Abstract
The most common destination for breast cancer metastases is bone. If bone metastases are detected, treatment algorithms should include the continuation of cytoreductive therapy with the addition of treatment to counter skeletal-related events, including bone pain. The range of current treatment options includes bisphosphonates, surgical intervention to improve structural integrity and palliative focal radiotherapy. This article focuses on the role of bisphosphonate therapy in metastatic breast cancer. Bisphosphonate therapy significantly reduces the impact of skeletal-related events, reduces bone pain and increases patient quality of life. Our current understanding is that bisphosphonates may also disrupt the metastatic process and reduce the development of bone lesions. Ultimately, this may lead to further expansion of bisphosphonate-based therapy in the future.
Keywords
Over the last three decades, improved therapy options and an increase in screening and educational programs have contributed to a modest but steady decline in the breast cancer-related death rate [1,2]. Research shows that early diagnosis of breast cancer results in a good prognosis, with a current 5-year survival rate of approximately 98% for stages 0 or 1 [1,2]. However, although breast cancer is curable when identified early, approximately 50% of patients will develop metastatic disease, and 6–10% of patients with breast cancer have this at initial diagnosis [3]. In contrast to the good prognosis of patients with early-stage metastatic breast cancer (MBC) is incurable, with only 20% of patients surviving after 5 years (median survival range: 24–48 months) [3,4]. The most common site for metastasis of breast cancer is the skeleton, with 65–75% of MBC patients developing bone metastases [5]. Furthermore, following mastectomy, 12–47% of patients will have the first relapse of cancer in bone [3,6].
In patients with metastatic bone disease (MBD), the normal process of bone remodeling is disrupted, producing clinical complications such as metastatic bone pain (MBP), skeletal fractures and spinal cord compression. These symptoms greatly impact on patients' wellbeing and substantially erode quality of life (QoL). Bisphosphonates are the current standard of care for MBD and are usually combined with cytoreductive agents (i.e., cytostatic, immune, hormonal therapy and radiotherapy) targeted against the tumor cells. Bisphosphonates represent a broad group of compounds with a complex pharmacologic profile, varying in chemical structure and biological properties [4,7]. The main therapeutic effect of bisphosphonates is inhibition of osteoclast activity resulting in decreased bone resorption and a reduction of MBD-related complications. Nitrogen-containing bisphosphonates, such as pamidronate, zoledronic acid and ibandronate, are many times more potent (
The clinical course of MBC is diverse and reflects the heterogeneity of patients and of their tumors. Multiple factors such as age, comorbidities, menopausal status and tumor characteristics contribute to prognosis and influence therapeutic strategy [10,11]. This article evaluates treatment options for breast cancer and bone metastases, with a particular focus on the role of bisphosphonate therapy.
Diagnosing metastatic bone disease
It is well established that bone screening of asymptomatic breast cancer patients after surgical intervention does not prolong survival or improve QoL [12]. Thus, screening for bone metastases has no place in the regular follow-up of asymptomatic breast cancer patients who have received surgery. However, if a hitherto asymptomatic patient complains of bone pain, a resolute search for possible neoplastic bone involvement has to be performed. Imaging techniques that are available for the diagnosis of neoplastic bone involvement include radiopharmaceutical methods (i.e., skeletal scintigraphy [SS; a bone scan] and PET) and radiologic methods (i.e., plain radiography, computed tomography [CT] and MRI).
Bone scans involve conjugates to a bisphosphonate in order to enable the radionuclide (Tc99) to bind to sites of high bone turnover. Bone scans have a high sensitivity to increased bone remodeling due to MBD. Even following excessive pre-treatment with bisphosphonates, bone scans do not produce false-negative results [13]. However, increased bone remodeling can also be caused by other diseases affecting the bone (e.g., inflammation or fracture) [14]. These conditions decrease the specificity of the SS. Thus, diagnosis of MBD cannot rely on bone scintigraphy alone and may require further investigation using other techniques. The same is true for 8-fluorodeoxyglucose PET, which appears to be more sensitive than SS for osteolytic or mixed bone lesions but of lower sensitivity for osteoblastic bone lesions [15]. In [18F]-2-fluoro-2-deoxyglucose PET (18-FDG PET), a radio-labeled glucose analog is avidly taken up by tumor cells (e.g., breast cancer cells). Increased tumoral uptake of 18-FDG reflects elevated glucose consumption by tumor cells, as evidenced by the overexpression of glucose transporter proteins at the cells' surface and increased levels of active hexokinase demonstrated in many tumors. In comparison to normal cells, neoplastic cells take up higher amounts of glucose, since they rely to a higher degree on anaerobic glycolysis, which results in a poorer energy yield [16].
Suspected MBD, detected by radiopharmaceutical methods, can be confirmed by standard radiography, CT scans or MRI

Methods to detect and/or monitor bone metastases.
