Abstract
Background:
Bladder pain syndrome/interstitial cystitis (BPS/IC) is a persistent pain perceived in the urinary bladder region, accompanied by at least one symptom, such as pain worsening with bladder filling and daytime or nighttime urinary frequency without any proven infection or obvious pathology. The aim of this study is to evaluate the efficacy and safety of pentosan polysulfate (PPS) in patients with BPS/IC.
Methods:
Systematic search was performed by PRISMA checklist. Electronic databases, including PubMed and Cochrane library, were checked until 2021 using keywords: ‘pentosan polysulfate’, ‘pain syndrome’, ‘interstitial cystitis’, and bibliography of relevant papers was checked.
Inclusion criteria:
Patients with confirmed diagnosis of BPS/IC and cystoscopy criteria – Hunner’s lesions. Exclusion criteria included hypersensitivity, pregnancy, lactation, and oral therapy for BPS/IC in the period of 1 month before the study and abstracts or unpublished papers.
Results:
In total, 13 clinical trials were included in systematic review and 7 were included in meta-analysis. Studies evaluated the effectiveness and safety of oral PPS versus placebo or other treatment options. In the first meta-analysis, three studies compared oral PPS with placebo: [relative risk (RR) = 2.07, 95% confidence interval (CI): 1.37–3.13, p = 0.0006]. The second meta-analysis of two studies compared oral PPS with another treatment options (intravesical liposome and CyA): (RR = 0.44, 95% CI: 0.10–1.93, p = 0.28). The third meta-analysis of two studies included intravesical regimen of PPS compared with intravesical placebo: (RR = 1.09, 95% CI: 0.54–2.22, p = 0.80). The majority of studies do not report any particular serious side effects.
Conclusion:
PPS treatment has a statistically significant effect over placebo on the subjective improvement of patients with BPS/IC. There was no difference between PPS and other treatment options. Intravesical regimen of PPS had no significant impact on response rates. None of included studies reported severe side effects after intervention.
Introduction
Bladder pain syndrome/interstitial cystitis (BPS/IC) is defined as persistent or recurrent pain perceived in the urinary bladder region, accompanied by at least one other symptom, such as pain worsening with bladder filling and daytime or nighttime urinary frequency without any proven infection or other obvious local pathology. 1 Patients more often use the term ‘interstitial cystitis’ to refer to BPS. Thus, the use of both terms in parallel is allowed to denote this disease. 2 Even though IC has been known and recognized for a long time, the causes and etiology are poorly understood at the moment. In this regard, approaches to the diagnosis and treatment of BPS/IC are controversial. The generally accepted initial treatment for BPS/IC is a conservative approach. Pharmacological therapy is prescribed in cases of no effectiveness or manifestation of severe symptoms in which conservative treatment is unlikely. It implies the use of including analgesics, antibiotics, and antidepressants. If such treatment is unsuccessful, surgical intervention is considered. 3 Despite the various pharmacological therapies, pentosan polysulfate (PPS) stands out from the rest. 4 The first articles devoted to the study of PPS have been known since the last century. However, there is still insufficient evidence on the efficacy and safety of its use. Several randomized, placebo-controlled trials have reviewed the use of oral PPS in patients with BPS/IC, resulting in appropriate efficacy of the drug compared with placebo.5–7 This study aims to evaluate the efficacy and safety of PPS in patients with BPS/IC.
Materials and methods
The present systematic review included all published research articles that evaluated the effectiveness of PPS in terms of quality of life, subjective, and objective symptoms regression in patients with BPS/IC. Furthermore, a secondary outcome measure evaluated side effects and safety after the intervention. Our systematic review was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses 2020 checklist. 8
The included studies were randomized clinical trials (RCT) and non-RCTs (prospective controlled, prospective cohort, retrospective studies, and other types of studies) that included a minimum of 10 patients. Only articles written in English were included. Abstracts of conferences and unpublished papers were not included. Institutional Review Board (IRB) approval was not requested as this study is a review of published studies. The present systematic review has been registered in the PROSPERO international prospective register of systematic reviews by the National Institute for Health Research (NIHR). The registration number is PROSPERO 2021 CRD42021270600. 9
An electronic database search was conducted using PubMed, MEDLINE (last searched August 5, 2021), the Cochrane Library (last searched August 7, 2021), Google Scholar, and ClinicalTrials.gov to identify articles published until August 2021. The search used a combination of the following terms: ‘pentosan polysulfate’, ‘bladder pain syndrome’, and ‘interstitial cystitis’. In addition, MeSH terms were used in PubMed: (‘Cystitis, Interstitial/therapy’[Mesh]) AND ‘Pentosan Sulfuric Polyester’[Mesh]; in The Cochrane Library the following additional terms were used: [mh ‘Cystitis, Interstitial’/DT, TH] and [mh ‘Pentosan Sulfuric Polyester’] in Trials.
