Abstract
Within the last decade, several studies have investigated whether adjuvant treatment of breast cancer affects cognitive function. A number of prospective studies have demonstrated inconsistent results regarding whether chemotherapy affects cognitive function. Approximately half of the studies demonstrated subtle cognitive decline in a wide range of domains among some breast cancer patients following chemotherapy, and half did not. Concomitant changes in brain structure and function have been identified in neuroimaging and neurophysiologic studies. Estrogenic therapy has been specifically associated with deterioration in verbal memory and processing speed. However, evidence is mostly based on smaller studies with cross-sectional data. Breast cancer patients who underwent both chemotherapy and estrogenic therapy showed the most deterioration and the most persistant decline in cognitive function. Since cognitive impairment is subtle, if evident at all, discrepant findings are due to hormonal, physiological, psychological or temporal confounding variables and differences in study design. Neuropsychological training has been demonstrated to improve cognitive dysfunction experienced by breast cancer patients after chemotherapy. Future research may examine the unique impact of endocrine therapy on cognitive function with prospective, controlled trials, as well as the role of further confounding variables (e.g., menopausal status, cytokine deregulation, cortisol and concurrent medication).
Keywords
Breast cancer therapy consists of a combination of different treatment modalities, including surgery, radiation, chemotherapy and hormonal therapy, which represent standard treatment options. These treatments involve a range of side effects affecting women's quality of life, with potential effects on body image, mood, the occurence of hot flashes and induced menopause being among those highly prevalent. Furthermore, consequences of breast cancer treatment on cognitive function have been reported. A growing body of research suggests that chemotherapy has an impact on cognitive function in some breast cancer patients. Since estrogen has neuroprotective effects [1], endocrine therapy may influence cognitive function in addition to chemotherapy. Therefore, the present review will assess the consequences of breast cancer treatment on women's cognitive function, with a focus on the impact of chemotherapy and endocrine therapy, and is based on a literature search of PubMed and PsycInfo databases (until January 2009) of published English language articles. Studies that described only subsamples of later studies have not been reported, so as not to bias the summary of results.
Chemotherapy & cognitive function