Although methods to detect MBD from breast cancer are clearly defined and have been evaluated in numerous studies, monitoring MBD remains problematic. The most frequently available methods (bone scan and plain radiography) have limited value in determining the course of MBD, which can be further complicated if lytic and blastic lesions coexist (mixed bone metastases). Thus, imaging techniques may only provide valuable supportive evidence. In clinical practice, the assessment of progression or regression of bone metastases is usually achieved by monitoring changes in the severity of MBD-related symptoms (e.g., pain or restriction of mobility). However, this situation might change in the near future. Preliminary results suggest that uptake of 18-FDG in PET reflects tumor activity and response to cytostatic treatment. Confirmation in further studies is required [18]. Furthermore, success of treatment can also be estimated using tumor markers and serum levels of markers of bone turnover (resorption and formation, discussed below).
Treating bone metastases in breast cancer patients
Surgery
Surgical intervention for MBD is most often carried out in patients who have an overt or impending long bone fracture. In these cases, surgery should precede radiotherapy in order to prevent or delay bone fracture [19]. Surgical interventions of the spine are usually only performed to stabilize the vertebral column or to alleviate symptoms of spinal cord compression.
Several kinds of surgical interventions for MBD can be performed. It is beyond the scope of this article to discuss them in detail. Reviews on this research area have been published elsewhere [20–22].
Kyphoplastia and vertebroplastia are relatively new interventions for the treatment of impending or overt vertebral fractures. In kyphoplastia, a collapsed vertebral body is initially raised up again by the use of a balloon catheter and then acrylic-based cement is injected locally to stabilize the previously broken bone. In vertebroplastia, cement is injected into the collapsed vertebral body without prior reconstitution of its previous height. Both methods lead to an almost immediate relief of pain in the treated region [23]. The clinical impact of these interventions in the treatment of MBD-related vertebral collapses cannot be fully assessed: there are no prospective and randomized trials available that evaluate the advantage of vertebroplasty or kyphoplasty in comparison with conservative treatment of vertebral compression fractures. With regard to compression fractures due to osteoporosis, some reports have demonstrated that an increased incidence of secondary vertebral fractures in the vicinity of the stabilized vertebral body [24]. Thus, although vertebroplasty and kyphoplasty are frequently performed, it is presently not clear whether patients really benefit from these methods.
Radiotherapy
Radiation therapy is still the mainstay for palliating pain due to bone metastases from breast cancer. The delivery of ionizing radiation to a bone containing metastatic tumor can be achieved using either radiation from external x-ray or γ-ray beam, or injected radioisotopes that localize to bone [25–27]. Both methods can be accompanied by bisphosphonate therapy [28]. In external-beam radiation, cost-sparing benefits promote single-fraction radiotherapy over multiple-fraction radiotherapy, since both regimens provide equal pain palliation and QoL benefits [29–31]. The onset of pain relief after radiation is not immediate, with only a few patients experiencing analgesia within the first 24–28 h, but with 50% of patients having some pain relief within 14 days [32].
In contrast with systemic radioisotope therapy with, for example, strontium-89, rhenium-186 or samarium-153, external-beam radiation is not only used for reducing pain, but is frequently also used for preventing pathological fractures in the weight-bearing bones, such as the spinal column and long bones. Moreover, bones that had been stabilized by osteosynthesis to prevent an impending fracture or to treat a manifest fracture due to neoplastic lesions are irradiated to inhibit further tumor growth in this area. An impending or overt spinal cord compression is defined as an oncologic emergency situation. Spinal canal compression arises most often from neoplastic involvement of the epidural space. However, in approximately 25% of cases this complication is due to metastases in the osseous parts of the vertebral column [25]. In breast cancer patients with this debilitating complication, effective treatment should follow the first appearance of neurologic deficits; otherwise the spinal cord compression will result in irreparable loss of function. An excellent review of the pathophysiology and treatment of spinal cord compression has been published by Byrne [33]. Surgery to the spine has an advantage over rapidly initiated radiotherapy in most cases of myelon compression [34].
Bisphosphonate therapy
Bisphosphonates are the standard of care for bone metastases from breast cancer. US treatment guidelines recommend that pamidronate or zoledronic acid (the approved options in the USA) should be initiated at first diagnosis of bone lesions and continued in patients with bone metastases [35,201]. In Europe and the rest of the world, excluding the USA, four bisphosphonates are approved for patients with breast cancer and skeletal lesions: clodronate, pamidronate, zoledronic acid and ibandronate
Bisphosphonate dosing regimens for bone metastases from breast cancer.

iv.: Intravenous.
