Abstract
Women are delaying starting their families. High-profile women such as Cherie Blair and Madonna are having successful pregnancies with healthy babies in their forties. Is this sending out messages to women that delaying conception during their twenties and thirties is a good idea? Fertility treatment can overcome certain age-related hurdles but is certainly not the magic wand some expect it to be.
Keywords
The average life expectancy for a woman born in 2002 in the UK is 81 years, compared with 45 years in 1901. More women now have careers and are sometimes voluntarily delaying starting a family until they are established. Between 1976 and 1998 the conception rates for women aged 35–39 years increased by 82% [101].
The average number of children born per couple in 2004 was less than two, compared with nearly three in the 1960s. This decline is more marked in areas of higher socioeconomic status within the UK [101].
Some women may have not yet met a partner whom they wish to have a child with. Some women wish to undergo fertility treatment alone and live as a single-parent family. In addition, with the current high divorce rate, many people consider embarking on a ‘new family’ with a new partner at an older age and possibly following sterilisation. The number of women marrying younger men has doubled over the last 10 years. Based on current statistics, one in five women will never have a child [1].
From a financial perspective it has been calculated that it costs approximately £50,000 to bring up a child [102]. If a woman delays childbearing until her career is established, this financial outlay may be more easily achievable compared with women having children prior to gaining financial security.
Infertility treatment may be able to overcome certain problems but it still cannot significantly overcome aging. This can be demonstrated in the live-birth rate following in vitro fertilization (IVF) quoted by the recent National Institute for Clinical Excellence (NICE) guidelines [2]. A recent review by Klipstein and colleagues of 2705 women aged 40 years and over undergoing IVF cycles with their own oocytes showed a live-birth rate of 13.9% at 40 years, 1% at 45 years and 0% at 46 years [3].
The association between advanced maternal age and reduced reproductive potential is well established.
A study published in 1957 examined the relationship between the age of the female partner and fertility [4]. The findings of this study are outlined in
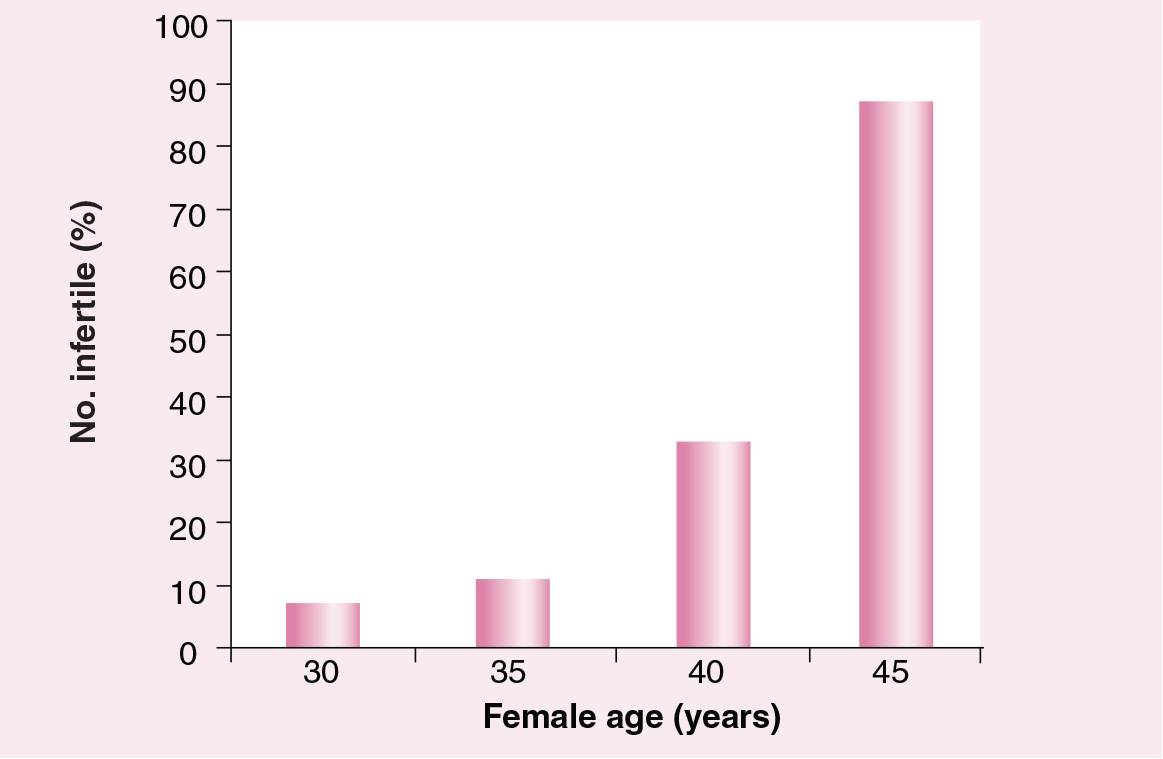
Female age and infertility rates.