The search was conducted independently by two investigators (B.G. and L.P.). Following the search, all articles were rechecked based on their titles and abstracts. The full texts of the studies that appeared to be appropriate according to their titles and abstracts were then reviewed. The reference lists of eligible trials were searched for additional potential studies.
Two investigators (B.G. and L.P.) independently read the full texts of the preselected articles to verify the eligibility of the articles based on the presence of comparisons between PPS and any other treatment options for BPS/IC. After this step, studies were excluded if there were duplicate datasets.
Any disagreements regarding the inclusion or exclusion of preselected studies and any other disagreements during the review process were resolved with the help of the third author (G.K.). The included studies were independently collected by two authors (B.G. and L.P.) using a standardized data extraction procedure (authors, publication year, study design, patient characteristics, intervention, and outcomes).
The primary analysis aimed to assess the objective or subjective effectiveness of achieving symptoms regression in patients with BPS/IC. Subsequently, subjective efficacy was analyzed as a dichotomous variable (efficacious versus non-efficacious) based on the patient’s subjective opinion after treatment and therefore the quality of life. Secondary analysis aimed to assess safety.
The inclusion criteria for this systematic review were clinical studies (cohort and randomized) that evaluate adult patients of both sexes with a clinically confirmed diagnosis of BPS/IC, bladder pain duration of more than 6 months, and cystoscopic criteria – petechial hemorrhages or Hunner’s lesions. Articles with a statistically significant difference in the mean patient age in the study groups were excluded. Furthermore, the exclusion criteria were studies that included pregnant and lactating patients, patients with hypersensitivity to the drug, and people who received oral therapy for BPS/IC in the period of 1 month before the study.
Risk-of-bias assessment was conducted for each of the studies included using the Cochrane Handbook for Systematic Reviews of Interventions. 10 Two investigators (B.G. and L.P.) independently assessed the quality of the selected studies. A third investigator (G.K.) was involved when disagreements occurred. Following the Cochrane Handbook for Systematic Reviews of Interventions, the RoB 2 tool 11 was used to assess the risk of bias in randomized-controlled studies, and ROBINS-I 12 was used for non-randomized studies (prospective controlled, prospective cohort, retrospective studies, and other types of studies). These tools were also used to assess the risk of bias arising from reporting biases due to missing results in synthesis.
Results
We used an electronic search of PubMed, MEDLINE, Google Scholar, Clinical Trials, and the Cochrane Library databases and received 2559 articles. After removing duplicates and searching for the title and abstract of the articles, 54 publications were selected. Of these, 36 articles were excluded after reading the full texts. Two studies were comments to the authors, nine were reviews, one article was a guideline, two were not in English, and another was an abstract. Also, due to the absence of relevant inclusion criteria, 24 studies were excluded, and 2 articles were excluded due to the absence of the results. Regarding the studies without appropriate inclusion criteria (n = 24), they contain five case series studies, four that evaluated PPS dosage, three articles included heparin, two times evaluated to start PPS, in two studies, PPS was used for chronic non-bacterial prostatitis, and one to prevent recurrent urinary tract infections. One study assessed epidermal growth factor and interleukin-6 levels, one aimed to evaluate cost-effectiveness, vision side effects, validation of the O’Leary-Sant interstitial cystitis symptom index, and one included animals. Two studies were excluded because there were no cystoscopy criteria.
Furthermore, we checked the references of the selected articles for acceptable studies (n = 251). We selected 52 articles for full-text review, but 51 of them were duplicates, and 1 publication was excluded due to noncompliance with our criteria (Figure 1).
