Abstract
Interstitial cystitis is a condition that affects the urinary bladder and may be more prevalent than commonly thought. Symptoms range widely in severity and typically include urinary urgency, frequency and pelvic pain. Therapies usually target the known pathophysiology of this condition. Oral agents commonly include bladder surface mucin analogues, antihistamines, narcotics, tricyclic antidepressants and anticonvulsants. Intravesical therapy is usually used as second-line treatment. Multiple forms of pharmacotherapy are often provided simultaneously to improve efficacy and time to response.
Keywords
Interstitial cystitis (IC) is a clinical syndrome of unclear etiology and varying severity affecting the urinary bladder. Presenting symptoms include urinary frequency, urgency and/or pelvic pain in the absence of any other identifiable pathology such as urinary tract infection, bladder carcinoma or radiation cystitis [1–3]. Often progressive in nature, IC can present with a pattern of flares and remissions [4]. Although providing a diagnostic challenge at times, symptom improvement can be achieved in most instances with a proper medical regimen and early diagnosis.
Variations in clinical presentation commonly provide a diagnostic dilemma to physicians. ‘Classic IC’ presents as bladder pain, urgency, and the cystoscopic finding of Hunner's ulcers, ulcerative patches signifying a transmural inflammatory process (Figure 1A). Identification of a Hunner's ulcer is made in less than 5–10% of cases [3]. The majority of patients present as ‘nonclassic’ IC, where symptoms exist but no cystoscopic abnormalities are seen. Over-distention of the bladder (hydrodistention) under anesthesia may demonstrate a lower anesthetic bladder capacity and submucosal hemorrhages, termed ‘glomerulations’ (Figure 1B). In an attempt to clarify this dilemma, the International Continence Society (ICS) created and characterized new terminology, painful bladder syndrome (PBS). PBS is defined as “the complaint of suprapubic pain related to bladder filling, accompanied by other symptoms such as increased daytime and night-time frequency, in the absence of proven urinary infection or other obvious pathology”. The term IC would be utilized when typical cystoscopic and histological features are apparent [5].
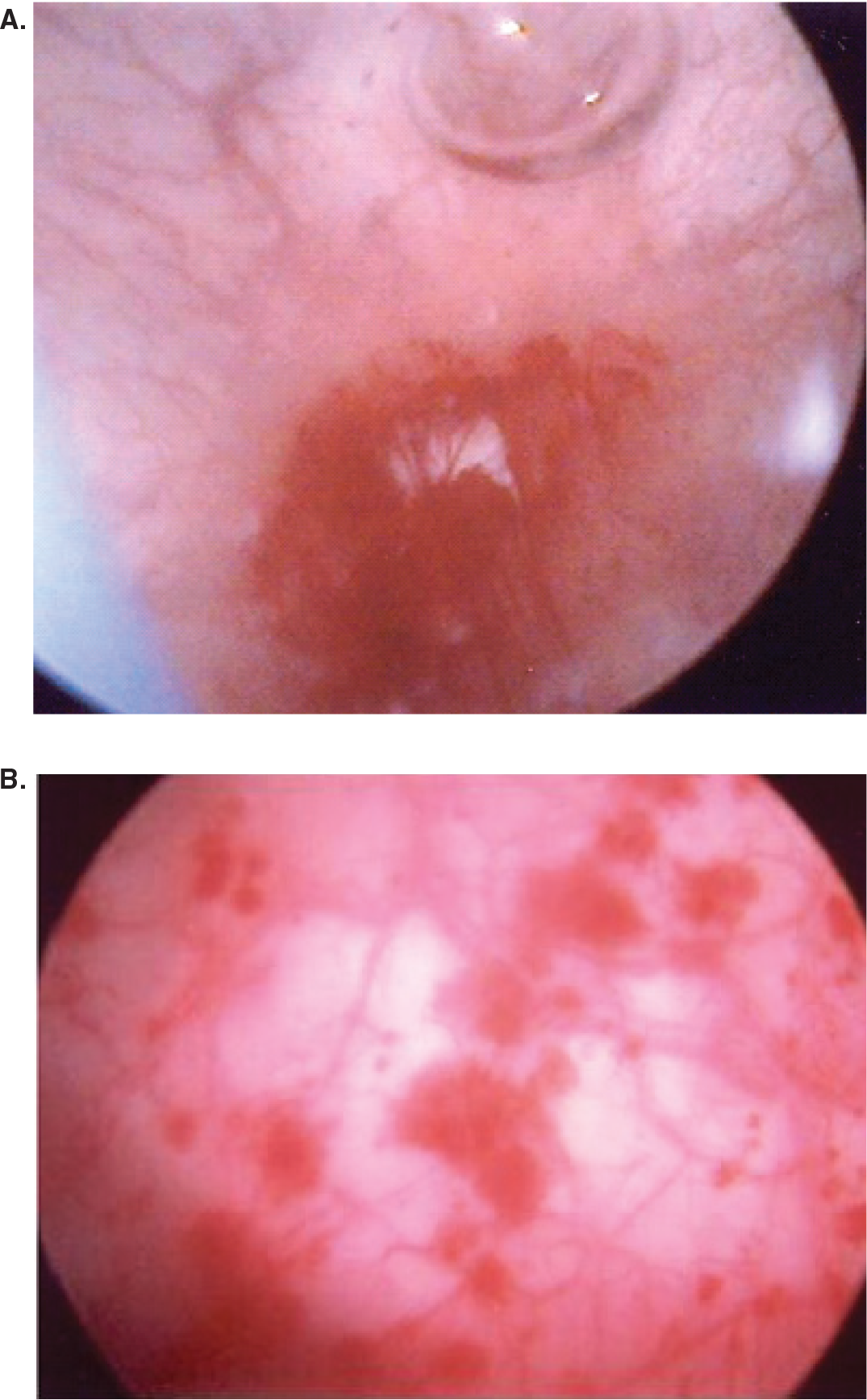
(A) Hunner's ulcers, (B) glomerulations noted during cystoscopy and hydrodistension.
