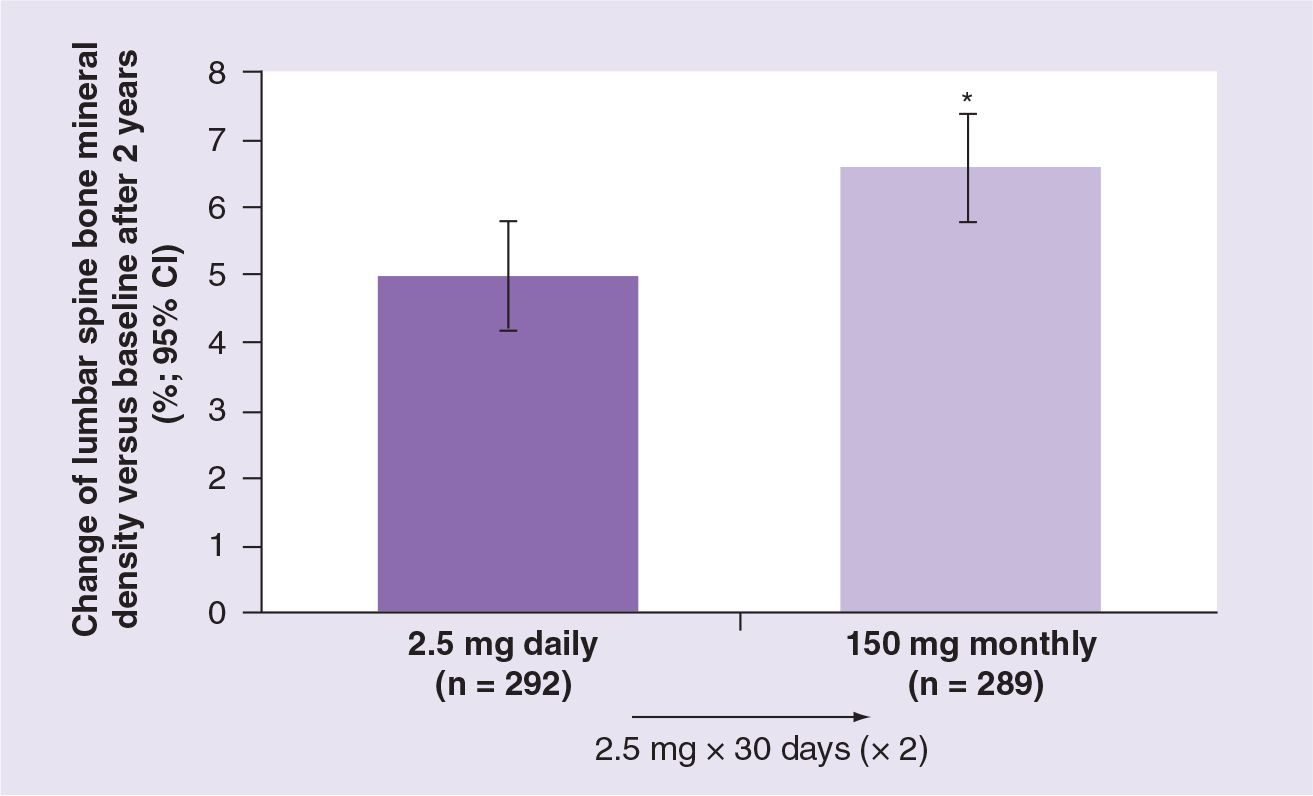
Increase of bone mineral density by once-monthly treatment with ibandronate 150 mg compared with ibandronate 2.5 mg daily (MOBILE study).
After 24 months of treatment with 35 mg/week or 5 mg/day risedronate, mean lumbar spine BMD increased by 4.74 and 5.17%, respectively, meeting the pre-specified noninferiority criteria without achieving superiority [12]. Similar BMD increases were also observed after 24 months of treatment with alendronate 70 mg once-weekly and alendronate 10 mg daily (6.8 and 7.3%, respectively) [14].
Meta-analyses
A recently published meta-analysis of the individual patient data of eight randomized studies also provided evidence for a superior nonvertebral antifracture efficacy of higher ibandronate dosing regimens based on ACE [7]. The ACE concept allows comparison of dosing regimens and yearly doses by considering differences of bioavailability of oral and intravenous ibandronate, the time course of application (daily, monthly oral or quarterly intravenous) and the dose itself.
Patients receiving high doses of ibandronate (including the 150 mg monthly oral and the quarterly intravenous 2 and 3 mg formulation: ACE ≥ 10.8 mg) had a 38% lower relative nonvertebral fracture risk than patients receiving 2.5 mg daily (analogical to an ACE of 5.5 mg) oral ibandronate (HR: 0.621; 95% CI: 0.396–0.974; p = 0.038;

Canadian meta-analysis: significantly superior nonvertebral efficacy of a higher annual cumulative ibandronate exposure compared with a lower annual cumulative exposure (p = 0.036; log-rank).
