Abstract
Pre-eclampsia is a multisystem disorder of pregnancy, usually characterized by the appearance of high blood pressure and the excretion of protein in the urine of a previously healthy woman. Symptoms and signs vary in intensity from woman to woman; from a borderline rise in blood pressure, to convulsions (eclampsia), stroke and death. The disease remits following removal of the placenta and so the mainstay of current treatment is timely delivery. A pathophysiological framework of the disease has been established, beginning with failures in placental development, inducing oxidative stress and release of compounds that lead to endothelial activation, vasoconstriction and glomerular endotheliosis. A combination of epidemiological, biophysical and biochemical tests now allow most patients at-risk to be identified by midpregnancy, whilst minimizing false-positive prediction. It is hoped that earlier classification of patients at-risk of the disease, on the basis of pathophysiological changes, will enable specific therapies to be developed targeting these changes.
Keywords
Pre-eclampsia is a major complication of pregnancy characterized by proteinuria, increased vascular resistance and hypertension during the last trimester of pregnancy and is usually associated with edema and hyperuricemia. It occurs only in the presence of the placenta, even when there is no fetus, as in the hydatidiform mole, and remits postpartum. If untreated, eclampsia may occur, with seizures, severe hypertension and ultimately cerebrovascular accident and death. There are no satisfactory preventative treatments, although measures are available that can modify the severity of the condition (low-dose aspirin) and prevent eclamptic seizures (magnesium sulphate); early delivery remains the only successful treatment [1].
Pre-eclampsia is a major cause of maternal and fetal mortality and morbidity in both developed and developing countries. In the UK, hypertensive diseases of pregnancy rank second only to thromboembolic disease as cause of direct maternal deaths [2], whereas in developing countries they lie behind hemorrhage (25%) and infection (15%), equal third with unsafe abortion (13%) [3]. Pre-eclampsia is a direct cause of 3.9% of all stillbirths in the UK [4], and also leads to significant neonatal mortality and morbidity associated with severe prematurity and intrauterine growth restriction (IUGR).
Pre-eclampsia and eclampsia are associated with a poor fetal outcome owing to IUGR, intrauterine asphyxia and iatrogenic prematurity [5–7], whereas gestational (nonproteinuric) hypertension (GH) in general, is not [8,9]. Ferrazzani et al. [10] estimated that hypertensive disease complicated by significant proteinuria was associated with a perinatal mortality rate four-times higher than nonproteinuric hypertension. They also noted that IUGR occurred in 52% of cases with pre-eclampsia, compared with only 18% in GH and 12% in patients with chronic hypertension. Salafia et al. concluded from their study that the degree of proteinuria was the single most important determinant of intrauterine fetal nutritional deprivation [11]. However, this good prognosis of GH has been disputed by Brown and Buddie [12]. They found that although there was a higher prevalence of all maternal complications, and perinatal mortality was higher and birth-weight lower amongst patients with proteinuria (>1g/l), similar maternal complications still occurred in 4–13% of women without proteinuria.
Most maternal (80%) and fetal (65%) deaths attributed to pre-eclampsia are associated with substandard care, either:
Failure to identify and act on known risk factors at booking; or
Failure to recognize and act appropriately on signs and symptoms from 20 weeks’ gestation, where the level of care contributed significantly to the death, and where different management would reasonably have been expected to alter the outcome [13,14].
Pathogenesis
Pre-eclampsia is primarily a disease affecting nuliparous pregnant women, although multiparous women who have changed their sexual partner are also at risk [15]. Roberts and Hubel suggest a two-stage model of pre-eclampsia [16]:
“The initial stage is reduced placental perfusion due to abnormal implantation or other pathological disorders. The second stage is the production, by a maternal response to the reduced placental perfusion, of the maternal syndrome. The maternal response is influenced by maternal constitutional factors – genetic, behavioral or environmental.”
Pre-eclampsia is associated with characteristic lesions in the kidney known as glomerular capillary endotheliosis, which result in glomerular dysfunction and the development of proteinuria. Urinary protein excretion varies widely from 300 μg to 8–10 g/day in established pre-eclampsia. Mild endotheliosis may also exist in patients with GH without proteinuria, suggesting that some of these cases have an earlier or milder form of the same syndrome. Proteinuria usually accompanies or follows the development of hypertension and both disappear during the puerperium, along with the resolution of glomerular endotheliosis [17].
