Abstract
Stress urinary incontinence affects women of all ages, becoming more prevalent with increasing age. While many nonsurgical therapies are available for the treatment of stress urinary incontinence, options are limited for women who fail to respond to these therapies yet may not be candidates for, or wish to avoid, surgery. This limitation prompted efforts to develop less-invasive procedures for treating stress urinary incontinence patients, including bulking-agent injections and a new transurethral, radiofrequency collagen-denaturation system. This new treatment (Renessa®) reduces tissue compliance without necrosis or stricture, thus differentiating it from a surgical radiofrequency treatment, transvaginal radiofrequency tissue ablation. The advent of new treatment options for stress urinary incontinence allows physicians to offer their patients a broader choice of treatment options, underscoring the importance of educating patients regarding all available therapies, including success rates and risk for complications. This is particularly important for women who have not responded to prior treatment strategies.
Keywords
Urinary incontinence, a common medical condition, occurs in women of all ages, increasing in prevalence with advancing age. It is estimated to occur daily in 12.2% of women aged 60–64 years, and daily prevalence increases with age to 20.9% in women aged 85 years and older [1]. Stress urinary incontinence (SUI) is the involuntary loss of urine that occurs during coughing, sneezing or physical exertion such as sporting activity or sudden changes of position. In severe cases, there may also be gravitational loss of urine with postural changes [101], a condition that is due to sphincter abnormalities [2]. While intrinsic sphincter deficiency (ISD) is one cause of SUI [3–5], the most common type of SUI is associated with urethral (bladder-outlet) hypermobility [2]. The laxity of the pelvic floor muscles that support the bladder may be attributable to childbirth, prior pelvic surgery or fracture, neurologic dysfunction and, possibly, the loss of estrogen during menopause [101].
There are many treatments available to women with SUI, but there is a distinct gap in the range of options for those who fail to respond to traditional nonsurgical options and may not be candidates for, or desire not to undergo, surgery. When evaluating incontinence procedures for treatment response, it is important to note that the definition of response is not defined consistently in clinical trials. Although the Urodynamics Society has issued guidelines on terminology such as dry, cured and successful [2,6], the definitions of such words vary greatly between studies. This paper briefly reviews various nonsurgical therapies and surgical treatments for SUI, with a focus on new nonsurgical procedures that may bridge the gap between them.
Conventional nonsurgical therapy
Nonpharmacologic treatment
As they are noninvasive, behavioral and physical treatments are typical first-line therapies for patients with SUI. The wide range of options available includes biofeedback, pelvic floor conditioning, pelvic floor electrical stimulation, bladder training, the use of pessaries or occlusive devices, vaginal cones, lifestyle modification, intermittent self-catheterization, and treatment of medical comorbidities that may contribute to the condition [101]. Clinical trials have demonstrated the effectiveness of each of these treatments and many women can experience symptom improvement; however other women fail to benefit from them. Results from clinical studies indicate that pelvic floor muscle strengthening is effective in improving incontinence in approximately 44–69% of women with SUI [7,8]. One study indicated that a third of patients either using or not using a specially designed audiotape during pelvic floor exercises remained improved at 6 months [9]. However, in a study that assessed the effects of bladder training, pelvic floor muscle training or a combination of these treatments, outcomes did not substantially change in 67% of women with SUI [10]. In another study, 47% of women with SUI experienced long-term (2-year) benefit with biofeedback-based therapy [11]. Long-term compliance is essential with conventional nonsurgical therapies, as outcomes rely heavily on patient persistence.
