Abstract
A combined oral contraceptive pill containing 20 μg of ethinyl estradiol and 3 mg of the progestin drospirenone in a novel dose regimen (24 active pills followed by 4 placebo pills), has demonstrated efficacy for the symptoms of premenstrual dysphoric disorder, a severe form of premenstrual syndrome, with an emphasis on the affective symptoms. Drospirenone has progestagenic, anti-androgenic and anti-aldosterone properties, which differ from earlier generations of progestins, and reducing the hormone pill-free interval allows for better suppression of ovarian steroid production.
Premenstrual disorders, including premenstrual syndrome (PMS) and premenstrual dysphoric disorder (PMDD), are cyclic luteal-phase events that encompass physical, psychological and behavioral symptoms. For PMS, these symptoms range from moderate to severe [1–4]. PMDD, by definition, is a severe form of PMS characterized as a distinct affective disorder that causes social and/or occupational impairment [5,6]. It has been estimated that 20–40% of ovulatory women have symptoms that can be classified as PMS [7], whereas 3–8% of reproductive women have symptoms that meet criteria for PMDD [6].
The criteria for both PMS and PMDD stipulate that symptoms must be present during the 5 days prior to menses, resolve by the end of menses, occur for at least 2–3 consecutive menstrual cycles, and not be an exacerbation of an underlying medical or psychiatric disorder [8,9]. The diagnosis of PMDD is outlined in the Diagnostic and Statistical Manual for Mental Disorders (Box 1) [8]. Since there is no specific assay to diagnose these disorders, prospective daily rating by the patient is imperative in the diagnostic process. There is currently no general accepted consensus regarding the diagnostic criteria for PMS other than those of the American College of Obstetricians and Gynecologists (ACOG), although these criteria were generally reaffirmed by a recent expert panel convened to refine the criteria for the diagnosis of PMS [10]. ACOG criteria require one or more bothersome affective or somatic symptoms, including irritability, depressed mood, angry outbursts, anxiety, confusion, social withdrawal, breast tenderness, abdominal bloating, headache and swelling of extremities, which interfere with social or occupational domains [9].
PMS and PMDD share a complex psychoneuroendocrine etiology hypothesized to involve alterations in neurotransmitter systems and neuropeptides, including but not limited to the serotonin, γ-amino butyric acid (GABA), and endogenous opioids [11–13]. However, clinically, the symptoms are triggered by the rise and fall of estrogen and progesterone related to ovulation in genetically or otherwise predisposed individuals. PMS-like symptoms can also be evoked by exogenous steroid (estradiol or progesterone) administration in women with PMS, but not control women [14].
An obvious therapeutic approach to reliably produce anovulation is the combined estrogen/progestin-containing contraceptive pill (COC). COCs have long been indicated as a safe, reversible form of contraception, but the evolution of the pills' active ingredients has allowed them to be considered for the treatment of cycle-related disorders such as PMS and PMDD. The new class of COCs that has been recently shown to be effective for treating PMDD contains a low dose of ethinyl estradiol and a new progestin called drospirenone in a novel dose regimen [15,16]. Drospirenone is related to the diuretic spironolactone and has anti-androgenic and anti-aldosterone properties. Shortening the pill-free interval from 7 to 4 days provides better suppression of endogenous steroid production.
Overview of the market
Current pharmacological options for treatment of PMDD are limited and no specific pharmacological treatments have been expressly indicated for PMS. Serotonergic agents such as selective serotonin reuptake inhibitors (SSRIs), serotonin/norepinepherine reuptake inhibitors (SNRIs) and serotonergic tricyclics have been shown to be effective in reducing PMS/PMDD symptoms by at least 50% in over 60% of study participants in many randomized, controlled trials. The three SSRIs that are approved by the US FDA for PMDD include: fluoxetine, sertraline and paroxetine controlled-release [17–19]. These three SSRIs are approved for both daily (continuous) and luteal-phase (intermittent) dosing. One smaller placebo-controlled study of the SNRI venlafaxine has also shown efficacy [20].
Diagnostic and Statistical Manual IV Diagnostic Criteria for Premenstrual Dysphoric Disorder.
In most menstrual cycles during the past year, five (or more) of the following symptoms were present for most of the time during the last week of the luteal phase, began to remit within a few days after the onset of the follicular phase, and were absent in the week postmenses, with at least one of the symptoms being either (1), (2), (3) or (4):
Markedly depressed mood, feelings of hopelessness, or self-deprecating thoughts
Marked anxiety, tension, feelings of being ‘keyed up’ or ‘on edge’
Marked affective lability (e.g., feeling suddenly sad or tearful or increased sensitivity to rejection)
Persistent and marked anger or irritability or increased interpersonal conflicts
Decreased interest in usual activities (e.g., work, school, friends, hobbies)
Subjective sense of difficulty in concentrating
Lethargy, easy fatigability or marked lack of energy
Marked change in appetite, overeating or specific food cravings
Hypersomnia or insomnia
A subjective sense of being overwhelmed or out of control
Other physical symptoms, such as breast tenderness or swelling, headaches, joint or muscle pain, a sensation of ‘bloating’ or weight gain
The disturbance markedly interferes with work or school or with usual social activities and relationships with others (e.g., avoidance of social activities, decreased productivity and efficiency at work or school).