Placental hypoxia–reperfusion
In normal pregnancy the uteroplacental arteries are invaded by trophoblast and converted into dilated, inelastic tubes with no maternal vasomotor control. In abnormal pregnancies such as those with pre-eclampsia and some cases of IUGR, there is a defect in placental development (the cause of which remains elusive) that results in the maintenance of high uteroplacental vascular resistance [18]. This leads to placental hypoxia-reperfusion with the generation of excessive reactive oxygen species (ROS) and cytokines. ROS are inactivated either by circulating or tissue antioxidants until these defense systems are overwhelmed. Once this defense threshold is breached, chain reaction peroxidation of lipids and proteins (including DNA) occurs. This in turn leads to generalized endothelial activation (see Box 1) within the maternal vascular system, with subsequent increased or prolonged vasoconstriction [19,20]. Endothelial activation may already be present in women with diabetes mellitus or chronic hypertension. Other placental proteins may be involved in endothelial activation, inducing the release of vasoconstrictors such as endothelin, thromboxane and angiotensin II. These agents stimulate protein kinase C activity, causing enhanced muscle sensitivity to intracellular calcium ion concentration. In addition to endothelial cell activation, expression of the full clinical picture of pre-eclampsia requires the release of the cytokines TNF-α, IL-6 and IL-8 [21].
Endothelial activation
Endothelial activation: changes in cellular function in response to environmental stimuli, such as oxidants, radiation, cytokines, thrombin and bacterial endotoxins, which interact with cell-surface receptors.
F2-isoprostanes are produced by ROS-induced peroxidation of arachidonic acid in cell membranes and are considered the most reliable measure of oxidative stress in biological systems. When released into the circulation, F2-isoprostanes exert a potent vasoconstrictor action by stimulating thromboxane formation [22]. Physiological concentrations of 8-iso-prostag-landin F2α (8-iso-PGF2α), the most abundant F2-isoprostane, have been shown to stimulate cell proliferation, DNA synthesis and endothelin-1 mRNA and protein expression by endothelial cells [23]. In pregnancy, F2-isoprostanes are formed and secreted by the placenta and production is abnormally increased in the placentas of pre-eclamptic women [24]. These circulating factors also increase vascular permeability, and can affect sympathetic tone and the neuronal control of blood pressure [25]. Recently, a placental protein called soluble fms-like tyrosine kinase 1 (sFlt-1) has been shown to produce all the systemic manifestations of pre-eclampsia by antagonizing the vasodilatory effects of vascular endothelial growth factor (VEGF) and placental growth factor (PIGF) [26]: sFlt-1 production is upregulated by hypoxia and raised tenfold in patients with pre-eclampsia [27] and has been suggested as a biomarker for the subsequent development of pre-eclampsia [28]. Unfortunately, although a differential increase in serum levels of sFlt1 is seen at 20-weeks gestation, it does not become significant before the onset of the clinical disease as shown in Figure 1 [28].

Measurement of sFlt1 throughout pregnancy in pre-eclamptic and normotensive controls.
Nutrition
Patients with poor nutrition may be deficient in antioxidants such as vitamin C, vitamin E, folate and vitamin B complex, as well as in trace elements such as iron, zinc, magnesium, manganese and selenium [29], thus accelerating the disease process and enhancing the severity of the disease. Supplementation of diet by vitamins and minerals in high-risk patients may prove successful in modulating the severity of complications of pre-eclampsia, or at least delay the onset of the clinical syndrome, allowing longer fetal development before delivery becomes necessary [30]. Bodnar et al. observed that regular use of multivitamins in the periconceptional period was associated with a 45% reduction in pre-eclampsia risk [31]. However, their study was observational, not a randomized, controlled trial (RCT). At present, the results of preventative studies with vitamins A and E have been disappointing, although most suffer from methodological deficiencies related to patient selection and institution of treatment after the critical stage of placental development when a beneficial effect might be anticipated [32]. The recent Vitamins in Pregnancy (VIP) trial actually showed an increase in the number of low-birth-weight babies amongst women supplemented with vitamins A and E from 14 weeks of pregnancy and no difference in the incidence of pre-eclampsia [33], whereas multimicronutrient supplementation of undernourished women during pregnancy has been shown to reduce the incidence of IUGR [34].
