Abstract
Vaginal and vulvar cancers are less prevalent than their cervical counterparts, and collectively account for approximately 6% of all diagnosed gynecological cancers [1]. The American Cancer Society estimates that in the year 2007 there will be 3490 new cases of vulvar cancer with 880 mortalities, and 2140 new cases of vaginal cancer with an additional 880 mortalities. Considering the rarity of these gynecological cancers, literature on the topic is less than that available for cervical disease. Despite their relative rarity, both in situ and invasive vulvar carcinomas have increased their incidence by an estimated 411 and 20%, respectively, between 1973 and 2000 [2]. in discussing vaginal and vulvar dysplasia and cancer, one cannot overlook the long-suspected association between human papillomavirus virus (HPV) infection and disease, which has been a topic of focus in gynecologic oncology for at least 20 years [3,4]. Studies using PCR methodology have found the percentage of vaginal cancers positive for HPV to be 60–65%, and that of vulvar cancers to be 20–50% [5]. In this review, we will examine data regarding the prevalence of HPV infection in cases of vaginal/vulvar neoplasias and cancers, and consider what proportion of these diseases may be prevented through the recently developed quadrivalent HPV vaccine for types 6, 11, 16 and 18.
HPV epidemiology
The commonality of HPV among sexually active adults, and the likelihood that sexually active adults will become infected within their lifetime was another major factor behind the development of the vaccine. HPV is the most prevalent sexually transmitted infection in the USA, and the CDC has estimated the lifetime risk of contracting a genital infection from HPV to be at least 50% for sexually active men and women [6]. In the USA, there are an estimated 6.2 million people infected with the virus each year. It is believed that approximately 20 million Americans are currently infected.
Considering the annual rate of infection, it is apparent that many cases are cleared from detectable levels after some time [6]. A study of 608 college women in 1998 discovered that 70% of new HPV infections were cleared within 12 months, and 91% were cleared by 24 months after the time of initial infection. It was determined that the median duration of HPV infection in this cohort was approximately 8 months, depending on various factors regarding immune function and virus type. High-risk (oncogenic) HPV types, such as 16, 18, 61 and 73 were more likely to persist than nononcogenic types; however, the majority of high-risk HPV infections were cleared within 24 months. Virtually all transmission of the virus is via sexual intercourse with an infected individual, but data from lesbians suggest that intercourse in which an infected penis penetrates the vagina is not a strict requirement for seroconversion [7,8].
Cervical transformation zone
It is important to understand that HPV does not infect all regions of the female genital tract at the same frequency. We know, for example, that the clinical prevalence of dysplastic lesions in the various epithelia of the female genital tract (cervix, vagina and vulva) are quite varied. The incidence of cervical intraepithelial neoplasia (CIN) is tenfold higher than the incidence of dysplasia in the vagina or vulva, and the incidence of cervical dysplasia and their derived carcinomas peak approximately 10 years earlier than that of the vagina. Despite the high frequency of HPV infection events that occur throughout the female genital tract, it seems that the cervical transformation zone, as pictured in Figure 1, is of particular risk to HPV carcinogenicity [6,9].

1. Cervical transformation zone depicting columnar epithelium above and squamous epithelium below.
Molecular transformation events
The molecular mechanisms through which host cells are transformed by HPV are well known, and are executed largely via the E6 and E7 viral oncoproteins carried in the genomes of oncogenic HPV strains. The E6 and E7 proteins are implicated in the transformation of a normal cell to a cancerous one by respectively guiding the cell to evade apoptosis, and making it insensitive to antigrowth signals.