Some evidence supports the notion that chemotherapy regimens pass the blood–brain barrier [2]. Accordingly, treatment with chemotherapy may have some consequences for breast cancer patients' cognitive function (see
Observational studies on chemotherapy and cognitive function.
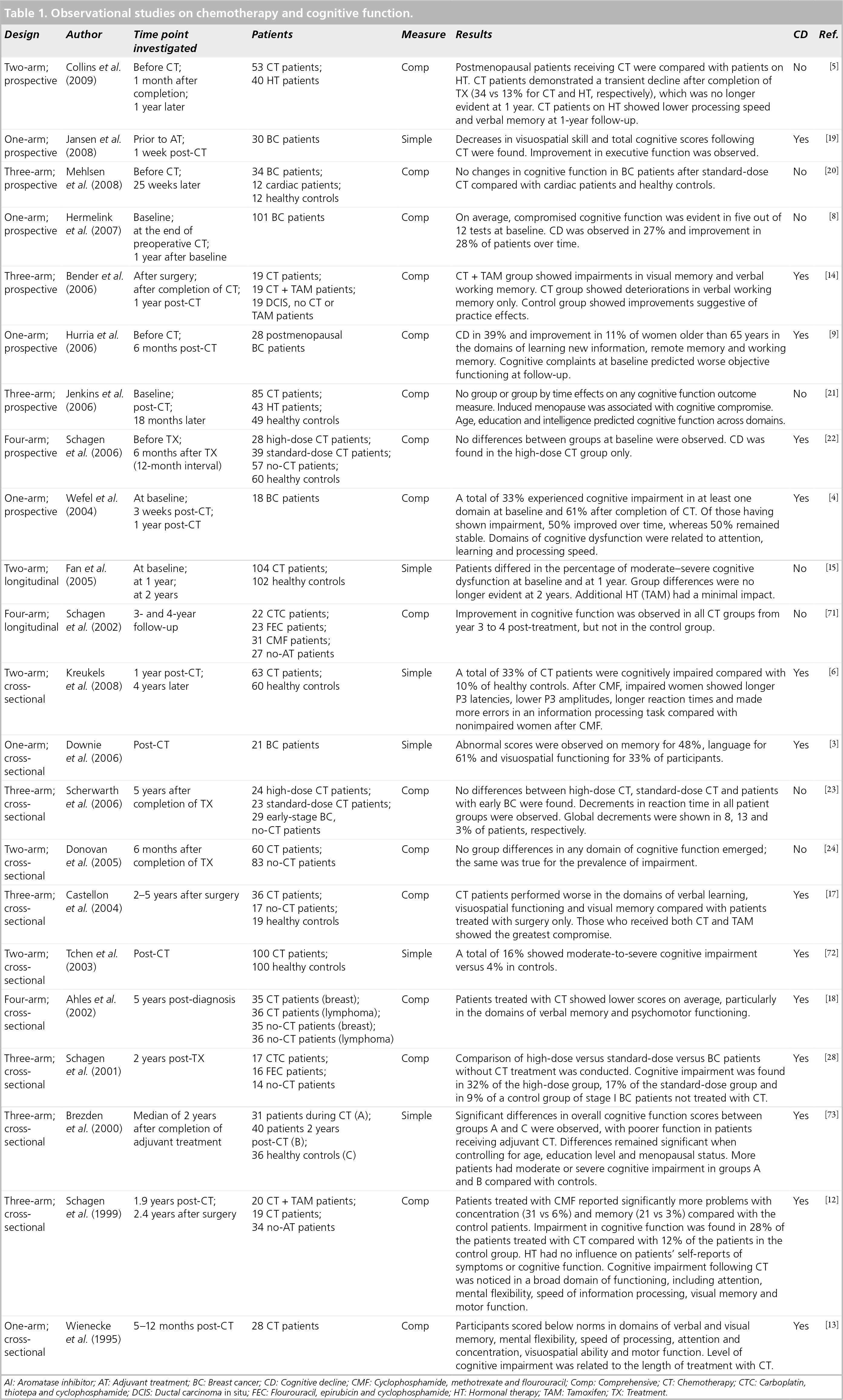
AI: Aromatase inhibitor; AT: Adjuvant treatment; BC: Breast cancer; CD: Cognitive decline; CMF: Cyclophosphamide, methotrexate and flourouracil; Comp: Comprehensive; CT: Chemotherapy; CTC: Carboplatin, thiotepa and cyclophosphamide; DCIS: Ductal carcinoma in situ; FEC: Flourouracil, epirubicin and cyclophosphamide; HT: Hormonal therapy; TAM: Tamoxifen; TX: Treatment.
Cognitive impairment has been found in multiple domains of cognitive function; many studies demonstrated longer reaction times [6,11] and slower information processing [4–6,9,12,13] in patients treated with chemotherapy. Furthermore, frequent domains of cognitive function affected by chemotherapy are working memory [9,14–16] and verbal memory [5,13,16–18]. In addition, some studies also provide evidence of associations between chemotherapy and deteriorations in visuospatial memory [3,12–14,17,19], remote memory [9] and attention [4,12,13].
A few studies have also shown a dose–response relationship with breast cancer patients treated with high-dose chemotherapy demonstrating more severe cognitive impairment compared with women treated with standard-dose chemotherapy [10,12,18].
While most studies with a cross-sectional design concluded that breast cancer patients who had undergone chemotherapy provided evidence of some degree of cognitive impairment in different domains of functioning, the nine existing prospective studies provide a different picture [4,5,8,9,14,19–22]. Of these, five studies reported overall decrements in cognitive function [4,9,14,19,22]. However, three [4,9,19] of these five studies are characterized as one-arm trials with 30 participants or less. Four prospective studies did not provide evidence of overall cognitive decline [8,20,23,24]. Furthermore, four of the nine studies also reported improvement of cognitive functioning in some patients [4,8,14,19].
Several factors may explain these discrepant findings between cross-sectional and prospective trials. Since some studies (see
Cognitive function prior to/without adjuvant treatment.

AT: Adjuvant treatment; BC: Breast cancer; CD: Cognitive decline; Comp: Comprehensive.
One possible explanation for decrements in objective cognitive function, even before commencement of chemotherapy, could be a ‘diagnostic shock’ following a diagnosis of cancer. Several studies have shown that objective cognitive function is not related to subjective cognitive complaints. Objective cognitive function has been predicted by age, education and IQ [7,21,26], whereas subjective cognitive function has been associated with negative affect and fatigue [17,26].