The disturbance is not merely an exacerbation of the symptoms of another disorder, such as major depressive disorder, panic disorder, dysthymic disorder or a personality disorder (although it may be superimposed on any of these disorders).
Criteria
NOTE: In menstruating females, the luteal phase corresponds to the period between ovulation and the onset of menses, and the follicular phase begins with menses. In non-menstruating females (e.g., those who have had a hysterectomy), the timing of luteal and follicular phases may require measurement of circulating reproductive hormones.
Reprinted with permission from the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision ©2000, American Psychiatric Association.
Although these medications reduce symptoms, patient compliance must be considered when evaluating treatment. Intermittent (luteal-phase) administration may improve compliance [21–23]. SSRIs improve physical symptoms as well as psychological and behavioral symptoms, although higher doses may be required to relieve physical symptoms [24,25]. A Cochrane review of SSRIs as treatment for premenstrual disorders revealed that patients taking SSRIs are 2.5-times more likely than women using placebo to withdraw from a study due to side effects [26], especially sexual dysfunction and weight gain [27,28]. The most common side effects of SSRIs when used to treat PMDD included anxiety, insomnia and nausea. In addition, many women never fill the prescription due to the stigma of taking a psychiatric medication or fear of addiction. Another study found a discontinuation rate of over 60% after 2 years [29].
Other treatment options studied for PMS include the diuretic spironolactone, an aldosterone receptor antagonist that demonstrated efficacy in two small studies. Spironolactone given for 7 days prior to menses reduced the occurrence of PMS symptoms in 80% of cycles [30]. A crossover study compared luteal-phase spironolactone with placebo and concluded spironolactone significantly improved depression, breast pain, swelling and food craving [31].
Alprazolam 0.5–2 mg up to three-times daily during the luteal phase has had conflicting results for PMS in three small studies, with only two of the three studies demonstrating efficacy [32–34]. The side effects, including drowsiness, reduced cognitive abilities and the risk of addiction, limit the utility of alprazolam. Alprazolam should only be given in the luteal phase to avoid addiction, and the dose should be tapered during the first few days of menses to avoid symptoms of withdrawal.
Gonadotropin-releasing hormone (GnRH) agonists create a ‘chemical menopause’ by decreasing pulsatile GnRH release and thereby inhibiting follicle-stimulating hormone (FSH) and luteinizing hormone (LH), resulting in suppression of ovarian estradiol and progesterone production. Medical oophorectomy (utilizing GnRH analogs) [35–38] or surgical oophorectomy [39,40] is effective but costly and is associated with long-term cardiovascular and osteoporosis risks, even with appropriate estrogen (in the case of hysterectomy with oophorectomy), low-dose GnRH analog or estrogen/progesterone hormone add-back [41,42].
COCs suppress ovulation, the hormonal event that interacts with the neurotransmitter alterations, and should be effective for PMDD, but most studies have not demonstrated efficacy. A study of over 600 women who had used different COCs for at least 3 months found little change in premenstrual symptoms, and in 16.3% of the women there was a worsening of their mood, although approximately 12% did report improvement [43]. Many women on COCs have bothersome physical and emotional symptoms during the 3 weeks of hormone exposure and others have symptoms during the week off active drug, just before menstruation. The mechanism underlying these symptoms remains unclear, but it has been suggested that PMS-like symptoms may accrue from:
Adverse central and/or peripheral effects of the hormonal constituents of the COCs, including but not limited to somatic symptoms from fluid retention mediated by ethinyl estradiol acting on the renin–angiotensin–aldosterone system (RAAS);
Exposure to and/or withdrawal from endogenous sex steroids, in predisposed women;
Exposure to and/or withdrawal from the exogenous steroids comprising the COC, in predisposed women [44].
COCs comprised of progestins other than drospirenone do not have antimineralocorticoid properties, so the estrogen effects predominate and could lead to symptoms of bloating and breast tenderness. Drospirenone is a novel progestin derived from 17α-spirolactone with antiandrogenic and antimineralocorticoid activity similar to the spirolactone derivative, spironolactone. An oral contraceptive pill containing drospirenone and ethinyl estradiol has been studied in two different formulations for PMDD. Drospirenone 3 mg/ethinyl estradiol 30 μg in a 21/7 dosing strategy showed significant reductions in premenstrual symptoms in an open-label, case–control trials [45,46]. However, a double-blind, placebo-controlled trial of women with PMDD demonstrated statistically significant differences for the symptoms of increased appetite, acne and food cravings [47].