Assessing the efficacy of bisphosphonate therapy in patients with bone metastases is not always straightforward. Owing to the inherent difficulties in visualizing bone metastases by radiography, it is not practicable to directly evaluate the effect of bisphosphonates on the progression or regression of bone lesions using radiographs. A further complication is that lesions do not always occur in isolation. Diffuse involvement of the skeleton by both lytic and blastic lesions is a more likely scenario. Therefore, to assess the efficacy of bisphosphonates in clinical studies, several sequelae to bone lesions are considered including the incidence of skeletal fractures, the need for radiotherapy or surgery to bone, serum levels of biochemical markers for bone turnover, bone pain and patient QoL. The clinical benefits of bisphosphonate-based therapy on these outcomes are well documented [5,7,36–42].
Reducing skeletal-related events
The number and rate of skeletal-related events (SREs) are frequently used as primary efficacy measures in bisphosphonate clinical trials. The SRE is typically a composite end point, and can include fractures, surgery and radiation to bone, spinal cord compression and/or hypercalcemia of malignancy, with the exact measure differing between studies. The time period for this efficacy end point also varies between trials, for example, the skeletal morbidity rate (as used in trials of zoledronic acid), examines the number of SREs per year, whereas the skeletal morbidity period rate (as used in trials of ibandronate) examines the number of 12-week periods in which a patient experiences a new bone event divided by the number of periods in the study. Multiple event analyses are also employed to consider the reductions in the number of complications or the delay in time to new bone events with bisphosphonates, as a measure of overall skeletal morbidity [43]. The differences in the assessments used in different bisphosphonate trials make it difficult to compare the available agents, and further comparative trials are needed.
Efficacy results from pivotal Phase III clinical trials of clodronate, pamidronate, zoledronic acid and ibandronate are shown in
Phase III skeletal-related events data for aminobisphosphonates in patients with bone metastases from breast cancer.

End point definitions for SREs differ between trials. In zoledronic acid trials, the 8 mg dose is due to renal toxicity.
MBC: Metastatic breast cancer; NS: Not significant; QoL: Quality of life; SMPR: Skeletal morbidity period rate (number of SREs occurring in a 12-week period, divided by time on trial); SMR: Skeletal morbidity rate (number of SRES divided by time on trial); SRE: Skeletal-related event.
A Phase III comparative trial of patients with MBC and bone metastases or multiple myeloma has been conducted for zoledronic acid 4 mg and pamidronate 90 mg, showing comparable reductions in the proportion of patients experiencing at least one skeletal event at 13 months [50]. At the 25-month follow-up, a multiple-events analysis showed that zoledronic acid reduced the risk of developing a SREs by 16% versus pamidronate in the overall patient population (p = 0.03) [51]. In a subset analysis of patients with hormone-sensitive MBC from the trial, the risk of developing an SRE (according to multiple-events analysis) was reduced by 30% versus pamidronate (p = 0.025) [52]. Moreover, time to the first skeletal event was also significantly prolonged in patients who were treated with hormonal therapy and zoledronic acid, when compared with those who received hormonal therapy and pamidronate (415 vs 370 days, respectively; p = 0.047). In 2005, Kohno
Phase III trials have demonstrated that in patients with MBC, intravenous ibandronate 6 mg significantly reduced mean skeletal morbidity period rate (SMPR; p = 0.004 vs placebo). An analysis of the different SREs that contributed to the composite end point SMPR, showed that the number of vertebral fractures (p = 0.023) and the need of radiotherapy were also significantly lower in the ibandronate group compared with the placebo group (p = 0.012) [37,54], although the study was not powered to show statistical significance on the individual SMPR components. Similarly, oral ibandronate 50 mg significantly reduced mean SMPR (p = 0.041 vs placebo) and the number of bone events requiring radiotherapy (p < 0.004 vs placebo) [55]. Oral ibandronate also reduced the mean number of SREs (1.15 vs 1.85 with placebo; p = 0.008) and the number of 12-week periods with events per patient (SMPR; 0.71 vs 0.99 with placebo; p = 0.015) [55].
Reducing bone marker turnover
Biochemical markers of bone turnover are released into the circulation following resorption or formation of bone matrix, and show promise as surrogates of disease progression, prognostic factors of survival and predictors of fractures in patients with MBD. Although screening for bone metabolism markers is not readily available in the clinical setting, the technique may allow patients at particular risk of bone metastases to be identified at an earlier stage, thereby improving prognosis [56]. Additional reasons to explain why monitoring response with bone markers may be of particular importance include individual patient resistance to bisphosphonates for unknown reasons and poor compliance with oral regimens because of the required dosing restrictions. Examples of bone resorption markers are covalent pyridinoline and deoxypyridinoline crosslinks, N-terminal crosslinked type I collagen telopeptide (NTX), C-terminal crosslinked type I collagen telopeptide (CTX) and carboxy-terminal type I collagen telopeptide [57,58]. Markers of bone formation include bone-specific alkaline phosphatase, serum total alkaline phosphatase, osteocalcin and procollagen type I C-terminal peptide and procollagen type I N-terminal peptide [57–59].