Dietary calcium and magnesium deficiencies in particular have been related to a higher incidence of pre-eclampsia; recent studies demonstrate an inverse relationship between calcium intake and severity of disease [35]. The role of calcium in the pathogenesis of pre-eclampsia is obscure, but appears to be a secondary effect related to cell-membrane damage by lipid peroxidation. Calcium helps maintain vasodilation, so deficiency encourages vasoconstriction. Magnesium stabilizes vascular smooth muscles and helps regulate vascular tone.
Genetic inheritance & the immune system
Pre-eclampsia occurs in 26% of the daughters and 16% of the granddaughters of women with eclampsia [36]. Genetic and immunologic conflict between the mother and fetus may predispose a woman to develop pre-eclampsia. The risk of pre-eclampsia is increased with oocyte donation, where the fetus differs genetically from the mother [37]. The immune system has to be modified in pregnancy to allow the fetus to grow within the mother, despite having a different antigenic structure (partly paternal origin), which would normally result in rejection. In pre-eclampsia, there is evidence of activation of the maternal immune system (the immune system believes that blood vessels have been damaged) directed against elements of the placental trophoblast. In trying to repair the vascular damage, therefore, the immune system accelerates the disease process. Women with preeclampsia demonstrate a fivefold increase in circulating fetal DNA in comparison with controls, suggesting either increased fetal–maternal cell traffic or a defect in maternal antigen clearance [38]. Wimalasundera et al. recently noted that patients with HIV1-related immune deficiency demonstrated a low rate of pre-eclampsia, but that the rate in women treated with triple antiretroviral therapy was restored to the expected rate [39]. They concluded that ‘this indicates a pivotal role of the immune system in the pathogenesis of pre-eclampsia.’
Definition (diagnosis) of pre-eclampsia
The clinical picture of pre-eclampsia is variable, and no precise definitions are universally accepted for commonly used terms such as pregnancy-induced hypertension (PIH), GH or severe pre-eclampsia. Definitions used for research purposes provide strict criteria to ensure that only women with pre-eclampsia are included, whereas clinical management criteria seek to identify pregnant women at higher maternal and/or fetal risk [40]. For the purposes of this review the definitions used in the recently published pre-eclampsia community guideline (PRECOG) will be assumed (Table 1) [41]. These definitions are based on recommendations from Redman and Jefferies (1988) [42] and Davey and MacGillivray (1988) [43].
Definitions of terms used in the pre-eclampsia community guideline.

Precision of definitions
Even with these simple definitions errors can occur. Pregnancy itself causes changes in blood pressure that are gestation dependent. Therefore, a patient with borderline pre-existing hypertension may be classed as normotensive when first seen by an obstetrician in the first or second trimester, and subsequently be diagnosed as having PIH when her blood pressure rises to 140/90 mmHg or above, even if this is a ‘normal’ physiological level for her. Conversely, a patient with low baseline blood pressure may have a significant rise in blood pressure during pregnancy, reflecting pathological changes of GH or pre-eclampsia, but not be diagnosed as such because her highest readings fall short of the critical value of 140/90 mmHg. Low baseline levels are particularly common in women of slight build where nutrition is poor, such as in the Asian subcontinent.
As pregnancy usually occurs in young women who may not have visited a doctor or clinic in recent years, it is impossible to state with certainty that no hypertensive disorder pre-existed. The probability of pre-existing hypertensive disease, especially essential hypertension, increases with age. Thus, the older the pregnant woman is, the higher the chance of an error in diagnosis.
Problems with blood pressure measurement
The conventional definition of hypertension (diastolic blood pressure of > 90mmHg; systolic blood pressure > l40mmHg) is based on measurement in a resting state, sitting with 30 inclination using a mercury sphygmomanometer. During pregnancy, diastolic blood pressure cannot be reliably measured using the Phase 1V Korotkoff sound (when the sounds muffle) as recommended by WHO for the nonpregnant state: using Phase V (when sounds disappear) provides an estimate closer to true diastolic pressure during pregnancy [44] and is also more reproducible [45].
In recent years the use of automated blood pressure monitoring devices has largely replaced the mercury sphygmomanometer on hospital wards. Many of these devices systematically underestimate blood pressure during pregnancy, and particularly in those patients with preeclampsia. They should therefore be frequently recalibrated against measurements obtained with a mercury sphygmomanometer.