Recently, a COC containing ethinyl estradiol 20 μg and drospirenone 3 mg in a novel dosing regimen consisting of 24 active pills followed by a shortened hormone-free interval of only 4 days (24/4; Yaz®) has been shown to significantly reduce symptoms of PMDD in two large randomized, controlled trials [15,16] and received FDA approval for the treatment of the emotional and physical symptoms of PMDD in women who desire hormonal contraceptive for birth control. The ethinyl estradiol/drospirenone 24/4 COC appears to be as effective as the class of SSRIs [15,16,48]; however, no head-to-head trials exist.
Introduction to the ethinyl estradiol 20 μg/drospirenone 3 mg
The 24/4 regimen COC with the indication for the treatment of symptoms in women with PMDD contains the steroid hormones drospirenone 3 mg/ethinyl estradiol 20 μg within a β-cyclodetrin (betadex) clathrate. This improves the stability and thus prolongs the shelf life of the hormonal formulation [49].
For the chemical structure of the compound, see Figure 1.

The chemical structures of drospirenone, ethinyl estradiol and betadex.
Pharmacodynamics
Similar to other COCs, COCs containing ethinyl estradiol/drospirenone prevent pregnancy via inhibition of ovulation. The inhibition of GnRH secretion from the hypothalamus in turn prevents the release of FSH and the maturation of the ovarian follicle. The progestin also prevents the LH surge, which is typically responsible for ovulation [50,51].
The estrogenic component used in this COC, ethinyl estradiol 20 μg, is lower than the 30–35 μg dose used in many formulations [101]. Drospirenone, an analog of spirolactone, has both antimineralocorticoid and anti-androgenic properties, unlike the earlier generations of COCs formulated with the 19-nortestosterone derived progestins that potentially have a range of androgen-like effects [52–55].
COCs containing drospirenone/ethinyl estradiol are marketed in the USA in two different regimens: Yaz® contains 24 active pills of drospirenone 3 mg/ethinyl estradiol 20 μg and four hormone-free pills and Yasmin® contains 21 active pills of drospirenone 3 mg/ethinyl estradiol 30 μg accompanied by seven hormone-free pills. The Pearl Index, a measure of contraceptive efficacy, is commensurate with that of other COCs [56], but only the 24/4 regimen has the indication for PMDD. The novel regimen with the shortened pill-free interval may be at least in part responsible for the efficacy for PMDD. There is an increase in physical premenstrual symptoms during the hormone-free interval in women receiving a 21/7 COC regimen [57]. Since the COC inhibits ovulation, the symptoms presented during the hormone-free interval may be due to failure of complete ovarian suppression. Reducing the hormone-free interval results in a more complete suppression of FSH and estradiol [57–59].
Pharmacokinetics & metabolism
The ethinyl estradiol and drospirenone information that follows was obtained from studies of the combination ethinyl estradiol 20 μg/drospirenone 3 mg 24/4 formulation. The ethinyl estradiol in single combination tablet has a bioavailability of 40% due to presystemic conjugation and first-pass metabolism. Upon oral administration, ethinyl estradiol undergoes rapid absorption; peak serum levels are found 1–2 h postconsumption, with little daily variation. Although ethinyl estradiol does not bind sex hormone-binding globulin (SHBG), a dose of ethinyl estradiol 20–30 μg is responsible for doubling the levels of SHBG; this is not affected by drospirenone. The ethinyl estradiol in this COC is 98.5% bound to serum albumin and is rapidly metabolized. The excreted metabolites are seen as both glucuronide and sulfate conjugates in urine and feces and undergo enterohepatic circulation [60].
Drospirenone is also rapidly absorbed following oral administration. Serum drospirenone concentrations are linearly related to the amount of drospirenone in a single oral dose with a mean bioavailability of 76%. Daily administration of a drospirenone 3 mg/ethinyl estradiol 20 μg tablet reaches steady-state conditions after 8 days [49].
Circulating drospirenone binds serum proteins to an extent of nearly 98.5%, but does not bind SHBG or corticosteroid-binding globulin (CBG) [49,60,61]. Unlike drospirenone, other progestins such as norethindrone, levonorgestrel, desogestrel and gestodene all have binding affinities to SHBG, resulting in less availability of SHBG for androgen binding [62].
