Abstract
Breast cancer is the most common cancer in women and is a major killer. Progress in treating the disease has been spectacular in recent decades and mortality is now declining in Western countries. This article describes the development of conservative surgical treatments for breast cancer and indicates future directions. Since they respect the integrity of the woman's body, conservative treatments have encouraged women to participate en masse in screening programs, plausibly resulting in earlier disease detection and greater curability.
Keywords
In no area of oncology has our understanding of prevention, diagnosis and treatment expanded so quickly in recent years as in breast cancer. A major aspect of this trend is the recognition that quality of life is important for the breast cancer patient, and this has driven a series of paradigm changes:
From ‘maximum tolerated treatment’ to ‘minimum effective treatment’
From an anatomical concept of cancer spread towards a biological concept of spread
From aggressive surgery and radiotherapy to targeted conservative treatments
Modern breast cancer treatment, therefore, requires limited surgery instead of mutilating operations, and more targeted radiotherapy instead of wide-field irradiation that includes the regional lymph nodes and less aggressive chemotherapy. These changes may in turn have motivated women to participate in early detection programs, plausibly explaining the recent and encouraging reduction in breast cancer mortality in Western countries.
Furthermore, new developments in imaging technologies have made it possible to detect breast cancers at increasingly early stages, probably further contributing to improvements in curability; at least this is the situation in Western countries. The situation regarding breast cancer is much more worrying in the developing countries, where incidence is increasing but the resources are not available to plan prevention programs or introduce new technologies to improve the treatment of the disease [1].
Surgery
The revolution in breast cancer treatment began in the 1970s with Umberto Veronesi's Milan I trial. This trial randomized 701 patients to either breast-conserving surgery (quadrantectomy, complete axillary dissection (AD) and breast radiotherapy, QUART) or the traditional Halsted mastectomy [2]. Eligible patients had infiltrating breast cancer up to 2 cm in diameter, but no clinically evident involvement of the axilla. After sufficient follow-up, no difference was found in survival between the two study arms and it was this result that ushered in the era of conservative surgery for breast cancer and, eventually, for all other types of cancer.
The most recent analysis of the Milan I results, after more than 20 years of follow-up [3], confirmed that survival curves for the two arms were indistinguishable

Overall survival after 20 years of follow-up in the Milan I trial.
A second major advance in the conservative treatment of breast cancer, also developed by Veronesi and his group in Milan, was the sentinel node (SN) biopsy (SNB)

Anatomy of lymph nodes and localization of sentinel lymph node.
Sentinel node biopsy was developed as a minimally invasive method of determining the presence of axillary lymph-node involvement in women with early breast cancer. The concept is that the SN is the first lymph node to pick up malignant cells that detach from the breast lesion and travel in the lymph ducts. If the SN is free of disease, then the whole of the axilla is also free of the disease, so complete AD can be avoided. A single-center randomized trial was performed at the European Institute of Oncology from March 1998 to December 1999 to verify this principle [7]. Patients with primary breast cancer consisting of a diameter up to 2 cm were randomized to undergo either SNB plus total AD (AD group) or SNB but followed by AD only if the SN contained metastatic cells (SN group), with no further treatment to the axilla if the SN was negative. The aim was to determine how many patients developed axillary metastases in the group with a negative SN and, hence, the power of SNB to correctly predict the disease status of the whole axilla. A new method for the intraoperative histological examination of the SN was developed for the study. After removal, the entire sentinel lymph node was frozen and a large number of sections (between 30 and 60) were cut and examined under the microscope. The surgeons waited for the result and proceeded with AD if the SN was reported to be metastatic.
Of the 257 patients in the AD group, 83 had a positive SN (32.3%) and 174 had a negative SN (67.7%). Of the 259 patients in the SN group, 92 had a positive SN (35.5%) and 167 had a negative SN (64.5%). No cases of overt clinical axillary metastases were observed among the 167 patients of the SN group not subjected to AD, although eight cases were expected based on past experience. After a median follow-up of 60 months, the overall survival of patients was 97.6% in the AD group and 98.6% in the SN group [7].