To date, only one automated device has been validated both for home monitoring in pregnancy and for pre-eclampsia; the Microlife® 3BTO monitor (Microlife AG, Switzerland) [46]. A few others have been validated for static measurement in pregnancy in the hospital/research setting (Spacelabs 90207 [47], Omron-MIT [48]).
Blood pressure measurement in pregnancy is influenced by gestation: blood pressure falls throughout early pregnancy until approximately 24 weeks of gestation, after which it begins to rise again, reaching prepregnant levels around term [49]. In addition, progressive weight gain throughout pregnancy may introduce errors in measurement if a larger sphygmomanometer cuff size is not used.
Despite the variability of blood pressure measurement during the second trimester of pregnancy, women who will ultimately develop preeclampsia do tend to have a higher blood pressure than those who will remain normotensive. Blood pressure measurement therefore remains the mainstay of clinical risk assessment as it is cheap and widely available.
Statistical problems
The ultimate aim of prediction is to identify a disease state before it endangers the patient and in time for effective treatment to be implemented. Because assays have to be performed before symptom onset, many more normal patients are recruited than for an RCT. Prediction research therefore involves greater expense than an RCT.
Research into predictors of pre-eclampsia takes two forms; the survey of large low-risk populations and the examination of selected high-risk populations. With the relative infrequency of unfavorable outcomes, the latter approach is obviously more cost effective from the researcher's point of view. However, it provides limited useful information as the patients recruited are already known to be at risk of the complication.
Owing to the relative infrequency of preeclampsia, there is a tendency for researchers to aggregate cases of pre-eclampsia, GH and even IUGR into one group so as to increase the numbers with an unfavorable (pathological) outcome. IUGR cases are included on the basis that this condition is also associated with placental hypoxia and disturbed endovascular trophoblast invasion. Unfortunately, small babies are also caused by fetal genetic defects and even hereditary factors (small women tend to have small babies); therefore, these individuals will not be predicted by any factor related to poor placental perfusion. This dilution of the outcome group with ‘normal’ cases obviously adversely affects the statistical performance of any test (predictor) by reducing the sensitivity.
Massive physiological changes occur between conception and midpregnancy, many of which begin to reverse in later pregnancy. This means that biophysical and biochemical measurements performed in early pregnancy will vary considerably depending on the exact gestational age – that is, a particular measurement may be normal at 10 weeks but outside the normal range for 20 weeks, or vice versa. It also means that the aggregation of data from women at different gestations, say 18, 19, 20, 21 and 22 weeks, may introduce a problem in selecting a cutoff value if significant variation occurs even in normal pregnancy. This problem can be resolved by taking a multiple of median (MoM) approach to prediction, as used in Down's syndrome screening [50]. However, this involves considerable expense as gestational age assessment must be precise and is only applicable if median values are well defined in a large population. It is only applicable therefore once a reliable predictor has been identified.
Identification of patients at risk, or in the early stage of disease, may prompt obstetric intervention, thus either modifying the severity of disease or effecting delivery before the full clinical picture develops. This will increase the false-positive rate of the test and reduce its apparent specificity. Treatment that may also affect the relationship between predictor and outcome is known as treatment paradox [51]. Random events (preterm labor) or interventions based on other factors may cause similar miss-classifications if delivery occurs before the full clinical picture develops. Higgins and de Swiet have argued that [4]:
“Given the current high expectations for the outcome of pregnancy, the definition (of pre-eclampsia) should be as all-encompassing as practical, even if it has a high false-positive rate i.e., women will be included where the excess risk is small, if there is any at all”
This argument is acceptable until an effective treatment becomes available. If the treatment poses any risk to the patient, or has unpleasant side effects, its use in large numbers of women who are not really at risk (false positives) will be unacceptable. For example, low-dose aspirin has been shown to have some preventative effect on development of pre-eclampsia (approximately a 10% reduction) [52]. However, aspirin may also increase the incidence of postpartum hemorrhage, so treating the 50% of the pregnant population identified as at risk on epidemiological grounds may not be a realistic option. Under these circumstances the ability of the test to identify those not at risk (specificity) also becomes an important factor.
The appropriate statistics for reports on prediction are the sensitivity and specificity of the predictor for the disease, as these are unaffected by the prevalence of the disease in the sample tested (unlike positive and negative predictive values). Comparisons between different predictors can therefore be made even if research has been performed on independent datasets with variable disease prevalence. Comparisons can also be made between predictive performance whether subjects are preselected on the basis of risk factors or not, provided the predictor and the risk factor are independent of one another (otherwise bias may be introduced).