Pharmacotherapy
Pharmacologic intervention is often the second step in the treatment of patients who fail initial treatment with nondrug therapies. Although no pharmacologic treatment for SUI has been approved by the US FDA, several medications are prescribed by some physicians. Duloxetine, a dual norepinephrine–serotonin-reuptake inhibitor, is commonly prescribed in Europe. One 12-week study of 227 women with SUI who received duloxetine 40 mg twice-daily revealed a median decrease of 54% in the frequency of incontinence episodes and a median increase of 20.4 min in voiding intervals. Investigators also reported a median ten-point increase in quality-of-life questionnaire scores. However, as with many drugs, long-term use may be limited by side effects. In addition, many women prefer not to take a twice-daily drug on a long-term basis for a benign condition [12]. Although there was optimism that duloxetine would be approved by the FDA for the treatment of patients with SUI, the manufacturer withdrew its application for approval. At this time, it is unclear if duloxetine will ever become FDA approved for the treatment of SUI in the USA.
A variety of other agents, including tricyclic antidepressants (TCAs), α- and β-adrenoceptor agonists, β-adrenoceptor antagonists and estrogen-replacement therapy, have been used to treat patients with SUI. However, these medications are not consistently effective and patients may experience adverse events that can include elevated blood pressure, exacerbation of abnormal cardiac rhythms (α-adrenoceptor agonists), cardiac decompensation (β-adrenoceptor antagonists), anticholinergic symptoms, orthostatic hypotension and arrhythmias (TCAs) [13–15]. Muscarinic cholinergic receptor antagonists have been shown to be effective in the treatment of overactive bladder and urge urinary incontinence, but have not demonstrated efficacy in patients with SUI [15]. However, these agents may be helpful in cases of mixed urinary incontinence.
Surgical intervention
As a result of the lack of pharmacologic options for SUI patients, women who currently fail treatment with nonsurgical interventions frequently undergo surgery. Of the two major types of surgical interventions for women with SUI, neither the suburethral sling procedure nor colposuspension is accepted as first-line treatment for all patients, although slings have demonstrated long-term efficacy. Traditional pubovaginal sling operations have been performed using a wide range of autologous and synthetic materials. The development of tension-free vaginal tape has substantially increased the popularity of this procedure. The aim of this intervention is to place the tape in a U shape around the midurethra, or as a backboard, as in the obturator procedures, to provide a hammock against which the urethra can be compressed when there are sudden changes in abdominal pressure [16].
Colposuspension may be carried out as either an open or laparoscopic procedure. This surgery involves lifting the tissues near the bladder neck and proximal urethra in the area behind the anterior pubic bones to correct deficient urethral closure. A recent Cochrane review indicated that open colposuspension resulted in overall cure rates ranging from 68.9 to 88.0% [17]. A similar range of success rates has been reported for the laparoscopic procedure [18].
Transvaginal radiofrequency bladder-neck suspension (SURx® Transvaginal System, Cooper Surgical, CT, USA) has been used as a surgical option for women with SUI [19–21]. This ablation technique has typically been performed transvaginally in a hospital setting under general anesthesia, but has also been reported in an outpatient setting, performed laparoscopically, using local anesthesia with intravenous sedation. An incision is made through the vagina, lateral to the urethra, exposing the endopelvic fascia. Radiofrequency energy is then applied in a slow, sweeping motion over the endopelvic fascia at high temperatures, resulting in the blanching and gross shrinkage of a large area of tissue secondary to scarring and necrosis [19,20]. Buchsbaum and colleagues reported that, owing to low cure rates, low patient satisfaction levels and the frequent need for additional treatment with this procedure, her practice has discontinued offering SURx as a treatment option [21]. Owing to similar results across the country, the popularity of this procedure has declined.
While some surgical procedures have demonstrated high cure rates, there are potential complications with any surgery. Immediate complications of surgical intervention for SUI may include urinary retention, accidental bladder injury and excessive bleeding. Delayed complications can include wound infection, urinary tract infection, chronic pain, erosion and de novo detrusor overactivity [22–24]. The overall rate of complications for patients from the Nationwide Inpatient Sample (n = 147,473) who underwent a surgical intervention for SUI was reported to be 13.0% [25].
