Abstract
Serotonin (5-HT) is one of the key mediators of gut motility, secretion and sensation. Most 5-HT is localized in the gastrointestinal tract. Particularly important for gut function and regulation are the 5-HT1P, 5-HT3 and 5-HT4 receptors. These receptors have been the focus of research evaluating the pathophysiologic mechanisms of irritable bowel syndrome (IBS) as well as targets for the development of novel agents to treat irritable bowel syndrome. Alosetron is one of three 5-HT3 antagonists currently available. The other two, ondansetron and granisetron, are primarily used in the treatment of chemotherapy-induced nausea and vomiting. Alosetron, which slows gut transit, has been approved for the treatment of severe diarrhea-predominant IBS (IBS-D) in women. This review will examine the common, yet therapeutically challenging, disorder IBS, as well as the role of alosetron in the treatment of IBS-D.
Irritable bowel syndrome (IBS) is a common, female-predominant disorder (sex ratio: 2–4:1) that is estimated to affect up to 20% of the population [1,2]. Although controversy exists, for the purpose of clinical management, IBS is often subdivided by predominant bowel type: diarrhea (IBS-D), constipation (IBS-C) and alternating diarrhea and constipation (IBS-A or IBS-M). Studies have also demonstrated the dramatic burden that this disorder has on the quality of life of patients compared with the general population and individuals with other chronic health conditions [3,4]. In addition, IBS patients utilize healthcare for both gastrointestinal (GI) and non-GI complaints more than non-IBS patients [5]. This, in turn, impacts the productivity of IBS patients so that absenteeism from work or school has been found to be three-times higher than that of non-IBS patients [6]. Unsurprisingly, economic studies have demonstrated that the disorder is costly to the healthcare and economic systems, creating an annual total cost estimated to be as high as US$30 billion [7–9]. IBS is a disorder for which there are seemingly many available treatments. However, apart from a few exceptions, the use of these therapies are not supported by high-quality, randomized, double-blind, placebo-controlled clinical trials [10]. Currently, only two IBS treatments approved by the US FDA can claim this distinction: tegaserod (Zelnorm®) and alosetron (Lotronex®). Tegaserod is indicated for the treatment of IBS-C and chronic constipation. Alosetron still remains the only FDA-approved treatment for IBS-D.
Overview of the IBS market
As one of the most common disorders seen by gastroenterologists and primary care providers, there is a need for the development of more effective treatments because IBS patients are often not satisfied with traditional therapies [3]. In addition, a recent position paper summarized the findings by the American College of Gastroenterology Functional GI Disorders Task Force when they examined clinical trials evaluating available therapies for IBS. The Task Force found that the quality of these studies did not strongly support the efficacy of treatments commonly used by physicians to treat IBS [10]. However, this systematic review determined that currently only the serotonergic agents, the serotonin receptor 3 (5-HT3) antagonist alosetron and the serotonin receptor 4 (5-HT4) agonist tegaserod, have undergone rigorous testing in randomized, double-blind, placebo-controlled trials for the treatment of IBS and were demonstrated to be efficacious for IBS-D and IBS-C, respectively [10].
The development of serotonergic agents for the treatment of GI conditions resulted from the recognition that 5-HT plays a key role in gut motility, sensation and secretion. Another 5-HT3 antagonist, cilansetron, has been developed for the treatment of IBS-D and has completed Phase III trials. Although the data appeared promising, this medication has not been approved by the FDA and is currently not available [11,12]. Another serotonergic drug, renzapride, which is a combination 5-HT4 agonist/5-HT3 antagonist, is currently being studied in Phase III trials for IBS-C [13,14].
Other novel nonserotonergic agents are in various phases of development (targeting other receptor sites) [15]. Lubiprostone is a chloride channel activator that has been recently approved by the FDA for the treatment of chronic idiopathic constipation in men and women and is currently being studied in IBS-C in a Phase III clinical trial. Neurokinin antagonists, a corticotrophin-releasing factor receptor 1 (CRF1) antagonist, dextofisopam (a 2,3-benzodiazepine), and a cyclic guanylate cyclase-C agonist are among the drugs in development that are undergoing investigation in Phase II IBS trials. Dextofisopam showed benefit in Phase II trials for IBS-D and IBS-A [16]. The CRF1 antagonist shows promise for regulating gut function [17]. The neuroendocrine hormone CRF and three related peptides (urocortin 1, 2, and 3) bind to CRF1 and CRF2, which are distinct G-protein-coupled receptors. [18]. Interaction of CRF with the pituitary CRF1 receptor is essential for the increase in glucocorticoid that is induced by stress. It appears that understanding CRF signaling pathways both centrally and peripherally in the intestine is key to understanding the impact of stress. It is known that activation of brain CRF1 receptors is involved in stress responsiveness and mediates stress-induced upper and lower GI tract motility as well as visceral sensitization [19–21]. It further appears that CRF1 is key because CRF1 receptors exist on enteric cholinergic neurons and probably also contribute to colonic motor responses to stress [21]. It is hypothesized that the comorbidity of IBS with anxiety and depression might be explained by hyperactivity in the CRF signaling pathways in the brain. Therefore, the role of a CRF antagonist would be to modulate alterations in GI function occurring in stress-related conditions such as IBS.