Study results are mixed regarding whether cognitive disturbances are transient [4,5] or permanent [16,21]. Although many recent studies are prospective in nature, they do not include a long-term follow-up and/or lack a control group of patients who did not undergo adjuvant treatment or, alternatively, healthy controls. Hence, the strongest evidence can be derived from prospective studies with long-term follow-up (≥1 year after completion of treatment with chemotherapy) that include a control group of women with breast cancer who received local treatment only. While one of these high-quality studies suggests no impact of chemotherapy on cognitive function [21], other studies provide evidence of cognitive disruption as being transient in nature [5] or demonstrate long-term persistence of cognitive decline in breast cancer patients treated with chemotherapy [14]. Hence, study results remain inconclusive at present as to whether cognitive disruption is transient or permanent. The majority of studies, if evident at all, suggest subtle impairment in cognitive functioning in a subgroup of breast cancer patients treated with chemotherapy. In order to determine whether cognitive dysfunction is a consequence of chemotherapy treatment that persists in breast cancer patients, studies also need to control for the effect of endocrine treatment on cognitive function. Since many patients who have undergone chemotherapy usually continue with endocrine treatment for up to 5 years, interaction effects of chemotherapy and endocrine therapy need to be taken into account. Indeed, a few studies suggest that patients who had undergone both treatment modalities experienced the most pronounced cognitive disruption [5,14,17], and the domains of cognitive impairment appear to be distinct to the type of endocrine therapy [27]. Therefore, future studies should control for hormonal treatment.
Mechanisms
A number of studies have examined potential mechanisms by which treatment with chemotherapy may result in cognitive dysfunction. Among these are studies using electrophysiological, neuroimaging and genetic techniques, as well as studies investigating immune responses.
Event-related potential (ERP) studies assessed during a cognitive task detected electrophysiological alterations, such as reduced P3 latencies [28,29]. Differences were also observed between patients treated with different chemotherapy regimens 3–6 years post-treatment [29]. The extent of alteration was related to the degree of cognitive disruption, although not all studies were confirmatory [30]. Longer P3 latencies have been demonstrated for simple and complex cognitive tasks in patients who received treatment 5 years before the examination [30,31]. Schagen and co-authors found no correlation between neurophysiologic parameters and neuropsychological performance, except for an overall relation between longer P300 latencies and the total number of deviant test scores [28]. Furthermore, asymmetry of the α-rhythm of 0.5 Hz or more was detected in seven patients treated with high-dose chemotherapy, in two patients who had undergone standard chemotherapy and in none of the control patients. No differences were found between the groups with regard to frequency of α-rhythm, α-blocking or latency of P300. The absence of group differences in N1 suggests that early perceptual processes are not affected by chemotherapy [29].
Brain imaging studies conducted via functional MRI during a short-term recall task in breast cancer survivors 5–10 years after completion of chemotherapy demonstrated specific alterations in the activity of the frontal cortex, the cerebellum and the basal ganglia [32]. However, effects on the basal ganglia were only evident in patients treated with both chemotherapy and tamoxifen (TAM). Positron emission tomography imaging studies demonstrated lower resting brain metabolism and a larger area of frontal cortex activation in breast cancer patients treated with chemotherapy [32].
White matter changes have been shown via structural MRI in women treated with high-dose chemotherapy [33]. Furthermore, a twin case study indicated small differences in neuropsychological test performance but striking contrasts in self-reported cognitive complaints as well as structural and functional MRI differences. Specifically, the twin who underwent chemotherapy had substantially more subjective cognitive complaints and more white matter hyperintensities on structural MRI. Using functional MRI, an expanded spatial extent of brain activation during working memory processing was found compared with the non-affected twin [34]. This study suggests that chemotherapy may not only be related to abnormalities in brain structure, but also to abnormalities in brain functioning. Furthermore, after completion of treatment with chemotherapy, compensatory processes may occur since differences in brain structure and functioning did not (or no longer) translate into discrepant neuropsychological performance.
Genetic investigations demonstrated that survivors with at least one ∊4 allele of the APOE gene scored significantly lower in visual memory and spatial ability domains, as well as tended to score lower in the psychomotor functioning domain when compared with survivors who did not carry an ∊4 allele of this gene [35]. The results of this study provide preliminary support for the hypothesis that the ∊4 allele of the APOE gene may be a potential genetic marker for increased vulnerability to chemotherapy-induced cognitive decline.
Both cognitive dysfunction [11,25] and elevated levels of proinflammatory cytokines [36,37] have also been demonstrated in breast cancer patients before commencement of chemotherapy. Hence, cytokines released from tumor tissue might cause cognitive disorders before, during and after treatment [38]. Consequently, cognitive changes may also occur by inflammation, and treatment with chemotherapy may add to cognitive dysfunction through different pathways.