The E6 viral protein confers oncogenic phenotypes to its host cell primarily through its mediated degradation of p53, the most commonly altered gene in human cancers, one that is mutated in over 50% of all tumors. Often referred to as ‘the guardian of the genome’, p53 is a transcription factor responsible for the stimulation of many genes, including the apoptotic BAX genes, the G1 growth arrest p21 gene, and several members of the relevantly named p53-induced genes (PIGs), of which all are coinvolved with BAX in executing apoptosis [10]. In 1990, it was shown that the E6 proteins from HPV-16 and HPV-18 have a strong affinity for E6AP, a ubiquitation ligase. The E6/E6AP complex binds to p53, resulting in the ubiquitination of the p53–E6/E6AP complex, which is followed by a subsequent proteosome-mediated degradation of the tumor suppressor. This loss of p53 function leaves the cell unable to stimulate the apoptotic pathways, and also disrupts its ability to arrest the cell cycle when DNA damage is apparent (Figure 2). A significant observation with gynecologic cancers was made in 1991, when it was discovered that, unlike most other human cancers, p53 was found to be ‘almost invariably wildtype’ in cervical carcinomas, meaning that there was little to no deviation in base-pair sequence within p53 loci when compared with ‘normal’ cells producing functional p53 gene product. This wild-type p53 protein is then marked for degredation by the E6 protein, resulting in the loss of function of p53 as seen in other cancers. This fact emphasizes the role that E6 viral oncoproteins play in p53 downregulation and cervical carcinogenesis. However, it is worth noting that once cervical tumor cells metastasize, mutations within p53 become more frequently observed with time [11].

Interaction between human papillomavirus oncoprotein E6 & p53.
The E7 gene product is a viral oncoprotein carried in the genomes of both low- and highrisk HPV types. E7 proteins are structurally and functionally similar to the adenovirus E1A (Ad E1A) proteins and the simian virus 40 large tumor antigen (SV40 TAg), and cooperate in shifting a cell from G1 to S phase. Specifically, the E7 oncoprotein has been shown to bind to retinoblastoma protein (pRb), disrupting the pRb.E2F complex and resulting in a subsequent release of free, unbound E2F transcription factor. The E2F factors bind their promoters, and are responsible for the stimulation of genes involved in the G1 → S transition (Figure 3). In normal host cells, soluble antigrowth signals and those tethered to the extracellular matrix of nearby cells effectively block cell division upon binding their appropriate ligands. The pRb and its two relatives, p107 and p130, have been implicated in channeling many, if not all, of the known antiproliferation signals, and thus are a primary target for transformation. Studies that have compared the E7 oncoproteins of low- and high-risk HPVs have found differences in their binding affinity for pRb, and have thus shed light on some of the molecular differences key to explaining the different oncogenic potentials of HPV strains. The E7 proteins of high- and low-risk HPV strains have a single amino acid difference from one another in their pRb binding site (aspartic acid 21 in HPV-16 E7 vs glycine 22 in HPV-6 E7) that result in a seven- to ten-fold increase in binding affinity for high-risk strains such as HPV-16 as compared with lower-risk strains such as HPV-6 [12,13]. High-risk HPV E7 proteins do not simply bind to pRb, but also target pRb for degredation.