Where a continuous variable is used in prediction, sensitivity and specificity bear an inverse relationship to one another depending on the cutoff used to define positive and negative tests. This relationship can be plotted for any variable over the entire range of possible cutoff values, generating a receiver operating characteristic (ROC) curve for the test. The ROC curve can be used to identify the optimum cutoff value, or the area under ROC curves (AUC) can be used to compare the predictive performances of different tests. Point estimates of sensitivity and specificity (optimum performance) are usually quoted by researchers rather than the AUC, which can lead to erroneous conclusions, particularly where small numbers of pre-eclampsia patients have been studied. The alternative of quoting sensitivity at a fixed level of specificity (usually 95:5% false-positive rate) results in huge variation in reported results, as this is invariably the steepest part of the ROC curve for a good test (Figure 2). This also assumes, incorrectly, that specificity is more important than sensitivity: a more useful approach to obstetric prediction is to maximize sensitivity as suggested by Higgins and de Swiet [4] and then look for ways of improving precision by reducing the false-positive prediction rate.

Receiver operating characteristic curve for second trimester mean arterial pressure predicting pre-eclampsia.
Current status of prediction of pre-eclampsia
The majority of women at risk can, in fact, already be identified on epidemiological grounds (primigravidity/primipaternity; very young or very old; obesity, pre-existing medical disorders such as renal disease, diabetes or hypertension; poor or malnourished, multiple pregnancy). There has been an extensive search in recent years for better biophysical and biochemical predictors of pre-eclampsia.
Biophysical predictors
The increased availability of ultrasound in recent years has prompted numerous investigations into the vascular changes in early pregnancy. As preeclampsia is thought to occur as a consequence of poor trophoblast invasion of the maternal vascular system, leading to inadequate perfusion of the placenta, this would appear a logical start in the search for an early predictor. Pregnancies destined to develop pre-eclampsia have raised placental vascular resistance, which can be detected with uterine artery Doppler ultrasound. This increased resistance often results in a characteristic ‘notching’ of the vessel's waveform as pregnancy advances. Doppler studies of low-risk women typically report sensitivities for predicting preeclampsia in the region of 50–70% for specificities between 80 and 90%. The presence of bilateral notches offers the most accurate prediction (compared with resistance or pulsatility indices) [53]. However, when compared against the ROC curve for mean arterial blood pressure measurement (Figure 2) [54] (under research conditions), Doppler ultrasound does not appear to have any predictive advantage. Combined results for bilateral notching from Conde-Agudelo's systematic review [40] and also results from the large study by Yu et al. [55], in which Doppler measurements were integrated with maternal factors in a logistic regression model, are superimposed on Figure 2. The sensitivity of uterine artery Doppler for other adverse pregnancy outcomes such as IUGR is lower than for pre-eclampsia, as a half to a third of IUGR cases are not associated with placental vascular abnormalities [56].
Biochemical predictors
Biophysical measurements tend to only identify crude changes, which reflect an already advanced disease process. Therefore, there has been more concentration in recent years on identifying more subtle biochemical changes that occur at much earlier stages of the disease process and also genetic and proteomic changes that are related to disease pathogenesis. From the previous discussion on pathogenesis, it is probable that evaluation of oxidative stress will provide significant improvements in our ability to predict GH and pre-eclampsia.
We have recently evaluated fasting plasma 8-iso-PGF2α (produced by ROS conversion of arachidonic acid, in situ, in cell membranes), measured midtrimester as a predictor of preeclampsia. Figure 3 shows the ROC curve for this analysis and confirms that 100% sensitivity was attained at a reasonably high level of specificity (78.5%). The Wilcoxon estimate of area under this ROC curve was 0.94 (DeLong standard error = 0.02: 95% CI = 0.90–0.98). Others have obtained similar predictive accuracy with less specific markers of oxidative stress such as malondyaldehyde (MDA), but only in measurements taken in the third trimester [57]. Uterine hypoxia also induces a rapid and sustained elevation of fetal activin A levels [58]: Emine et al. studied serum measurements of activin A and inhibin A at 16–18 weeks of gestation and found AUC of 0.94 and 0.87, respectively. As can be seen from Figure 3, the point values for activin A taken from this paper correspond almost exactly with our ROC curve for F2-isoprostane, with 100% sensitivity achieved at approximately 80% specificity. Spencer et al., sampling slightly later between 20 and 22 weeks of gestation, achieved 44% sensitivity for activin A at 95% specificity, which again lies on the same curve [59].