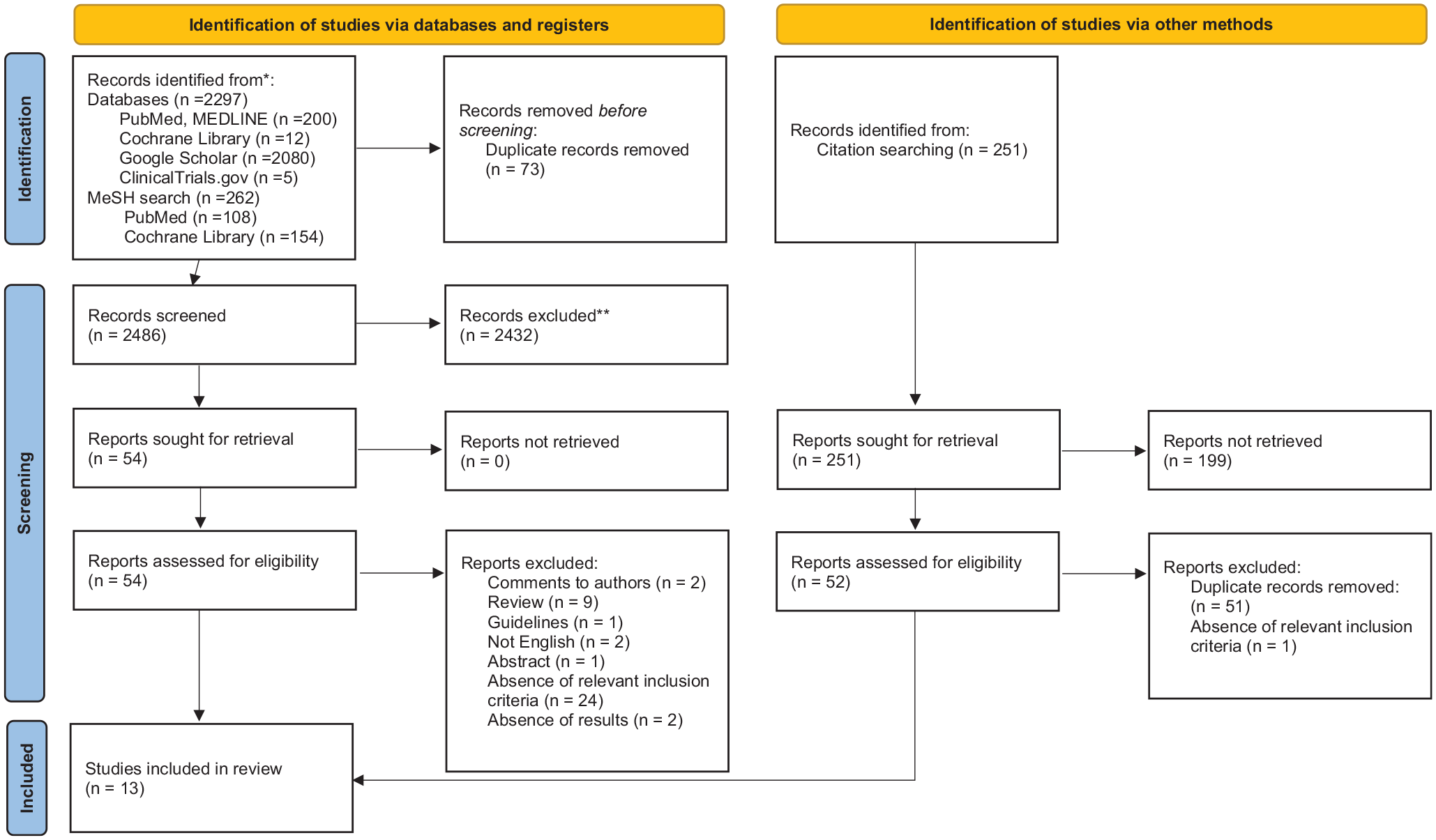
PRISMA flow diagram.
In conclusion, 13 clinical trials were included in our systematic review and 7 in the meta-analysis. Nine of them were randomized-controlled trials,4–7,13–17 one prospective longitudinal study, 18 and three retrospective studies19–21 (Table 1).
Description of the included articles.