The effectiveness of surgical procedures for SUI can decline somewhat over time. In a long-term study of pubovaginal slings, 108 patients underwent the procedure with polypropylene mesh. Follow-up ranged from 24 to 72 months and showed that 71 (65.7%) patients remained continent [26]. In a separate trial, follow-up of 82 SUI patients who underwent a modified pubovaginal sling procedure and responded to an outcomes questionnaire (mean follow-up: 3.4 years; range: 0.5–8 years) revealed that 40 (49.3%) patients were completely continent while 18 (21.9%) reported partial continence [27]. In what is considered to be a landmark study, Ward et al. conducted a multicenter trial, randomizing 341 women with SUI to undergo a transvaginal tape midurethral sling or a Burch colposuspension. At the end of 2 years, objective cures were found in 63% of women who had undergone a sling procedure and 51 % of the women who had undergone a colposuspension [28].
Treatment gap
For women with SUI who fail traditional nonsurgical treatment for SUI, surgery is often considered the only effective treatment option. However, not all of these patients are suitable candidates for invasive surgical procedures, and many are reluctant to undergo surgery. Results from a study of women (n = 267) aged 75 years and older who underwent urogynecologic procedures revealed that 25.8% of patients experienced significant perioperative complications, including blood transfusions or significant blood loss, pulmonary edema and postoperative congestive heart failure [29]. Factors associated with an increased risk for adverse events included coronary artery disease and peripheral vascular disease. Some patients who are fearful of surgery avoid seeking treatment for SUI because they worry that a surgical procedure might be recommended [30]. A recent survey revealed that only 38% of women consider a surgical intervention that has an 85% cure rate with a 2% risk of self-catheterization to be acceptable treatment [12].
Filling the treatment gap
Considerable effort has been directed toward developing nonsurgical procedures for women who do not respond to conservative SUI therapy but who are not candidates for, or who wish to avoid, invasive surgical procedures. The goal has been to identify an option that can be performed under local anesthesia, with minimal associated trauma and a success rate comparable to that of the sling procedure or colposuspension, although with reduced morbidity [3,16]. These procedures should be simple, quick and easily learned by surgeons with varied skill levels [16]. The desire for less invasive interventions for women with SUI has increased interest in injectable periurethral bulking agents and in a nonsurgical radiofrequency treatment.
Injectable bulking agents
Urethral bulking agents are most suitable for patients with ISD [3–5], and all FDA-approved agents are approved for this indication only. The bulking agent is injected with the use of a cystoscope, and multiple injections may be required to achieve and maintain continence (Figure 1). The compounds most commonly used in the USA are carbon-coated zirconium beads (Durasphere®; Carbon Medical Technologies, Inc., MN, USA) and glutaraldehyde cross-linked (GAX) bovine collagen (Contigen®; CR Bard, GA, USA) [3]. The declining efficacy of most bulking agents over time can limit their use, and cost–effectiveness can become an issue with repeated procedures. Initial rates of improved or complete continence with these injectables are approximately 60–80% at 1–3 months [31,32]; this rate drops to approximately 50% after 2 years [32,33]. A study of 60 patients injected with GAX collagen showed that 37 (61%) patients were considered continent or improved at 3 months. This number declined to 32 (54%) at 12 months and 29 (48%) at 24 months [32]. In a study of 25 women with SUI who received paraurethral injections of GAX collagen, 20 (80%) women were considered cured or improved at 3 months [31]. A separate study of 34 women with SUI treated with paraurethral injections of GAX collagen showed that 16 (48%) patients were continent and three (9%) were improved at 2 years [33]. In an 18-month follow-up report of 46 women with SUI who were injected with carbon-coated zirconium beads, 18 women (50%) were considered cured, two (5.5%) were improved and 16 (44.4%) were deemed as treatment failures [34].