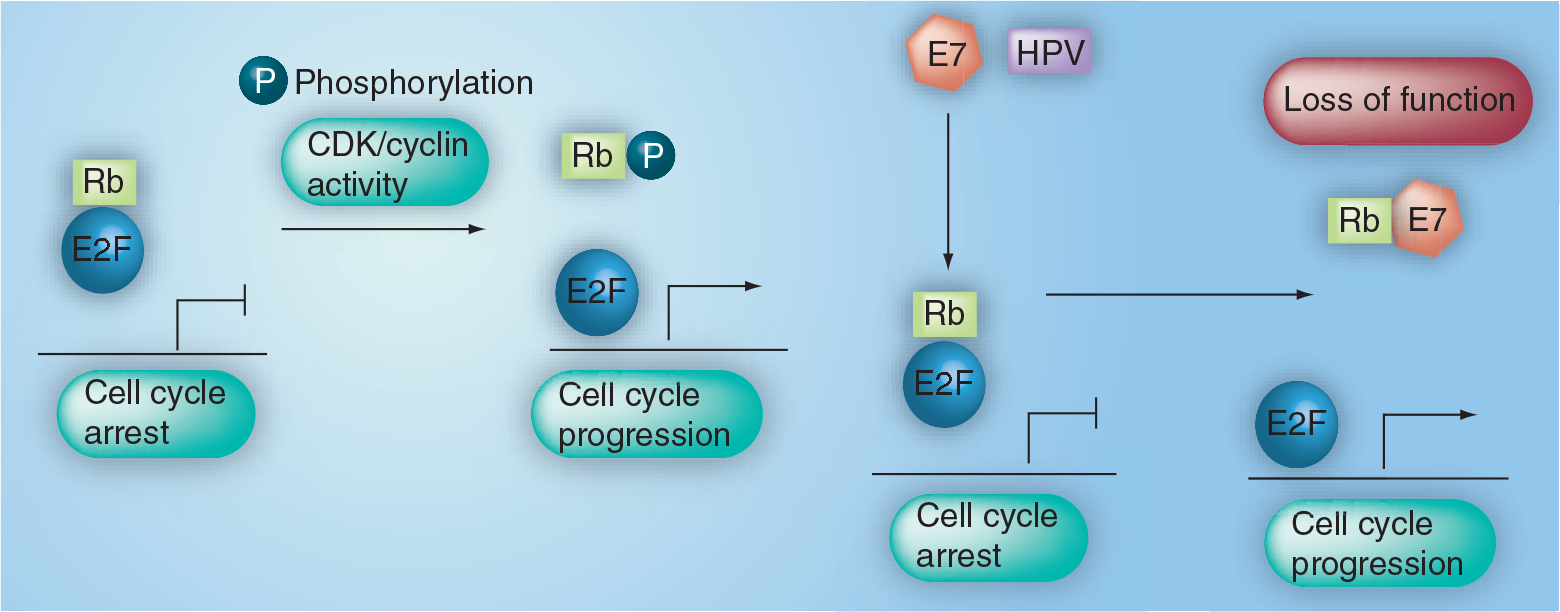
Interaction between human papillomavirus protein E7 & retinoblastoma protein.
Eliciting an immune response for disease prevention
While understanding the oncogenesis of HPV is important, its prevention relies upon understanding HPV immune function. The production of virus-like particles (VLPs) with exquisite immunogenicity has lead to the development of type-specific VLP-based vaccines.
Gardasil® (MERCK & Co., Inc., NJ, USA) is a three-dose prophylactic vaccine given at 0, 2 and 6 months. It is a sterile liquid suspension prepared from highly purified VLPs, more specifically, from recombinant L1 capsid proteins of HPV types 6, 11, 16 and 18. The L1 proteins are produced in recombinant Saccharomyces cerevisiae (yeast), purified and adsorbed on aluminum-containing adjuvant (amorphous aluminum hydroxyphosphate sulfate). Studies regarding the cell-mediated immune response to L1 VLP vaccination have found that vaccination significantly increased B-cell response, and also L1-specific T-cell response as detected by CD4+ and CD8+ proliferation. In addition, it has been found that significant increases in cytokine (IFN-γ, IL-5 and IL-10) responses to L1 VLPs are apparent in vaccinated individuals [14].
Connection between cervical & vaginal/vulvar lesions
To better understand the relationship between cervical disease and lesions of the vagina and vulva, researchers in Germany studied 1500 women with anogenital disease. Considering the rarity of vaginal and vulvar lesions as compared with those of the cervix, only seven patients from this cohort had high-grade vaginal or vulvar lesions and all had a previous history of cervical disease (five with prior high-grade cervical dysplasia, and two with a history of cervical cancer). Since it is well known that high-risk oncogenic papillomavirus genomes often integrate randomly into host-cell genomes, HPV-16 and HPV-18 viral integration sites were examined in multiple metachronous lesions of these patients' lower genital tracts, and compared with those of their previous cervical disease. It was later determined that four of the five patients with a previous history of high-grade cervical dysplasia had identical HPV integration sites for their vaginal and/or vulvar lesions, when compared with their previous cervical disease. Both of the patients with a previous history of cervical cancer also showed identical viral integration sites between their vaginal and/or vulvar lesions and their previous cervical tumors. These data suggest that perhaps a large majority of high-grade lesions in the female genital tract emerge as monoclonal cell populations derived from the cervical transormation zone [9]. Owing to this fact, we must consider vaccine efficacy in preventing cervical lesions in our estimate of how vaginal and vulvar epidemiology may change.
HPV vaccination & cervical disease
Gardasil (Merck & Co., Inc., NJ, USA) is a quadrivalent vaccine for HPV types 6, 11, 16, and 18, and is the first vaccine approved by the US FDA for prevention of a gynecologic cancer; specifically targeting that of the cervix. In fact, it has been said that the association between oncogenic HPV types and cancer of the cervix is unique in cancer research, and that there is no human cancer yet to be found with such a clearly defined causative agent [15]. The FUTURE I and FUTURE II clinical trials of the vaccine highlight its efficacy in prevention of cervical dysplasia and invasive carcinoma associated with those HPV types administered.