However, in women of the same age, individual variations exist with regard to their ovarian responses. Chronological female age is universally seen as indicative of the qualitative alterations in the remaining follicle pool but cannot serve as the only marker for ovarian status. More recently attention has also focussed on the male and the possibility that male age should not be overlooked when counseling patients regarding potential outcome of a pregnancy.
In this article we will review the current literature in this area and also explore potential strategies to overcome the impact of aging on reproduction, and strategies that may improve clinical pregnancy rates in older women.
Fecundity & age
Fecundity, the ability to bear a child, decreases with increasing female age. It has long been recognised [5] and appears to involve several factors:
Germ cells in the female are not replenished during life, therefore attrition and utilization of follicles leads to a decline in the number of oocytes from birth to menopause;
The quality of existing oocytes diminishes with age [6];
Intercourse frequency may decline with age.
However, age alone does not completely determine the potential response to and outcome of fertility treatment.
Physiology of the aging ovary
In the UK the average age for the menopause is 51 years. The ovary obtains the maximum number of germ cells during fetal development. At birth a woman has an average of 2 million oocytes, which will gradually decline until the menopause is reached. In addition, the quality of the oocytes will also decline with age, leading to lower fertilization rates and also an increasing risk of aneuploidy in embryos. The risk of having a baby with Down's syndrome at 40 years is 1 in 105, rising to 1 in 35 at 44 years (
Relationship of Down's syndrome incidence to mother's age.
Assessing ovarian reserve
Ovarian reserve can be defined as the total number of oocytes remaining in the ovary. This gives an indication of how well an ovary will respond to exogenous stimulation such as that given during an IVF cycle.
Predictors of ovarian reserve can be divided into biochemical and imaging modalities. Imaging methods include the antral follicle count and measurement of ovarian volume. Biochemical markers include follicle-stimulating hormone (FSH), inhibin B, anti-mullerian hormone (AMH) and estradiol [6]. However, none of the biochemical or ultrasound markers reflect the quality of the oocyte and therefore are not good predictors of live-birth rate.
Antral follicle count & ovarian volume
This is assumed to correlate with the number of primordial follicles within the ovary. If there are less than five antral follicles the ovarian reserve is said to be reduced; this correlates well with ovarian response to stimulation in IVF cycles [8].
With regard to ovarian volume, the bulk of the ovary in the cortex is made up of antral follicles and, in the absence of a corpus luteum, the total volume correlates well with the total number of antral follicles. When the ovarian volume is less than 3 ml the ovarian reserve is said to be reduced [9]. Ovarian volume may be better calculated using 3D ultrasound imaging.
Biochemical markers
FSH is a hormone that is released from the anterior pituitary gland. It is one of the longest established parameters in assessing ovarian reserve. Elevated FSH seems to be associated with a higher rate of pregnancy loss and reduced pregnancy rate in all age groups [10]. Its regulation is by negative feedback from estradiol and inhibin.