These results were confirmed by studies conducted elsewhere and, as a result, SNB is now a routine procedure for early breast cancer [5,6]. At the European Institute of Oncology, more than 14,903 patients with breast cancer have undergone SNB, and only if the SN is positive do they receive AD.
New technologies are being developed and perfected to determine the status of the axilla and make possible more targeted approaches to diagnosing and treating breast cancer. PET is one such technology. In PET, a harmless quantity of radioactive (positron-emitting) substance is injected into the body. The substance is metabolized by the body, and tissue areas at higher metabolic rate take up more of the substance than tissue areas at low metabolic rate. In breast cancer, PET is carried out with 18-fluorodeoxyglucose (FDG) as the radioactive material. FDG-PET can detect multifocal breast disease not detected by conventional imaging. In a pilot study at the European Institute of Oncology, it was found that when a SN was ‘positive’ by PET it was always metastatic on examination, suggesting that, in these cases, SNB is not necessary and one should proceed directly to AD. However, at the moment the technique can not replace AD or SNB because it is not sensitive enough for small metastases (micrometastases) [8].
Radioguided occult lesion localization
As breast cancers are diagnosed at an earlier stage, more and more cancers are occult or nonpalpable and can only be detected using instruments, such as ultrasound or mammography. Once an occult lesion is detected it must be ‘marked’ so that it can be found by the surgeon during the operation.
Radioguided occult lesion localization (ROLL) is a sophisticated surgical technique developed at the European Institute of Oncology for the location of occult breast lesions [9]. It involves the injection of an immobile radioactive tracer (as opposed to the mobile radiotracer used to identify SNs) directly into the center of the lesion under mammographic or ultrasonographic control. During the operation, a handheld γ-ray detector is used to locate the lesion and guide its removal. ROLL was first introduced at the European Institute of Oncology in 1996 and, by 2007, a total of 5169 ROLL procedures had been performed. It is a simple and accurate technique [9], and has proven to be an excellent solution to the problem of locating occult breast for excision (Figure 3).

Results of study on efficacy of radioguided occult lesion localization.
New radiotherapy techniques: TARGIT & ELIOT
As radiotherapy technologies advance, it has become possible to progressively reduce the area of tissue being irradiated so as to better concentrate the radiation on the area of the excized tumor (tumor bed) and a circumscribed area around it. For women with breast cancer, partial breast irradiation is becoming a realistic prospect for the future. Targeted intraoperative radiotherapy (TARGIT) is being investigated in international trials comparing the efficacy of a lowenergy radiation source used intraoperatively with the conventional postoperative approach as the only radiotherapy treatment in selected patients, and as a tumor bed boost in patients with higher risk of recurrence [10,11].
The European Institute of Oncology has pioneered a technique known as electron intraoperative therapy (ELIOT). This is a method of delivering a high dose of electron radiation to the breast during surgery (after removal of the primary tumor). Data demonstrate that tumors recur mainly at the resection site, and that relapses in other quadrants of the same breast are much rarer. ELIOT delivers high-dose radiotherapy to a very limited area – the part of the breast involved by the tumor (Figure 4) – while sparing adjacent and underlying tissues. Since 1999, the European Institute of Oncology has used ELIOT on over 3000 breast cancer patients either as a boost to traditional radiotherapy or as the only radiation treatment. Implementing ELIOT as the only radiation treatment is being investigated at the European Institute of Oncology in a prospective, randomized trial in comparison with conventional radiotherapy in women with early-stage breast cancer [12]. ELIOT/TARGIT have several advantages over conventional radiotherapy to the residual breast: they cost less, solve the problem of difficult access to radiotherapy centers and have a beneficial effect on the quality of life of the patient. In addition, ELIOT/TARGIT does not irradiate the skin and contralateral breast, while irradiation to the lung and the heart is greatly reduced because of the surgical insertion of radiation shields under the breast.