Drospirenone binds to aldosterone receptors in the kidney, blocking the effects of aldosterone and resulting in moderately increased sodium and water excretion (Figure 2) [53,54,61,63,64]. Drospirenone also binds the androgen receptor (AR) in peripheral tissues, blocking the effects of testosterone (Figure 3) [53,54,61,64].
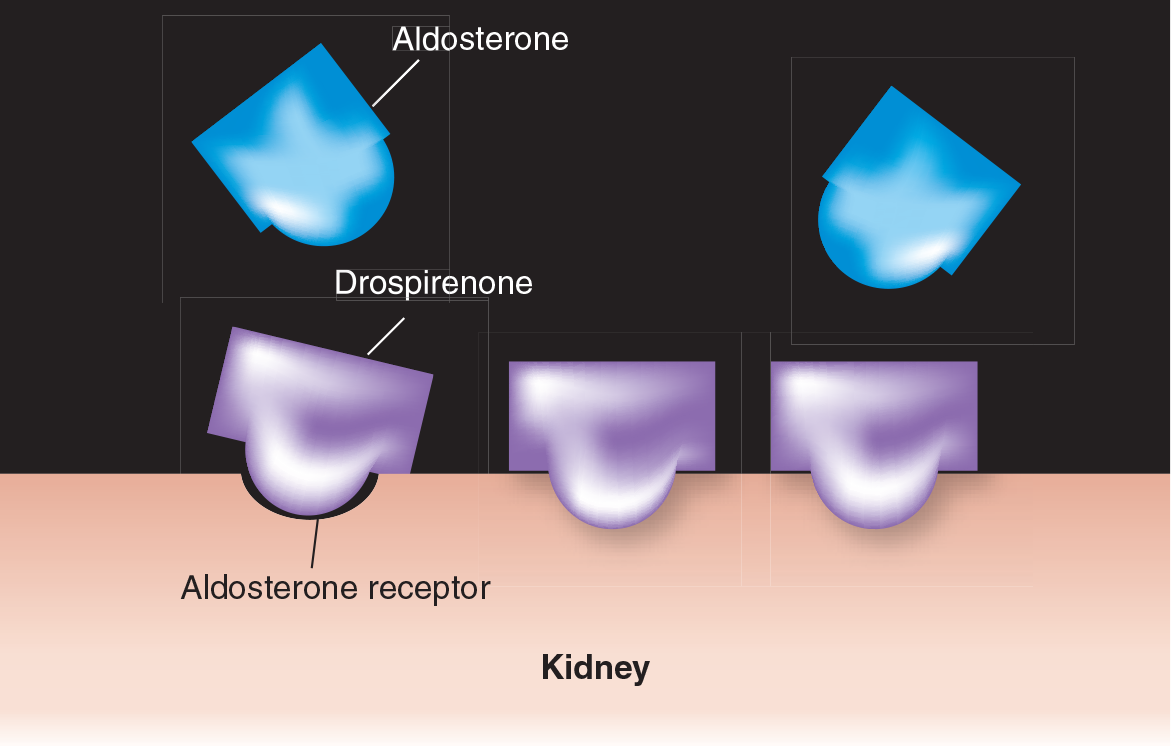
Antimineralocorticoid effect of drospirenone.

Anti-androgenic effect of drospirenone.
Drospirenone is metabolized in the liver to the acid conjugate and 4,5-dihydrodrospirenone-3-sulfate, which are formed independently of the cytochrome P450 system. These main plasma metabolites are considered pharmacologically inactive and excreted in urine or feces, with almost complete excretion occurring 10 days after administration of single and multiple dose regimens. Plasma levels decline biphasically with a plasma distribution phase half-life of 2 h and a terminal disposition half-life of 30 h [49].
The rate of systemic absorption of the combination drospirenone/ethinyl estradiol tablet was shown to be affected by food consumption of a high-fat meal when compared with fasting conditions. The Cmax values of both drospirenone and ethinyl estradiol decreased by approximately 40% after a high-fat meal, but the effect was not considered clinical relevant. Therefore, fasting conditions are not necessary for administration [49,60].
Clinical efficacy
Animal studies
The activity of drospirenone has been analyzed in animal models. Like progesterone, drospirenone was found to efficiently promote the maintenance of pregnancy and inhibit ovulation in rats. Antimineralocorticoid and long-lasting natiuretic activity, as well as anti-androgenic activity of drospirenone was also demonstrated in the rat. Drospirenone did not display androgenic, estrogenic, glucocorticoid or antiglucocorticoid activity. Possible interactions between drospirenone and ethinyl estradiol were also studied. Ethinyl estradiol did not interfere with the progestogenic or antimineralocortocoid activity of drospirenone [64].