FSH reflects the number of early antral follicles that can be recruited. It is known that the serum levels begin to increase approximately a decade prior to the menopause; however, there can be marked intercycle variation. Although FSH is widely used in an attempt to predict ovarian response, this intercycle variation can lead to difficulty in interpreting results [11].
Inhibin B is produced by early antral follicles. Its action is to have an inhibitory effect on pituitary secretion and synthesis of FSH.
Taken on day three of the menstrual cycle, the levels correlate with ovarian reserve and higher levels are associated with better outcomes [12]. The ability of inhibin B to predict pregnancy has not been confirmed.
AMH is produced from granulosa cells of the pre- and antral follicles. It is a novel serum indicator of ovarian reserve and influences ovarian sensitivity to FSH.
Recent studies have shown an association between reduced baseline AMH and poor response to IVF. AMH appears to correlate more strongly with antral follicle count than the other biochemical markers discussed earlier. There is an additional advantage in that there is minimal intercycle variation in AMH levels. AMH is not used routinely as a marker for ovarian reserve in the UK at present, but may become more widespread in the future [13].
Over the counter tests for ovarian reserve
Over the counter tests and tests available via the internet allow women to assess their ovarian reserve in their own home. Many of the tests that can now be bought in chemists and via the internet measure a number of hormones including FSH, AMH and inhibin B as a predictor of ‘fertility’.
An example of such a test is the ‘Plan Ahead Fertility Test’ [103], which claims to estimate how close a woman is to her menopause. The cost of this test over the internet is £179.99. The hormone levels measured in this test and others like it can give women misleading results, as, although the quantity of eggs remaining in the ovary is important, these tests cannot predict or represent the declining egg quality with age and the associated aneuploidy rates.
Aneuploidy & age
The incidence of aneuploidy is one of the key factors that determines oocyte quality. With increasing maternal age, aneuploidy increases. At the age of 25 years the risk of a Down's syndrome fetus at mid-pregnancy is 1 in 1081. At 45 years this increases to 1 in 22.8. In addition, both trisomy 18 (Edward's syndrome) and trisomy 13 (Patau's syndrome) increase with advancing maternal age.
In women who are considering fertility treatment at advancing age, these statistics are important, not only because fetuses with aneuploidy are more likely to miscarry but also because if a baby with an aneuploidy such as Down's syndrome is born to older parents, there are long-term care implications for that child. Furthermore, diagnostic testing, such as amniocentesis, during the pregnancy carries a risk of miscarriage of 1% [14].
Paternal age
It has been suggested that while the age of 35 years is the ‘amber light’ for a woman's reproductive life, the age of 40 years should be the ‘amber light’ for men. There is evidence from IVF studies that the ability to conceive is reduced when the male partner is over 40 years of age [15].
It is known that the quality and quantity of sperm can decline with age. This may be a reflection of the reduction in Leydig cells with age and also additional factors such as arteriosclerotic lesions and thickening of the seminiferous tubules.
In addition, some studies have shown an increased risk in fetal death if the male partner is over 50 years old. This association appears to be stronger for late fetal death than early fetal death [16].
There does not appear to be an increase in aneuploidy in children born to men who are over 40 years; however, some studies have shown an increased risk of congenital malformation of the extremities such as syndactyly, polydactyly and limb-reduction defects [17].
Pregnancy & increasing maternal age
With regard to the endometrium, this does not appear to be a limiting factor for successful pregnancy in women of advanced maternal age. Evidence for this comes from donor egg cycles where the endometrium is initially prepared with estrogens and progestogens [2].
The miscarriage rate is known to increase with maternal age. Between 25 and 29 years the miscarriage rate is 10%, whereas the miscarriage rate between 40 and 44 years is 33% [18]. This is largely a reflection of the increase in chromosomal abnormalities with advancing age. At 25 years of age, the total risk of chromosomal abnormalities is 1 in 476, while at 45 years it is 1 in 21 [19].