Biochemical markers of bone turnover have also been used to evaluate patient responses to bisphosphonate therapy. Particularly useful surrogates of the effect of bisphosphonates on SREs appear to be NTX and CTX [60–66]. Reductions in levels of urinary CTX and NTX have also been found to correlate with improvements in bone pain score following bisphosphonate therapy in patients with MBD [64,66]. A recent
In a recent Phase III trial, bone markers were used to compare the effects of oral ibandronate and intravenous zoledronic acid in suppressing tumor-induced bone resorption in women with breast cancer and bone metastases [68]. Their noninferiority on this measure was confirmed. At week 12, oral ibandronate 50 mg (daily) reduced the level of serum CTX, by 76%, and intravenous zoledronic acid (4 mg infusion every 4 weeks) led to a 73% decrease compared with baseline (p < 0.001 for both treatment groups); urinary NTX was reduced by 78 and 86% (p < 0.001), respectively. Oral ibandronate and intravenous zoledronic acid were also comparable in their effects on the markers of bone formation bone-specific alkaline phosphatase, procollagen type I N-terminal peptide and osteocalcin.
Results from a head-to-head comparative trial are required to confirm whether these bone marker effects translate into similar reductions in SREs with oral ibandronate and intravenous zoledronic acid in MBC patients with skeletal lesions. Two large Phase III trials are underway, recruiting patients in order to compare the effects of oral ibandronate and intravenous zoledronic acid on SREs in breast cancer patients with bone metastases. In the Zoledronic Acid Versus Ibandronate Comparative Evaluation (ZICE) trial, 1400 patients being randomized to oral ibandronate 50 mg or intravenous zoledronic acid 4 mg for 2 years. The primary end point is the frequency and timing of SREs during the 2-year treatment period. Secondary end points include the proportion of patients experiencing new SREs, time to first SRE, QoL, resource utilization and safety [69]. The South Western Oncology Group (SWOG) trial is randomizing 488 patients with breast cancer and bone metastases to oral ibandronate (50 mg/day) or zoledronic acid (4 mg) by infusion every 4 weeks, for 18 months. The primary end point is the proportion of patients with new SREs, with secondary end points including time to first SRE, QoL, overall survival and safety [70].
Palliation of bone pain
Bone pain is usually a secondary end point in clinical trials of bisphosphonates, but is often the first manifestation of metastatic disease and negatively impacts QoL. Approximately 70% of breast cancer patients diagnosed with bone metastases have MBP [71]. In clinical studies, a variety of instruments have been used to evaluate bone pain, including a visual analog scale (VAS), rating scales and the composite bone pain inventory (BPI). The efficacy of aminobisphosphonates such as pamidronate, zoledronic acid and ibandronate in reducing bone pain (measured by VAS) is superior to non-nitrogen-containing bisphosphonates such as clodronate [72], which suggests that these agents should be preferred in patients who report a high pain score.
In a Phase III study of intravenous pamidronate (90 mg) versus placebo, pain scores and analgesia use increased from baseline in both treatment arms, with a significantly greater increase in the placebo group (p ≤ 0.05) [47]. A smaller increase in mean pain score (0.5 vs 1.6 for pamidronate and placebo, respectively; p = 0.007) was observed in another Phase III trial, and the number of patients requiring radiation for bone pain relief was significantly reduced (25% pamidronate vs 34% placebo; p = 0.01) [48]. Although there is evidence for a reduction in bone pain with clodronate, some trials also report the absence of a palliative effect on the VAS [73,74]. In a comparative study, pain was assessed via a pain questionnaire in which pamidronate was more effective in reducing bone pain than clodronate [75].
In an open-label study of patients with breast cancer (n = 259) and other tumor types, zoledronic acid (4 mg) reduced bone pain in breast cancer patients with bone metastases over 6 months of treatment (mean change in pain score [via 100 mm VAS] was −7.1 at end point; p < 0.05) [76]. In a placebo-controlled registration trial in Japan, zoledronic acid reduced bone pain (BPI) by approximately −0.8 relative to baseline at 1 year [53]. Furthermore, in a Phase III comparative trial, both zoledronic acid and pamidronate reduced bone pain below baseline levels over 13 months (pain measured via BPI) [50]. A more recent report demonstrates that patients with breast cancer who switched to second-line zoledronic acid (from clodronate or pamidronate) had significantly improved pain control after 8 weeks (p < 0.001) [77].