In the primary analysis group of the FUTURE I clinical trials, the per-protocol population, women aged between 16 and 24 years were randomly assigned three doses of either HPV-6/11/16/18 vaccine or placebo, at day 1, month 2 and month 6. All the women in this group had no virologic evidence of HPV types 6, 11, 16 and 18, from the time of accrual to 1 month after the third dose. Out of the 2258 women in the per-protocol placebo group screened for cervical lesions CIN 1–3/adenocarcinoma in situ (CIN1–3/AIS), there were 65 cases of dysplasia, while the 2241 women in the vaccine group remained disease-free. Under these optimal conditions, vaccine efficacy was 100% (95% CI: 94–100) [16].
To gauge vaccine efficacy under less than perfect conditions, women were enrolled who were HPV negative at accrual, but who may have violated testing protocols and contracted the disease between dose one and 1 month after dose three were studied. In this group, there were two cases of cervical dysplasia in the vaccine group (N: 2667), and 89 cases in the placebo group (N: 2684), to yield a 98% efficacy (95% CI: 92–100). Finally, to test efficacy for CIN1–3 and AIS in an intention-to-treat population, which included women who had previously been exposed to HPV, 2723 women were given vaccine and 2732 were given a placebo. Considering that there were 71 dysplastic events in the vaccine group and 155 in the placebo group, vaccination was found to be 55% effective under these conditions (95% CI: 40–66). It should be noted that the FUTURE I trials also quantified incidences of vulvar intraepithelial neoplasia (VIN) and vaginal intraepithelial neoplasia (VaIN), and these data are provided in Table 1 and Table 2 [16].
FUTURE I study group.

Summary of efficacy of HPV vaccination in preventing various anogenital dysplasias within the studied populations from the FUTURE I clinical trials.
AIS: Adenocarcinoma in situ; CIN: Cervical intraepithelial neoplasia.
FUTURE II study group.

Summary of efficacy of HPV vaccination in preventing various anogenital dysplasias within the studied populations from the FUTURE II clinical trials.
AIS: Adenocarcinoma in situ; CIN: Cervical intraepithelial neoplasia; HPV: Human papillomavirus.
In the FUTURE II trials, women between the ages of 15 and 26 years were assigned to receive three doses of either the HPV-6/ 11/16/18 vaccine or a placebo. Of the 10,565 women who were seronegative and DNA negative on cervical swab for HPV-16 and HPV-18 at accrual, and remained so 1 month after dose three, and who were also screened for cervical lesions (CIN2–3 and AIS), 5305 were given vaccine and 5360 placebo. In this per-protocol population, there was one case of cervical dysplasia in the vaccine group, and 42 cases in the placebo group, giving a vaccine efficacy of 98% (95% CI: 86–100). In women who were HPV seronegative and DNA negative (for HPV-16/18) at accrual, but may have become infected between dose one and 1 month past dose three, vaccination was found to be 95% effective (95% CI: 85–99). Finally, in the FUTURE II intention-to-treat population, which included women who may or may not have been previously infected by HPV, there were 83 cases of dysplasia among the 6087 women given vaccine, and 148 cases among the 6080 given placebo. Under these conditions, vaccination was found to be 44% effective (95% CI: 26–58) (Table 1) [17].
HPV vaccination & vaginal/vulvar disease
To gauge the effect that vaccination may have on the incidence of vulvar and vaginal cancers, we refer now to three clinical trials carried out between 2000 and 2007. A total of 18,174 women between the ages of 16 and 26 years were divided amongst three double-blind, placebo-controlled, randomized trials. Women were enrolled from 157 locations in 24 countries in the Americas, Europe and Asia. All participants were nonpregnant, healthy, had no previous abnormal pap smear, and had a lifetime history of four or fewer sexual partners. Participants underwent anogenital screening at day 1, at 1 month after administration of the third dose, and at 6–12 month intervals for up to 48 months. Vaccination efficacy under optimal circumstances was analyzed in the per-protocol susceptible population [1].