The affinity of drospirenone to steroid receptors was studied in the male rat model [53]. The results of steroid-binding assays obtained with drospirenone were compared with the same assays for the natural steroid hormone progesterone. Drospirenone binds the progesterone receptor (PR) and mineralocorticoid receptor (MR) with high affinity. Drospirenone binds the AR and glucocorticoid receptor (GR) with low affinity, while progesterone showed greater binding affinity to GR; neither progesterone nor drospirenone binds to the estrogen receptor (ER). Drospirenone also displayed anti-androgenic activity by the inhibition of transcription events mediated by the activation of MR. Drospirenone displayed antimineralocorticoid activity [53].
Clinical studies
Clinical studies conclude that drospirenone is effective for ovulation suppression, manifests anti-androgenic activity with favorable outcomes regarding blood pressure, body weight, glucose tolerance, lipid metabolism, acne, hirsutism, seborrhea and antiproliferation of the human endometrium [65–67].
In an open-label, multicenter study, 1018 women evaluated during 13 treatment cycles on drospirenone/ethinyl estradiol 20 μg 24/4, showed no statistically significant changes in weight or systolic or diastolic blood pressure [68]. In an open-label study of drospirenone/ethinyl estradiol 30 μg 21/7, blood pressure remained within normal limits and no significant changes from baseline occurred in either systolic or diastolic pressure. Lipids also remained within normal but total cholesterol, triglycerides, high-density lipoprotein (HDL) and the ratio of HDL to low-density lipoprotein (LDL), showed significant increases from baseline at cycle 13 (p < 0.0001) [69].
In two large, randomized, multicenter studies, which compared drospirenone/ethinyl estradiol 30 μg with desogestrel 150 μg/ethinyl estradiol 30 μg, there were no negative effects on lipids or carbohydrate metabolism and blood pressure was essentially unchanged [70].
In a study of four groups of women, three receiving drospirenone 3 mg with ethinyl estradiol 30, 20 and 15 μg, respectively, and one group receiving levonorgestrel 150 μg/ethinyl estradiol 30 μg, systolic and diastolic blood pressures fell by 1–4 mmHG in the drospirenone groups and rose by 1–2 mmHg in the levonorgestrel/ethinyl estradiol group. In the drospirenone groups, HDL cholesterol rose (p < 0.05) compared with the levonorgestrel/ethinyl estradiol group. LDL cholesterol fell (p > 0.05), while triglyceride levels rose in the drospirenone groups (p < 0.05) compared with the levonorgestrel group. Basal glucose levels remained essentially the same in all groups [71].
In 15 hirsute polycystic ovarian syndrome patients treated with drospirenone 3 mg/ethinyl estradiol 30 μg for 12 cycles, blood pressure remained stable and there was no effect on glycoinsulinemic homeostasis. Total cholesterol, triglycerides, and HDL and LDL were all normal with a trend toward increase with cholesterol/HDL and HDL:LDL ratios remaining stable [72].
Additional studies evaluating the efficacy and tolerability of the drospirenone/ethinyl estradiol combination pill found the ethinyl estradiol 30 μg and 20 μg, and drospirenone 3 mg combinations to be highly effective in suppressing ovulation [73], with good cycle control [68,70].
The combination of drospirenone/ethinyl estradiol is also helpful for women experiencing premenstrual symptoms, as summarized in Table 1. Open-label cohort studies evaluated the COC containing drospirenone compared with one containing levonorgestrel or desogestrol. The results demonstrated a significant decrease in body weight in women in the drospirenone 3 mg/ethinyl estradiol 30 μg group compared with those given desogestrol/ethinyl estradiol [70]. In another open-label study, after cycle 6, women receiving drospirenone/ethinyl estradiol had approximately a 50% decrease in baseline premenstrual symptoms including: water retention, weight gain, irritability, anxiety and feeling blue, whereas the comparison group receiving levonorgestrel/ethinyl estradiol experienced no changes in their premenstrual symptoms [74]. Drospirenone 3 μg/ ethinyl estradiol 30 μg was more effective in a regimen extended for 42–126 days compared in another open-label study, with a standard cyclic regimen [75]. Open-label studies validated these findings in both new users and women switching from COCs containing progestins [45,46].
Summary of clinical studies of drospirinone/estradiol combined oral contraceptive pill: effect on premenstrual symptoms.
OC: Oral contraceptive; PMDD: Premenstrual dysphoric disorder; PMS: Premenstrual syndrome; RCT: Randomized, controlled trial.
A randomized, controlled trial was performed in women with PMDD using drospirenone 3 mg/ethinyl estradiol 30 μg 21/7 regimen. The tool used to evaluate the improvement of symptoms in this trial was the Calendar of Premenstrual Experiences rating scale [76]. Overall, most items showed improvement from baseline but statistical significance was only found for acne, appetite and food cravings [47].