In two Phase III trials (data pooling prespecified), oral ibandronate significantly reduced and maintained bone pain (5-point patient rating scale) below baseline for 2 years compared with placebo (the end point bone-pain score was −0.1 with ibandronate vs +0.2 for placebo; p = 0.001) [54]. Treatment with ibandronate resulted in significantly lower use of analgesia (p = 0.019) compared with placebo and had a positive effect on QoL (p ≤ 0.05 vs placebo). Intravenous ibandronate also significantly improved bone pain scores (patient-rated 4-point scale) and QoL in a Phase III study (p = 0.001 and p = 0.005, respectively, vs placebo) [78]. As seen previously for intravenous zoledronic acid [77], a 12-week Phase II study recently demonstrated that switching from oral clodronate or intravenous pamidronate to oral ibandronate significantly improved bone pain control (defined by ≥2-point reduction in worst pain score) in patients with breast cancer and bone metastases [79].
Intravenous infusion of the bisphosphonates clodronate, pamidronate or ibandronate using nonstandard, intensive regimens has been shown to relieve MBP in patients with various solid tumors, including breast cancer [72,80–82]. Patients received considerably higher doses than recommended for MBD, either as a single infusion or repeated infusions (or ‘loading doses’) each day for 3–4 days. Significant pain relief was typically observed within 3–7 days of the initial dose. This effect has even been demonstrated for patients with opioid-resistant bone pain at baseline [83]. Since there are no guideline recommendations for the use of high-dose bisphosphonates for metastatic bone pain, further research is required to elucidate their role for the palliation of this symptom in patients with MBC. However, the value of standard bisphosphonate doses to reduce bone pain is recognized by new treatment recommendations provided by an expert panel [43].
Bisphosphonates as adjuvant therapy
In addition to the well-established role of bisphosphonates in treating bone metastases, these agents have potential in preventing bone metastases. Bisphosphonates bind to bone mineral and rebalance bone remodeling through an inhibiting effect on osteoclasts, which subsequently lose their orientation and detach from the bone surface [84]. The decrease in osseous break down reduces the concentration of potential tumor growth factors (e.g., fibroblast growth factor or transforming growth factor β), which are released to the intercellular space during bone resorption. This effect and the high drug concentration of bisphosphonates in the osseous tissue makes it likely that the potential antitumor action of bisphosphonates is more concentrated within the bone and its close vicinity (bone marrow), than elsewhere. Preclinical data suggest that early intervention with bisphosphonates may reduce the risk of osteolytic lesions in patients with breast cancer by inducing apoptosis and inhibiti ng bone tumor progression
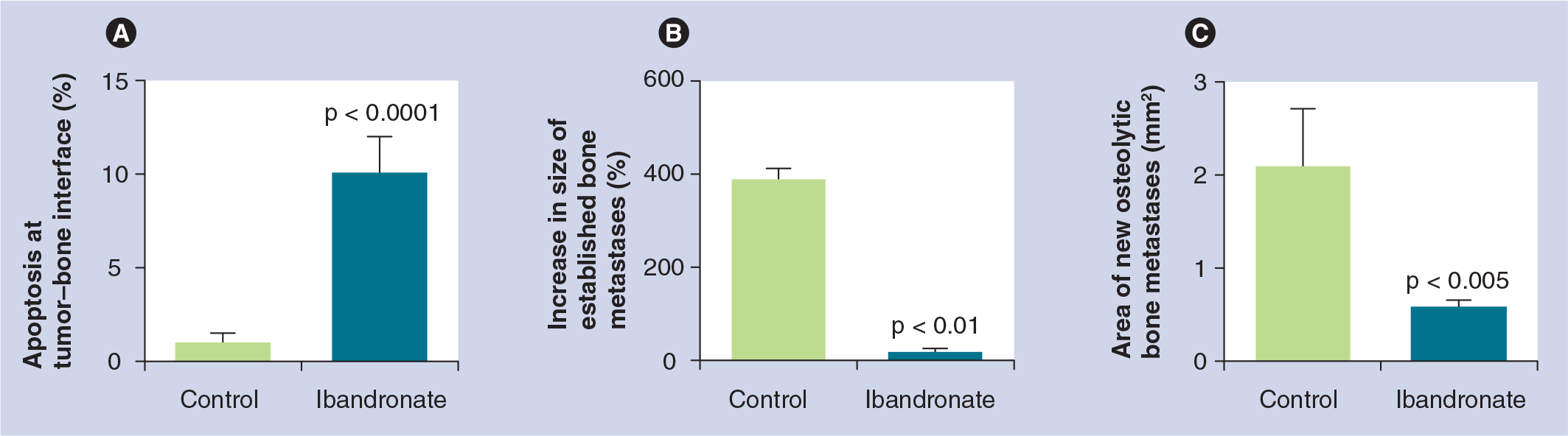
Preclinical breast cancer models.
