Abstract
Keywords
Osteoporosis – low bone mass and microarchitectural deterioration predisposing to fractures –poses hazards to postmenopausal women's health and mobility. The lifetime risk of an osteoporotic fracture (one in six) exceeds lifetime risk of contracting breast cancer (one in nine) for the average Caucasian woman worldwide [1]. Fragility fractures inflict potentially devastating human and economic costs. Within 1 year after a hip fracture, a third of sufferers lose their independence permanently and 20–24% die [2]. Approximately 150,000 US nursing home placements annually result from osteoporotic fractures [3]. In a recent US modeling study, direct costs attributable to osteoporosis were estimated at US$ 13.7–20.3 billion (adjusted to 2005 currency); 89% of this cost burden was borne by women over 65 years of age [4].
Timely diagnosis of postmenopausal osteoporosis allows effective treatment to prevent fractures and their untoward consequences. Measurement of bone mineral density (BMD) by dual-energy x-ray absorptiometry (DXA) is the standard diagnostic method [5]. According to the WHO [6], osteoporosis is diagnosed when BMD values fall below the sex-specific young adult mean by greater than or equal to 2.5 standard deviations. In the presence of corticosteroid use, calcium/vitamin D deficiency or BMD-independent risk factors, such as personal or parental fracture history, low body weight or smoking, fracture risk may exist at higher BMD values [6].
Nitrogen-containing bisphosphonates are standard first-line pharmacotherapy for osteoporosis, along with calcium and vitamin D supplementation, physical training and fall prevention. Oral bisphosphonates (daily or weekly alendronate [7]; daily [8], weekly [9] or intermittent risedronate [10]; or monthly ibandronate [11]) are widely prescribed in osteoporosis. However, stringent dosing guidelines of fasting and upright posture are necessary with all oral bisphosphonates to maximize absorption and reduce gastrointestinal (GI) irritation [12]. Patients who cannot remain upright for 30–60 min and those with esophageal disorders require a parenteral dosing alternative. Parenteral treatment also may improve adherence for patients with high daily pill burdens, cognitive impairments or residency in long-term care facilities [13].
Quarterly intravenous (i.v.) ibandronate injection (3 mg/3 months) [14] became, in 2006, the first i.v. bisphosphonate to be approved for postmenopausal osteoporosis in the USA and the EU. This was followed in 2007 by the approval of yearly i.v. zoledronic acid infusion (5 mg/year) [15]. The risk of renal toxicity [16] requires zoledronic acid to be administered as a slow i.v. infusion over 15–30 min. By contrast, quarterly i.v. ibandronate has a renal safety profile similar to placebo [14] and is administered as a 15-s injection that can be given in a primary care practice.
This paper integrates evidence on the efficacy, safety and tolerability of the i.v. ibandronate 3 mg/3 months injection regimen approved by the US FDA and European Medicines Agency (EMEA) in 2006 for treatment of postmenopausal osteoporosis.
Pharmacology of intravenous ibandronate
Overview
Detailed information on the pharmacokinetics and pharmacodynamics of monthly and quarterly ibandronate regimens has been reviewed previously in this journal [17]. Briefly, ibandronate, (3[N-methyl-N-pentyl]amino-1-hydroxypropane-1.1-diphosphonic acid monosodium salt monohydrate) (Figure 1), is the only nitrogenous bisphosphonate with a tertiary nitrogen group. In rat models assayed by 45Ca kinetics, this structural feature confers greater antiresorptive potency than risedronate, alendronate or pamidronate [18,19]. Pharmacokinetic studies of i.v. ibandronate (0.5–6 mg) in healthy postmenopausal women [20] revealed a total area under the serum concentration–time curve of 316–908 ng/hml, a total clearance of approximately 90–130 ml/min (renal: ~60 ml/min) and terminal elimination half-life of 10–23 h. Circulating ibandronate that does not enter bone is excreted in urine.

Ibandronate.
Up to half (40–50%) of circulating ibandronate binds rapidly to bone mineral and is partially internalized by osteoclasts [21]. Within osteoclasts, ibandronate inhibits farnesyl diphosphate synthase, preventing protein prenylation and, thus, disrupting multiple processes required for bone resorption [22]. Ibandronate's inhibitory action is largely confined to osteoclasts, with the occasional exception of highly endocytic blood cells such as monocytes [22]. By-products of prenylation inhibition may accumulate in such cells and become small-molecule immune stimuli. This may be the source of transient flu-like symptoms occurring in a subset of patients on initial i.v. bisphosphonate treatment [22].