Endocrine therapy & cognitive function
A large body of research suggests that estrogen increases neuroplasticity, enhances or maintains verbal memory and reduces the risk of Alzheimer's disease [39]. By contrast, results of the Women's Health Initiative Memory Study (WHIMS) examining the use of hormone-replacement therapy (HRT) on cognitive function have been discouraging [40] and several theoretical and clinical issues with regard to the use and initiation of HRT have not yet been clarified [39]. In this trial women who underwent HRT performed worse in tests of cognitive function than participants who underwent natural menopause [40]. Derived from the WHIMS, the timing of HRT is considered critical for neuroprotective effects leading to the ‘critical period hypothesis’. This hypothesis suggests that the initiation of HRT should start around the time of the onset of menopause in order to display neuroprotective effects, whereas the initiation of HRT after estrogen depletion would result in null or even detrimental effects on memory [1].
In cancer patients, the effects of endocrine therapies on cognitive function have been studied to a lesser degree. Endocrine therapy for breast cancer includes treatment with selective estrogen receptor modulators (SERMs) and aromatase inhibitors (AIs). TAM and raloxifene (RLX) represent common SERMs. Unlike estrogen, SERMs have estrogen agonist effects on some tissues and estrogen antagonist effects on other tissues. TAM has estrogen antagonist effects on the breast but estrogen agonist effects on other tissues, such as the endometrium. The mode of action of TAM on the brain is not well understood.
Raloxifene is a drug that is not specifically for breast cancer patients but for postmenopausal women with osteoporosis. Similar to estrogen, RLX has agonist effects on bone and lipid metabolism, but estrogen antagonist effects on reproductive tissues. Hence, it does not increase the risk of breast and uterine cancer.
A second group of drugs, AIs, are used in postmenopausal women with breast cancer. AIs represent newer agents of endocrine therapy. While TAM blocks receptors of female hormones, AIs prevent the transformation (aromatization) of androgens into estrogen.
Observational studies of endocrine therapies on cognitive function in patients with breast cancer are described in more detail in
Observational studies on endocrine therapy and cognitive function.
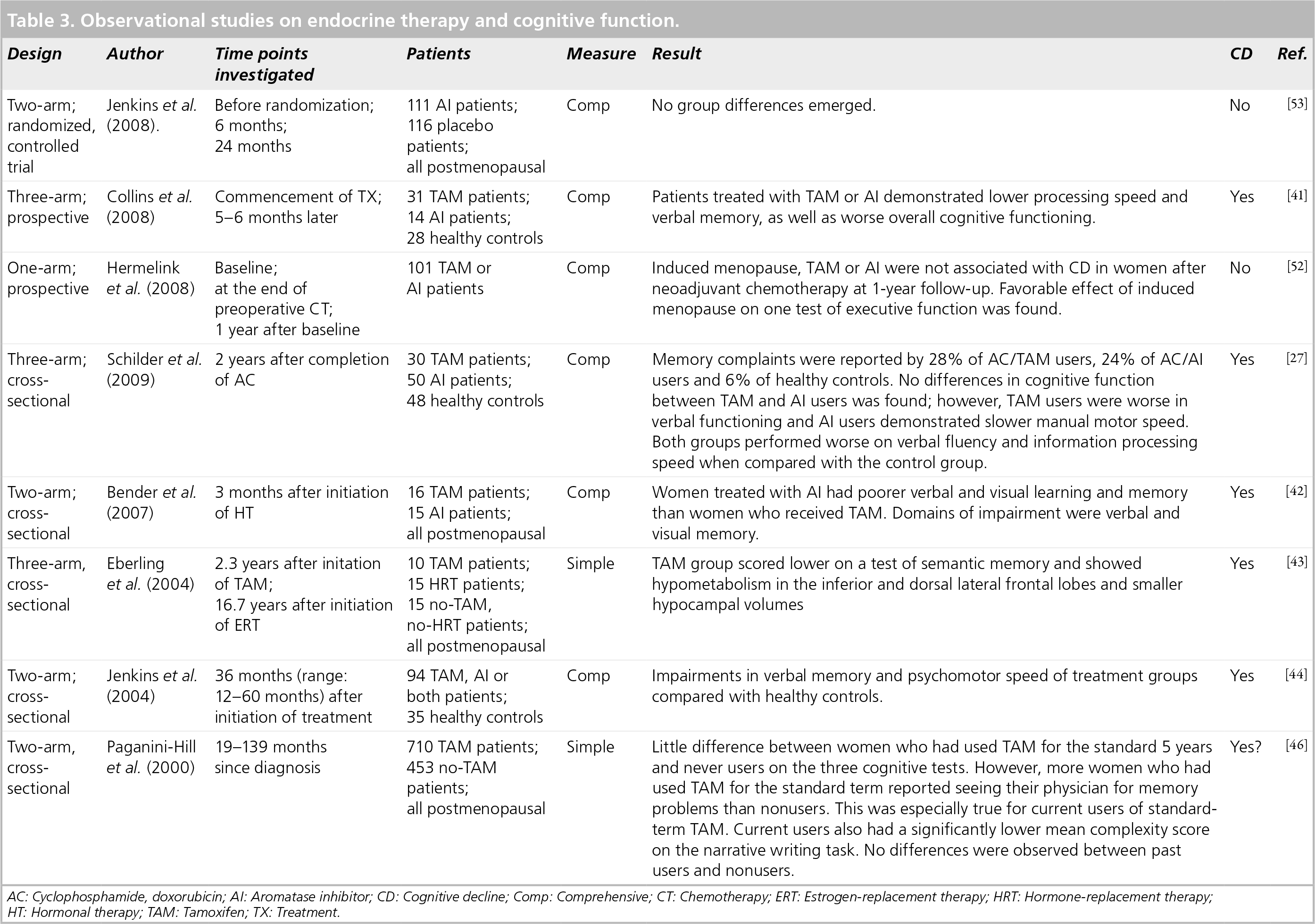
AC: Cyclophosphamide, doxorubicin; Al: Aromatase inhibitor; CD: Cognitive decline; Comp: Comprehensive; CT: Chemotherapy; ERT: Estrogen-replacement therapy; HRT: Hormone-replacement therapy