The pill containing drospirenone 3 mg/ethinyl estradiol 20 μg in a 24/4 regimen is an effective and well tolerated COC [68]. The effects of this new formulation and regimen on symptoms of PMDD were evaluated in two large multicenter randomized, controlled trials. In both studies, the primary outcome was measured by decrease in severity of the 21 symptoms of the Daily Record of Severity of Problems [77]. The Endicott Quality of Life Enjoyment and Satisfaction Questionnaire (Q-LES-Q) was also utilized [78].
A multicenter, double-blind, placebo-controlled crossover study evaluated 64 participants who met the criteria for PMDD and were randomized to active treatment and placebo pills for three cycles. Total symptom scores showed significantly greater improvement in the treatment group compared with placebo [15]. In the model incorporating both periods of the crossover trial, the differences between groups in the change from baseline of the total Daily Record of Severity of Problem total symptom score were −22.94 for active treatment and −10.6 for placebo, for a mean difference of −12.5%. Drospirenone/ethinyl estradiol, was also statistically significantly superior to the placebo for all of the functional impairment items on the Daily Record of Severity of Problems. Rates of response were defined on the basis of percentage of subjects with Clinical Global Improvement by Investigator scale (CGI-I) [79] scores of 1 or 2, of a possible 7, where 1 = very much improved, 2 = much improved and 7 = very much worse. ‘Responders’ were significantly more likely during the active treatment arms: 61.7% responded while using drospirenone/ethinyl estradiol compared with 31.8% while using placebo. There were also statistically significant differences favoring active treatment for 15 of the 16 items on the Q-LES-Q.
Adverse events (AEs) considered to be related to the drug were noted in 48.2% of subjects using drospirenone/ethinyl estradiol and 28.6% of subjects using placebo, and included nausea, upper respiratory infection, headache, intermenstrual bleeding, breast pain, nervousness, asthenia, back pain and sinusitis. Many of these symptoms, in particular breakthrough bleeding, improve after continued use [15].
The effect size, based on difference from baseline, was 1.2, which is considered a large effect. For example, effect sizes for treatment studies of PMDD with SSRIs range between 0.13 to 1.2, although these numbers would need to be confirmed from head-to-head trials [15].
A parallel design study was performed comparing the same regimen of drospirenone 3 mg/ethinyl estradiol 20 μg 24/4 in 449 subjects with prospectively documented PMDD [16]. The results from this study of three cycles indicated that total scores decreased by 47% from a mean of 77.4 during the baseline cycles to an average of 41.2 for the three treatment cycles in the active treatment group compared with 38%, from 78.1 to 48.1, in the group receiving placebo. Clustered scores for mood symptoms and physical symptoms also demonstrated significantly greater decrease in the drospirenone/ethinyl estradiol group (−19.2 for mood symptoms in the active treatment group compared with −15.3 for the placebo group, and −10.7 for the physical symptoms in the active treatment group compared with −8.6 in the placebo group). Each of the 11 distinct items included in the drospirenone had an estimated improvement that was significantly greater for subjects receiving drospirenone/ethinyl estradiol compared with placebo [16]. The adjusted mean difference in treatment scores between drospirenone/ethinyl estradiol and placebo was −0.8 for depression, −0.4 for anxiety, −1.0 for mood swings, −0.9 for angry/irritability and −1.4 for the physical symptoms item, which combined breast tenderness, bloating, headache and muscle pain. Active treatment was associated with a 49% decrease in depression, which the authors state is notable because some studies have suggested COCs can worsen depression [16].
The most commonly reported AEs in the treatment group included nausea, intermenstrual bleeding and asthenia [16]. Taken together, these results suggest that the 24/4 regimen of drospirenone 3 mg/ethinyl estradiol 20 μg is effective in treating psychological, physical and behavioral symptoms of PMDD. The results led to the US FDA approval for the indication of PMDD in women who desire hormonal contraception.
Postmarketing surveillance
Since the 1960's, serious vascular side effects including myocardial infarction, thromboembolism and stroke have been recognized in association with COCs. These serious adverse events are related, in a dose–response fashion, primarily to the estrogen component [80]. Low-dose COCs with ethinyl estradiol 20–35 μg confer less risk than COCs containing ethinyl estradiol 50 μg. It is accepted that there is an increased risk of venous thromboembolism (VTE) in women using low-dose COCs. However, the relative risk (RR) of VTE associated with a low-dose COC (RR: 3–4) is less than the thrombolembolic risk associated with term pregnancy (RR: 6–12) [80].