Bone turnover biomarker suppression
Ibandronate induces marked suppression of biochemical markers of bone turnover. Biomarkers commonly measured during follow-up of bisphosphonate treatment include the collagen breakdown products collagen C-telopeptide (CTX) and collagen N-telopeptide (NTX) as well as the bone formation markers osteocalcin and bone-specific alkaline phosphatase [23]. Unlike BMD, which requires up to 2 years' treatment before clinically meaningful changes can be detected [24], serum biomarker suppression begins within 3 days of ibandronate administration and reaches a nadir by 7 days postdose [25]. Early decreases of the resorption marker CTX during bisphosphonate therapy are strongly correlated with bisphosphonate-induced BMD increases at 6 months and 1 year[23]. Thus, serum biomarkers allow early monitoring and prediction of therapeutic response.
During the interdose periods of intermittent bisphosphonate regimens, biomarker levels fluctuate and gradually return toward predose levels. However, as long as interdose biomarker levels remain within the premenopausal range, fluctuations do not appear to affect efficacy materially [26].
Pharmacodynamics & efficacy
Biomarker pharmacodynamics
Bone turnover marker results of key i.v. ibandronate injection studies are summarized in Table 1. The first large-scale clinical trial of quarterly i.v. ibandronate, the Intravenous Fracture Reduction Study [27], used dosing regimens of 0.5mg/3 months and 1mg/3 months. Suppression of bone resorption and formation markers showed clear dose dependence but a modest treatment effect versus placebo. Thus, increased i.v. doses were tested in subsequent studies. In the Phase II/III Intermittent Regimen Intravenous Ibandronate Study (IRIS) [28], ibandronate 2 mg/3 months suppressed bone resorption and formation markers significantly more than a 1 mg/3 months regimen from the first measured timepoint through to the end of the study. In parallel with the biomarker results, the BMD outcomes of IRIS were dose dependent but suboptimal, prompting the higher-dose and shorter-interval regimens tested in the Phase III Dosing Intravenous Administration (DIVA) study [14,29].
Pharmacodynamic results (bone turnover biomarker changes) in key studies of quarterly intravenous ibandronate for osteoporosis.