HT: Hormonal therapy; TAM: Tamoxifen; TX: Treatment.
By contrast, another SERM, RLX has demonstrated null [47–49] or small [50,51] neuroprotective effects on the brain. In a large trial investigating treatment with RLX for 3 years, the drug was associated with reduced risk of cognitive impairment [51].
Aromatase inhibitors have been compared with TAM in terms of their effects on cognitive function in four studies. Of these, one study suggests that AIs have a comparable impact to TAM on cognition [44], while Bender et al. reported poorer verbal and visual memory in AI users compared with TAM users [42], and Schilder et al. found that TAM users fared worse in verbal functioning and AI users were slower on motor speed [27]. Concomitant lower plasma estrogen levels in women treated with AI compared with TAM could suggest a more profound effect of AI on cognitive function.
To date, only three prospective studies examining the effects of TAM and/or AI on cognitive function exist [41,52,53]. While Collins et al. found decrements in verbal memory and processing speed as well as in overall cognitive functioning in women with breast cancer treated with TAM or AI compared with healthy controls [41], another study [52] found no effect of TAM or AI treatment on memory in women who had previously undergone neoadjuvant chemotherapy. However, this study did not include a comparison with groups of women with endocrine treatment only or chemotherapy treatment only.
In contrast to the aforementioned studies, a double-blind, placebo-controlled, randomized trial investigating AI versus placebo in postmenopausal women at risk for breast cancer [53] did not demonstrate differences in cognitive function for women treated with AI compared with placebo at 24-month follow-up. While this study was not conducted in breast cancer patients, its significance is based on a randomized, controlled trial design. Owing to the large sample size, it is not likely that an existing group difference could have been overlooked. In addition, confounders, such as previous treatment with a different chemotherapy regimen or menopausal status, were excluded.
Other hormonal agents, such as analogs of gonadotropin-releasing hormone, have not been investigated for their impact on cognitive function. These drugs (e.g., goserelin) are used in premenopausal women with breast cancer to ablate ovarian function. However, a few studies in healthy women suggest that cognitive function might not be affected by usage of gonadotropin-releasing hormone [54–56].