As it shares similar symptom characteristics with other diseases, IC is often a diagnosis of exclusion [6,7]. A thorough history and physical examination is prerequisite for proper diagnosis. Variation of pain with bladder filling and emptying, nocturia and anterior vaginal wall tenderness are frequently seen. Questionnaires such as the O'Leary–Sant symptom and problem index and the pain, urgency and frequency (PUF) may further quantify patient symptoms [8]. Diagnostic testing includes urinalysis and culture and urine cytology when appropriate. Further diagnostic exams are performed to exclude other pelvic-based pathologies at the clinician is discretion. Procedures such as the potassium sensitivity test or cytoscopy with hydrodistension may be performed to better clarify a bladder-based disorder; however, the diagnosis of IC is frequently made without these more invasive exams.
Due to the wide range of symptom severity and differences in clinical presentation, the prevalence of IC is difficult to determine. It is estimated that between 700,000 and 1 million women in the USA have IC. The female to male ratio is approximately 10:1 [3,9]. Criteria developed by the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) to identify IC patients may be too restrictive for epidemiological studies (
The healthcare costs of IC can be significant. Wu and colleagues assessed the direct medical cost and indirect costs of work loss for IC patients in the first year after diagnosis [11]. Compared with a control group of non-IC patients, the average IC patient had 130% higher direct costs (p < 0.05) and 84% higher indirect costs (p < 0.05). The IC patient group also had higher relative risk rates of concomitant comorbidities such as endometriosis, vulvodynia, chronic pelvic pain, depression and anxiety [11].
Current pharmacotherapy entails the administration of oral and intravesical agents, often in combination. New avenues for possible treatment include the use of botulinum toxin type A (Botox®) or vanilloid receptor-binding agents. Familiarity with the pathophysiologies of IC enables the clinician to choose the most appropriate management strategies.
Theories of pathophysiology
The etiology of IC is not completely known and may be multifactorial. Several theories have been presented, ranging from dysfunction of the urothelium to mast-cell activation to neurogenic causes [6,12].