Electron intraoperative therapy apparatus for intraoperative delivery of radiotherapy.
The ELIOT technique is also being used in nipple-sparing mastectomy followed by immediate breast reconstruction. In women with multicentric cancer or large intraductal cancer, breast conservation is not possible and mastectomy has to be performed. Nipple-sparing mastectomy is a painstaking surgical technique, which, as the name suggests, preserves the skin of the nipple-areola complex that is incorporated in the reconstructed breast. If this area is treated intraoperatively by ELIOT, it kills any occult cancer cells, thus, increasing the chances of a successful outcome.
A particular advantage of ELIOT/TARGIT is that they do not interfere with systemic therapy (chemotherapy) when this is indicated.
Toward a new TNM classification
The TNM classification of breast cancer specifies cancer size (T), axillary lymph-node involvement (N) and presence of distant metastases (M) (Table 1). It was devised some 60 years ago to classify breast cancers according to their prognoses. At that time, tumor size (T) was of major prognostic importance; however, this is less of the case today. Furthermore, the entities lobular carcinoma in situ (LCIS) and ductal carcinoma in situ (DCIS) have always been problematic within the TNM classification because, by definition, malignant tumors (specifically including carcinomas) are those that invade adjacent tissue and metastasize. In situ lesions do not show these behaviors so ‘carcinoma’ is inappropriate. A better classification is that proposed by Tavassoli [13], which replaces the terms LCIS and DCIS by lobular intraepithelial neoplasia and ductal intraepithelial neoplasia, respectively. As these neoplasias do not metastasize, the categories N and M of the TNM classification do not apply. Similarly, there is no reason to add the adjectives ‘invasive’ or ‘infiltrating’ to carcinomas that are not intraductal.
The European Institute of Oncology TNM classification.
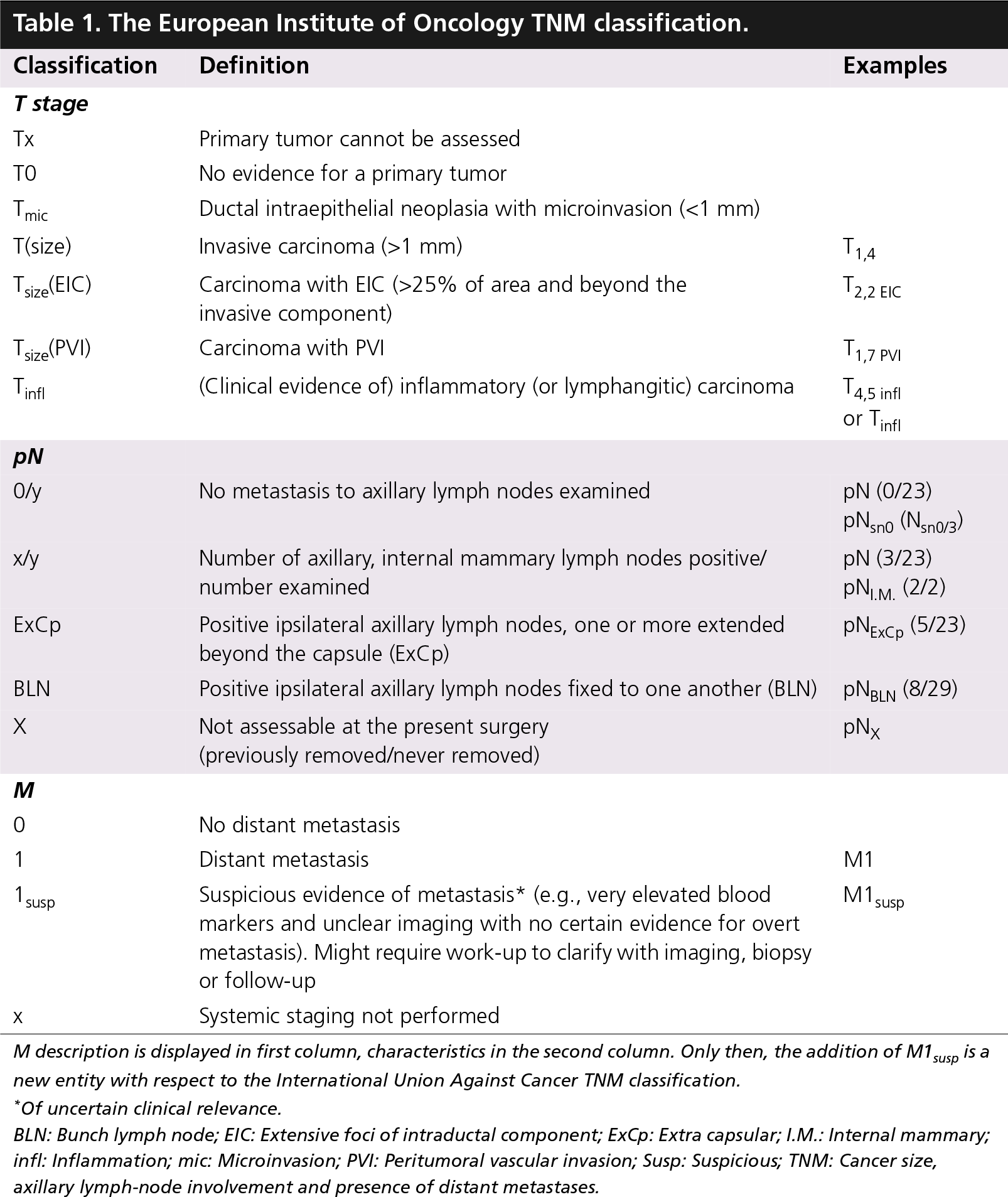
M description is displayed in first column, characteristics in the second column. Only then, the addition of M1susp is a new entity with respect to the International Union Against Cancer TNM classification.