Mechanisms
A few studies have examined the potential mechanisms of SERMs on brain function using MRI and positron emission tomography methods. Two studies compared three groups of postmenopausal women, women who had received either TAM, estrogen or neither [43,57]. Ernst and co-authors found that the groups treated with TAM or estrogen both showed lower concentrations on one of four metabolites in the basal ganglia [57]. The authors, therefore, concluded that TAM has similar effects to estrogen. By contrast, Eberling et al. demonstrated TAM users to have widespread hypometabolism in the interior, dorsal lateral and frontal lobes as well as smaller right and left hippocampal volumes [43]. Hippocampal volumes of women using neither TAM nor estrogen were intermediate to the two other groups. Nonusers also demonstrated lower metabolism in the inferior frontal and temporal cortexes compared with the estrogen users. Consequently, the two studies provide conflicting results. While one study [57] concludes that TAM has a similar effect to estrogen on brain metabolism, the other [43] showed effects of TAM that were distinct from estrogen users and nonusers. An animal study of ovarectomized rats investigating the effects of different SERMs and estrogen on hippocampal neurons found SERMs to have different dose-dependent neuroprotective effects that were distinct from estradiol [58], supporting the Eberling et al. findings in humans [43]. Differential effects of specific SERMs on brain function in an animal study [58] also correspond to neuropsychological findings suggesting that TAM probably has detrimental effects, whereas RLX probably has slightly favorable effects on some aspects of cognitive function.
Distinct effects of AIs on brain function have not been studied as yet. To conclude, mechanisms of estrogenic therapy for breast cancer on brain function have been scarcely studied and it remains to be clarified in what respect their effects are distinct versus comparable to the use of HRT.
Treatment of cognitive function
A few drugs as well as psychological interventions have been investigated to prevent cognitive decline in breast cancer patients (who were treated with chemotherapy). While one study suggests that erytropoetin, a drug for the prevention and treatment of anemia and fatigue in cancer patients, may have attenuated the cognitive impairment and fatigue that occurred during adjuvant breast cancer chemotherapy [59], in another study, no effect of this drug on cognitive function was observed [60]. In addition, methylphenidate, a CNS stimulant approved for attention-deficit/hyperactivity disorder and narcolepsy, did not show beneficial effects on memory [61]. A recent study demonstrated that modafinil, a drug used to treat sleep apnea, modestly improved short-term memory in breast cancer patients [62].
By contrast, behavioral interventions appear to have more potential to improve cognitive dysfunction in breast cancer patients who have undergone chemotherapy. Neuropsychological training, so called Memory and Attention Adaptation Training (MAAT), demonstrated improvements in objective measures of cognitive function and also improved stress management in dealing with memory problems in everyday life at 2- and 6-month follow-up [63]. The MAAT consists of a participant workbook, four individual monthly visits of 30–50 min and phone contacts between visits. The four cognitive–behavioral components include:
Education on memory and attention;
Self-awareness training;
Self-regulation emphasizing arousal reduction through relaxation training, activity scheduling and pacing;
Training of cognitive compensatory strategies.
The application of strategies was considered the homework between visits. The telephone contacts were intended to assist in applying the strategies.
With regard to the inhibition of mechanisms contributing to inflammation, a randomized, controlled trial using cognitive–behavioral stress management demonstrated increased benefit finding and reduced cortisol levels. Effects on cortisol were mediated by benefit finding [64]. A different trial demonstrated improved lymphocyte proliferation under the same treatment condition [65]. Neurodamaging effects of stress and inflammation have been indicated in a number of studies [66–68]. Hence, behavioral interventions that usually target other goals may also prevent cognitive impairment in breast cancer patients.
Future perspective
Considering the inconsistent findings with regard to cognitive dysfunction due to treatment with chemotherapy, the characterization of subgroups at risk for cognitive decline would be an important target for future research. The identification of vulnerable subgroups would allow an early intervention targeted at maintaining cognitive function in these patients. Looking at studies that demonstrated neurophysiologic compensation [34], it can be assumed that prevention of cognitive impairment may be possible. As neuropsychological training and stress-management techniques have been effective in reversing cognitive decline, its preventive and rehabilitative application could have some merit for clinical practice.
Otherwise, in cases where reasonable choices exist between different chemotherapy regimens, there could be a preference to opt for the less aggressive regimen since evidence suggests a dose–response relationship with women treated with high-dose chemotherapy, demonstrating more pronounced cognitive symptoms compared with patients treated with standard-dose chemotherapy [10,12,18]. However, decision making in this regard should be carried out following extended counseling of the patient that allows her to weigh the pros and cons of each decisional option.
Owing to the fact that results on chemotherapy-related cognitive dysfunction were discrepant regarding whether they are transient or persistent, future studies should include longer follow-up to more thoroughly address this issue.
Several biological mechanisms have been identified that may underlie cognitive decline in breast cancer survivors treated with chemotherapy. If cognitive changes were transient in nature, it would be interesting to see whether biological alterations would also improve, or, in cases where neurophysiologic compensation is occurring, whether these effects are also associated with improvements in cognitive function assessed by neuropsychological testing.