The occurrence of VTE associated with drospirenone/ethinyl estradiol was noted in two reports published in 2002 and 2003 [81,82]. In 2002, 40 cases of VTE, with two deaths, were reported, while in 2003 five VTE cases were reported. These were not scientific studies, nor was there a baseline population identified in order to determine the incidence of VTE or permit an epidemiologic evaluation. The second report concluded that “Physicians may prefer a new type of combined pill, like ethinyl estradiol and drospirenone, assuming that these are safer. However, an association of these drugs with a lower risk of thromboembolism has not been proved by research, and our cases show that newer contraceptive pills may have a risk of thromboembolism” [81]. This report indicates that the expectation was that drospirenone-containing COCs had no risk or a lower risk of thromboembolism. The fundamental reasoning is incorrect because it is not the progestin, rather it is the estrogen, ethinyl estradiol, contained in all 20–35 μg COC, that is thought to be associated with changes in clotting factors that contribute to VTE and therefore the risk should be the same [80]. Subsequently, the regulatory agency for The Netherlands concluded that there was no increased risk of VTE in patients taking this hormone combination compared with other COCs [102].
More data than ever before are available (140,000 women years) from the European Active Surveillance (EURAS) study. The final results of the EURAS study have been presented (International Society for Pharmacoepidemiology [ISPE] 2006, International Federation of Gynecology and Obstetrics [FIGO] 2006) and published [83]. There was no increased risk of adverse cardiovascular or thromboembolic events for drospirenone-containing COCs compared with other COCs [83].
Several postmarketing surveillance studies of the 21/7 regimen drospirenone/ethinyl estradiol were designed to evaluate the risk of DVT and pulmonary embolism for patients taking this medication. A 1-year study based on follow-up of COC prescriptions written by general practitioners over a 6–12-month period was conducted. All physicians who had written prescriptions were sent a questionnaire asking about ‘health-related events’ that subsequently occurred. An incidence rate of 13.7 VTE cases/10,000 women in users of this COC was determined. However, this statistic was based on a 51% response rate to the questionnaires, thus the validity of this finding is uncertain. While there was a positive association between ethinyl estradiol/drospirenone and VTE, the results were subject to selective prescribing bias and “should be interpreted with caution” [84]. Among the VTE cases were women with congenital thrombophilias, smokers, trauma and other potential risk factors [84]. In fact, the background incidence of VTE may be higher than previously estimated, and more in the range of 5–10 per 10,000 women [85].
Safety & tolerability
A full discussion of the side effects and safety, including contraindications, for the COC drug class exists in the published literature [49,60,80]. However, the focus on myocardial infarction and stroke, in addition to VTE, as possible side effects of COC deserves mentioning [86]. Studies have shown that healthy, nonsmoking women taking prescription oral contraceptives are not at an increased risk for myocardial infarction or stroke [87]. Women who smoke and are over the age of 35 years have a higher risk of myocardial infarction [86,87]. COCs are contraindicated in heavy smokers (>15 cigarettes daily) who are over the age of 35 years. Women with underlying risk factors, such as hypertension, hyperlipidemia, obesity and diabetes predisposing to coronary artery disease may also be at increased risk for myocardial infarction.
Drospirenone is unique as a progestin because of its antimineralocortiocid activity, and therefore there is the theoretic potential for hyperkalemia. The 3 mg of drospirenone is comparable with 25 mg of the weak potassium-sparing diuretic, spironolactone. The labeling states that COCs containing drospirenone should not be prescribed to women with renal insufficiency, hepatic dysfunction or adrenal insufficiency [49,56,60]. A study was performed to evaluate potassium and drospirenone levels after 3 mg drospirenone was given for 14 days to groups of women with normal renal function, and mild and moderate renal impairment. There was no effect on potassium or serum levels of drospirenone in women with normal renal function (creatine clearance [CrCl] >80 ml/min) or mild renal dysfunction (CrCl 50–80 ml/min). In women with moderate renal dysfunction (CrCl 30–50 ml/min), there was a 37% increase in serum drospirenone levels compared with women with normal renal function, due to results from one subject. However, overall pharmacokinetics was minimally effected and clinically increased potassiun (>5.5 mEq/l) was not a problem [88]. Labeling states that women chronically using medications that may increase serum potassium should have serum potassium checked during the first treatment cycle. Such medications include angiotensin-converting enzyme inhibitors, angiotensin II receptor antagonists, potassium-sparing diuretics, heparin, aldosterone antagonists and chronic daily use of any nonsteroidal anti-inflammatory agents (NSAIDs) [49,56,60]. Of note is the fact that occasional, or even chronic, use of NSAIDs was not limited in the pivotal clinical trials [44]. Hepatic disease is a contraindication based on metabolic characteristics rather than clinical characteristics since drospirenone is metabolized in the liver.
Issues of tolerability include, but are not limited to, adverse events such as menstrual cycle control (number of days of bleeding and spotting), nausea and headache, all of which can account for premature discontinuation. These results with drospirenone/ethinyl estradiol are consistent with other low-dose COC formulations.