Urothelial dysfunction
The urothelium is a highly effective barrier against the movement of water and ionized substances across the bladder wall. It consists of three layers: a basal cell layer attached to the basement membrane, an intermediate layer and a superficial apical layer composed of large hexagonal umbrella cells up to 250 μm in diameter [13]. The urothelium has specialized sensory and signaling characteristics, enabling it to actively participate in the chemical and physical environment of the detrusor muscle. These characteristics involve:
Expression of various receptors such as nicotinic, muscarinic, tachykinin, adrenergic and vanilloid;
Sensitivity to neurotransmitters of afferent and efferent nerves;
Mechanoreceptive properties;
Close proximity to afferent nerves;
The ability to release chemical transmitters such as ATP and nitric oxide, which can regulate the action of local nerves, cause vascular changes, and/or produce reflex detrusor contractions [13].
The leaky bladder urothelium theory postulates that bladder urothethial surface is normally lined by bladder surface mucin, a heterogenous matrix composed of glycoaminoglycans and glycoproteins. This provides protection against various organisms and components of urine [2,3]. An alternation in the bladder surface glycosaminoglycan layer, possibly combined with an inability to repair itself, results in increased permeability of substances, such as potassium, causing inflammation and pain [14].
Current experimental evidence suggests that other components of the urothelium may have a more pronounced role. ATP released from damaged urothelial cells during an inflammatory response can initiate painful sensations through excitation of P2X receptors on sensory fibers [13]. Sun and colleagues analyzed urinary ATP levels and concentrations of ATP released from stretched bladder wall biopsies in patients with IC [15]. Compared with a control group, mean urinary ATP levels and concentrations of released ATP during muscle stretch were significantly higher in the IC group.
NIDDK research definition of interstitial cystitis.
Symptoms: bladder pain or urinary urgency
Cystoscopy: glomerulations or classic Hunner's ulcer
Bladder capacity >350 cc on awake cystometry
Absence of intense urge to void with bladder filled to 100cc during cystometry with a fill rate of 30–100 ml/min
Demonstration of phasic involuntary bladder contractions on cystometry
Duration of symptoms <9 months
Absence of nocturia
Symptoms relieved by antimicrobials, urinary antiseptics, anticholingerics, or antispasmodics
Frequency of urination while awake < 8x/day
Diagnosis of bacterial cystitis or prostatits within a 3-month period
Bladder or ureteral calculi
Active gential herpes
Uterine, cervical, vaginal or urethral cancer
Urethral diverticulum
Cyclophosphamide or any type of chemical cystitis
Tuberculous cystitis
Radiation cystitis
Benign or malignant bladder tumors
Vaginitis
Aged under18 years
NIDDK: National Institute of Diabetes and Digestive and Kidney Diseases.
Along with ATP, antiproliferative factor (APF) presents another possibility for IC treatment and diagnosis. APF, a nonapeptide glycoprotein, is present in high levels in the urine of IC patients and acts by inhibiting normal urothelial cell proliferation [16]. The production of APF occurs in bladder urothelial cells and alters the levels of two growth factors, epidermal growth factor (EGF) and heparin-binding EGF-like growth factor [17]. APF may also have some utility as a specific marker for IC, as it is not associated with other diseases of the lower urinary tract [18,19].
Mast-cell activation
Mast cells arise from the bone marrow and are known to participate in allergic and type I hypersensitivity reactions. These cells contain numerous vasoactive and inflammatory substances such as histamines, leukotrienes, prostaglandins and tryptases. Generally, triggers for mast cells are many and include anaphylatoxins (complement C3a and C5a), cytokines (i.e., interleukin [IL]-1, IL-2, tumor necrosis factor [TNF]-α, etc.), growth factors, bacteria, certain neuropeptides and free radicals [20].
Numerous studies have reported increased numbers of mast cells in the bladders of IC patients, especially when associated with Hunner's ulcers [20]. The factors responsible for proliferation of mast cells in IC are unknown. Damaged urothelial cells can produce cytokines such as stem cell factor (SCF), which can cause activation of the mast cells [21]. The various vasoactive and inflammatory contents of activated mast cells may lead to neuronal sensitization and secretion of substances, such as acetylcholine or substance P. As discussed below, the release of substance P further activates mast cells and contributes to a vicious cycle [20].