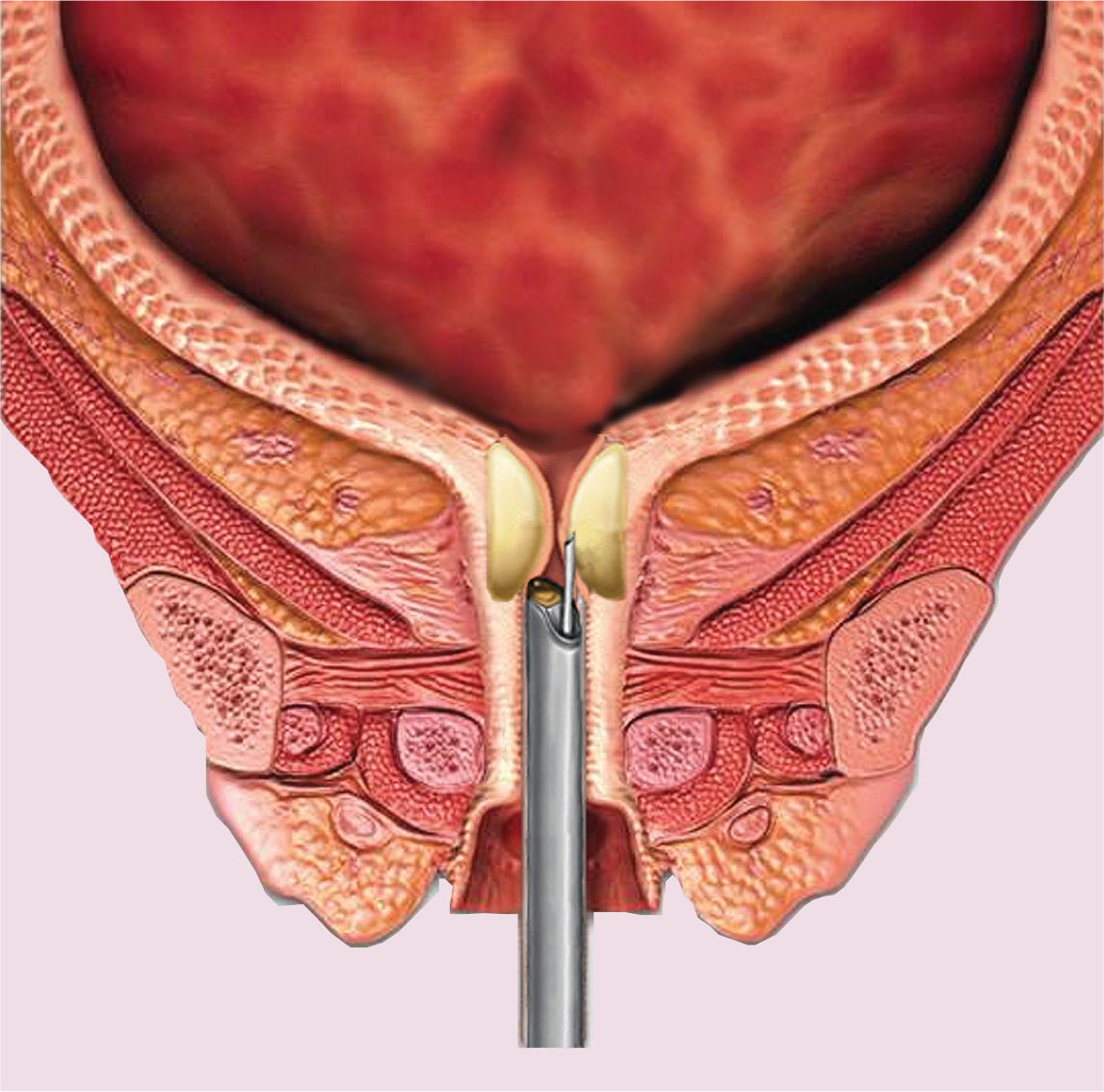
Placement of needle for periurethral injection of a bulking agent.