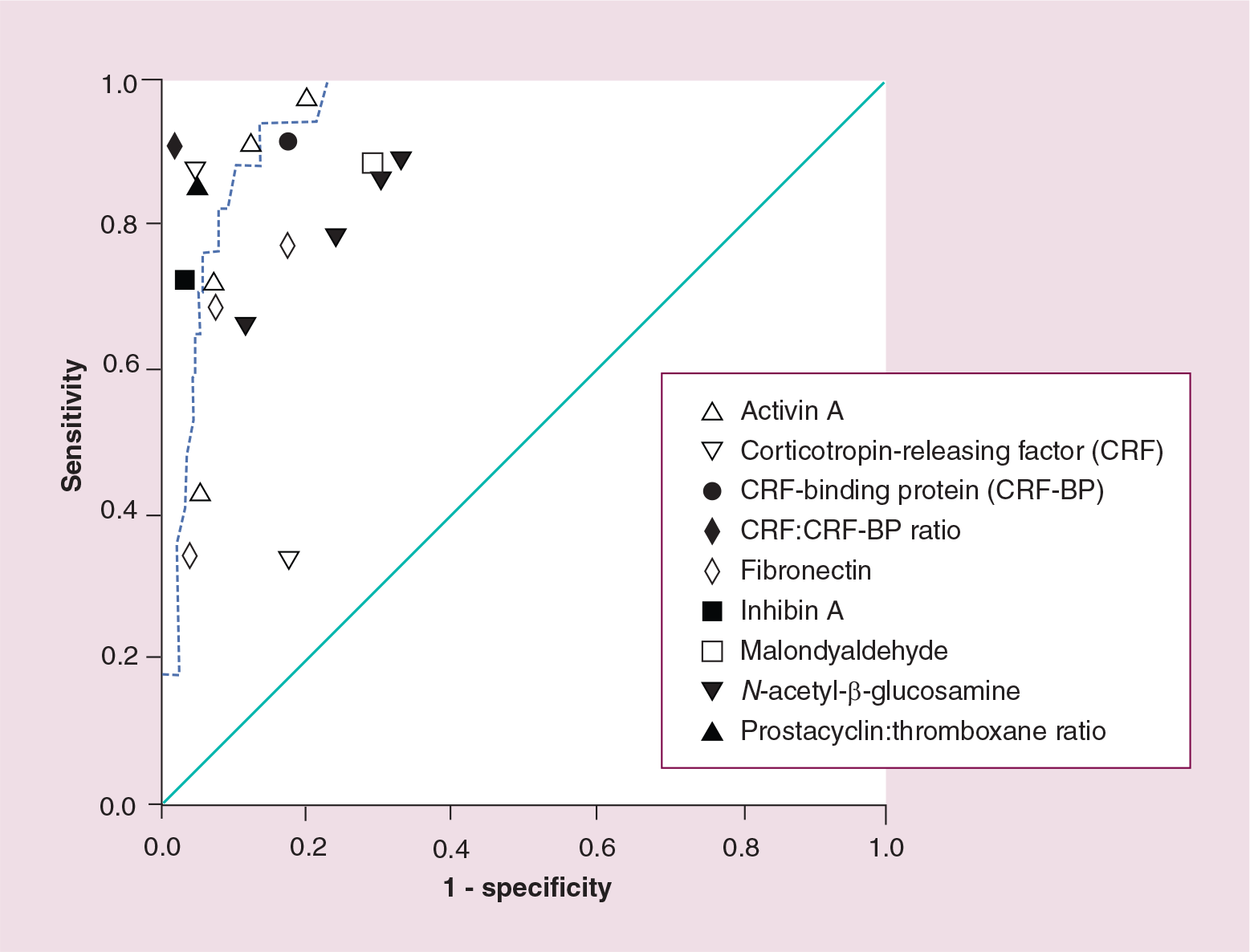
Receiver operating characteristic curve for 8-iso-prostaglandinf2α predicting pre-eclampsia.