Several trials of adjuvant bisphosphonate therapy have been conducted with oral clodronate [91–94], with positive results in two of three trials. In a 5-year follow-up of a 2-year study of 1069 patients with stage I–III breast cancer, who received oral clodronate 1600 mg/day in addition to surgery, radiotherapy, adjuvant chemotherapy and/or tamoxifen, treatment with this bisphosphonate significantly reduced the risk of bone metastases (p = 0.043) and improved overall survival (p = 0.048) [92]. In a German study of 302 patients with primary breast cancer, the risk of osseous and visceral metastases was significantly reduced (p = 0.003) after 2 years of treatment with oral clodronate. In this study, an improvement in overall survival was demonstrated for the clodronate group, even in the most recent follow-up with a median time since inclusion in the study of 8.5 years, although the initially significant reduction in the occurrence of bone and visceral metastases was no longer present [90,93]. In a Finnish trial of adjuvant oral clodronate treatment for early-stage breast cancer, 10-year follow-up demonstrated that clodronate-treated patients were significantly more likely to experience nonskeletal metastases compared with controls, although the incidence of bone metastases was similar between groups [94]. Multiple events analysis also failed to find an impact of clodronate on overall survival [90]. A meta-analysis of oral clodronate therapy found no evidence of any statistically significant difference in overall survival, bone metastasis-free survival or nonskeletal metastasis-free survival in advanced breast cancer patients receiving clodronate therapy or early breast cancer patients receiving adjuvant clodronate treatment compared with those who did not receive any osteoclast-inhibiting treatment [95]. The role of adjuvant clodronate therapy is currently being further assessed in the National Surgical Adjuvant Breast Project (NSABP, B-34), which has just completed the recruitment of 3400 patients.
Recently, the results for oral pamidronate 150 mg twice daily (n = 460) have been reported [96]. This trial did not support the use of adjuvant pamidronate to reduce the occurrence of bone metastases or fractures in patients with primary breast cancer. Intravenous pamidronate (45 mg every 2 weeks) was used in an adjuvant trial in breast cancer patients with four or more positive lymph nodes, who were also treated with standard antineoplastic treatment. Although the trial was not randomized (the patients decided whether they received pamidronate or not significantly fewer patients in the pamidronate group developed bone metastases than in the control group [97].
It is hoped that newer, more potent bisphosphonates than clodronate or pamidronate will not only be better tolerated than these drugs, but will also be associated with a clear improvement in clinical outcome. Several trials with zoledronic acid and ibandronate are currently underway or planned in the adjuvant setting [90,98]. For example, the Adjuvant Zoledronic Acid to Reduce Recurrance (AZURE) study has just completed the recruitment of 3360 patients with stage II/III disease, and will assess disease-free survival as the primary end point, in addition to time to bone and nonskeletal metastases, overall survival and skeletal morbidity. The SWOG/Intergroup/National Surgical Adjuvant Breast and Bowel Project (SO307) trial is comparing the efficacy and safety of oral ibandronate with oral clodronate and intravenous zoledronic acid. It contains a 3-year treatment phase and a 3-year follow-up phase, and is currently enrolling 4500 women with histologically confirmed stage I, II or III non-MBC. The primary efficacy end point is disease-free survival. Secondary end points include overall survival, distribution of sites of first recurrence and adverse events, such as renal toxicity and osteonecrosis of the jaw and maxilla [99]. Other adjuvant studies include the of oral/intravenous Ibandronate with or without Capecitabine (six cycles) for 2 years in 1400 Elderly Patients with Early Breast Cancer trial (ICE trial, expected completion date: 2010) and the German Adjuvant Intergroup Node-Positive (GAIN) study, in which 3000 patients with node-positive breast cancer are receiving epirubicin, paclitaxel and cyclophosphamide with or without ibandronate or epirubicin, paclitaxel, cyclophosphamide and capecitabine with or without ibandronate (final analysis due in 2012). A German study known as SUCCESS is investigating disease-free survival in early breast cancer patients receiving zoledronic acid for 2 or 5 years following adjuvant chemotherapy [43]. If these trial results are positive, then targeting women who are at high risk of metastases (e.g., those that are node positive) sooner with bisphosphonate therapy may prevent or delay bone metastases.
Bisphosphonates for cancer treatment-induced bone loss
For normal bone development and for the preservation of bone strength and quality, the presence of sexual hormones is an essential prerequisite [100]. Thus, both the late development of a normal menstrual cycle and also the early menopause predispose a person to an increased risk of fracture [101]. However, several treatments of breast cancer are associated with intended or nonintended estrogen deprivation.