The authors of the analysis concluded that an ACE greater than or equal to ibandronate 10.8 mg has a significantly higher antifracture efficacy than lower ibandronate doses, presumably owing to the superior effects of the intermittent therapy with an ACE greater than or equal to ibandronate 10.8 mg on bone resorption and BMD.
Safety
The clinical studies demonstrated the good safety and tolerability profile of ibandronate. In BONE, oral ibandronate was well tolerated, with a similar adverse event incidence in active treatment and placebo groups [3]. Most frequently reported adverse events were dyspepsia (ibandronate 11.4%; placebo 9.1%), gastroenteritis (ibandronate 5.5%; placebo 5.5%), nausea (ibandronate 4.2%; placebo 6.3%), vomiting (ibandronate 3.0%; placebo 2.5%), gastritis (ibandronate 2.3%; placebo 2.2%) and gastrointestinal pain (ibandronate 1.9%; placebo 2.6%). The MOBILE study observed a good tolerability of ibandronate with a well-balanced incidence of adverse events in the four treatment arms. Therefore, ibandronate 150 mg once-monthly shows a similar tolerability profile than ibandronate 2.5 mg daily, which was similar to placebo.
Intravenous treatment
After successfully establishing the once-monthly tablet in the treatment of postmenopausal osteoporosis, intravenous ibandronate (3 mg every 3 months) was introduced into the market in 2006. Currently, ibandronate is the only nitrogen-containing bisphosphonate available for intravenous application as well as for oral use.
Efficacy: DIVA study
Evidence for the efficacy and tolerability of intravenous ibandronate was delivered primarily by the DIVA study [6]. This double-blind, double-dummy, noninferiority study randomized 1395 postmenopausal women with osteoporosis to intravenous ibandronate (2 mg every 2 months or 3 mg every 3 months) plus daily oral placebo, or to oral ibandronate 2.5 mg daily plus intravenous placebo 2- or 3-monthly. Efficacy end points were the changes in lumbar spine and proximal femur BMD measured by dual-energy x-ray absorptiometry (DEXA) as well as bone resorption markers.
After 2 years of randomized treatment, lumbar spine BMD had increased by 6.4% (95% CI: 5.9–6.9; n = 320) and 6.3% (95% CI: 5.7–6.8; n = 334) in the 2 mg every 2 months group and 3 mg every 3 months group, respectively, compared with 4.8% (95% CI: 4.3–5.4; n = 334) in the group receiving 2 mg/day orally. The first year results of this study confirmed the noninferiority of both intravenous treatments versus daily oral dosing. In addition, a prospectively planned statistical analysis demonstrated a superior efficacy of both intravenous ibandronate regimens compared with oral treatment on lumbar spine BMD. Differences between the two intravenous dosing schedules were not observed.
Proximal femur BMD (total hip, femoral neck and trochanter) increased in a similar way with both intravenous treatments, and both treatments resulted in superior BMD gains in total hip and trochanter BMD at 2 years (post-analysis: p < 0.001; all intravenous treatments versus daily oral treatment) and noninferiority for femoral neck. Bone resorption (measured by sCTX) decreased with all three ibandronate regimens within 3 months of treatment initiation and the lower bone resorption was maintained consistently throughout the study (sCTX decreases at 2 years in the per-protocol population: 55.6, 53.4 and 59.9% with 2 mg every 2 months, 3 mg every 3 months and 2.5 mg/day orally, respectively).
Therefore, the licensed intravenous ibandronate injection of 3 mg every 3 months is an effective and useful additional option in the treatment of postmenopausal osteoporosis.
Safety & tolerability of intravenous ibandronate
The DIVA study demonstrated a safety and tolerability of intravenous ibandronate comparable to daily oral treatment [6]. Intravenous ibandronate was associated with a comparable incidence of renal adverse events than oral ibandronate and the DIVA investigators reported no case of acute renal failure. After 2 years, the overall incidence of renal adverse events achieved was 3.9% with daily oral ibandronate treatment, 3.2% with ibandronate 3 mg intravenously quarterly, and 4.5% with ibandronate 2 mg intravenously every 2 months. Decreases in creatinine clearance were similar among the treatment groups at all time points (21, 23 and 21% of participants with 2 mg every 2 months, 3 mg every 3 months and 2.5 mg daily, respectively). No participants, were considered to have a drug-related change in creatinine clearance. In DIVA, no osteonecrosis of the jaw occurred in patients treated with oral or intravenous ibandronate. Furthermore, no cardiac safety issues were reported. Influenza-like symptoms were observed in a reasonable proportion of patients treated intravenously (4.9% with 3 mg every 3 months). There was no increased number of cases of atrial fibrillation in patients taking intraveneous ibandronate.
Owing to the excellent tolerability, ibandronate 3 mg can be injected as an intravenous bolus within 15–30 s [15], whereas other intravenous bisphosphonates have to be infused for a more prolonged period (e.g., zoledronate for at least 15 min [16]).