Several new bulking agents have received FDA approval for treating women with SUI due to ISD. Hydroxylapatite particles suspended in an aqueous gel carrier (Coaptite®; Boston Scientific, MA, USA) can be injected transurethrally, periurethrally or suprapubicly. In a study of 27 women with SUI injected with this agent, 1-year results showed an 80% reduction in pad use [35]. The second new agent, a copolymer implant (formerly called URYX, now Tegress™; CR Bard, Inc.), was periurethrally injected into 253 women with SUI. At 12 months, 47 (18.4%) patients reported complete continence, 123 (48.7%) reported improvement in incontinence and 120 (47.5%) reported no improvement [102]. However, Tegress was withdrawn from the market by the manufacturer in January 2007, less than 2 years after its approval by the FDA.
The newest bulking agent to receive FDA approval for SUI due to ISD is polydimethylsiloxane (Macroplastique®; Uroplasty, Inc., MN, USA), which has been used for more than 10 years in Europe and other countries. A follow-up of 21 Brazilian women with SUI who underwent periurethral injection of polydimethylsiloxane [36] revealed that at 2 years, 12 patients (57.1%) were considered cured, two (9.5%) were improved and seven (33.3%) were incontinent based on pad weight testing. A 60-month follow-up of 15 of these women determined that six (40%) remained continent, five (33.3%) were improved and four (26.7%) were incontinent.
Although injectable bulking agents are associated with a low rate of complications, a recent retrospective study found a late complication in some patients who were treated with periurethral injections of carbon-coated zirconium beads [37]. Four (3%) of 135 patients whose charts were examined had developed a periurethral mass 12–18 months post-treatment that required transvaginal endoscopic drainage or transvaginal excision. The investigators concluded that pelvic pain or obstructive or irritative voiding symptoms could indicate periurethral mass formation in patients who have received periurethral treatment with a bulking agent. However, the true incidence of this delayed adverse event is not yet known.
Transurethral collagen denaturation: a new option
Recently, the FDA approved a nonsurgical transurethral radiofrequency collagen-denaturation system (Renessa®, Novasys Medical, Inc., CA, USA) for use in women with SUI due to urethral hypermobility. This procedure differs from surgical transvaginal radiofrequency bladder-neck suspension in two important ways. First, the newer collagen-denaturation treatment is a nonsurgical procedure that does not require an incision and can be performed by trained specialists in an outpatient or office setting under local anesthesia. Typically, women can return to most normal daily activities the same or the next day. In addition, while radiofrequency bladder-neck suspension is an ablation procedure that causes necrosis and gross tissue shrinkage, transurethral radiofrequency collagen denaturation remodels tissue without causing significant damage. During the collagen denaturation procedure, a transurethral probe (Figure 2) heats very small areas of submucosal tissue in the bladder neck and proximal urethra to high temperatures (approximately 65°C), causing the patient's collagen to remodel and denature without resulting in necrosis or small-vessel thrombosis. Upon healing, collagen in the treated areas renatures in a more compact and less compliant structural pattern. The healed microscopic sites are organized in a helical pattern around the proximal urethra and bladder neck to increase tissue resistance to involuntary leakage at times of increased intra-abdominal pressure, thereby reducing SUI episodes [38].

Transurethral collagen denaturation radiofrequency probe.
A 1-year controlled US multicenter trial in 173 women with SUI prospectively randomized patients to undergo either active treatment (n = 110) or a sham procedure (n = 63) [39]. Computer-generated randomization targeted a 2:1 treatment ratio. The sham treatment was identical to the active treatment except that the probes lacked needle electrodes and the generator did not actually deliver radiofrequency energy. Results showed that in treated patients, 84 (76%) experienced a reduction in daily incontinence episodes and 75 (68%) used fewer pads each day, including 64 (58%) who no longer used any pads [39,40]. Results also showed that 80 (73%) patients experienced a reduction in leak severity, including 39 (35%) women who became continent as a result of this treatment. This study also utilized the Incontinence Quality of Life (I-QOL) instrument, wherein scores are associated with specific improvements. For example, a ten-point increase in overall score correlates with a 25% or greater reduction in incontinence pad weight and a 25% or greater reduction in incontinence episode frequency [41]. Among both postmenopausal and premenopausal women in this study with moderate to severe SUI symptoms at baseline, approximately 128 (74%) demonstrated a ten-point or greater increase (improvement) in I-QOL scores at 12 months [38,39]. The most common adverse events were temporary dysuria (9.6 vs 1.6% for sham treatment), wet overactive bladder (10.0 vs 9.5%) and dry overactive bladder (7.3 vs 3.2%). No serious adverse events were reported [38,39,42].