Considering complications during the pregnancy, in women aged over 40 years hypertensive complications are twice as common as among younger women, as is the risk of delivering a low birthweight or preterm infant [20].
A number of reports have also shown that women aged over 40 years are more likely to have operative vaginal delivery and cesarean section than younger women. This may be a reflection of the anxiety felt by medical practitioners when dealing with pregnancies in women of advanced maternal age.
Pregnancy in older women does, therefore, appear to become ‘high risk’ and involves increased obstetric surveillance and, often, intervention.
Can we overcome the aging process with IVF?
IVF with advanced maternal age
The success rates of IVF in the UK using the patients' own eggs show a rapid decline with advancing maternal age [2]. The live-birth rate per treatment cycle with increasing age is shown in

Chances of a live-birth rate per treatment cycle with in vitro fertilization.
This decline is primarily due to a reduction in oocyte quality. Currently the UK NHS funds approximately 25% of all IVF cycles [104].
Recent NICE guidelines advised an upper age limit of 39 years for NHS-funded treatment based on efficacy, although some Primary Care Trusts have limited funding to women less than 39 years as a rationing tool. The majority of IVF units within the UK would decline IVF treatment in women aged over 44 years on the basis of the poor predicted outcomes with the patient's own eggs.
In order to address this significant decline in live-birth rates in older women following fertility treatment, a number of strategies have been proposed. These include using donor eggs from younger women, as this significantly improves the live-birth rate in older women [21]. In addition, as we are aware that aneuploidy in older women may account for the lower success rates through poorer implantation rates and increased likelihood of miscarriage, techniques such as preimplantation genetic screening (PGS) have been developed in an attempt to detect aneuploid embryos prior to transfer [22].
Some women are choosing to store their eggs at younger ages in an attempt to overcome the increase in aneuploidy with age [23]. In addition, new strategies to manipulate oocytes to correct aneuploidy are being developed [24]. These are discussed below.
Donor eggs
Using eggs from donors who are younger is a potential option for older women, as the success rates with assisted reproductive techniques significantly improve with ‘younger’ eggs.

Live births per transfer for ART cycles using fresh embryos from own and donor eggs, by ART patient's age (2003).
The oldest reported woman who has conceived using donor eggs is Adriana Iliescu, a 66-year-old Romanian woman who gave birth to a baby girl named Eliza Maria in 2005 [105]. The oldest British woman who has conceived using donor eggs is Patricia Rashbrook, who is 63 years old and received the treatment in Russia [106]. She is due to give birth later this year.
In April 2005 the law in the UK changed to remove donor anonymity, allowing donor-conceived people the opportunity to find information about their biological origins. Donor oocytes have always been in short supply but following this legislation it has become more of a problem [107].
With the small number of donor eggs available, how do the units decide who should receive them? Should older women take priority over women who have had ovaries removed due to malignancy or have undergone chemotherapy which has lead to a premature menopause? The NICE guidelines have indicated that the use of oocyte donation after the start of the natural menopause (between the ages of 45 and 55 years) is controversial [2].
Overcoming aneuploidy
Selection of an embryo that has the highest implantation potential remains a significant challenge. Several strategies have been employed to address this, including morphological characteristics of the embryo, culture of the embryo to blastocyst and genetic diagnosis.
Morphological criteria include fragmentation of the embryo, cell number, cell size and early cleavage. Culture to blastocyst has been postulated to allow self selection of the embryos that are capable of blastulation and exclusion of embryos showing cell arrest. A recent study by Papinikolaou and colleagues compared pregnancy and delivery rates following IVF with single-blastocyst-stage versus single-cleavage-stage embryos [26]. The results showed a higher rate of delivery in the blastocyst-stage group of 32 versus 21.6% in the single-cleavage-stage embryo group.