Endocrine therapy has been demonstrated to have detrimental effects on cognitive function, specifically with regard to verbal memory and processing speed in most studies. However, these studies were mostly cross-sectional and one of the three prospective studies is a secondary data analysis of breast cancer patients that received prior treatment with chemotherapy [52]. Collins and co-authors found that postmenopausal women with breast cancer treated with TAM or AI demonstrated lower scores on verbal memory, processing speed and overall cognitive functioning compared with healthy controls [41]. By contrast, the International Breast Cancer Intervention Study (IBIS)-II trial [53], a double-blind, randomized, controlled trial with a large sample of women at high risk for breast cancer, found no detrimental effect of AI on cognitive function.
While the results of the two latter studies are conficting, both studies are free of some major confounds, such as pretreatment with chemotherapy and mixed menopausal status. Consequently, effects of endocrine treatment on cognitive function need to be studied with more robust research designs, such as prospective trials with larger sample sizes controlling for major confounds (e.g., chemotherapy pretreatment, hormone-replacement pretreatment and menopausal status).
In some studies, patients treated with endocrine therapy in addition to chemotherapy performed worse cognitively compared with breast cancer patients treated with chemotherapy alone, or the detrimental effects on cognition were more permanent than in patients treated with chemotherapy alone [5,14,17]. These findings may support the argument for a shared vulnerability of some women who experience cognitive decline as a consequence of chemotherapy and/or endocrine therapy.
Little is known about whether menopausal status of patients affects the extent to which breast cancer patients experience cognitive decline in response to adjuvant treatment. Several studies were completed in postmenopausal women. Nevertheless, most research was conducted on samples of both pre- and post-menopausal women and, in most studies, a significant proportion of postmenopausal patients at some point underwent HRT. However, the small sample sizes of most existing controlled, prospective trials deal with problems of statistical power and hence may not reliably detect significant moderator effects. While early estrogen deprivation in premenopausal women may have more profound effects on long-term cognitive function compared with older women, the latter group's potential for compensation of cognitive dysfunction may be less likely since estrogen deprivation is ongoing after completion of adjuvant treatment. Similarly, with regard to the effects of chemotherapy on cognitive function, a higher potential for compensation in younger women may be assumed. Furthermore, few studies have included data on the effects of treatment-induced menopause on cognitive function [21,52] and their results are inconsistent. In addition, evidence from noncancer samples is inconclusive regarding whether induced menopause has detrimental effects on cognition [69].
Limitations of prior research include several methodological issues. The majority of existing research is based on a comprehensive assessment of cognitive function with a wide set of neuropsychological tests. The use of less sophisticated assessments captures the risk of low sensitivity to detect cognitive decline. Therefore, the use of the high-sensitivity cognitive screen appears to be inappropriate to measure cognitive function in breast cancer patients, since the instrument may not capture subtle cognitive decline [3].
Furthermore, meaningful results can only be drawn from prospective studies that contain a baseline assessment of cognitive function and include a control group. The choice of a control group of breast cancer patients who only received local treatment has the advantage that women across groups share the experience of a cancer diagnosis with its emotional turmoil and potential effects on cognition (since some studies [11,25] reported cognitive dysfunction even before commencement of adjuvant therapy). However, this choice of a control group may also represent patients with a better prognosis and better physical health. In addition, treatment of breast cancer is becoming more and more individualized, which makes the control of a myriad of potential confounds a challenge. For example, the use of antibody therapy has not been studied so far in terms of its consequences on cognitive function.
Findings concerning the effects of endocrine treatment on cognitive function suggest that breast cancer patients undergoing endocrine therapy also do not appear to represent an appropriate control group to examine the effects of chemotherapy on cognitive function.
Several studies used a matched comparison group of healthy women as a control group. This choice of control group does not account for illness-related confounds and hence, preferably, should be included in addition to a disease control group. Nonetheless, the inclusion of a healthy control group holds the possibility to account for practice effects or normal aging (for longer time intervals) as well as the possibility of heightening the sensitivity of the measurement of change in cognitive function [70]. Ouimet and co-authors suggest a standardized regression-based approach for individual-based analyses [70]. This method uses change scores of a matched control group of healthy individuals to account for practice effects in order to more reliably measure cognitive change in patients with breast cancer. This method has been shown to be more sensitive compared with group analyses and the reliable change index method [70].
No studies to date have investigated whether newer molecular-targeted therapies of breast cancer (e.g., trastuzumab) impact cognitive function of breast cancer survivors in the long term. Future research may address their impact on cognitive function or at least control their effects as moderators of traditional cancer treatment on cognitive function.