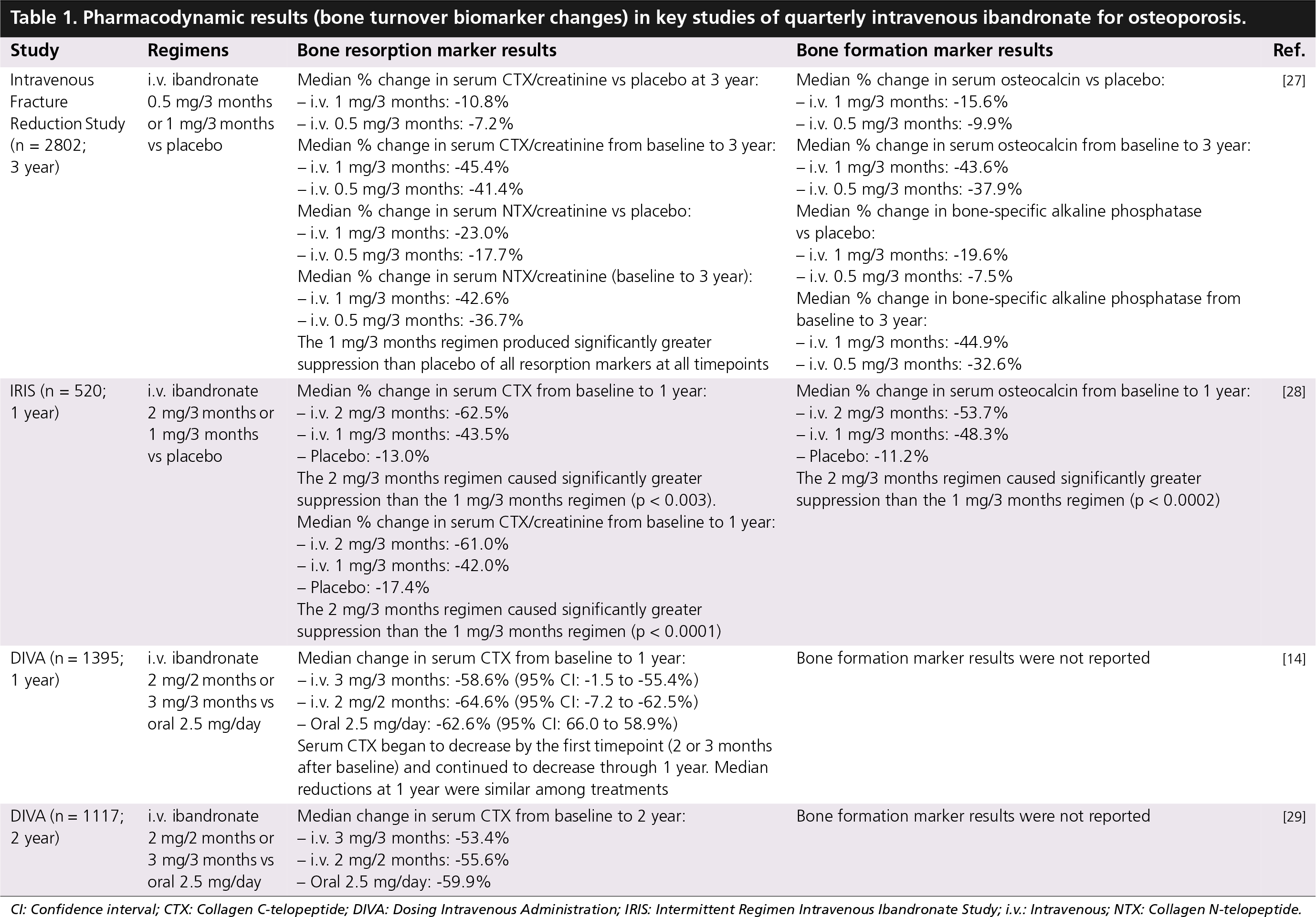
CI: Confidence interval; CTX: Collagen C-telopeptide; DIVA: Dosing Intravenous Administration; IRIS: Intermittent Regimen Intravenous Ibandronate Study; i.v.: Intravenous; NTX: Collagen N-telopeptide.
The DIVA study was designed as a bridging study, comparing the efficacy of ibandronate 3 mg/3 months or 2 mg/2 months i.v. injections with that of the oral ibandronate 2.5 mg/day regimen approved in 2003 [30]. Reductions in serum CTX observed at the first timepoint (2 or 3 months after first dose) were sustained throughout the 2-year study [29]. Median serum CTX decreases from baseline to 1 year [14] and from baseline to 2 years [29] were statistically similar among all three treatments (Table 1). Thus, the 3 mg/3 months and 2 mg/2 months i.v. regimens maintained residual bone turnover suppression levels comparable to those achieved with the previously approved daily oral regimen.
BMD efficacy
The primary efficacy measure of DIVA was mean change from baseline to 1 year [14] or 2 years [29] in lumbar spine BMD (Table 2). Both i.v. regimens were found to be as efficacious as daily oral ibandronate [14,29]. Subsequent prespecified analyses of variance demonstrated statistical superiority of the i.v. regimens to daily oral treatment at the lumbar BMD primary end point. Similarly, proximal femur BMD parameters showed significantly greater increases with i.v. than with the daily oral regimen.
Key efficacy studies of quarterly intravenous ibandronate in osteoporosis.

ANOVA: Analysis of variance; BMD: Bone mineral density; DIVA: Dosing Intravenous Administration; IRIS: Intermittent Regimen Intravenous Ibandronate Study; i.v.: Intravenous.
At the completion of the 2-year DIVA study [29], BMD responder rates were significantly greater for both i.v. regimens than for daily oral ibandronate. For example, 92.8% of 3mg/3 months i.v. recipients, but only 84.7% of 2.5 mg/day oral recipients (p ≤ 0.004), had measurable lumbar BMD increases baseline at 2 years; 53.1% of 3 mg/3 months i.v. recipients and 37.7% of 2.5 mg/day oral recipients (p ≤ 0.002) achieved less than or equal to 6% lumbar BMD increases [29].
The demonstration of DIVA's superior BMD efficacy and response rates with 3 mg/3 months i.v. ibandronate injection led to the approval of this quarterly i.v. regimen in early 2006.