IBS symptoms can come and go over time as well as overlap with other functional disorders. Although IBS has often been divided into IBS-D and IBS-C, it has been recognized that these designations have been made so that investigators can select patients for treatment trials targeting a specific bowel pattern. In addition, the validity and stability of such subtypes is uncertain and is in need of further investigation. Supportive symptoms that were not part of the diagnostic criteria for IBS for Rome II were used to designate patient's bowel symptoms. Such supportive symptoms included:
Less than three bowel movements/week
More than three bowel movements/week
Lumpy or hard stool
Loose/watery stool
Defecation straining
Urgency or feeling of incomplete evacuation
IBS-D patients had one or more of symptoms b, d, or f but none of a, c, e, or more than two of b, d, or f and one of a or e (c, hard lumpy stool excluded). Similarly, IBS-C was more than one of a, c, e and none of b, d, f, or more than two of a, d, e and one of b, d, or f. Box 1 summarizes the new Rome III Criteria for IBS as well as the subtyping by predominant stool pattern [22]. The major difference in these criteria is the recognition that stool consistency may be a more reliable way of determining bowel subtype. In addition, it is suggested that the Bristol Stool Form Scale (Figure 1) [70] be used clinically to help clinicians and patients recognize the stool type. In addition to IBS-C, -D and -M, a fourth designation of IBS-unsubtyped (IBS-U) has been added.
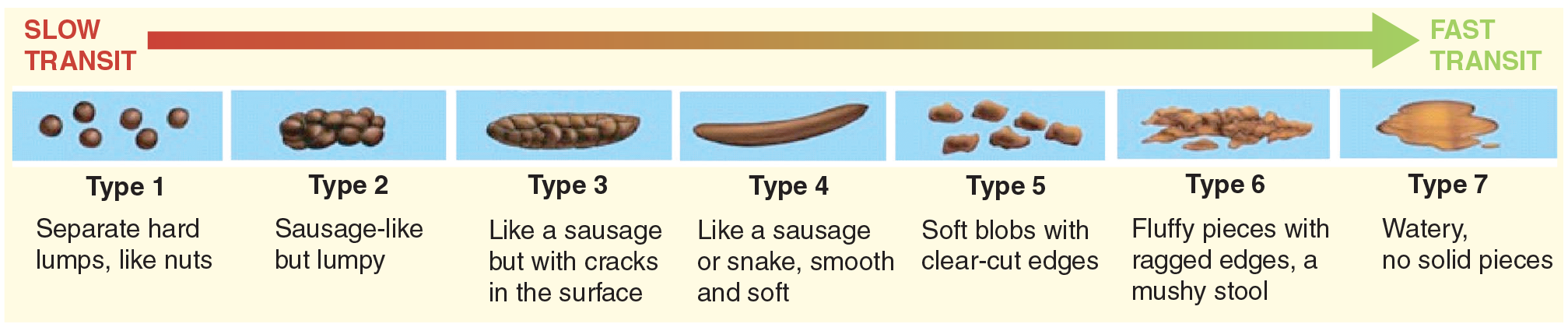
Bristol Stool Form Scale.
Two emerging areas of research are the roles of inflammation and mucosal–microbial interactions in IBS, which may potentially influence therapeutic options in IBS. Postinfectious IBS has been identified in 7–30% of patients with a recent history of established bacterial gastroenteritis [23]. It has been demonstrated that the inability to downregulate the immune response to infection may result in a variety of abnormalities in the colon, such as increased proinflammatory cytokines, lymphocytes, mast cells and enterochromaffin (EC) cells [24]. An area receiving particular attention is the role of inflammation on the mucosal expression of the serotonin reuptake transporter (SERT). In the gut, SERT terminates the actions of 5-HT by removing it from the interstitial space. Linden and colleagues demonstrated that in an animal model of colitis, there are distinct changes in EC cell numbers and 5-HT content, release and uptake [25]. Most notably the amount of 5-HT is increased in colitis due to EC hyperplasia, but the amount of SERT reuptake was decreased, resulting in decreased motility of the colon. Decreased motility was thought to be due to both receptor desensitization and reduction in SERT expression. Further work in this area by Coates and colleagues examined the components of serotonergic signaling in human rectal mucosal biopsy specimens derived from healthy controls and from individuals who met stringent diagnostic criteria for ulcerative colitis (UC), IBS-D and IBS-C [26]. In this model, 5-HT signaling was tested by examining mucosal 5-HT, tryptophan hydroxylase 1 messenger RNA, 5-HT transporter messenger RNA, and 5-HT transporter immunoreactivity. These were all found to be significantly reduced in UC, IBS-C and IBS-D. In contrast to 2,4,6-trinitrobenzene sulfonic acid (TNBS)-induced colitis in the guinea pig, Coates and colleagues found that in severe UC the population of EC cells was reduced, but was unchanged in IBS-C and IBS-D. Serotonin release was investigated under basal and mechanical stimulation conditions, and no changes were detected in any of the disease groups relative to controls. Therefore, similar molecular changes in serotonergic signaling mechanisms are seen in UC and IBS, while UC and IBS have distinct pathophysiological properties. These data suggest that shared defects in 5-HT signaling may underlie the altered motility, secretion and sensation seen in these disorders. The extent to which these changes occurs in the small bowel has not yet been demonstrated [26].