The included studies evaluated the effectiveness and safety of oral PPS versus placebo5,6,14–16 or other treatment options. For example, intravesical liposome, 18 oral cyclosporine A (CyA), 17 intravesical chondroitin sulfate, 21 and oral hydroxyzine. 7 In the Waters et al.’s 19 retrospective study, the PPS group was compared with the control group, which had to be taking at least one oral medication as treatment for their symptoms of BPS/IC. Simsir et al. 20 evaluated the effectiveness of oral PPS in comparison with combination therapy of PPS and hydrodistension. Moreover, a randomized study by Sant et al. 7 estimated a combination of oral PPS and hydroxyzine and their effectiveness.
Intravesical PPS was evaluated in two randomized placebo-controlled studies.4,13
Oral PPS has shown a statistically significant improvement in BPS/IC in five RCTs. Criteria for efficacy evaluation were the average O’Leary-Sant Interstitial Cystitis Symptom Score (ICSI) points change in comparison with initial data7,14 and the patient’s subjective improvement.6,15,16
However, Holm-Bentzen et al. in 1987 published a randomized multicenter placebo-controlled trial on 115 patients with BPS. The authors used two different protocols to evaluate the effectiveness of PPS. Protocol A included 43 patients with clinically and pathologically verified interstitial cystitis (28 or more mast cells per mm2), and protocol B included 72 patients with a painful bladder and unspecific histological findings. The patients received treatment (PPS 400 mg/day or placebo) during a period of 4 months. The results of this trial showed no difference between the pre- and post-intervention values in both groups and both protocols regarding symptoms, urodynamic parameters, cystoscopic appearance, and mast cell counts. However, bladder capacity increased in the PPS group compared to placebo, but the authors have not mentioned the statistical significance. 5 Another prospective study showed no promising results after the intervention. In 2009, Chuang et al. evaluated the safety and efficacy of intravesical liposomes compared to oral PPS for BPS/IC. Intravesical liposomes are mucosal protective agents that can positively affect the dysfunctional epithelium of the bladder. In total, 24 patients were monitored at baseline, 4, and 8 weeks. Intravesical LP-08 80 mg/40 cc distilled water was administered once weekly and oral PPS (100 mg) three times daily. After 4 weeks of treatment, oral PPS and intravesical liposome instillation achieved similar efficacy. 18
Regarding the randomized-controlled trials that have shown efficacy, the first was published in 1987 15 and the last in 2021. 14
Published in 1987, randomized study by Parsons et al. evaluated 62 patients with cystoscopically confirmed diagnosis of BPS/IC. They were randomized into two groups: oral PPS 100 mg three times a day and placebo. The drug did better at all end points. The average voided volumes were significantly improved on PPS therapy (p = 0.009), and subjective improvement was seen for all symptoms (p = 0.02). 15
Mulholland et al. 1990 included 110 patients in their multicentred trial for the same objective. Overall, 28% of the PPS group reported overall improvement (greater than 25%) and 13% of the placebo-treated patients (p = 0.03). 6
In 1993, Parsons et al. published a multicenter double-blind randomized trial including 148 patients who had pain, urgency, negative urinary cytology, and cystoscopic findings of petechial hemorrhages. After 3 months of treatment with oral PPS treatment (100 mg three times daily) and placebo, 24 of 74 patients in the treatment group and 12 of 74 in the placebo group evaluated subjective 50% improvement (p = 0.01). 16
In a pilot clinical trial of oral PPS and hydroxyzine, Sant et al. published their results to estimate effectiveness and safety. In total, 121 patients were enrolled with cystoscopically confirmed diagnosis of BPS/IC. The follow-up period included 10 months. There was no significant difference in the global response rate between the hydroxyzine (19 of 61, 31%) and no hydroxyzine treatment groups (12 of 60, 20%) (p = 0.26). However, there was a trend for a higher response rate in the PPS treatment arm (20 of 59, 34%) as compared with no PPS (11 of 62, 18%, p = 0.064). Moreover, oral PPS and hydroxyzine combined demonstrated the highest response rate (12 of 30, 40%). 7
The most recent study by Kasyan et al. in 2021 has shown successful results. The authors conducted the randomized, double-blind multicenter trial that included 90 patients with BPS/IC. They completed the ICSI questionnaire at baseline and after treatment. Statistical analysis showed that treatment in the pentosan group was significantly more effective than in the placebo group (p = 0.014). 14