Chavarría et al. monitored the prostacyclin/thromboxane ratio serially throughout pregnancy and noted that a ratio of less than 3 between 22 and 26 weeks of pregnancy gave a sensitivity of 87% with specificity of 97% [60]. In another study, Chavarría assessed the predictive value of serum fibronectin measurements, reporting sensitivities of 73% with 87% specificity and of 70% at specificity of 95% [61]. Dreyfus et al. found fibronectin had a sensitivity of 37.5% with specificity of 96.6%, 3–4 weeks before the onset of symptoms [62].
Florio et al. measured two placental neurohormones; corticotropin-releasing factor (CRF) and CRF-binding protein (CRF-BP) between 28 and 29 weeks of gestation [63]. Individually these compounds had sensitivities of 94.8 and 92.5% with specificities of 96.9 and 82.5% for prediction of pre-eclampsia; when both CRF and CRF-BP levels were raised, a sensitivity of 93.75% was achieved with 100% specificity if both were within the normal range. However, our own study of CRF in midpregnancy showed a sensitivity of only 38.1% with specificity of 79.0% for predicting pre-eclampsia [64].
Combination of biochemical & biophysical predictors
Ay et al. analyzed the predictive power of various combinations of maternal serum inhibin A, activin A, hCG, estriol, α-fetoprotein levels and uterine artery Doppler for pre-eclampsia in the second trimester of pregnancy. Inhibin A, activin A and uterine artery Doppler were the best predictors of pre-eclampsia. However, addition of the hormonal markers to the Doppler results only slightly improved the predictive efficacy, and the improvement appears to be clinically insignificant [65].
Early pregnancy studies
It can be argued, particularly in light of the poor results of recent antioxidant trials, that if interventions to prevent the syndrome of preeclampsia need to start in the first trimester we should also be looking for biochemical predictors in the first trimester. Several markers being investigated as part of antenatal screening for congenital abnormalities have also been evaluated as predictors of late pregnancy complications including pre-eclampsia and IUGR. These include serum sex hormone-binding globulin (SHBG), placental protein 13 (PP13), pregnancy-associated placental protein A (PAPP-A), proangiogenic factors such as VEGF and PlGF, and antiangiogenic factors such as sFlt-1 and endoglin. First trimester serum SHBG concentrations have no predictive value for pre-eclampsia, GH, IUGR or preterm delivery [66]. Nikolaides et al. have reported a sensitivity of 90% with 6% false-positive rate for predicting pre-eclampsia requiring delivery before 34 weeks using a two-stage screening procedure of serum PP13 followed by Doppler study of the 14% considered at highest risk [67]. Unfortunately, PP13 prediction of term severe pre-eclampsia is poor, with only 29% sensitivity at 80% specificity compared with 85% for early pre-eclampsia and 50% for the two combined [68]. Spencer et al. [69] have reported improvement in prediction of preeclampsia when first trimester PAPP-A concentrations are combined with second trimester Doppler flow studies, achieving sensitivity of 62.1% at 95% specificity. However, the sensitivity of first trimester PAPP-A alone was only 14.1%. Rana et al. have investigated the antiangiogenic factors sFlt-1 and soluble endoglin in the first and early second trimesters [70]. They noted that whilst there were no significant differences in the first trimester (as with Levine's study: Figure 1), both factors were significantly elevated in specimens obtained between 17 and 20 weeks in patients who subsequently developed pre-eclampsia. The effect was particularly marked in those who developed early pre-eclampsia. Unfortunately, sensitivity and specificity of the predictions are not available from this study.
Conclusion
It is apparent from reading the obstetric literature from the past decade that most researchers start out with a rather pessimistic outlook that:
We do not know the cause of pre-eclampsia;
We cannot predict patients who will develop the disease in advance of clinical signs;
We do not have any effective treatment even if we could predict patients.
When they confirm that their particular predictor cannot identify all cases of pre-eclampsia (i.e., with a sensitivity of 100%) they conclude it is not worth performing. This is interesting as the highest (and most recent) reported predictive accuracy of prenatal screening for Down's syndrome based on the optimal cutoff risk from the ROC curve analysis achieved a sensitivity of 90% with a 17.8% false-positive rate and an AUC of 0.92 [71]. This is almost identical to the results of second trimester activin A, and F2-isoprostane reported here for predicting preeclampsia. Research into Down's syndrome screening has been pursued vigorously (and at great cost) in developed countries over the last 20 years, despite considerably poorer predictive accuracy than can now be achieved. Why, therefore, have we not devoted similar effort to identifying the 2–5% of women who face a significant risk of dying during pregnancy and childbirth from eclampsia or other complications of this disease?