Fracture reduction efficacy
The efficacy of bisphosphonates for fracture prevention as well as for BMD improvement requires sustained suppression of bone turnover. Continuous bisphosphonate treatment, for example, weekly oral alendronate or risedronate, obtains pharmacodynamic effect with a linear multiple of the daily dose [31]. Intermittent ibandronate regimens (quarterly i.v. or monthly oral), by contrast, require nonlinear dose increases versus daily [26]. The efficacy of quarterly or monthly ibandronate regimens depends upon their annual cumulative skeletal exposure (ACE): the product of the dose amount, number of doses per year and an absorption factor (100% for i.v.; 0.6% for oral) [32]. In the pooled meta-analysis of Adachi et al., patients receiving ACE 12 mg (i.e., either of the DIVA i.v. regimens) had a 43% reduced risk of nonvertebral fractures and a significantly longer time to fracture versus ACE 5.5 mg (daily oral) [33]. These results accord with earlier analyses showing clinical and nonvertebral fracture risk reduction with ACE greater than 10.8 mg versus ACE 5.5 mg [34] or placebo [35]. Thus, quarterly i.v. ibandronate achieves nonvertebral fracture protection, in addition to the BMD gains reported in DIVA.
Safety & tolerability
A detailed review of monthly oral and quarterly i.v. ibandronate drug-related adverse events (AEs) has been presented previously in this journal [17]. This discussion will accordingly focus on safety and tolerability issues of current clinical interest.
GI tolerability
Quarterly i.v. ibandronate bypasses the potential for GI irritation associated with oral bisphosphonates and may improve tolerance and adherence. The PRIOR study enrolled 543 osteoporotic or osteopenic women who had discontinued prior daily or weekly oral bisphosphonates owing to GI intolerance [36]. Participants self-selected their study ibandronate regimens (150 mg/month oral [73%] or 3 mg/3 months i.v. [27%]) and were allowed to switch once. GI tolerance was assessed using the self-report GI Experience Survey. For both PRIOR treatment regimens, mean GI tolerance scores after 1 month of study treatment were significantly higher than at screening. Adherence to original study regimen was 83% for i.v. and 70% for oral. Only 3.9% of participants (13 oral and eight i.v.) withdrew because of a GI AE. Thus, quarterly i.v. ibandronate injection may improve GI safety, tolerance and adherence in patients with adverse GI histories associated with taking oral bisphosphonates.
Flu-like syndrome
Immune stimulation (γδ T-cell response) may result from prenylation inhibition, causing transient flu-like symptoms in occasional patients on initial exposure to i.v. bisphosphonates [22]. This syndrome, when present, generally begins within 3 days of dosing and resolves by 7 days postdose. Over the 2-year course of DIVA [29], investigators reported ‘influenza-like illness’ or ‘acute-phase reaction’ in 4.9% of the 3 mg/3 months group and 5.6% of the 2 mg/2 months group, versus 1.5% in the daily oral group. A DIVA exploratory analysis examined 33 symptoms potentially indicative of an acute-phase reaction (e.g., myalgia, arthralgia, headache, fatigue and malaise). Overall, 10.0% of 3 mg/3 months recipients, 15.6% of 2 mg/2 months recipients, and 4.5% of daily oral recipients had any individual AE of this set [29]. In PRIOR, 2% of i.v. users and 1% of oral users of ibandronate reported flu-like symptoms [36]. Experience suggests that these symptoms are transient, treatable with antipyretics and generally tolerable.
Cardiac, dental & renal adverse events
Quarterly i.v. ibandronate injection has a favorable profile with respect to AEs of clinical concern for i.v. bisphosphonate use. Serious atrial fibrillation was significantly more frequent with zoledronic acid than placebo in the pivotal Health Outcomes and Reduced Incidence with Zoledronic acid Once yearly (HORIZON) osteoporosis trial [15]; however, when overall atrial fibrillation events (including nonserious) were considered, treatment groups did not differ. In DIVA [29], no serious atrial fibrillation events related to ibandronate were reported.