Of uncertain clinical relevance.
BLN: Bunch lymph node; EIC: Extensive foci of intraductal component; ExCp: Extra capsular; I.M.: Internal mammary; infl: Inflammation; mic: Microinvasion; PVI: Peritumoral vascular invasion; Susp: Suspicious; TNM: Cancer size, axillary lymph-node involvement and presence of distant metastases.
These concepts have been incorporated into a new ‘dynamic’ TNM classification proposed by the European Institute of Oncology (TNMEIO). This new classification also proposes to change how size (T) is specified. Instead of the present four broad T categories, each representing a range of sizes and range of prognoses, it is proposed to precisely specify the maximum diameter of each cancer. Thus, if a cancer has a maximum diameter of 1.3 cm, it would be classified as T1.3; if it has a diameter of 4.5 cm it would be T4.5.
A similar classification criterion could also apply to lymph-node involvement (N) by specifying both the number of nodes examined and the number found to be involved. Thus, N0/21 would indicate no involved nodes out of 21 examined, and N3/19 would indicate that three of the 19 examined nodes were metastatic. Similarly, the code Ns0/2 would indicate that two SNs were examined and both were negative for metastasis.
It is also important to specify the biological characteristics of the breast cancer because these too have a major influence on prognosis. For example, endocrine receptor expression, HER2 expression and peritumoral vascular invasion would all be specified within the TNMEIO.
The TNMEIO classification indicates the presence of distant metastases with M, as in previous classifications, but adds an acronym specifying the organ or organs involved, together with an indication of whether the involvement is macroscopic or microscopic.
The new classification is specific for breast cancer and aims to both summarize the features of the cancer that are of therapeutic relevance and to categorize patients for clinical trials of specific treatment modalities. However, the main novelty of TNMEIO is that it is dynamic and flexible in that it allows for the inclusion of new prognostic and predictive information as treatments improve, and new knowledge of the molecular bases for response and resistance is acquired [14].