Neurogenic inflammation & occult infections
Sensory nerves may secrete inflammatory mediators, thereby causing pain and local inflammation. Substance P is a short-chain polypeptide and main participant in this process. With nociceptive and inflammatory properties, substance P is a neurotransmitter that is mainly released by peripheral nerves associated with pain known as C-fibers. Once released, an inflammatory cascade occurs and results in mast-cell degranulation and activation of adjacent nerve terminals [2]. An increased number of C-fibers and substance P is demonstrated in IC patients and may contribute to symptoms of pelvic pain [22].
Urinary tract infections and IC share similar symptoms and, as such, early experimental investigations targeted an infectious etiology. However, pathogenic bacteria are not routinely demonstrated in urine cultures and IC patients do not respond to antibiotic treatment. Current evidence does not support an infectious etiology [23]. However, after documented bacterial cystitis, some IC patients have the start of their chronic symptoms; this may entail that an acute event of bacterial cystitis may lead to permanent ‘bladder injury’ and the subsequent development of IC in susceptible patients [2].
Medical treatment options for interstitial cystitis
Many of the medical therapies attempt to target the pathophysiological aspects of IC, primarily focusing on the symptoms of pain and replacing various constituents of the bladder surface mucin. With the exception of pentosan polysulfate, most of the medications are not US FDA approved for the treatment of IC. Prior to initiation of therapy, patients' expectations should be addressed. Counseling of patients necessitates delineation of several goals:
Reduction of symptoms;
Improvement of quality of life;
Possible use of different and/or multiple treatments;
Possible long-term therapy.
In severe cases, combinations of oral and intravesical therapies may be required to achieve relief. Due to the chronic nature of IC, complete remission is possible, but not to be expected.
Oral therapy
Bladder surface mucin analogues
Pentosan polysulfate sodium
Pentosan polysulfate sodium (PPS) (Elmiron®, Ortho-McNeil Pharmaceuticals, NJ, USA) is a synthetic, highly sulfated polysaccharide similar in structure to heparin sulfate, one of the glycosaminoglycan components of the bladder surface mucin. The actual mechanism of action is not clearly elucidated, but it is believed to augment the glycosaminoglycan layer, thereby decreasing the altered permeability of the bladder surface. This oral heparinoid is also the only US FDA-approved oral medication for IC. Several double-blind, randomized studies have shown its efficacy [24–26], with response rates ranging from 28–32% versus 13–16% for placebo. A meta-analysis by Hwang and colleagues showed PPS to be more efficacious than placebo in the treatment of pain, frequency and urgency but there was no significant difference in the treatment of nocturia [26].
The usual dosage for PPS is 100 mg three times a day, and may take up to 4 months to produce pain relief and up to 6 months to decrease urinary frequency [4]. Common side effects include diarrhea, headache and reversible alopecia. Nickel and colleagues recently evaluated the onset of effect and dose–response effect of PPS by comparing three dosages (300, 600 and 900 mg daily) in a randomized, double-blind, parallel-group multicenter study [28]. Utilizing the O'Leary–Sant Interstitial Cystitis Symptom Index and the Patient's Overall Rating of Symptoms Index, improvements in scores were noted within 4 weeks and steadily improved over time. Overall, the responses to the three different dosages, as measured by study end points at 32 weeks, were not statistically different. The study concluded that response to treatment with PPS correlates with duration rather than dosage. Diarrhea and mild rectal bleeding were also noted at the higher dose ranges [28].
PPS has also been evaluated with concurrent administration of subcutaneous low-dose heparin [29]. A total of 41 patients were divided into three groups of minor, intermediate and major responders to PPS and were given 5000 U of subcutaneous heparin every 8 h for 2 days followed by 5000 U every 12 h for 12 days along with oral PPS. Compared with a small group of patients receiving PPS alone, 10 patients (24.4%) were identified as responders (moderately or marked improved) at 3 months and at 6 months, 9 patients (21.9%) (p ≤ 0.001). In the subset analysis, the minor response to PPS group had a significant amelioration of pain intensity at 3 and 6 months and reported a statistically significant overall improvement in their wellbeing [29].