Rome III diagnostic criteria for irritable bowel syndrome (IBS) and subtyping of IBS by predominant stool pattern.
At least 3 months, with onset at least 6 months previously, of recurrent abdominal pain or discomfort* associated with two or more of the following:
Improvement with defecation; and/or
Onset associated with a change in frequency of stool; and/or
Onset associated with a change in form (appearance) of stool
IBS-C: Hard/lumpy stools > 25% of the time and loose (mushy)/watery stools < 25% of the time
IBS-D: Loose/watery stools > 25% of the time and hard or lumpy stools < 25% of the time
IBS-M: Hard/lumpy stools > 25% of the time and loose/watery stools > 25% of the time
IBS-U: Insufficient abnormality of stool consistency to meet criteria for IBS-C, D, or M
‘Discomfort’ means an uncomfortable sensation not described as pain.
IBS: Irritable bowel syndrome; IBS-C: IBS with constipation; IBS-D: IBS with diarrhea
IBS-M: IBS with mixed stool type; IBS-U: Unsubtyped IBS.
Other research has explored the role of the microflora, particularly in the small intestine, as a possible pathophysiologic mechanism causing IBS. One group has proposed that a high proportion of IBS patients have symptoms on the basis of the presence of small intestinal bacterial overgrowth as measured by lactulose breath test [27]. Both neomycin and, more recently, rifaximin have been used to treat IBS symptoms in short-term, placebo-controlled experimental studies [28–30]. However, other means of breath testing for bacterial overgrowth, such as the glucose breath test, do not find the same high rate of bacterial overgrowth in IBS patients [31,32]. While antibiotic therapy may be an effective therapy for a subgroup of patients, high-quality studies are needed to determine the exact role of small intestinal bacterial overgrowth in IBS. The other area of growing interest is the use of probiotics as a possible immune modulator in IBS. The work of O'Mahoney and colleagues demonstrated that Bifidobacterium infantis not only helped relieve IBS symptoms both by improving daily symptom scores and bloating but also noted that the probiotic had a beneficial effect in normalizing the ratio of anti-inflammatory to proinflammatory cytokine levels in the blood. However, cytokine levels were not measured in the colonic tissue [33]. More prospective research is needed to evaluate whether the potentially immune-modulating effect of probiotics affects gut function and if this correlates with symptom response in IBS.
Introduction to alosetron
Alosetron is one of three available 5-HT3 antagonists on the market today. The other compounds, granisetron and ondansetron, are indicated for the treatment of nausea and vomiting, and are not as potent in the lower GI tract as alosetron. As mentioned previously, the 5-HT3 antagonist cilansetron is not currently available. Thus, this review summarizes the pharmacology and clinical efficacy of alosetron in functional GI disorders. Although alosetron is approved only for women with severe IBS-D, a recent review of the data suggests that the drug may be also efficacious in men [34], and may also have therapeutic efficacy for functional dyspepsia (FD) [35].
Chemistry
Alosetron hydrochloride (HCl) is a potent and selective antagonist of the serotonin 5-HT3 receptor type. Chemically, alosetron is designated as 2,3,4,5-tetrahydro-5-methyl-2-[(5-methyl-1H-imidazol-4-yl)methyl]-1 H-pyrido [4,3-b]indol1-one monohydrochloride. Alosetron is achiral (i.e., is superimposable on its molecular mirror image) and has the empirical formula: C17H18N4O•HCl, representing a molecular weight of 330.8. Alosetron is a white to beige solid that is very soluble in water (61 mg/ml). Its solubility in a very acidic solution is better than its solubility in basic solution (42 mg/ml in 0.1 M HCl vs < 0.1 mg/ml in pH 8 phosphate buffer). The chemical structure of alosetron is shown in

Alosetron.