Adjuvant chemotherapies
Systemic cytotoxic chemotherapy is given as an adjunct to primary treatment (surgery plus radiotherapy to the residual breast) to eradicate micrometastatic disease, which appears to be present in most patients with invasive breast cancer. However, there is concern that these drugs are given to women who are either cured by local treatment or in whom the small risk of developing overt disease is adequately controlled by hormone treatment. Patients selected for adjuvant cytotoxic chemotherapy are generally those whose cancers do not express hormone receptors and, hence, do not respond to hormone treatment and are at an increased risk of relapse [15].
In the past, premenopausal women whose cancers express hormone receptors were offered tamoxifen with or without suppression of ovarian function. Nonsteroidal (anastrozole and letrozole) and steroidal (exemestane) aromatase inhibitors are new and effective alternatives to tamoxifen for postmenopausal women (whose ovaries no longer produce estrogen). These drugs are usually given after 2–3 years of tamoxifen, cutting short the standard 5-year tamoxifen treatment. There are indications, however, that aromatase inhibitors alone are superior than tamoxifen [16].
Overexpression of the epithelial growth factor receptor ErbB2 on cancer cell membranes is a strong predictor of response to trastuzumab, a monoclonal antibody that binds to the ErbB2 receptor and deactivates it. Trastuzumab is particularly effective when given to women with ErbB2-positive cancers in combination with or after a cytotoxic drug [16].
Cancer prevention & genetics
The sequencing of the human genome and development of techniques for monitoring the expression of thousands of genes by a tissue fragment has made a new approach to the prevention of cancer possible. Individuals susceptible to developing cancer can be identified by their gene-expression profile. Gene-expression profiling will open the way to significant improvements in the efficacy of cancer-screening programs, and will stimulate the development of molecular interventions in those found to be at risk in order to prevent them developing cancer.
Cancer prevention may be primary, secondary or tertiary. In primary prevention, the aim is to prevent disease onset by interventions to healthy persons whose environmental, lifestyle (e.g., lack of exercise and inappropriate diet) or familial/ genetic characteristics indicate they are at increased risk of cancer. Secondary prevention treats groups with premalignant conditions, such as in situ neoplasia, in order to block evolution to invasive disease. Tertiary interventions aim to protect persons against second primary cancers after they have been cured of an initial cancer. To increase the cost–benefit ratio of prevention interventions, it is essential to identify target populations at increased risk of cancer.
Consultation is part of the wider process of counseling, the first step of which is investigation of the family or genetic history of cancer to assess the risk of genetic predisposition. If the risk is high then genetic testing may be the next step to confirm or refute a genetic predisposition. If the result of the genetic test is positive, personalized followup programs are agreed with family members at risk. There are three main approaches to women and their families genetically predisposed to breast cancer: intensive surveillance, pharmacological prevention and surgical prophylaxis. Confirmed carriers of BRCA1 or BRCA2 mutations may well be counselled about surgical prophylaxis involving bilateral mastectomy and ovariectomy [17]. MRI is being investigated as an examination for young women with suspected or confirmed mutations predisposing to breast cancer. High-risk persons are also offered participation in clinical trials for the early detection of cancer (e.g., ductal lavage) or the reduction of cancer risk (including chemoprevention, diet and lifestyle interventions). To identify effective chemoprevention agents, it is – in theory – necessary to perform long and expensive studies on very large groups of patients in order to determine whether the risk of cancer is indeed reduced in the group taking the chemopreventive agent. One way around this – reducing study time, trial sample size and costs, and permitting the screening of many promising agents – is to study markers of cancer risk rather the development of cancer itself. Examples of such markers include levels of circulating proteins or their expression in tissue samples. Such markers can be used as intermediate end points (surrogate end point biomarkers [SEBs]) to test investigational agents.