Antihistamines
Hydroxyzine
Mast-cell degranulation involves histamine release, and antihistamines have been used with variable success. The most commonly used antihistamine is hydroxyzine (Atarax®, Vistaril®), which functions as an antagonist at the H-1 receptor and blocks mast-cell secretion [30]. Hydroxyzine may also have a neurogenic mechanism. Minnogiannis and colleagues demonstrated in rat bladder studies a decrease in carbachol-induced serotonin levels by hydroxyzine through a mechanism different from its H-1 receptor antagonistic properties, possibly through inhibition of substance P release [31].
The dose of hydroxyzine ranges from 10–75 mg/day. An open-label study of hydroxyzine treatment showed a 40% reduction in symptom scores from baseline. A further reduction (55%) was noted in patients with history of allergies [32]. A recent pilot study comparing hydroxyzine alone with PPS or in combination showed no significant benefit compared with placebo [33].
Tricyclic antidepressants
Amitriptyline
The tricyclic antidepressant amitriptyline (Elavil®) is often used for pain management in IC patients. Other tricyclic antidepressants commonly used include nortriptyline (Pamelor®) or doxepin (Sinequan®). Amitriptyline has several properties well-suited for IC therapy, including:
Moderate anticholinergic and sedative effects;
Serotonergic effects by its inhibition of 5-hydroxytryptamine reuptake;
Mast-cell stabilization.
The starting dosage for amitriptyline is 10–25 mg and may be increased to 75–100 mg on a weekly basis. Common side effects include dry mouth, weight gain, constipation and decrease in libido. Administration at bedtime may minimize side effects as well as provide restful sleep.
In a randomized, double-blind, placebo-controlled study, amitriptyline significantly demonstrated improvement in pain and urgency symptoms (p < 0.001). Although frequency and functional bladder capacity improved in the amitriptyline group compared with the placebo group, the results were not statistically significant [34].
Long-term results of amitriptyline therapy were analyzed in a prospective, open-label study [35]. Amitriptyline was given at bedtime to 94 patients, divided into a NIDDK criteria group and a non-NIDDK group, in an established self-titration protocol. After a mean follow-up time of 19.0 ± 12.5 months, a response rate of 64% was noted (60 patients) while the dropout rate was 31% (29 patients), primarily due to lack of response followed by side effects [35]. The overall mean dose was 55 mg (range 12.5–150) and side effects, such as dry mouth and weight gain, occurred in 79 patients (84%). Improvement in IC symptoms was statistically significant compared with baseline and responses were uniformly observed in those fulfilling NIDDK criteria and those with clinical diagnosis [35].
Second-line oral agents
Narcotics
As pain is a significant component of IC, and if suboptimal relief is obtained with nonsteroidal anti-inflammatory drugs (NSAIDs), chronic use of opioids may be required. Narcotics are believed to affect the postganglionic pain C-fibers. Long-acting narcotics and patches have been described as standard of care [2]. Adverse effects, i.e., respiratory depression, sedation, chronic constipation, associated urinary retention and physical dependency, should be kept in consideration.
H-2 receptor antagonists
Oral cimetidine is used primarily for the treatment of peptic ulcer disease and is the only medication in this group that is utilized for IC. Dosing consists of 600 mg per day in divided doses and its mechanism of action is unknown. Initial small, nonrandomized studies demonstrated symptomatic improvement rates ranging from 66–71% [36,37]. Thilagarajah and colleagues analyzed the efficacy of oral cimetidine in 36 patients with PBS or nonulcerative IC in a prospective, randomized, double-blind, placebo-controlled study [37]. Among the group receiving cimetidine, significant improvement in symptoms was demonstrated, as well as a significant decrease in suprapubic pain and nocturia. Histologically, no qualitative changes were apparent in the glycoaminoglycan layer or basement membrane after treatment. The authors surmise cimetidine may have an immune-modulating effect on the bladder mucosa by restraining suppressor T-cells. Consequently, the histamine effect is blocked and the inflammatory response is inhibited [37].
Antiseizure medications
Gabapentin
Gabapentin (Neurotin®) is an anticonvulsant that has documented efficacy in neuropathic pain conditions such as diabetic neuropathy, reflex sympathetic dystrophy and postherpetic neuralgia [30]. Although it is structurally related to γ-aminobutyric acid (GABA), a neurotransmitter, its mechanism of action is unclear. It is not known to bind to GABA receptors or have any effect on GABA itself. Its analgesic effect may be related to modulation of calcium channels, mostly at the α−2-γ subunit [30].