The positive results with adjuvant chemotherapy in breast cancer were always believed to be only related to its direct cytoreductive activity. However, it has become clear that the majority of premenopausal women (63–96%) with breast cancer who are receiving postoperative adjuvant chemotherapy regimens develop ovarian insufficiency [102]. Chemotherapy in these patients may lead to ovarian injury and premature menopause with low levels of circulating estrogen. This chemical castration has an additional growth inhibiting effect on breast cancer cells, but also puts the patients at increased risk of osteoporosis. In the recent years, adjuvant therapy with luteinizing hormone-releasing hormone agonists or surgical ovarian ablation (both interventions reduce concentrations of circulating estrogens to a postmenopausal level) has also been demonstrated to be comparably effective to standard chemotherapy schedules in premenopausal patients with hormone receptor-positive breast cancers [103]. Consequently, a major detrimental effect of long-term endocrine-and chemotherapy-based adjuvant treatment in MBC is accelerated bone loss that leads to osteopenia or osteoporosis [104]. Interestingly, treatment with tamoxifen, the main representative of selective estrogen receptor-modifying drugs (SERMs), which are also widely used in the treatment of postmenopausal osteoporosis [105], can prevent bone loss in postmenopausal but not premenopausal patients [106]. In contrast to SERMs, treatment with aromatase inhibitors leads to bone loss and fractures, irrespective of the menopausal status.
Guidelines for bone health in breast cancer recommend the use of bisphosphonates to preserve bone density in all patients regardless of the stage of their disease [36]. Several studies have demonstrated that bisphosphonate therapy was successful in combating loss of bone mineral density in patients receiving adjuvant hormone-chemotherapy [46,107–111], and that early intervention is particularly beneficial [112]. Ongoing trials are investigating the efficacy of the aminobisphosphonates in this setting, including the Z-FAST, ZO-FAST, and Austrian Breast and Colon Cancer Study Group (ABCSG-012) studies of zoledronic acid plus letrozole in postmenopausal women with early-stage breast cancer [98,113,114]. Preliminary results demonstrate that zoledronic acid is able to increase bone mineral density in these patients with first-line administration (compared with delayed treatment) [56,98,99]. An updated analysis of the ABCSG-012 study was presented at the American Society of Clinical Oncology Meeting in 2008. Patients receiving both adjuvant endocrine and zoledronate treatment did not have only an improved bone mineral density compared with the patients receiving endrocrine therapy alone, but they also had improved disease-free survival and relapse-free survival [115].
Safety considerations in bisphosphonate therapy
Patients with MBC may already be experiencing adverse events caused by anticancer therapy. Since patients with bone metastases from breast cancer are likely to survive for several years, the impact of adverse events on QoL should be minimized. Three adverse events associated with bisphosphonates are osteonecrosis of the jaw (ONJ), hypocalcemia and renal toxicity. Over 200 cases of ONJ associated with bisphosphonate therapy have been described since 2003 [116], with an incidence of up to 2.5% estimated for breast cancer patients [117]. Risk factors include the duration of therapy and previous dental surgery [118] and, to date, most reports have been linked to zoledronic acid and pamidronate therapy, most likely reflecting long-term experience with these agents [119,120]. ONJ is an area that has received much interest of late, and, as such, has become a key topic. However, it is likely that good dental prophylaxis would preclude the occurrence of such events. The debate continues regarding the necessity to cease bisphosphonate therapy prior to dental treatment and no definitive answer has yet been reached. Hypocalcemia is rarely a clinically relevant problem. However, it is important to provide calcium and vitamin D supplements in order to reduce the risk of secondary hyperparathyroidism with the nitrogen-containing bisphosphonates now in common usage. Most patients with bone metastases are profoundly deficient in total body calcium and may also be low on vitamin D. Renal toxicity was first reported following high doses and rapidly administered intravenous bisphosphonates [121,122], but has also occurred with standard doses of bisphosphonates infused every 3–4 weeks [50,51,123,124]. In rare cases, patients in clinical practice have progressed to renal failure and required dialysis [125]. In order to reduce the impact on renal function, it is recommended that serum creatinine is monitored prior to each dose of zoledronic acid, with stepwise dose reductions for patients with impaired renal function (based on creatinine clearance). Using these recommendations, renal tolerability is improved [43,126]. Intravenous ibandronate has been shown to have an incidence of renal adverse events comparable to placebo in a Phase III trial of patients with bone metastases from breast cancer [37,43], with no adverse effects observed with treatment for up to 4 years [127]. A recent trial assessing a 15-min infusion time for ibandronate 6 mg demonstrated no adverse renal events, leading to an updated label recommendation for the infusion time of this bisphosphonate (previously ≥1 h) [55].
Other adverse events associated with intravenous bisphosphonates are an acute flu-like syndrome (acute-phase response; associated with aminobisphosphonates only), with symptoms of joint pain, pyrexia, fever and nausea [116], and local injection-site reactions (e.g., phlebitis, pain, local swelling and ulceration). Oral bisphosphonates are associated with adverse gastrointestinal (GI) events. Bisphosphonates are generally considered to be poorly absorbed in the GI tract and high doses of these drugs cause GI problems. For example, oral clodronate is associated with GI disturbances such as diarrhea [128]. The incidence of adverse GI tract events with long-term oral ibandronate was similar to placebo in Phase III trials [54,129]. It is still important to follow recommendations for administration of oral bisphosphonates (e.g., fasting prior to dosing, taken upright with a glass of water) in order to increase bioavailability and reduce the risk of esophagitis and other GI adverse events.