The researchers' per-protocol susceptible population was comprised of 15,596 women, 7811 of whom were given the vaccine, and 7785 of whom were given the placebo (both containing 225 mg of amorphous aluminum hydroxyphosphate sulfate adjuvant, and both administered at months 0, 2 and 6). All women in the per-protocol susceptible population were established at day 1 to be both seronegative and HPV DNA negative by PCR to either HPV-16 or HPV-18, and all remained both seronegative and HPV DNA negative to that same HPV type through 1 month after dose three. Of the 15 women in this group who developed histologically confirmed VIN2–3 or VaIN2–3 associated with HPV-16 and HPV-18, all had been given placebo, and quadrivalent vaccination was found to be 100% effective against VIN2–3 or VaIN2–3 for women under these conditions [1].
To look at vaccination efficacy under slightly more lenient restrictions, another group of women was identified and termed the unrestricted susceptible population. In this group, all individuals were also established to be both PCR-negative and seronegative to either HPV-16 or HPV-18 at the time the first dose was administered; however, individuals may have violated testing protocols, such as proper use of contraception throughout the trial, and may have contracted HPV between dose one and 1 month past dose three. In the unrestricted susceptible population, 8757 women were given the vaccine, and 8774 were given placebo. There were 29 cases of HPV-related VIN2–3 or VaIN2–3 in the placebo group, and a single case of VIN2–3 (HPV-16 positive) in the vaccine group, rendering the vaccine 97% effective under these conditions [1].
Finally, to estimate vaccination efficacy without regard to baseline HPV status, or previous HPV-related anogenital disease, an intention-to-treat population was examined. In the intention-to-treat population, 9087 women were given the vaccine, and 9087 were given placebo. In this group, there were 31 cases of HPV-related VIN2–3 and/or VaIN2–3 in the placebo group, and nine cases in the vaccine group, reducing the incidence of VaIN2–3 and VIN2–3 by 71%. While initially the health benefits associated with vaccinating the intention-to-treat population may have been clouded by prevalent infection or disease, the prevention of new HPV-16 and HPV-18 infections led to a marked decrease in the incidence of VIN2–3 and VaIN2–3 with time. Furthermore, after all high-grade lesions were examined in this group, and disregarding whether or not HPV was detected in the lesion, 27 cases were observed in the vaccine group, and 53 in the placebo group, yielding a 49% efficacy under these conditions [1]. Table 3 summarizes the data reviewed regarding the efficacy of the quadrivalent HPV vaccine in preventing vaginal and vulvar dysplasia.
Combined analysis of three clinical trials.

Summary of the reviewed data regarding efficacy of the HPV vaccine in preventing vulvar and vaginal high-grade intraepithelial neoplasia.
HPV: Human papillomavirus; VaIN: Vaginal intraepithelial neoplasia; VIN: Vulvar intraepithelial neoplasia.
Despite the fact that the mean age of incidence for VIN2–3 has decreased from 50 to 39 years in recent decades, current clinical trials still exclude women at the peak age of incidence for disease [14]. Owing to the fact that the majority of vulvar and vaginal dysplasias and cancers are in older patients, these studies may underestimate the ultimate benefits of vaccination. While other gynecologic diseases, such as invasive cervical carcinoma, have been established to be nearly 100% attributable to oncogenic HPV types, vaginal and vulvar dysplasias and carcinomas seem to be slightly less attributable to the virus.
Executive summary
An estimated 6.2 million Americans are infected with the human papillomavirus (HPV) annually.
It is believed that approximately 20 million Americans are currently infected.
Studies have shown that an estimated 70% of new infections are cleared by the immune system within 12 months postinfection, and an estimated 91 % are cleared within 24 months.
Oncogenic HPV types such as 16, 18, 61 and 73 were more likely to persist than nononcogenic types, however the majority of oncogenic HPV infections were also cleared within 24 months.
The incidence of cervical intraepithelial neoplasia (CIN) is tenfold higher than the incidence of dysplasia in the vagina or vulva, and the incidence of cervical dysplasia and their derived carcinomas peak approximately 10 years earlier than that of the vagina.
Despite the high frequency of HPV infection events that occur throughout the female genital tract, the cervical epithelium experiences transformation events at a much higher frequency than vaginal and vulvar epithelium.
A large majority of high-grade lesions in the female genital tract emerge as monoclonal cell populations derived from the cervical transformation zone.
The molecular mechanisms through which host cells are transformed by HPV are well known, and are executed largely via the E6 and E7 viral oncoproteins carried in the genomes of oncogenic HPV strains.