Expert commentary
In clinical trials and meta-analyses, the potent bisphosphonate ibandronate demonstrates an excellent antifracture efficacy together with a good safety and tolerability profile. Ibandronate has a rapid effect and remains effective for at least 3 years. The vertebral antifracture efficacy of ibandronate is numerically the highest in comparison with other oral bisphosphonates. Ibandronate is the only nitrogen-containing bisphosphonate that can be administered orally and intravenously. The intravenous administration has a safety and tolerability profile comparable to the oral application.
The BONE study demonstrated a very high and consistent vertebral antifracture efficacy of oral ibandronate, reducing the relative vertebral fracture risk by 62% [3]. The BONE study also provided evidence for a similar antifracture efficacy of intermittent and daily oral ibandronate treatment schedules. A post hoc analysis of patients with a high risk for nonvertebral fractures (femoral neck T score ≤ −3.0) showed a 69% risk reduction.
The BONE study demonstrated a rapid onset of action: the risk for moderate and severe vertebral fractures was reduced by 59% within the first year. Furthermore, antifracture efficacy remained constant over the whole 3-year study period.
The MOBILE study demonstrated that the BMD increases observed in BONE with daily oral ibandronate treatment can be exceeded by a once-monthly ibandronate treatment regime [5]. In MOBILE, a strong association between ibandronate doses and BMD increases was observed. Treatment with the highest oral ibandronate dose (150 mg once-monthly) resulted in significantly higher BMD increases than ibandronate therapy with 2.5 mg daily.
The recently published Canadian meta-analysis confirms that higher doses of ibandronate resulted in an improved nonvertebral antifracture efficacy [7].
The currently marketed ibandronate treatment regimens (150 mg monthly oral and 3 mg quarterly intravenous) both belong to the higher doses showing superior nonvertebral antifracture efficacy compared with a lower ibandronate dose in this meta-analysis [7]. The demonstrated dose-dependent nonvertebral effect is a strong indicator for the efficacy of ibandronate and is in line with the dose-dependent gain in BMD in the pivotal bridging studies MOBILE and DIVA. It should be emphasized that intravenous ibandronate demonstrated an excellent safety and tolerability profile. In clinical studies, no cardiac side effects and no osteonecrosis (especially no osteonecrosis of the jaw) were reported [6]. Influenza-like symptoms occurred at a reasonable rate after ibandronate injections (in approximately 4.9% of patients), which is considerably less than with other intravenous bisphosphonates [16].
The availability of two administration forms (intravenous and oral) enables an easy change from oral to intravenous application in case of compliance or tolerability problems. In addition the intravenous quarterly injection allows an assessable conduct of the patient.
New studies conducted in women who have undergone surgical treatment for breast cancer and who are treated with an aromatase inhibitor suggest that ibandronate is also effective in preventing bone loss. These women were postmenopausal and treated with ibandronate 150 mg once-monthly or placebo for 2 years. Women receiving ibandronate gained BMD in their lumbar spine and hip, whereas women on placebo lost BMD [17].
Conclusion
Ibandronate is the only bisphosphonate available for oral and intravenous treatment demonstrating an excellent safety and tolerability profile with both application routes. Oral as well as intravenous ibandronate shows convincing vertebral and nonvertebral antifracture efficacy in combination with very good tolerability. Its efficacy starts rapidly and remains effective for a duration of at least 3 years. The currently marketed ibandronate doses correspond to the higher ACE doses, which showed a superior nonvertebral antifracture efficacy compared with lower ACE doses in a recently published meta-analysis.
These results confirm the clinical long-term experience with ibandronate in terms of antifracture efficacy and safety.
Executive summary
Ibandronate shows an excellent antifracture efficacy combined with a very good tolerability profile.
The vertebral and nonvertebral antifracture efficacy of ibandronate was demonstrated by randomized, controlled clinical trials and meta-analyses.
Ibandronate is the only nitrogen-containing bisphosphonate available for oral and intravenous administration.
The Ibandronate Osteoporosis Vertebral Fracture Trial in North America and Europe (BONE) study demonstrated a strong and consistent vertebral antifracture efficacy of the potent bisphosphonate ibandronate. After 3 years of oral ibandronate treatment the incidence of new vertebral fractures decreased by 62%.
The efficacy of ibandronate starts rapidly and remains effective for a duration of at least 3 years.
The currently marketed oral and intravenous ibandronate doses correspond to the higher doses, which showed in a recently published meta-analysis, a superior nonvertebral antifracture efficacy compared with lower ibandronate doses.
Intravenous ibandronate delivered every 3 months demonstrated noninferiority on lumbar spine and proximal femur bone mineral density (BMD) compared with daily oral treatment. In addition, a prospectively planned analysis showed a superior efficacy on the lumbar spine BMD with quarterly injected intravenous ibandronate.
Intravenous ibandronate has a safety and tolerability profile comparable to daily oral treatment, which was similar to placebo except for influenza-like symptoms. These were observed in a reasonable proportion of patients treated intravenously.
Footnotes
Acknowledgements
The authors thank Christoph Müller-Löbnitz, Forchheim, Germany, for his assistance in the preparation of this manuscript.
This publication was supported by GlaxoSmithKline, Munich, Germany. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
Medical writing assistance was funded unconditionally by GlaxoSmithKline, Munich, Germany.