In addition, three retrospective studies were included in our qualitative synthesis. Published in 2000, retrospective study by Waters et al. included 54 patients with BPS/IC who were treated in 1:1 drug-control groups from May 1998. These patients were treated for 9.3 months, while the control group had taken at least one oral drug. Changes in frequency, urgency, and pain were greater in the treatment group and statistically significant (p = 0.11, p = 0.49, and p = 0.004, respectively). 19
Also, Sannan et al. published the results of their research in 2017. Of 80 patients, 51 received oral PPS and 29 received intravesical chondroitin sulfate. Both options had shown encouraging results after 6 months of treatment. However, the symptom score, the bother score, and the total score improved significantly after 3 months in the PPS group (p = 0.027, p = 0.035, and p = 0. 026, respectively). 21
A most recent retrospective study by Simsir et al. was published in 2019. The authors reviewed 415 patients in a single center from November 2005 to July 2015, where 339 were eligible based on inclusion criteria. However, 157 patients with BPS/IC had oral PPS 300 mg/day (prescribed TID) and 182 received combination therapy with oral PPS and hydrodistension. The patients were examined at 3, 6, and 12 months after treatment. ICSI, ICPI (international cystitis problem index), and VAS (visual analog scale) were used to evaluate effectiveness. On the basis of these scales, combined therapy has shown positive results (p < 0.001, p = 0.02, p < 0.001, respectively). PPS treatment started just after hydrodistension was significantly more effective than PPS treatment alone, and combined treatment significantly reduced the rate of noncompliance. 20
One randomized 3- and 6-month clinical trial study has found more efficient treatment. Sairanen et al. included 87 patients in intravesical dimethyl sulfoxide (DMSO) and bacille Calmette-Guérin (BCG) groups during a 3-month trial and 64 participants in a 6-month study, in which 32 received oral PPS and 32 received oral CyA. According to the health-related quality of life (HRQoL) questionnaire, CyA had more impact on the majority of domains and, therefore, had more impact on emotional well-being, social functioning, activity limitation days, pain, and physical capacity than PPS treatment (p < 0.05). 17
Intravesical administration of PPS was discussed in two randomized-controlled trials. First, Bade et al. published an RCT that included 20 patients with a confirmed diagnosis of IC. Participants were invited twice a week to the hospital where health care practitioners performed intravesical instillation of PPS (300 mg in 50 ml of 0.9% NaCl) or placebo. Then, they started to perform self-catheterization at home. The duration of treatment was 3 months. As a result, 4 out of 10 in the treatment group and 2 out of 10 in the placebo group gained significant improvement in symptoms (p = 0.047). After 18 months from the beginning of the study, 8/10 in the PPS group and 4/10 in the placebo recorded subjective improvement. 13
After 11 years, Davis et al. published the results of their RCT in which 41 participants were enrolled. The first group (n = 21) received intravesical + oral PPS, and the second (n = 20) received intravesical placebo + oral PPS for 6 weeks. Points on the ICSI scale were measured at baseline, 6, 12, and 18 weeks. However, an overall improvement was shown in 18 patients in the treatment group and 18 in the placebo group (p = 0.6). 4
Two reviewers (B.G. and L.P.) evaluated the risk of bias in each of the studies included according to the Cochrane Handbook using RoB 2 for randomized-controlled trials and ROBINS-I for non-randomized trials. Any disagreements were resolved by a third reviewer (G.K.).
Visualization tools were created by the ROBVIS app. 22 This app created ‘traffic light’ plots of the domain-level judgments for each result and weighted bar plots of the distribution of risk-of-bias judgments within each bias domain.
Two non-randomized trials had a moderate risk of bias, one low and one serious, according to the ROBINS-I instrument (Figure 2). Also, based on RoB instrument 2 (Figure 3), two randomized trials were at high risk, of bias, four had some concerns, and three were low.

ROBINS-I tool for non-randomized studies of interventions (traffic light plot).

RoB2.0 tool for randomized-controlled trials (traffic light plot).
The primary analysis focused on the subjective effects of PPS in patients with BPS/IC. We have conducted three meta-analyses with different comparisons to avoid biases. In the first meta-analysis, three studies were included and compared oral PPS with placebo: [relative risk RR = 2.07, 95% confidence interval (CI): 1.37–3.13, p = 0.0006]. The heterogeneity for this comparison was 0%. Consequently, treatment with oral PPS showed efficacy over placebo in subjective improvement of BPS/IC symptoms (Figure 4).