Appell et al. recently published a retrospective follow-up of 21 patients from this 12-month study involving assessment of 3-day diaries and I-QOL scores [43]. At 3 years or more, patients reported a mean overall I-QOL score improvement of 12.7 (±26) compared with baseline. In addition, 56% of patients achieved a 50% or greater reduction in incontinence frequency, the same as at 1 year, indicating durability of response. No new adverse events arose during the course of follow-up compared with the 12-month results, and 81% of women did not go on to have a more invasive surgical procedure. Those who did have subsequent surgeries experienced no negative effects, suggesting that transurethral collagen denaturation does not preclude future, more invasive therapies should they be warranted.
In another study using this procedure in 41 women with SUI, the percentage of patients who experienced improvement in I-QOL scores at 6 months ranged from 75 to 80%. At 12 months, scores remained high, with 75–78% of patients experiencing an improvement from baseline [44,45]. Additional studies are being conducted to further define the role of this intervention in the treatment of SUI. These include a large, multicenter, 3-year durability trial and a randomized, sham-controlled study on the role of transurethral collagen denaturation in patients who have had previous SUI surgical procedures with suboptimal outcomes.
Selecting therapy: the importance of doctor–patient communication
Although incontinence is not a life-threatening condition, its symptoms can substantially impact a patient's quality of life. It is important for a physician to determine the symptoms that are most distressful to a patient, that is, stress, urge or mixed, before selecting treatment options. The decision regarding the most suitable treatment should then be made by the patient, with the guidance of her physician. Noninvasive behavioral and physical therapies are typically first-line treatments. To achieve optimal results, it is important that patients fully understand how the treatment works and how to perform any techniques involved. Invasive procedures are warranted if the patient's quality of life has significantly deteriorated due to SUI; however, most women, when given a choice, opt for conservative measures first [13]. Part of the physician's role in the overall management of women with SUI is educating them regarding available procedures and providing up-to-date information regarding success rates, durability and risks for complications. It is also important that an evaluation of the treatment's success be made from the patient's perspective, since patient and physician perceptions of successful therapy may differ substantially. In an evaluation of surgery for SUI conducted in 442 women [46], 66% of patients felt that after 1 year, results from the procedure had met or exceeded their expectations, and 68% would recommend the operation to a friend in a similar situation. By contrast, 85% of surgeons were satisfied with treatment outcomes after 1 year, and would again treat 94% of the women as they had done previously. This imbalance highlights the need for improvement in communication, as well as a better physician understanding of the patient's perspective.
Excutive summary
There are many traditional nonsurgical options available for the treatment of stress urinary incontinence (SUI), as well as many surgical options; however, for women who have failed traditional treatments and cannot or do not wish to undergo surgery, there are limited treatment options.
Options include behavioral therapy, biofeedback, pelvic floor stimulation, bladder training, use of pessaries or occlusive devices, lifestyle modification, intermittent self-catheterization, and treatment of medical comorbidities.
While these treatments can be beneficial for some patients, other women do not show substantial improvement with them.
No pharmacologic therapy is currently approved by the US FDA for the treatment of SUI.
Several medications, including duloxetine and tricyclic antidepressants, have been used off label to treat SUI symptoms, but these medications are not consistently effective and patients may experience side effects.