Executive summary
Pre-eclampsia is a major complication of pregnancy, which only occurs in the presence of placental tissue. If untreated, seizures (eclampsia) may occur leading ultimately to cerebrovascular accident and death. At present there are no preventative treatments, although low-dose aspirin can modify the severity of the disease and magnesium sulphate can prevent seizures: early delivery remains the only successful treatment.
Reduced placental perfusion owing to abnormal implantation leads to oxidative stress, which in turn causes systemic activation of the endothelial system resulting in vasoconstriction and hypertension. Glomerular endotheliosis leads to renal dysfunction which presents as proteinuria. The maternal response is influenced by genetic, behavioral and environmental factors.
The clinical picture of pre-eclampsia is variable both in time of presentation and in severity. Definitions used for research purposes provide strict criteria to ensure that only women with pre-eclampsia are included, whereas clinical management criteria seek to identify pregnant women at higher maternal and/or fetal risk.
The conventional definition of hypertension (diastolic blood pressure of >90 mmHg; systolic blood pressure >140 mmHg) is based on measurement using a mercury sphygmomanometer. Automated blood pressure monitoring devices may systematically underestimate blood pressure during pregnancy and particularly in patients with pre-eclampsia. To date, only one automated device (out of hundreds on the market) has been validated both in pregnancy and in patients with pre-eclampsia.
Blood pressure falls throughout early pregnancy until about 24 weeks of gestation, after which it begins to rise again, reaching prepregnant levels around term. Patients with mild pre-existing hypertension may therefore be normotensive at antenatal booking, but pass the diagnostic threshold for gestational hypertension as they approach term.
Massive physiological changes occur between conception and midpregnancy, many of which begin to reverse in later pregnancy. Biophysical and biochemical measurements performed in early pregnancy vary considerably depending on the exact gestational age. Resolving this problem using a multiple of median approach involves considerable expense as median values need to be well defined in a large population with precise gestational age assessment. Early detection may prompt obstetric intervention; either modifying the severity of disease or effecting delivery before the full clinical picture develops. This diminishes the relationship between predictor and outcome (treatment paradox).
Most patients who will develop pre-eclampsia can be identified either by epidemiological, biophysical or biochemical markers. However, wide physiological variability of these markers leads to high false-positive predictions. Some improvements in predictive accuracy can be obtained by statistical modeling; combining these three factors. The timing of testing is critical to the disease being predicted. First trimester screening is unlikely to give useful predictions of term onset pre-eclampsia, but can achieve excellent results for the more serious forms that require preterm delivery. Late onset disease can be predicted more accurately by second trimester measurements.
With our current understanding of the disease processes involved in pre-eclampsia and the application of combinations of epidemiological, biophysical and biochemical tests it is now possible to identify a reasonably small high-risk subset of the pregnant population for preventative and or therapeutic trials to begin, targeting different pathophysiological processes.
Future perspective
Our understanding of the pathogenesis of preeclampsia may not be complete but it is sufficient to provide a framework for setting up predictive models. We can identify the majority (95–100%) of cases by lowering cutoff values for whichever test (epidemiological, biophysical or biochemical) is chosen. The consequence of this is an increasing false-positive rate. However, multifactorial prediction models combining two, or all three, elements are able to decrease false-positive rates to acceptable levels. Identification of most cases at risk of preeclampsia is achievable by midpregnancy, (22–26 weeks), well before clinical signs appear. Those cases prone to develop early pre-eclampsia can often be detected at an earlier stage of pregnancy as their pathology is more advanced. Currently available therapies such as low-dose aspirin and antioxidants, which appear to at least have some efficacy in moderating disease severity, can therefore be rationally applied to high-risk subgroups of patients, minimizing treatment of patients with a false-positive test. Future treatments may be targeted at specific biochemical abnormalities responsible for maternal morbidity such as cytokines and the antiangiogenic factors sFlt-1 and endoglin, or at the processes that stimulate their release. The timing of these therapies will depend on the particular prediction model adopted and which presentation of the pre-eclamptic disease spectrum is being targeted.
Footnotes
The authors have no relevant financial interests including employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties related to this manuscript.