Pharmacodynamics
Alosetron is a potent and selective 5-HT3 receptor antagonist. Unlike 5-HT4 receptors, the 5-HT3 receptors are not involved in the initiation of the peristaltic and secretory reflexes. They also differ in that they are ligand-gated ion channel receptors, not G-protein-coupled receptors like the 5-HT4 receptors (and all other 5-HT receptors) [36]. The 5-HT3 receptors are extensively distributed on enteric neurons in the human GI tract, as well as other peripheral and central locations and they are known to be present on myenteric intrinsic primary afferent neurons. Activation of these channels with the resulting neuronal depolarizations affects the regulation of visceral pain, colonic transit and GI secretions. When the receptors are stimulated, they evoke fast inward currents that are responsible for mediating a subset of fast excitatory postsynaptic potentials in the enteric nervous system. 5-HT3 antagonists are known to inhibit activation of nonselective cation channels and block the 5-HT3-mediated depolarization of guinea pig myenteric and submucosal neurons in vitro in a dose-dependent fashion [37]. 5-HT3 antagonists do not normally block propulsion because propulsion is in large part mediated by acetylcholine. However, the noncholinergic component of fast excitatory synaptic potentials as recorded from myenteric neurons is mediated in part by 5-HT3 receptors [38]. Thus, in addition to alosetron's potency in the lower GI tract, it may act to inhibit propulsion to some degree [39] and slow colonic transit, particularly in the left colon [40]. Alosetron has also been shown to decrease chloride and water secretion [41].
The more potent effect of the 5-HT3 receptor is to activate extrinsic sensory nerves in the gut. The release of 5-HT from EC cells can stimulate 5-HT3 receptors on vagal afferents, which may result in nausea, for example, associated with cancer chemotherapy, and possibly nonpainful GI sensations such as bloating and fullness. The 5-HT3 antagonists thereby can reduce these sensory symptoms. The effect on visceral hypersensitivity may be due to both its peripheral effects on mechanoelastic properties [42] and its central inhibitory effect on brain regions concerned with central autonomic control of the gut and expression of emotion in the body. [43].
Pharmacokinetics
Alosetron is supplied as 0.5 and 1 mg tablets for oral administration. The pharmacokinetics of alosetron have been studied both after single oral doses of 0.05–16 mg in healthy men and women, and after repeated oral doses ranging of 1–8 mg twice daily in IBS patients. After a dose of 2 mg daily, alosetron has a bioavailability of 60% and an elimination half-life of 1.5 h [44]. The pharmacodynamic half-life is 6–10 h. The drug is rapidly absorbed after oral administration but, when taken with food, the absorption is decreased about 25%. However, the drug can be taken with or without food. Following oral administration of a 1 mg alosetron dose to young men, a peak plasma concentration of approximately 5 ng/ml occurred at 1 h. In young women, the mean peak plasma concentration is approximately 9 ng/ml with a similar time to peak concentration. Plasma concentrations are 30–50% lower and less variable in men compared with women given the same oral dose [45]. Population pharmacokinetic analysis in IBS patients confirmed that the drug concentration was affected by sex; specifically, clearance is 28% lower in women, resulting in approximately 30–50% higher concentrations in women compared with men for a given dose [46]. Also, in healthy men and women over the age of 64 years, plasma concentrations were increased approximately 40%. It is not entirely clear that these sex-and gender-related differences explain the more robust symptom relief findings associated with alosetron in women with IBS-D than men. Despite the fact that there is lower activity of cytochrome P450 (CYP)1A2 (a CYP isoform belonging to the CYP1A subfamily) in women, further work into the metabolism of this compound suggests that CYP1A2 activity is also correlated with race and body mass index (BMI) as well as age and sex [45]. Further work is necessary to explain the (previously reported) sex-related differences in metabolism. Restriction of dietary inducers and smoking may further explain higher differences in plasma concentration in females seen in a later study [45].
The metabolism of the drug is via the CYP enzymes 3A4, 1A2 and 2C9 [46]. Important drug interactions can occur with fluvoxamine (strong inhibitor of CYP1A2) and ketoconazole (strong inhibitor of CYP3A4). Both drugs significantly increased the area under the plasma–time curve (AUC) of alosetron in separate pharmacokinetic studies and therefore have the potential to prolong the half-life of the drug. Coadministration of these drugs while using alosetron is contraindicated [46].
Clearance of alosetron is not influenced by oral contraceptive pills (OCPs). In a study of 18 women on low-dose OCPs alosetron had no effect on serum concentrations of luteinizing hormone (LH) or follicle-stimulating hormone (FSH) [47]. Steady-state (day 21) 24-h AUC (AUC24), maximum concentration (Cmax), and time of maximum concentration (Tmax) of both levonorgestrel and ethinyl estradiol were similar during coadministration of alosetron with an OCP. Concentrations and activity of biochemical markers of thrombosis risk were not different in the presence of alosetron.
Approximately three-quarters of the drug is cleared by the kidney and the remainder can be recovered from the stool. About 7% is excreted as intact drug. The drug is to be avoided in severe hepatic and renal failure [48]. A summary of this information appears
Main pharmacokinetic characteristics of alosetron.
Caution is also advised with use with quinolone antibiotics, macrolide antibiotics, protease inhibitors and azoles.
Clinical efficacy
Over 3000 IBS patients have been enrolled in Phase II and Phase III clinical trials of alosetron. Seven published multicenter, double-blind, randomized, placebo-controlled, parallel-group studies have evaluated the efficacy of alosetron (
Summary of clinical trials evaluating the efficacy of alosetron for irritable bowel syndrome or dyspepsia.
Response is based on 1 mg b.i.d. when dose-ranging study was done to primary end point in most cases global relief of IBS pain and discomfort and where applicable to urgency.