PGS is a technique used to identify genetic defects in embryos created through IVF prior to transferring them into the uterus. In humans, PGS was first performed in the mid-1980s and by May 2001, 3000 PGS cycles had been reported. The procedure involves blastomere biopsy at the 6–8 cell stage of the embryo (day 3). These cells are then analysed using either polymerase chain reaction or fluorescence in situ hybridisation techniques [108].
Initially these techniques were used to detect embryos carrying abnormalities inherited from couples known to be at risk of transmitting a genetic disorder. More recently, use has extended to screening for aneuploidies in couples who are not known to carry a genetic disorder but are at increased risk due to advancing maternal age [27]. In a recent European Society of Human Reproduction and Embryology review, 33% of the PGS cycles performed were for detection of age-related aneuploidy [22].
If PGS were used to detect aneuploid embryos, theoretically we would expect to see an improvement in the pregnancy rate in older women if euploid embryos were replaced. Unfortunately, this as not been confirmed in the literature and pregnancy rates do not reflect this assumption [28]. There does appear to be some benefit in that if the majority of embryos created as a result of the treatment appear to be aneuploid it can direct couples to considering alternative strategies such as egg donation or adoption [29].
Egg freezing
Egg freezing is a relatively new technique. It was offered initially to women prior to undergoing chemotherapy or radiotherapy, or prior to oophorectomy [102]. A woman may be more likely to conceive successfully at the age of 40 years with her own ‘frozen’ 30 year-old eggs than with her own ‘fresh’ 40 year-old eggs. Drawbacks are analogous to embryo cryopresevation, as a stimulated cycle is required.
The first baby in the UK to be born using frozen eggs was a baby girl born in 2002 [109]. Subsequently some women without partners are choosing to store their frozen eggs in their early thirties in an attempt to prevent the age-related decline in egg quality. Approximately 200 babies have been born worldwide following oocyte preservation.
The results of this procedure worldwide have been variable, with a reported success rate of less than 2%, despite the improved success rate when combined with intracytoplasmic sperm injection [107].
A recent meta-analysis by Oktay and colleagues has suggested that by employing vitrification as a method of oocyte cryopresevation, the pregnancy rates may improve dramatically compared with the slow-freezing methods previously performed [30].
Correction of oocyte aneuploidy
This technique has been described in a paper by Palermo and colleagues and involves nuclear transplantation into mouse and human oocytes [24].
The background to this technique is the relationship that exists between oocyte aging and the nondysjunction of bivalent chromosomes during meiosis. In addition, mutations in mitochondrial DNA have been observed in the oocytes of older women. By transferring the germinal vesicle from an aged oocyte to a younger ooplast, aneuploidy may be prevented. This technique may offer the opportunity in the future to rescue oocytes with damaged mitochondria.
Conclusion
How old is too old for infertility treatment? Certainly with a patient's own eggs it has been demonstrated, by the NICE guidelines, that after the age of 40 years the success rates of IVF decline. With donor eggs the situation is significantly changed, with the success rates of IVF approaching that of younger women. With the decline in egg donors nationally, should we regulate who is receiving donor eggs? A pregnancy in an older woman is ‘high risk’, with inherent complications. This has cost and resource implications.
Future perspective
Life gives a woman a ‘fertility lifespan’. Perhaps we should concentrate on offering more flexible working patterns for women wanting to start a family at a younger age and educating women regarding their natural fertility, rather than trying to move the goal posts [110].
Executive summary
Fecundity decreases with increasing female age.
Biochemical and ultrasound modalities can indicate quantitative ovarian reserve.
Aneuploidy increases with increasing female age.
The success of in vitro fertilization (IVF) declines significantly in women aged over 40 years.
Preimplantation genetic screening can detect aneuploidy but does not improve live-birth rates.
Currently, egg freezing has a low success rate, but, by using vitrification to preserve eggs, higher live-birth rates may be achievable in the future.
Donor eggs significantly improve live-birth rates in older women following IVF, but the availability of donor eggs is not good.