Gabapentin may also have an effect by enhancing endogenous opioid release and having some activity at the N-methyl-d-aspartate (NMDA) receptor, both consequently decreasing the sensation of pain. Gabapentin is given at a starting dose of 100 mg daily and may be elevated to 3600 mg/day, usually divided into three doses a day. Unlike other anticonvulsants, serum levels of gabapentin do not need to be monitored and a common side effect is drowsiness. No randomized clinical trials have truly tested the efficacy of gabapentin, and evidence is based on small trials [38,39].
Immunosuppressive agents
Prednisone & cyclosporine A
Immunosuppressive agents can be utilized as second-line therapy as they are thought to reduce the inflammatory response in the bladder. Most commonly used are low-dose prednisone and cyclosporine A; some efficacy is shown with both agents through several small studies, but further analysis is needed with randomized studies.
Soucy and Gregoire evaluated the use of prednisone in 14 patients with chronic severe IC with Hunner's ulcers unresponsive to first-line therapies [40]. The dosage of prednisone was initially 25 mg daily for 1–2 months, followed by titration to the lowest possible dose for resolution of symptoms. Of the 14 patients, nine (64%) continued the regimen at the end of the study with a follow-up period of 16 months: a 38% reduction (p < 0.005) in the total score of the O'Leary index was observed and pain was decreased by 88% (p = 0.0001) [40]. Although there was a significant improvement of pain control, the use of prednisone may be currently limited, as this study had a small population of patients and did not incorporate IC patients without Hunner's ulcers.
Cyclosporine A, mostly utilized in organ transplantation, has also been assessed for the treatment of IC in several small trials. Recently, the long-term effects of cyclosporine was analyzed in 23 patients fulfilling NIDDK criteria, with a treatment duration of 1 year or longer [41]. Initial starting dose was 3 mg/kg/day and was decreased gradually to 1 mg/kg/day. The number of micturitions in 24 h was 20.8 ± 6.3 before treatment and decreased to 10.2 ± 3.8 (p < 0.001) after a year. Maximal bladder capacity increased from 161.8 ± 74.6 to 360.7 ± 99.3 ml in a year (p < 0.001). Mean voided volume increased from 101.4 ± 42.7 to 246.4 ± 97.9 ml (p < 0.001) [41]. A total of 20 patients did not report bladder pain, and nine out of 11 patients resumed cyclosporine A after cessation due to improvement of symptoms. Adverse effects included hypertension, gingival hyperplasia and increased hair growth.
Cyclosporine A was compared with PPS in a randomized prospective study [42]. A total of 64 patients meeting NIDDK criteria were divided into two groups: one receiving 1.5 mg/kg twice daily and the other 100 mg of PPS three times a day. Micturition frequency in 24 h was significantly reduced in the cyclosporine A arm compared with the PPS arm. The clinical response rate (according to subjective global response assessment) was 75% for cyclosporine A, compared with 19% for PPS (p < 0.001). Although cyclosporine was found more effective, higher rates of adverse effects were also noted [42].
Intravesical therapy
Heparin sulfate
Intravesical therapy, or administration of agents via urethral catheter, is used alone or in conjunction with oral therapy as second-line therapy. As it is a glycosaminoglycan component of bladder surface mucin, heparin sulfate can be utilized intravesically. It also has anti-inflammatory effects, prevents angiogenesis and inhibits fibroblast and smooth muscle proliferation [2]. In severe cases, alone or in conjunction with oral PPS, a solution containing 40,000 IU of heparin in 10 cc of water can be self-instilled daily by the patient [4].
Dimethylsulfoxide
Dimethylsulfoxide is an amphipathic molecule with a highly polar domain and two apolar methyl groups, making it soluble in both aqueous and organic media. It is one of the most common solvents for the in vivo administration of several water-insoluble substances [43]. Dimethylsulfoxide is US FDA-approved for use in IC, and one of the mainstay medications of intravesical treatment.