F: Female; b.i.d.: Twice daily; IBS: Irritable bowel syndrome; t.i.d.: Three-times daily.
In these clinical trials, 90% of the study patients were Caucasian and the mean age was 45 years. Except for the single male-only study [54], the study populations of these trials were predominantly female. The primary efficacy end point in six of the eight studies was the adequate relief of IBS pain and discomfort [49–51,53–55].
Two studies by Lembo and colleagues enrolled women with relatively more severe IBS-D, who lacked satisfactory control of urgency on at least 50% of days [52]. Urgency is of particular consequence for these IBS patients due to the potential concomitant issue of fecal incontinence. Urgency was not defined by any particular criteria but seemed to correlate with consistency and frequency and not tenesmus [GlaxoSmithKline, Pers. Commun.]. In one study, a 7-point Likert scale with the global measure of relief ranging from substantially worse to substantially improved was used. Patients were asked to rate the degree of relief they experienced over the previous 4 weeks compared with the 3 months prior to the trial. This study also used a binary end point (i.e., yes/no response) for the primary outcome measure of satisfactory control of bowel urgency [52]. The second study evaluated the effect of alosetron on urgency in a more comprehensive manner. In this trial, the primary end point was the percentage of days with satisfactory control of bowel urgency and the response rate for global improvement was a secondary end point. Patients who were global responders recorded either moderate or severe symptoms relative to the way they felt before entering the study. Further analyses were performed on a subset of patients who had at least 10 of 14 days during screening (≥71% of days) with a lack of satisfactory control of bowel urgency. These patients had severe chronic IBS and 89% had IBS-D (11% had IBS-A). Alosetron resulted in a greater percentage of days with satisfactory control of urgency compared with placebo (69 vs 56%, respectively; p < 0.001). Greater percentages of alosetron-treated patients were global responders at 4, 8 and 12 weeks compared with placebo (59 vs 41%, 63 vs 41%, and 68 vs 46%, respectively; p < 0.001). Patients with more frequent urgency had similar results, with the median percentage of satisfactory control of urgency for this group being 66 versus 43% for placebo (p < 0.001) [56].
In addition to its short-term efficacy, there is significantly better long-term efficacy of the medication compared with placebo. A recent multicenter, randomized, placebo-controlled clinical trial demonstrated the long-term efficacy (i.e., 48 weeks) of alosetron (1 mg twice daily) in women with IBS-D [55]. The primary end point was relief of IBS pain and discomfort. Alosetron-treated patients also reported significantly greater control of urgency than placebo-treated patients (p < 0.001).
Men were under-represented in the initial Phase II clinical trials, and therefore the effectiveness in men was not established at the time of FDA approval. One subsequent dose-ranging clinical trial evaluated the efficacy of alosetron in 662 men with IBS-D [54]. The men were randomized to treatment with alosetron 0.5, 1.0, 2.0, 4.0 mg or placebo twice daily for 12 weeks, followed by a 4-week post-treatment period. Adequate relief of IBS pain and discomfort during weeks 5–12 of the treatment phase was the primary end point and the completed study found that a dose of 1 mg twice daily provided significantly greater relief of this symptom (53 vs 40% for placebo; p = 0.04). Relief of secondary end points included bowel urgency, stool frequency and consistency, incomplete evacuation, bloating, and pain-free days were also examined. All doses of alosetron significantly reduced stool consistency scores (p < 0.001), indicating more formed stools. No significant effects of alosetron were seen with regard to the other secondary symptom end points.
Only one trial evaluated the efficacy of alosetron in FD [57]. Alosetron provided greater relief of upper abdominal pain and discomfort than placebo in a multicenter randomized, controlled trial in women with FD. The reasons for its beneficial effects in FD are not entirely clear; however, this medication likely provides symptom relief via its central and peripheral effects in visceral sensory and motor function, which have been shown in IBS. While alosetron seemed to have no effect on gastric accommodation, it appeared to reduce postprandial bloating and nausea after satiation was achieved during a nutrient drink test [58]. Further FDA approval was not sought for this indication when the side effects of serious complications of constipation and ischemic colitis became apparent. No cases of ischemic colitis were reported in this FD study.
Safety & tolerability
In clinical trials, the two primary side effects of clinical concern were constipation and ischemic colitis. Other side effects were also mostly GI-related such as abdominal discomfort, nausea, distention, regurgitation/reflux and hemorrhoids. All occurred rarely; specifically, less than 2–7% occurrence, which was not significantly different from the placebo group. At the 1 mg twice-daily dose, the drug produced constipation as an adverse event in approximately 28% of patients but only 11% in those taking 0.5 mg twice daily. In Phase II trials, constipation caused approximately 11% of the patients to prematurely withdraw from the studies. The incidence of serious complications of constipation, defined as ileus, bowel obstruction, toxic megacolon, fecal impaction or perforation, in women was approximately 1 in 1000. Elderly, debilitated patients or those individuals receiving additional medications that decrease GI motility may be at greater risk for complications of constipation. It is currently recommended that alosetron be discontinued immediately in patients who develop constipation. Restarting at a lower or similar dose can be considered when the diarrhea recurs, which typically occurs within 2 days.