Conclusion & future perspective
Once bone metastases from breast cancer are detected, bisphosphonate therapy is essential to reduce skeletal complications. Several options are available to the clinician and there is extensive clinical evidence to support reductions in SREs and bone pain, both important measures of efficacy that lead to improved patient QoL and long-term clinical benefit. Current guidelines recommend initiating bisphosphonate therapy once bone lesions are detected and continuing treatment until a patient's performance status has substantially declined (i.e., for at least 2 years, and longer if possible) [43]. It is generally agreed that oral formulations of older bisphosphonates, such as clodronate, are less effective than intravenous administration of newer aminobisphosphonates [43,130]; however, the oral formulation of the aminobisphosphonate, ibandronate has been shown to have similar effects on markers of bone resorption to intravenous zoledronic acid. SRE data from ongoing comparative head-to-head trials are needed before conclusions can be made on whether these two bisphosphonates have similar effects on bone events in patients with established skeletal metastases. Having this information will help clinicians to make a better informed choice regarding which bisphosphonate to prescribe, although the choice for an oral or intravenous agent will also depend on patient preference and individual circumstances (e.g., hospital-based or outpatient home care).
Since treatment for bone metastases is likely to be required for several years, long-term safety data must be considered when selecting drugs. Although generally well tolerated, the renal and GI safety profiles appear different between bisphosphonates, and the possibility of ONJ must also be taken into account when initiating and continuing therapy.
For patients with early breast cancer, evidence suggests that adjuvant bisphosphonates can reduce the risk of skeletal and nonskeletal metastases, although this requires further investigation. Bisphosphonates have also demonstrated a protective effect against cancer treatment-induced bone loss. Further research is ongoing and the final results of large randomized trials are eagerly awaited.
Executive summary
Although survival rate is improved for patients diagnosed with early-stage breast cancer, 50% of patients develop incurable metastatic disease, with bone the most common site of metastasis. Advanced metastases to bone leads to increased osteoclast activity that reduces skeletal integrity and results in hypercalcemia. The current standard of treatment for bone metastases is bisphosphonate therapy.
Therapies targeted against primary breast tumors are insufficient to control metastases to bone.
Positive diagnosis of bone metastases and subsequent monitoring of progression can be made using several imaging techniques, including radiography, computed tomography and MRI.
Surgical intervention may be required to stabilize advanced bone lesions and can include prosthesis implants or inserted rods or plates. Systemic and focal radiotherapy is used to palliate bone pain.
Clodronate, pamidronate, zoledronic acid and ibandronate effectively reduce the risk of skeletal-related events in bone metastases patients with breast cancer. Zoledronic acid was more effective than pamidronate according to multiple-events analysis of the breast cancer subgroup from a Phase III trial. Ibandronate has apparently similar oral and intravenous efficacy (cross-trial comparison).
Bisphosphonate therapy is recommended for bone metastases in patients with breast cancer. Pamidronate appears to be more effective than clodronate in reducing bone pain; zoledronic acid and pamidronate had comparable (nonsignificant) effects over 1 year of treatment in a Phase III trial. Long-term (2-year) statistically significant palliation of bone pain (vs placebo) has been demonstrated with oral and intravenous ibandronate. Comparisons of ibandronate and zoledronic acid are in progress. One such example is the Zoledronic Acid Versus Ibandronate Comparative Evaluation trial, which aims to demonstrate noninferiority of oral ibandronate in comparison with intravenous zoledronic acid, with respect to tolerability and adverse events including pain/analgesic scores.
Serum levels of biochemical markers in bone metabolism are surrogate markers of efficacy. This emerging technology may help to identify high-risk patients at an earlier stage of disease. A preliminary study demonstrateed that ibandronate and zoledronic acid are equivalent in reducing bone marker turnover.
Bisphosphonate therapy is generally well tolerated. Hypocalcemia, gastrointestinal intolerance (oral agents) and renal toxicity are the main adverse events. There appear to be differences in the renal safety profile of different bisphosphonates, suggesting that nephrotoxicity is not a class effect.
Although established as treatment for metastases to bone, bisphosphonate therapy may play an additional role in preventing bone lesions. In preclinical studies and clinical trials in patients who are either postmenopausal or have osteoporosis, bisphosphonate therapy has been shown to reduce angiogenesis and breast cancer cell proliferation.
The current role of bisphosphonate therapy in metastases to bone from breast cancer is to reduce skeletal complications. Future treatment algorithms for high-risk patients may recommend introducing bisphosphonates earlier, with the aim of prevention rather than palliation.
Footnotes