Osteonecrosis of the jaw (ONJ) is a painful AE predominantly associated with potent i.v. bisphosphonates at the high and frequent dosing levels required to manage bone complications of cancer; it is much less common with osteoporosis treatment regimens. In a large ONJ review of 368 cases (comprising 350 cancer patients, 15 osteoporosis patients and three Paget's disease patients), 124 patients had received zoledronic acid [37]. Among the 15 ONJ cases in osteoporosis patients, one involved solely ibandronate, one involved ibandronate and zoledronic acid, and 13 involved alendronate, the agent that had been longest in marketed clinical use at the time of the case review [37]. Blinded safety adjudication of the HORIZON study revealed one case of ONJ each among zoledronic acid and placebo recipients [15]. No cases of ONJ have been reported in any controlled trial of ibandronate for postmenopausal osteoporosis [14,20,29].
Zoledronic acid's potential for nephrotoxicity requires it to be administered as a 15–30 min i.v. infusion, generally at a specialist infusion center (e.g., hematology–oncology clinic) [16]. Decreases in renal function occurred in 9–15% of patients receiving i.v. zoledronic acid or pamidronate in trials of treatment for bone metastases, and 72 cases of renal failure were reported in such patients in 2001–2003 [16]. In the HORIZON osteoporosis study, serum creatinine increased by less than 0.5 mg/dl 9–11 days postdose in significantly more zoledronic acid than placebo recipients [15]. These changes appeared to be transient, as creatinine levels returned to within 0.5 mg/dl of baseline within a month in most cases and did not differ significantly between zoledronic acid and placebo groups at the end of 3 years' follow-up [15]. Clinically relevant changes in serum creatinine affected 12 patients in the DIVA study, all of whom had comorbidities and concomitant medications capable of affecting kidney function; the changes were determined to be unrelated to ibandronate use [29]. Ibandronate's renal safety profile allows simple administration as a 15-s i.v. injection by a primary care practitioner.
Postmarketing surveillance
The US FDA issued an Early Communication of Safety Review in 2007 (shortly after the publication of HORIZON [15]) regarding concerns about atrial fibrillation with bisphosphonates; this document stated that prescribing patterns need not change at this time [38].
On January 7, 2008, the FDA issued an Information for Healthcare Professionals statement highlighting the possibility of severe musculoskeletal pain (distinct from the transient myalgia of flu-like syndrome) developing in patients receiving bisphosphonates [39]. The statement advised clinicians to consider a possible relationship to bisphosphonate use when patients present with such pain, to monitor these patients and to consider benefits and risks of bisphosphonate use. Further FDA evaluation is ongoing.
Regulatory affairs
Quarterly i.v. ibandronate injection (3 mg/3 months) received approval in 2006 from the FDA and the EMEA for the treatment of postmenopausal osteoporosis.
Conclusion
Quarterly i.v. ibandronate injection, approved in 2006 for the treatment of postmenopausal osteoporosis, offers safe and effective therapy that bypasses GI dosing requirements and allows physicians to monitor adherence. The DIVA study demonstrated superior BMD gains and response rates with the quarterly i.v. regimen versus daily oral ibandronate. Regimens with high ACE to ibandronate (including 3 mg quarterly i.v.) are associated with increased fracture protection as compared with daily oral ibandronate. Drug-related atrial fibrillation, a potential source of concern with i.v. zoledronic acid, has not been reported in trials of i.v. ibandronate, which also has a favorable renal safety profile allowing convenient, simple i.v. injection. Thus, quarterly i.v. ibandronate injection offers improved tolerability and convenience for patients with GI disorders, those who are bedfast and those with challenges to oral bisphosphonate adherence.
Future perspective
Directions inviting further research include combination osteoporosis therapies, potential extension of bisphosphonate indications to include prevention of osteoporosis, novel biological antiresorptive and anabolic agents, and application of a new WHO absolute fracture risk model to diagnosis and treatment.
Bisphosphonates ameliorate osteoporosis chiefly by inhibiting resorption by osteoclasts [22], although some evidence exists for additional effects on osteoblasts [40]. Thus, combining bisphosphonates with bone-forming therapies may achieve greater efficacy at lower doses of each agent. For example, alfacalcidol, a vitamin D hormone analog, combined with daily oral alendronate, has shown greater BMD increases and response rates than either agent alone [41,42]. Combinations of bisphosphonates with parathyroid hormone (PTH) or its truncated peptide teriparatide are being evaluated; interestingly, the sequencing of these agents affects their combined efficacy. Alendronate administered simultaneously with PTH [43] or teriparatide [44] blunts their efficacy; conversely, sequential treatment (1 year of PTH followed by 1 year of alendronate) maintains or increases BMD gains attained on PTH [45]. Bisphosphonate use after completion of PTH [44] or teriparatide [46] therapy is essential for BMD maintenance. Less frequent bisphosphonate regimens, such as quarterly i.v. ibandronate, may potentially extend the benefits of post-PTH or post-teriparatide bisphosphonate treatment to a wider range of patients.