Comparison between PPS and placebo: subjective effectiveness in patients with BPS/IC.
The second meta-analysis of two studies compared oral PPS with other treatment options (intravesical liposome 18 and CyA) 17 : (RR = 0.44, 95% CI: 0.10–1.93, p = 0.28). The heterogeneity for this comparison was 83%. Consequently, there was no significant difference between different treatment options in the subjective improvement of BPS/IC symptoms (Figure 5).

Comparison between oral PPS and other treatment options: subjective effectiveness in patients with BPS/IC.
The third meta-analysis of two studies included an intravesical regimen of PPS compared to an intravesical placebo: (RR = 1.09, 95% CI: 0.54–2.22, p = 0.80). The heterogeneity for this comparison was 34%. Hence, there was no effectiveness in intravesical administration of PPS in subjective improvement of BPS/IC symptoms (Figure 6).

Comparison between intravesical regimen of PPS and intravesical placebo: subjective effectiveness in patients with BPS/IC.
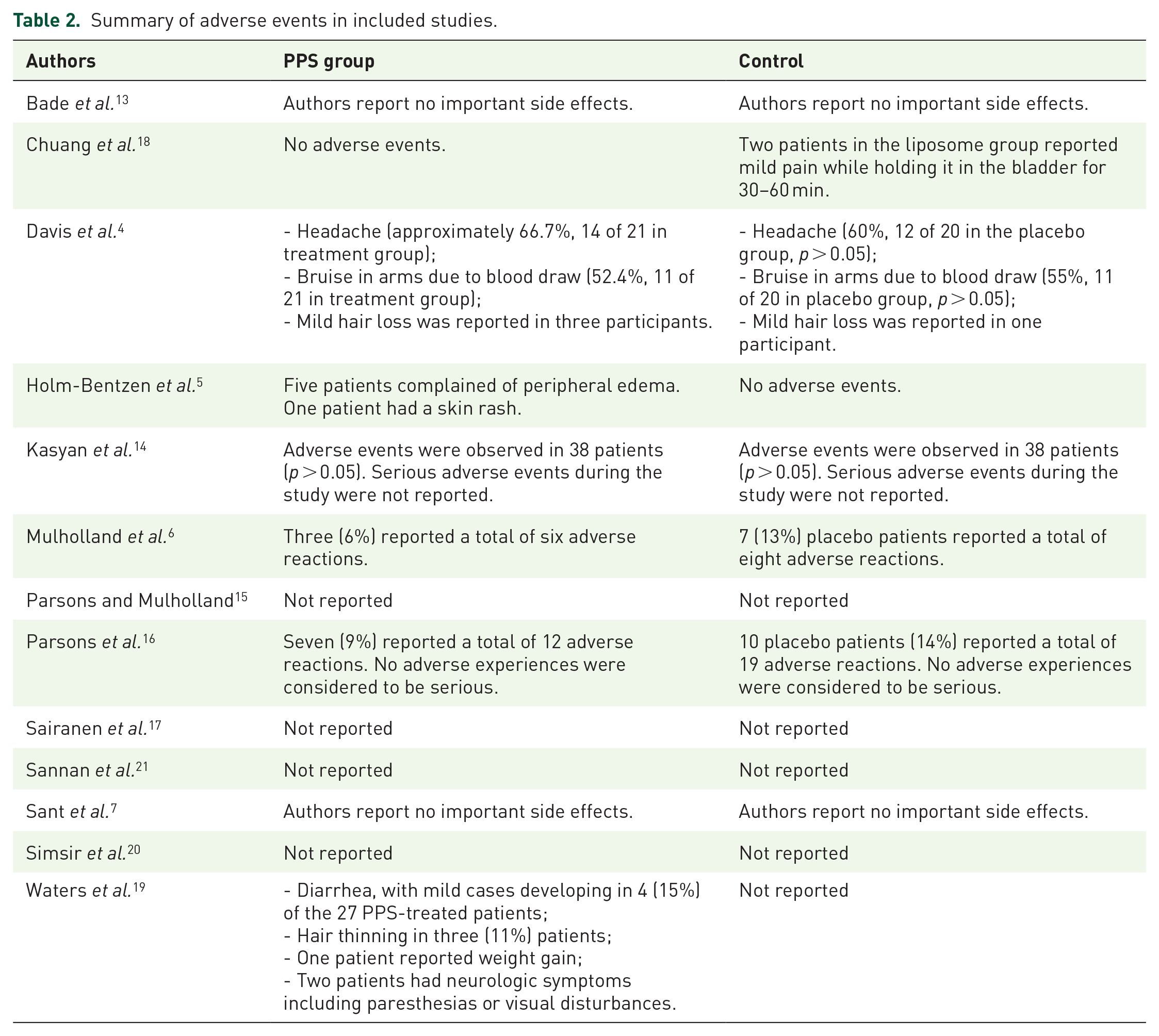
Secondary analysis aimed to assess safety. The adverse events in the included studies are summarized in Table 2.