Executive summary
Half of the published prospective trials support the assumption that subtle cognitive decline in multiple domains of cognitive function occurs in some breast cancer patients, whereas other prospective studies have not demonstrated cognitive impairment following chemotherapy.
A few studies suggest a dose–response relationship with patients treated with high-dose chemotherapy experiencing a more pronounced cognitive dysfunction compared with standard-dose chemotherapy.
Some patients showed cognitive decline before initiation of adjuvant treatment.
Results regarding whether cognitive alterations are transient or permanent are inconsistent.
Cognitive impairment is correlated with age, education and IQ. Subjective cognitive complaints are not associated with objective cognitive function.
Some evidence supports the notion that chemotherapy regimens pass the blood–brain barrier.
A direct neurotoxic effect may lead to atrophy of cerebral grey and white matter.
Long-term alterations in the activity of the frontal cortex, the cerebellum and basal ganglia have been shown in breast cancer patients after treatment with chemotherapy.
Breast cancer patients treated with chemotherapy demonstrated long-term lower resting brain metabolism compared with breast cancer patients who had never been treated with chemotherapy.
∊4 allele of APOE as a potential genetic risk marker for increased vulnerability to chemotherapy-induced cognitive decline.
Cross-sectional studies suggest cognitive impairment specifically in verbal memory and processing speed in women treated with tamoxifen or aromatase inhibitors.
The scarce prospective data are inconsistent. The International Breast Cancer Intervention Study (IBIS)-II, a double-blind, randomized, controlled trial in women at risk of breast cancer, did not show cognitive dysfunction in the aromatase inhibitor group compared with the control group.
Tamoxifen users have shown hypometabolism in the inferior and dorsal lateral frontal lobes and smaller hypocampal volumes. However, it remains to be clarified whether the effects are distinct for tamoxifen or are similar to the effects of estrogen on brain function.
The effects of aromatase inhibitors on brain function are unknown at present.
Neuropsychological training (using Memory and Attention Adaptation Training, a manual-based, structured intervention) demonstrated improvements in objective measures of cognitive function.
Cognitive–behavioral stress management demonstrated reductions in cortisol levels, a potential mediator of cognitive dysfunction.
No approved drugs are available at present.
Future research may examine the unique impact of endocrine therapy on cancer therapy-related cognitive change with prospective, controlled trials.
Identification and characterization of vulnerable subgroups for cancer therapy-related cognitive change, as well as which patients have the potential to compensate for cognitive impairment would be useful in the future.
Potential confounders that should be controlled in future studies are menopausal status/induced menopause, concurrent medication (affecting the CNS), distress, fatique, change in symptom severity over time, cytokine dysregulation and cortisol.
In terms of potential moderators of cognitive dysfunction, the examination of the role of stress and inflammation on cognitive outcomes in breast cancer survivors merits further empirical attention.
Testing of the efficacy of neuropsychological training and its potential for compensation should be conducted.
Exploration of the effects of analogs of gonadotropin-releasing hormone (e.g., goserelin) and molecular-targeted therapies for breast cancer (e.g., trastuzumab) on cognitive function should be carried out.
Alternative models of cancer therapy-related cognitive change (since some patients showed cognitive dysfunction before commencement of adjuvant therapy) should be studied.
In conclusion, owing to the mixed and conflicting findings, it is not yet clear whether adjuvant treatment of breast cancer is associated with cognitive impairment. Since some cancer patients show cognitive compromise even before initiation of adjuvant treatment, other unknown factors may contribute to the phenomenon of cognitive change associated with breast cancer therapy. For example, some vulnerable patients may experience prolonged or more pronounced effects of anesthesia following breast surgery. The stress response resulting from a recent cancer diagnosis may also prevent some new patients from fully engaging and performing well in cognitive assessments.
Breast cancer patients' quality of life is compromised for a significant time during and even sometimes after completion of chemotherapy. The reduced physical health as a consequence of chemotherapy may impact cognitive function, although fatigue has been repeatedly shown not to be associated with ‘chemo brain’.
Future research should further investigate the role of estrogenic therapy for the phenomenon of breast cancer therapy-associated cognitive change as well as the fact that some patients demonstrate transient cognitive impairment while others report long-term consequences.
Footnotes
Andrea Vodermaier was supported by an Emerging Team grant of the Canadian Institutes for Health Research (#AQC83559). There are no conflicts of interest or financial interests associated with this work. The author has no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
No writing assistance was utilized in the production of this manuscript.