As a local analgesic, dimethylsulfoxide may have several mechanisms of action. Its primary action may be the release and eventual depletion of substance P from the detrusor wall. Mast-cell degranulation may be another mode of action. It also may desensitize nociceptive pathways in the lower urinary tract by release of nitric oxide from afferent nerves [2]. Dimethylsulfoxide is traditionally administered as a 50% solution, but is often given as mixture containing 50 cc of 50% dimethylsulfoxide, 10 ml of sodium bicarbonate (75 mg/cc vial), 40 mg of triamcinolone and 10,000–20,000 IU of heparin sulfate. Instillations, where the solution is administered via catheter and patients are asked to hold it for 30 to 60 min, are performed on a weekly basis for 6–8 weeks followed by maintenance therapy every 2–4 weeks, if required, until amelioration of symptoms. Initial treatments may worsen patient symptoms [2].
A study Rossberger and colleagues recently evaluated 28 patients, 13 with classic IC and 15 with nonulcer disease, receiving at least one series of six instillations of DMSO [44]. Although a modest response was noted, side effects were generally well tolerated by patients. After instillations, treatment effects were noted to last 16–72 months [44].
Anesthetic cocktails
Cocktails are therapeutic solutions that seek to address several pathogenic processes of IC. One study analyzed the efficacy of a solution containing 40,000 U of heparin, 8 ml of 1 or 2% lidocaine and 3 ml of 8.4% sodium bicarbonate [45]. In the group with 1% lidocaine, 75% of 47 patients reported immediate relief while in the 2% lidocaine group, 94% of 35 patients reported relief. The differences in response rates were statistically significant (p < 0.01).
At our institution, the cocktail consists of 40 cc of a 1:1 mixture of 0.5% bupivacaine and 2% lidocaine jelly, to which 10,000 IU heparin sulfate, 40 mg triamcinolone and 80 mg of gentamycin are added [46]. Patients are instructed to hold the solution for 30 min and then void. In a recent study with 79 IC patients, intravesical instillation of this cocktail demonstrated high response rates in both groups of classical and nonclassical IC patients, 89 and 71%, respectively. The mean duration of response varied from 4–11 days. Short-term adverse effects included postinstillation pain and urinary retention. Overall, the patients responded favorably to the treatment [46].
Hyaluronic acid
Similar to heparin sulfate and pentosan polysulfate, hyaluronic acid (Cystistat™) is a glycosaminoglycan component of the bladder surface mucin and postulated to provide protection to the bladder surface. Kallestrup and colleagues recently analyzed 20 patients in a prospective, unblinded pilot study receiving hyaluronic acid instillations weekly for 1 month followed by monthly administration for 2 months [47]. Out of 13 patients (65%) who responded to treatment, four were complete responders who terminated therapy and seven were partial responders who continued treatment for a minimum of 3 years. Hyaluronic acid was well tolerated by patients and demonstrated reduction of pain and urinary frequency [47].
Bacillus Calmette–Guerin
Bacillus Calmette–Guerin (BCG) is an attenuated strain of Mycobacterium bovis and used primarily as an intravesical agent in the treatment of recurrent or multifocal bladder cancer. The mechanism of action is not fully elucidated, but it is thought to stimulate type 1 T-helper cells and decrease type 2 T-helper-cell-mediated allergic responses. Although it demonstrated some efficacy in smaller trials, a recent large randomized, controlled trial failed to show any benefit of BCG therapy [48]. A response rate in the patient global response assessment of 21% was noted in the BCG group, while the placebo group showed a rate of 12% (p = 0.062). Although a large number of adverse effects were reported in the BCG arm, there were no statistically significant differences in overall adverse event rates between both treatment groups [48].
Botox & resiniferatoxin
Resiniferatoxin is an ultrapotent capsaicin analog that acts to desensitize afferent pain C-fibers. Initial small studies showed some promise; however, a large, randomized, prospective clinical trial did not demonstrate any clinical efficacy [49]. A total of 163 participants were randomized into groups receiving a single 50cc intravesical dose containing resiniferatoxin 0.01 μm, 0.05 μm, 0.10 μm or placebo. At the 4-week follow-up, the placebo group had the highest proportion of patients (32%) with moderate or marked improvement in the global response assessment. Resiniferatoxin did not improve overall symptoms, pain, urgency, frequency or nocturia during the 12-week follow-up and was deemed to be ineffective for IC patients [49].