There are two major types of surgeries available for the treatment of SUI, the suburethral sling and the colposuspension, and although results can decline somewhat over time, the newer sling procedures have demonstrated long-term efficacy.
Women who have failed or achieved suboptimal results with traditional nonsurgical treatments, and who cannot or do not wish to undergo surgery, find themselves with a gap in SUI treatment options.
Urethral bulking agents are approved by the FDA for treatment of patients with intrinsic sphincter deficiency.
Multiple injections may be required to achieve continence and, while initial rates of improved or complete continence are good, efficacy can decrease over time and repeated procedures may be needed.
The collagen denaturation system is approved by the FDA for use in women with SUI due to hypermobility.
This incision-free procedure is performed transurethrally in a physician's office using local anesthesia.
A recent post hoc evaluation of patients who underwent transurethral collagen denaturation demonstrated sustained efficacy after 3 years or more.
It is important to determine the type of incontinence a patient is experiencing (i.e., stress, urge or mixed) and its cause (i.e., intrinsic sphincter deficiency or hypermobility) before selecting treatment options.
Physicians should ask their patients questions regarding urinary incontinence during checkups to determine which patients could benefit from treatment, and should discuss all treatment options, along with their success rates and risk of complications, with their SUI patients.
For women with SUI who cannot or do not wish to undergo invasive procedures, urethral bulking agents and transurethral collagen denaturation may be suitable treatment options, with collagen denaturation demonstrating durability for at least 3 years post-treatment.
Many women with SUI will desire or require a durable alternative to surgical treatment.
Injectable bulking agents will remain an acceptable alternative to surgery for patients with SUI due to intrinsic sphincter deficiency.
Further studies of newer agents will reveal durability of response.
Transurethral collagen denaturation could become an acceptable alternative to surgery for women with SUI due to urethral hypermobility.
Studies currently being conducted could broaden the indication for transurethral collagen denaturation to include women who experience suboptimal results with surgical SUI treatments.
Conclusion
SUI is a bothersome condition that negatively impacts the lives of millions of women. The lack of an effective medication for SUI has created a significant treatment gap for women who have failed treatment with behavioral interventions and physical therapy but who are reluctant to undergo invasive procedures or are not suitable candidates. Several new approaches may be employed by these patients, including the injection of bulking agents and treatment with a new transurethral collagen-denaturation treatment. Physicians should consider these options for patients who may not be suitable for, or may want to avoid, surgery. Most importantly, healthcare providers should communicate the full range of treatment options available to SUI patients at each stage of their care.
Future perspective
SUI will continue to be a medical problem experienced by approximately 20% of women during their lifetime, particularly as they age. Women who have tried and failed conservative nonsurgical treatments may desire or require a durable alternative to surgical treatment.
For women with ISD, bulking agents will remain an alternative to surgery. These agents are injected during a procedure performed in an office setting, and recovery time is minimal. Although the effectiveness of bulking agents typically declines over time, durability of response with the newer agents will be confirmed.
As transurethral collagen denaturation is also an office-based, nonsurgical procedure and is associated with a minimal recovery time, it could become an acceptable alternative to surgery for women with SUI due to urethral hypermobility. In addition, as transurethral collagen denaturation does not cause necrosis or stricture, surgery would not be precluded for women who might choose to pursue other treatment options in the future. A randomized, sham-controlled study is under way to expand the indication for transurethral collagen denaturation in patients who have undergone surgery for SUI and achieved suboptimal results. In addition, a current large, 3-year, prospective trial will further evaluate the durability of response with this procedure.
Footnotes
Acknowledgements
This manuscript was supported by Novasys Medical, Inc. Denise Elser has served as a consultant to Novasys Medical, Inc., and as a principal investigator for research involving Renessa®. The author has no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript, apart from those disclosed.
Writing assistance was provided by Peloton Advantage and funded by Novasys Medical. DME had editorial control over content and references.