Ischemic colitis is another adverse event that has been reported during the clinical trials and postmarketing period. There were 11,874 patients enrolled in the clinical trials and 17 reported cases of ischemic colitis. Nine patients were hospitalized and there were no surgeries or deaths. The postmarketing experience has demonstrated that, in 275,000 patients who had taken alosetron, there were 80 (0.02%) reported cases of ischemic colitis. A total of 74% of these cases occurred within the first month. It is generally accepted in medical practice that a diagnosis of ischemic colitis is only confirmed after positive endoscopic and histological findings. Cases of ischemic colitis have been classified as probable or possible on the basis that a diagnosis is supported by: clinical evidence and endoscopic and/or biopsy findings that were suggestive of, but not necessarily consistent with, the diagnosis of ischemic colitis. Of these cases, 55% were considered to be probable, 17.5% were possible, and 27.5% had insufficient evidence for ischemic colitis. Of these 80 cases, there were 48 hospitalizations, 6 surgeries and no deaths. Adverse events, particularly severe complications of constipation and ischemic colitis, most of which occurred during the first month of therapy, have led to its use under a comprehensive risk-management program. A recent systematic review concluded that there is a significantly increased rate of ischemic colitis among alosetron patients compared with placebo patients (0.15 vs 0.0%), but no significant difference in the rate of serious complications of constipation. All of the alosetron patients with ischemic colitis had a reversible colopathy without long-term sequelae. Based on postmarketing surveillance data, the postadjudication rate of ischemic colitis is 1.1 per 1000 patient-years of alosetron use and the rate of serious complications of constipation is 0.66 per 1000 patient-years of alosetron use [59].
The reasons for the association with ischemic colitis are uncertain. Recent studies indicate that IBS patients in general are more likely to be diagnosed with ischemic colitis compared with healthy controls [60–63]. The reasons for this are largely unknown but possible mechanisms for the association of alosetron and ischemic colitis include interference of a 5-HT-mediated mechanism regulating mucosal and submucosal blood flow and the development of severe constipation with associated colonic distention, which may infrequently contribute to the development of ischemic colitis. An additional factor that may play more of a role in increasing the risk for constipation than ischemia is genetic polymorphisms in the SERT. These genetic variants are an important factor influencing 5-HT availability to act on postsynaptic receptors and could affect the response to serotonergic medications such as tegaserod and alosetron for IBS, as well as selective serotonin reuptake inhibitors for the treatment of depression. One study demonstrated that slowing of colonic transit was greater in IBS-D patients with the long homozygous polymorphism of SERT, which results in greater reuptake of 5-HT than those with heterozygous or homozygous short polymorphism [64].
Owing to these complications of severe chronic constipation and ischemic colitis, the drug was voluntarily withdrawn from the market in November of 2000. However, it did return to the market in 2002 under the Restricted Use Program (see below).
Regulatory affairs
The FDA rereviewed the medication and approved alosetron for restricted use in June 2002. The FDA readdressed its original decision based on subsequent discussions with patients who had successfully taken the drug and the physicians who treated them. The medication became available again in December 2002 and is currently only available in the US. Under the Restricted Use Program, physicians who want to prescribe alosetron must fill out an attestation that they feel comfortable with the diagnosis and management of IBS. Once this form is completed the physician receives an educational folder that includes a patient–physician agreement form, a patient information sheet informing them of the signs and symptoms of constipation and ischemic colitis, and a special sticker for the prescriptions for each patient. The medication is indicated for women with IBS with severe diarrhea, with the following conditions:
Chronic IBS symptoms (generally lasting ≥6 months)
Anatomic or biochemical abnormalities of the GI tract have been excluded
Failed to respond to conventional therapy
IBS is considered severe if it includes diarrhea and one or more of the following:
Frequent and severe abdominal pain/discomfort
Frequent bowel urgency or fecal incontinence
Disability or restriction of daily activities due to IBS
The recommended starting dose is 0.5 mg twice daily to be increased to 1 mg orally twice daily after 1 month only if symptoms are not controlled on the lower dose. If the 1 mg twice-daily dose does not control the patient's symptoms after 4 weeks, it is recommended that the drug be stopped. Patients are generally advised to stop alosetron and call their physician if they develop constipation (e.g., no bowel movement for two consecutive days) or atypical or worsening abdominal pain or blood in their stools. Recently the ability to write refills on these prescriptions was restored.
As part of the conditions outlined by the FDA for reapproval of the drug, GlaxoSmithKline agreed to perform additional clinical trials of alosetron:
A low-dose, dose-ranging, safety and efficacy study comparing 0.5 mg once daily, 1 mg once daily and 1 mg twice daily
A study in patients using the drug only as needed
A patient survey to assess premedication counseling and appropriateness of the therapy prescribed
Analysis of these data is nearing completion and should be forthcoming shortly.