Although used in an off-label manner, urological indications for botulinum toxin type A (BTX-A) are mainly for neurogenic and non-neurogenic causes of detrusor overactivity. BTX-A inhibits the release of acetylcholine across the neuromuscular junction to cause muscle paralysis. However, data show that BTX-A may have possible antinocioceptive properties by effecting afferent neurotransmission [50]. The release of a sensory neuropeptide, calcitonin gene-related peptide, was decreased by 19% in whole rat bladders after the application of BTX-A [51]. Clinically, Smith and colleagues evaluated the BTX-A injected into bladder wall in 13 IC patients [52]. A response was noted in 6 patients (69%): significant decreases were noted in daytime frequency, nocturia and pain. Another small clinical trial with ten patients showed minimal but significant improvement in bladder capacity and pain score in two patients [53].
Future perspectives
IC may be part of a spectrum of chronic pelvic pain and, as the clinical definition becomes more clarified, it may be more common than once thought. A recent population-based study in Finland estimates the prevalence to be 300/100,000 to 680/100,000 women; older studies estimated prevalence to range from 4.5/100,000 to 870/100,000 [54]. With further research, diagnosis will be established with objective criteria rather than clinical criteria alone. Urinary markers will be identified to provide a noninvasive diagnostic modality. Uronic acid, a glycosaminoglycan component, was demonstrated to be elevated, in conjunction with sulfated glycosaminoglycans, in urine samples of patients with severe IC and may be a useful marker in monitoring IC [55]. APF, with further study, may be utilized clinically if efficacy is demonstrated.
As the pathophysiology of IC and neurophysiology of the lower urinary tract are elucidated, new targets for IC therapy will be produced. Evaluations of new agents to provide protection to damaged urothelium are underway. Liposomes are vesicles composed of concentric phospholipid bilayers separated by an aqueous compartment and can fuse with cells to provide a molecular film that can promote wound healing. Initial research shows a favorable response in rats with chemically induced bladder hyperactivity [56]. Other approaches for treatment include new agents for pain management (i.e., pregabalin, a relative of gabapentin, used in neuropathic and inflammatory pain) or newer medications against mast-cell activity (i.e. montelukast and suplatast tosilate). Ultimately, large, placebo-controlled, randomized studies will be needed to best access the most effective and safe strategies of care.
Executive summary
Interstitial cystitis (IC) is a clinical syndrome that presents as urinary frequency, urgency and/or pelvic pain in absence of any other identifiable pathology.
Prevalence may be higher than once believed.
Women more commonly affected than men (10:1 ratio), significant healthcare costs.
Symptoms reduction is possible in most patients.
Etiology for IC is not known and may involve defects in the urothelium, mast-cell activation and neurogenic causes.
Alterations in the bladder urothelial surface may cause increased permeability, leading to surface sensitization.
Increased numbers of mast cells are noted in bladders of IC patients and various vasoactive and inflammatory substances contents of mast cells may lead to neuronal sensitization.
Peripheral nerves associated with pain, or C-fibers and substance P are increased in number and may contribute to pelvic pain.
Target the pathophysiological aspects of IC and include oral and intravesical modalities.
In severe cases, combinations of oral and intravesical medications may be required to achieve relief.
First-line agents include pentosan polysulfate sodium (Elmiron®), hydroxyzine and amitriptyline.
Second-line agents include cimetidine, antiseizure medications (i.e., gabapentin), immunosuppressive agents (i.e., cyclosporine A), and narcotics.
Second-line therapy used in conjunction with oral medications.
Includes heparin sulfate, dimethylsulfoxide, hyaluronic acid and anesthetic cocktails.
Small studies with botulinum toxin type A show some promise.
Research will be directed toward a better comprehension of neurophysiology.
Urinary markers, such as uronic acid or antiproliferative factor, may be utilized to facilitate diagnosis of IC.
New possibilities for treatment include:
– liposomes, which are vesicular agents that may provide an alternative for repair of damaged urothelium, or
– pregabalin, a medication related to gabapentin, for pain management.
Large randomized, controlled studies are necessary to determine the efficacy of future treatments.