Several SEBs appear promising, including growth factors (e.g., the IGF family), hormones (estradiol, estrone, estrone sulfate, dehydroepiandrosterone and prolactin), atypical breast cells in cytological samples and mammographic density investigated using computerized techniques.
The synthetic retinoid fenretinide has shown promise as a cancer chemopreventative agent. In a randomized trial conducted by the National Cancer Institute in Milan, Italy, to test its efficacy in preventing secondary breast cancer in patients treated for primary breast cancer, no difference in the frequency of secondary breast cancer in the treatment and placebo arms was found [18]. However, a significantly reduced risk of secondary breast cancer was found in premenopausal women taking fenretinide, particularly in the youngest women. The reduction in risk persisted for at least 5 years after cessation of treatment.
In recent years, the Italian Tamoxifen Trial and four other chemoprevention studies have confirmed that tamoxifen is the most effective secondary chemopreventive currently available for women treated for breast cancer that is found to express estrogen receptors (ER+) – the most common form of breast cancer [18]. The subsequent Tamoxifen Prevention Trial [19] conducted at the European Institute of Oncology showed that low-dose tamoxifen combined with anastrozole (aromatase inhibitor) does not reduce anastrozole bioavailability (unlike in other studies in which tamoxifen was used at standard dose), and has beneficial effects on markers of bone turn-over like osteocalcin and C-telopeptide (particularly when given transdermally rather than orally), suggesting an overall beneficial effect in postmenopausal patients. Another important observation of the trial was that the protective effect of tamoxifen against breast cancer was more pronounced when combined with HRT, suggesting that the risk–benefit ratio of tamoxifen is particularly favorable in premenopausal women and HRT users, providing further justification for its wider use in breast cancer prevention. To confirm the benefits of the HRT/tamoxifen combination, the European Institute of Oncology is currently conducting the large national placebo-controlled HRT Opposed by Low Dose Tamoxifen (HOT) trial in healthy postmenopausal HRT users who had low-dose tamoxifen (5 mg/day) for 5 years.
The European Institute of Oncology is also investigating other chemopreventive agents. In women with ER+ intraepithelial neoplasia of the breast (a precancerous condition), the drug raloxifene (an ER inhibitor similar to tamoxifen) together with low-dose tamoxifen is being tested in premenopausal women, and exemestane plus celecoxib is being tested in postmenopausal women.
Conclusion
In Western countries, many of procedures and techniques described above for the treatment of breast cancer are already available; newer or experimental treatments can be expected to become available within a few years after trials have demonstrated them to be effective. Although breast cancer remains the most common cancer in women, the battle against the disease is being won by the early detection and conservative surgical approaches aided by science and technology. The problem lies in Eastern Europe and developing countries where, for economic reasons, the latest technologies cannot be applied but, more importantly, the attitudes of women and of physicians towards breast cancer remain what they were 30 years ago in Western Europe. Until attitudes change and mass screening is implemented, women will continue to die unnecessarily of breast cancer.
Breast cancer is the most common malignancy in women and is a major killer.
Over the last 40 years, increases in knowledge have driven a paradigm shift in breast cancer treatment from maximum tolerated treatment to minimum effective treatment.
Breast-conserving surgery has become established as an alternative to mutilating mastectomy.
Sentinel node biopsy makes it possible to avoid axillary dissection in a large proportion of breast cancer patients.
New imaging technologies make it possible to diagnose at an increasingly earlier stage.
Mass participation in screening also means that the disease can be diagnosed earlier.
New intraoperative radiotherapy techniques (electron intraoperative therapy and targeted intraoperative radiotherapy) may reduce or eliminate the need for long courses of postoperative radiotherapy.
A new cancer size, axillary lymph-node involvement and presence of distant metastases (TNM) classification is being developed to reflect the new and evolving knowledge of prognostic factors in breast cancer.
Footnotes
The author has no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, royalties.
No writing assistance was utilized in the production of this manuscript.