The E6 viral oncoprotein binds to p53, resulting in ubiquitination of the cellular protein, targeting p53 for proteosome-mediated degradation. p53 normally functions to gauge DNA damage, halt cell-cycle, and activate the apoptotic pathway.
The E7 viral oncoprotein has been shown to bind to retinoblastoma protein (Rb), releasing E2F from the Rb–E2F complex, and resulting in the E2F-mediated stimulation of genes involved in the G1 ŕ S transition.
The E7 proteins of high-and low-risk HPV strains share a specific amino acid difference in their pRb binding site (aspartic acid 21 in HPV-16 E7 vs glycine 22 in HPV-6 E7) that result in a seven- to ten-fold increase in binding affinity for high-risk strains such as HPV-16 as compared with lower-risk strains such as HPV-6. High-risk HPV E7 binds to and targets pRb for degredation.
The production of viral-like particles (VLPs) with exquisite imunigenisity has lead to the development of type-specific VLP-based vaccines.
Gardasil (MERCK & Co., Inc., NJ, USA) is prepared from the VLPs of L1 capsid proteins of HPV types 6, 11, 16 and 18, and are produced in recombinant Saccharomyces cerevisiae (yeast).
Studies regarding the cell-mediated immune response to L1 VLP vaccination have found that vaccination significantly increased B-cell and L1-specific T-cell responses, and also increased the activity of cytokines such as IFN, IL-5 and IL-10.
According to the FUTURE I clinical trial, women who were HPV negative and remained negative through 1 month after the third dose, the vaccine was 100% effective in preventing cervical intraepithelial neoplasia (CIN)1–3 and adenocarcinoma in situ (AIS) associated with HPV 16 and 18.
In the FUTURE I intention-to-treat group, which included women who may have previously been exposed to HPV, vaccination was found to be 55% effective against CIN2–3 and AIS associated with HPV 16 and 18.
Data from the FUTURE II trials suggest that in women who were HPV negative and remained so through 1 month after the third dose, vaccine efficacy is 98% against prevention of CIN2–3 and AIS associated with HPV 16 and 18.
The intention-to-treat group in the FUTURE II trial, which included women who may have previously been exposed to HPV, suggest that vaccination is 44% effective against all cases of CIN2–3 and AIS associated with HPV 16 and 18.
In women who were HPV negative prior to vaccination and remained HPV negative through 1 month after the third dose, the vaccine was found to be 100% effective in preventing vulvar intraepithelial neoplasia (VIN)2–3 and vaginal intraepithelial neoplasia (VaIN)2–3 associated with HPV16 and HPV18.
In women previously exposed to HPV, vaccination was found to reduce the incidence of VIN2–3 and VaIN2–3 by 71%.
A recent study published in 2006 examined 210 cases of intraepithelial neoplasia (VIN2–3, VaIN2–3 and AIN2–3), and 48 cases of vulvar carcinomas, and found HPV DNA to be in 92, 91, 89 and 60% of the VIN, VaIN, AIN and vulvar carcinoma samples, respectively [18]. More specifically, high-risk oncogenic HPV types 16 and 18 were found in 76, 64, 81 and 42% of the VIN2–3, VaIN2–3, AIN and vulvar carcinomas, respectively [18]. The proportion of cases seemingly attributable to HPV infection is substantial enough that we can expect the prevention of many pre-invasive neoplasias and invasive cancers of the vagina and vulva with proper vaccination.
Future perspective
Given the relative infancy of HPV vaccination, a limited amount of speculation can be made regarding the ultimate effect of the vaccine on the future epidemiology of HPV-related vaginal and vulvar disease. Within the next 5–10 years, there will undoubtedly be a greater availability of data regarding the duration of immunogenicity provided by the vaccine, and we will be able to assess how much benefit the vaccine will have in protecting women against vaginal and vulvar cancer as they age. Future studies should aim to evaluate the at-risk population, including older women and young women most at risk in the HIV-positive population, and others who are immunocompromised, such as renal transplant patients. Universal vaccination of women above the FDA-approved age limit could further impact the incidence of HPV-related vulvar and vaginal dysplasia, and ultimately cancer.
Footnotes
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending or royalties. No writing assistance was utilized in the production of this manuscript.