Conclusion
Various nonpharmacological alternatives can be used to treat mild symptoms of premenstrual disorders [48]. However, severe cases of PMS and PMDD are usually addressed pharmacologically. The medications with indication for treating PMDD include three SSRIs and one COC containing drospirenone 3 mg/ethinyl estradiol 20 μg in a regimen of 24 active pills followed by 4 inactive pills. The creation of a reliable and well-tolerated COC with an additional benefit of treating premenstrual disorders may be a welcomed alternative for women suffering from PMDD who are dissatisfied with the side-effect profile of SSRIs or fail to respond to SSRIs but have desire for hormonal contraception or at least, if not in need of contraception, lack contraindications for COCs.
Future perspective
The drospirenone-containing COC has recently been approved by the US FDA for the indication of PMDD for women who desire hormonal contraception. Since the response rate for PMDD is in the range of 60%, further studies will likely be performed to assess which women or which specific symptoms are most likely to respond to this COC. Oral contraceptives are reasonably safe medications and it is possible that physicians will offer ethinyl estradiol 20 μg/drospirenone 3 mg 24/4 to women who do not have a need for contraception but who have PMDD. Adolescents in particular will be unlikely to receive SSRIs as a first-line therapy, the other class of medications currently indicated for treatment of severe PMS/PMDD, due to recent concerns regarding suicidal behavior. This drospirenone-containing COC will possibly also be prescribed off-label for women with the whole range of PMS symptoms, mild to severe, somatic and psychological, and studies should be completed in this group of women. It is unclear which symptoms respond better to this COC compared with an SSRI, or whether the combination of the two approaches will improve treatment outcome, either increasing the number of responders or the magnitude of response. Additional possibilities for the treatment of resistant symptoms of PMS or PMDD could include other more extended regimens for drospirenone/ethinyl estradiol with a brief respite of 4 days every 3 months or with no break at all. Ethinyl estradiol 20 μg/drospirenone 3 mg 24/4 will possibly also be utilized for women with psychiatric diagnoses, who are receiving psycho-tropic medications but who, in spite of increasing medication dosages, still have premenstrual exacerbation of mood or anxiety disorders.
Drospirenone, an aldosterone antagonist, acts to oppose the stimulatory effect of estrogen on the RAAS and may be effective for the PMS symptoms that can recur when estrogen/progestin hormone therapy is given with a GnRH analog for PMS or pelvic pain, or for peri- and post-menopausal hormone therapy. The new drospirenone 0.5 mg/17 β-estradiol 1 mg-containing hormone therapy formulation (Angeliq®), recently approved in the USA for menopausal symptoms, may prove to be better tolerated than other currently available progestin-containing regimens for women with past history of PMS, postpartum depression or other affective disorders, but this is speculative. Women using these drospirenone/estrogen preparations for menopause are less likely to develop hypertension and weight gain over time [89,90]. This may also be the case with this COC for contraception; however, hypertension remains a relative contraindication for COCs at this time.
Executive summary
The 24/4 regimen of drospirenone 3 mg/ethinyl estradiol 20 μg combined estrogen/progestin-containing contraceptive pill (COC) has a Pearl Index equal to other COCs, and possibly better that other 20 μg COCs, due to the longer intake of active pills.
Drospirenone/ethinyl estradiol COCs are comparable with other previously approved COCs in regards to changes in glucose, blood pressure and lipids.
Drospirenone-containing COCs showed significant improvement in water retention, weight gain, negative affect, acne and increased appetite when compared with other COCs.
The 24/4 regimen with drospirenone 3 mg/ethinyl estradiol 20 μg has been approved for treatment of premenstrual dysphoric disorder (PMDD) in women desiring hormonal contraception.
Serious adverse events regarding COCs include venous thromboembolism, myocardial infarction and stroke, but these do not appear to occur more frequently with drospirenone-containing COCs.
Drospirenone and ethinyl estradiol are both rapidly absorbed following oral administration.
Steady-state condition is reached after eight days following daily administration of drospirenone 3 mg/ethinyl estradiol 20 μg tablets.
Drospirenone has anti-androgenic and antimineralocorticoid effects.
Drospirenone carries a potential risk of hyperkalemia, therefore should not be prescribed to women with renal or adrenal insufficiency or hepatic dysfunction.
Women on medications that may increase serum potassium should be monitored for hyperkalemia for the first few cycles on a drospirenone-containing COC.
Medications that can increase serum potassium include angiotensin-converting enzyme inhibitors, angiotensin II receptor antagonists, potassium sparing diuretics, heparin, aldosterone antagonists and chronic daily use of any nonsteroidal anti-inflammatory agents.