Summary of adverse events in included studies.
Discussion
The question of the effectiveness of pentosan was raised long ago, but it has not been adequately studied. One of the first to attempt a meta-analysis in a paper by Hwang et al. 23 in 1997 concluded that pentosan was more effective than placebo in the treatment of interstitial cystitis. Nevertheless, the number of articles used for their meta-analysis was rather low and required a further and more detailed study of this question. One of the most recent publications is the systematic review by Arndt van Ophoven et al. published in 2019. 24 This article was useful to study, as it considered only RCTs on the effectiveness of pentosan in the treatment of BPS/IC. As a result of this study, the authors also concluded that pentosan was more effective than placebo. But until now, there have been no systematic reviews in which the cystoscopy criterion was mandatory. It has a significant diagnostic value for the study of BPS/IC, allowing the differentiation of patients with Hunner’s lesions, which in turn is an essential aspect for determining the role of PPS used in therapy. The EAU2021 Guidelines 25 recommend cystoscopy due to its crucial diagnostic value, which allows one to obtain objective results and make a correct prognosis. It is noted that standardization will help improve the comparability and quality of studies. Thus, we excluded studies that lacked a cystoscopic criterion and focused on a narrower but more valuable and important diagnostic criterion. For example, we did not include the research by Nickel et al., 26 in which the authors did not adhere to the cystoscopic criterion but only included the ICSI scale. This leads to the risk of data bias because it is possible to include patients with other diagnoses, such as BPS/IC, which exposes the study quality to deterioration. This publication summarized that PPS did not have a significant therapeutic effect compared to placebo. Although in most cystoscopy studies, the authors concluded that PPS was clinically effective. If patients with normal bladder mucosa are included in the study, there is a high probability that the result may be distorted and biased.
As for the side effects of PPS, dizziness, vomiting, diarrhea, heartburn, and sleep disturbance were noted. In addition, there are known cases associated with long-term use of oral PPS, leading to maculopathy. This may be due to the cumulative effect of macular degeneration. Patients receiving doses greater than 1500 g of PPS are at increased risk of retinal atrophy due to macular toxicity. In this regard, it is recommended to use the drug with caution, and patients who are prescribed doses that exceed 500 g should undergo an eye examination with visualization of the retina.27–29 In addition, it is worth noting that some of the studies included in our review used intravesical administration of PPS. However, this route of drug administration did not show a significant difference or a better effect relative to the oral route. Moreover, the use of intravesical administration of PPS is rather controversial because it is inconvenient for the patient. Therefore, the use of PPS orally is more appropriate and comfortable.4,13
The limitations of our systematic review and meta-analysis include the low quality of some of the studies included. Most of the studies included had a small sample size and were published a long time ago. Regarding implications for future research, we recommend that researchers seek new therapeutic approaches (intravesical or oral) to manage this condition in patients with BPS/IC.30 Going forward, we need more robust, well-conducted, prospective and randomized studies that include cystoscopic criteria in the diagnosis of BPS/IC.
Conclusion
According to our systematic review and meta-analysis, oral PPS treatment has a statistically significant effect over placebo on subjective improvement in patients with BPS/IC. Also, it is important to note that there was no difference between PPS and other treatment options, although, there were only two studies in this comparison. In addition, the intravesical regimen of PPS had no significant impact on response rates. However, health care practitioners should be mindful of patient preference when considering intravesical insertion of PPS. Nevertheless, none of the included studies reported severe side effects after the intervention.
Footnotes
Author contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