Low-dose quarterly i.v. ibandronate (up to 2 mg/3 months) significantly improved BMD when used for prevention of bone loss in the immediate postmenopausal period [47]; expanding the indication for an i.v. preventive regimen can provide an alternative for women with osteopenia and other fracture risks who cannot or choose not to take oral agents.
Denosumab, an inhibitory monoclonal antibody against the RANKL, reduces osteoclast differentiation, activation and survival, and thus exerts antiresorptive activity. In a recent Phase II study, subcutaneous denosumab every 3 or 6 months for 12 months induced significantly greater BMD increases than placebo at the lumbar spine and proximal femur [48]. Unlike oral alendronate, denosumab also increased BMD at the distal radius, a site rich in cortical bone.
Sclerostin, the product of the SOST gene, is produced by osteocytes and potently inhibits bone formation by decreasing osteoblast number and activity [49]. Mechanical loading suppresses sclerostin production, allowing increased bone formation [50]. Monoclonal antibodies to sclerostin are in development. Studies in ovariectomized rat and monkey models [51,52] have shown improvements in bone density, quality and histomorphometric parameters with subcutaneous monoclonal antibodies to sclerostin, suggesting their potential as anabolic agents in the treatment of osteoporosis.
Executive summary
Ibandronate inhibits farnesyl diphosphate synthase, preventing protein prenylation and disrupting bone resorption by osteoclasts.
Prenylation inhibition may activate acute-phase responses with initial intravenous bisphosphonate administration.
After intravenous administration, circulating ibandronate binds rapidly to bone (40–60%) or is excreted in urine.
Ibandronate suppresses serum markers of bone turnover within 3 days of dosing.
Efficacy of intermittent ibandronate use is determined more by residual interdose bone turnover levels than by patterns of fluctuation.
The 2-year Dosing Intravenous Administration (DIVA) study demonstrated that 3 mg/3 months intravenous ibandronate injection achieved superior lumbar and hip bone mineral density gains compared with 2.5 mg/day oral ibandronate.
Meta-analyses have shown efficacy of this intravenous regimen against nonvertebral fractures.
In the PRIOR study, intravenous ibandronate achieved improved tolerance and adherence in women who had discontinued oral bisphosphonates as a result of gastrointestinal intolerance.
Quarterly intravenous ibandronate has a favorable renal safety profile, allowing 15-s intravenous injection.
No reports of drug-related atrial fibrillation are associated with intravenous ibandronate.
In clinical trials of intravenous ibandronate for osteoporosis, no cases of osteonecrosis of the jaw have been reported.
Flu-like symptoms occurring on first intravenous ibandronate use in a few patients are transient, treatable and tolerable.
Quarterly intravenous ibandronate (3 mg) is administered as a 15-s intravenous injection in a physician's office.
FRAX™, a new WHO-sponsored model to predict 10-year absolute fracture risk, incorporates as risk factors BMI, personal and parental history of fracture, glucocorticoid use, rheumatoid arthritis, smoking and excess alcohol intake, with or without BMD [53,54]. This model promises to improve osteoporosis diagnosis and treatment at both patient care and public health levels. Individualized assessment of absolute fracture risk will allow physicians to customize lifestyle, physiotherapeutic and pharmacotherapeutic interventions. Patient-friendly bisphosphonate regimens, such as quarterly i.v. ibandronate, as well as emerging combination, anabolic and biological therapies, will contribute importantly to multi-disciplinary osteoporosis management in the FRAX era.
Footnotes
Ronald Emkey is on the speakers bureau for GlaxoSmithKline, Merck, Roche and Novartis and is a consultant to Merck, GlaxoSmithKline, Roche and Novartis. The author has no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
The author acknowledges the editorial assistance of Kim Coleman Healy, PhD of Envision Pharma, USA, in the development of this manuscript. Editorial assistance was supported by an independent grant from Roche.