Conclusion
Alosetron remains at the time of writing the only FDA-approved treatment for severe IBS-D. Although other agents are available to treat IBS-D, such as diphenoxylate, loperamide and tricyclic antidepressants, their limited efficacy (i.e., antidiarrheal agents) or their side-effect profile (i.e., both antidiarrheal and tricyclic agents) often limit their utility for IBS patients. Further, randomized, double-blind, placebo-controlled trials of the efficacy of these agents in the treatment of IBS-D are either nonexistent or of poorer quality than the data available for alosetron. The randomized clinical trials have demonstrated that alosetron improves global IBS symptoms and individual symptoms of abdominal discomfort, stool frequency, stool consistency and urgency in non-constipated IBS patients. The drug exerts its beneficial effects on IBS symptoms through central and peripheral actions affecting motor and mechanoelastic properties of the gut [65,42], water and chloride secretion [41] and brain activation of ‘limbic’ brain regions, concerned with visceral perception and autonomic circuits [43]. Unfortunately, despite alosetron's statistically significant efficacy, the rare but serious side effects of constipation and ischemic colitis, in addition to the comprehensive risk-management program, have limited the number of prescriptions written since the drug was reapproved in 2002. Properly prescribed and carefully monitored, the drug has a favorable safety and tolerability profile and has improved the quality of life in many severely ill patients for whom no other therapy has been effective.
Future perspective
Theories explaining IBS over the past 50 years have ranged from that of a purely psychological disorder to primary abnormalities in GI motility to the concept of visceral hypersensitivity. However, animal and clinical data have increasingly supported a dysregulation in brain–gut interactions resulting in alterations in GI motility, secretion and sensation as the principal pathophysiological mechanism underlying IBS [1]. This altered brain–gut axis is mediated by many factors (both central and peripheral). These factors include genetic predisposition, chronic stress, environmental factors and a mounting body of evidence on inflammation/infection and intestinal microflora [66]. In addition, significant molecular alterations in the gut with relation to SERT expression and 5-HT signaling affecting 5-HT availability may also be playing a role in the development of the symptom complex of IBS [26]. Alteration in these factors subsequently leads to disturbances in visceral sensitivity, GI motility and mucosal immune response and permeability, producing the symptom complex of abdominal pain or discomfort and altered bowel function that are the defining characteristics of IBS [67]. Further studies are needed to better characterize these physiological alterations and to determine a biological marker that can be used as a diagnostic tool for IBS and one that correlates well with symptom severity and response to treatment. In addition, although the autonomic nervous system has been the focus of the integrated communication between the brain and the gut, recent evidence suggests that the hypothalamic–pituitary–adrenal axis may also play an important role, particularly in governing the gut's response to stress [68]. Further elucidation of this pathway may lead to further therapeutic options for IBS.
There are many neurotransmitters and hormones that mediate the bidirectional brain–gut communication. 5-HT is one of the key mediators of gut motility, secretion and sensation and 90–95% of the body's 5-HT is localized in the GI tract, primarily in EC cells and enteric neurons [36]. Particularly important for gut function and regulation are the 5-HT1P, 5-HT3 and 5-HT4 receptors. These receptors have been the focus of research evaluating the pathophysiological mechanisms of IBS as well as targets for the development of novel agents in the treatment of functional GI disorders. With regard to 5-HT-related mechanisms in functional GI disorders, future directions would include performing studies to:
More completely understand the role of 5-HT signaling pathways and SERT polymorphisms in the development of these disorders and their phenotypic presentations;
Characterize the clinical diagnostic criteria of ischemic colitis and the potential association with IBS in general and with serotonergic agents in particular.
Continuing developments in the understanding of the pathophysiology of IBS have resulted in other areas of potential interest for therapeutic development. These include:
Agents targeting the serotonergic receptors in combination (5-HT4 agonist, 5-HT3 antagonist agents), opioid receptor agonists and antagonists, neurokinin receptor agents, chloride channel activators and neurohormonal modulators [69];
Antibiotics and or probiotics modulating the influence of the intestinal microflora on complex interactions between the immune system and the enteric nervous system and CNS;
Progress in the area of neuroimaging and central pain processing that may result in the development of agents that more effectively modulate visceral pain alone or perhaps in combination with nonpharmacological therapies such as hypnosis and cognitive behavioral therapy, perhaps revealing receptor sites that have not yet been considered.
Information resources
The International Foundation of Functional Gastrointestinal Disorders (IFFGD) is a registered, nonprofit, education and research organization founded in 1991. IFFGD addresses the issues surrounding life with GI functional and motility disorders and increases awareness and provides education regarding functional GI and motility disorders among the general public, researchers, regulators, and the clinical care community.
The Rome Foundation is an international organization that developed a classification system of all functional GI disorders and by using the best evidence establishes criteria for their diagnoses. This comprehensive resource provides education and guidelines regarding diagnosis and treatment of these disorders that are followed by healthcare providers, and medical, research and regulatory organizations worldwide.
The American Gastroenterological Association (AGA) is a leading professional society representing gastroenterologists and hepatologists worldwide, which is dedicated to the mission of advancing the science and practice of gastroenterology. The AGA provides up-to-date information on research, meetings, education, policy and practice guidelines.
The American College of Gastroenterology (ACG) is another leading professional society that provides education, news and information regarding GI conditions to clinical GI practitioners.
The Functional Brain–Gut Research Group (FBG) is an organization of professionals that supports, promotes and advances multidisciplinary research and education in the basic science, clinical and behavioral aspects of brain–gut interactions.
The North American Society for Pediatric Gastroenterology, Hepatology, and Nutrition (NASPGHAN) strives to improve the care of infants, children and adolescents with digestive disorders by promoting advances in clinical care, research and education.
The National Digestive Diseases Information Clearinghouse (NDDIC) is a service of the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK). NIDDK is part of the National Institutes of Health (NIH) under the US Department of Health and Human Services, which provides information regarding digestive diseases to affected individuals and to their families, healthcare professionals and the public.
The FDA is responsible for protecting the public health by assuring the safety, efficacy and security of human and veterinary drugs, biological products, medical devices and the food supply in the USA. The FDA facilitates the availability of safe and effective products to the public and provides them with the accurate, science-based information they need to use medicines and foods to improve their health.
The GlaxoSmithKline website has up-to-date information regarding alosetron (Lotronex®), a medication that treats multiple GI symptoms in women with severe IBS-D, provides complete prescribing information for patients and healthcare providers.
Executive summary
Severe diarrhea-predominant irritable bowel syndrome (IBS-D) of at least 6 months duration, for which the trial of other standardly used medications has not proven therapeutic.
Mechanism not fully elucidated.
Drugs work at serotonin receptor (5-HT)3, a ligand-gated ion channel receptor, to slow colonic transit and decrease intestinal secretion.
May also have an effect on visceral pain sensation.
After a dose of 2 mg daily, alosetron has a bioavailability of 60% and an elimination half-life of 1.5 h.
Pharmacodynamic half-life is 6–10 h.
Drug is rapidly absorbed after oral administration but, when taken with food, the absorption is decreased by approximately 25%.
Plasma concentrations are 30–50% lower and less variable in men compared with women given the same oral dose, which may explain its more robust response in females.
Oral contraceptive pills (OCPs) do not influence drug concentration and can be coadministered with the drug.
Metabolism of the drug is via the cytochrome P450 (CYP) enzymes CYP3A4, 1A2 and 2C9.
Approximately three-quarters of the drug is cleared by the kidney and the remainder can be recovered from the stool.
Approximately 7% is excreted as intact drug.
The drug is to be avoided in severe hepatic and renal failure.
The efficacy of alosetron has been demonstrated in seven placebo-controlled clinical trials with more than 3000 patients with nonconstipated IBS at a dose of 1 mg twice daily.
Five of the seven studies demonstrated relief of IBS abdominal pain or discomfort.
Two of these studies demonstrated relief of IBS-related urgency.
Lower dose efficacy studies are pending.
In healthy men and women over the age of 64 years, plasma concentrations were increased by approximately 40%.
Safety and efficacy data do not exist in the pediatric population.
The two primary side effects of clinical concern are constipation and ischemic colitis.
Based on postmarketing surveillance data, the postadjudication rate of ischemic colitis is 1.1 per 1000 patient-years of alosetron use and the rate of serious complications of constipation is 0.66 per 1000 patient-years of alosetron use.
Other side effects were mostly gastrointestinal, such as abdominal discomfort, nausea, distention, regurgitation/reflux and hemorrhoids, and occurred rarely (2–7%).
Elderly, debilitated patients or those individuals receiving additional medications that decrease gastrointestinal motility may be at greater risk for complications.
Due to metabolism by the CYP450 system, important drug interactions can occur with fluvoxamine (strong inhibitor of CYP1A2) and ketoconazole (strong inhibitor of CYP3A4).
Concomitant use with moderate CYP1A2 inhibitors, e.g., quinolone antibiotics and cimetidine, is to be avoided due to potential increase in area under the plasma–time curve.
Caution should be exercised when prescribing alosetron with macrolide antibiotics, protease inhibitors and azole antifungal agents.
Available as 0.5 and 1 mg tablets for the treatment of severe IBS-D patients who have symptoms for more than 6 months.
Dosage initiation should start at 0.5 mg twice daily.
Titrate up in 4 weeks if IBS symptoms are not well controlled.
Discontinue drug if after 4 weeks of 1 mg twice daily, IBS symptoms are not well controlled.
To avoid serious consequences of chronic constipation or ischemic colitis patients should be instructed to stop drug if patient has no bowel movement for 48 h.
Patients should be instructed to stop drug immediately and notify physician if symptoms of ischemic colitis occur, e.g., new or worsening abdominal pain, rectal bleeding or bloody diarrhea.
Use with care in elderly debilitated patients, patients taking medications that decrease gastrointestinal motility and patients with hepatic impairment.
