Abstract
Tegaserod, a novel serotonin receptor agonist, has gained acceptance and widespread use for the treatment of women with irritable bowel syndrome and constipation. Development of this therapy evolved from the emerging role of serotonin in gastrointestinal motor, secretory and sensory functions. The efficacy and safety of tegaserod has been well established in women suffering from irritable bowel syndrome with constipation. Large, randomized, double-blind, placebo-controlled trials involving more than 3500 predominantly female patients with irritable bowel syndrome and constipation have demonstrated the superiority of tegaserod over placebo in improving global and individual symptoms. The most common side effects of tegaserod in clinical trials were diarrhea and headache. Recent data suggest that retreatment with tegaserod after a drug holiday is efficacious, opening the door to the possibility of intermittent therapy for patients with irritable bowel syndrome and constipation. Areas in need of further investigation include the role of tegaserod in the treatment of pain and bloating in irritable bowel syndrome, whether tegaserod has a role in male patients, the long-term efficacy of tegaserod, whether tolerance develops in a subset of patients with extended therapy and whether tegaserod is beneficial for the treatment of other functional gastrointestinal disorders.
Definition & symptomatology of irritable bowel syndrome
Irritable bowel syndrome (IBS) refers to a heterogeneous group of patients suffering with persistent or recurrent abdominal discomfort in association with altered bowel habits [1]. By definition, patients with IBS should have no identifiable organic cause for their symptoms. As there is no reliable anatomic, biochemical or physiologic marker, IBS is currently a symptom-based diagnosis. Several symptom-based diagnostic criteria have been developed in an attempt to identify a more uniform group of patients for clinical studies [2] (Box 1). Over time, symptom-based criteria have slowly become more accepted in clinical practice. The most current and widely used are the Rome II criteria, which include at least 12 weeks of abdominal pain or discomfort in the preceding 12 months (which need not be consecutive) accompanied by a minimum of two of the following features of abdominal discomfort or pain [3]:
Relieved with defecation
Associated with a change in bowel movement frequency
Associated with a change in the appearance of the stool
There are a number of additional gastrointestinal (GI) symptoms commonly reported by IBS patients, including straining, urgency, a feeling of incomplete evacuation, passage of mucus, bloating and abdominal distention. Furthermore, IBS patients often suffer from coexistent non-GI problems such as anxiety, depression, somatization, lethargy, poor sleep, backache, urinary frequency and dyspareunia [4]. Given such symptom diversity, attempts have been made to subgroup IBS patients based upon their predominant bowel symptom (constipation, diarrhea or mixed bowel habits, when there is a combination of diarrhea and constipation). It is currently believed that subgrouping may help to direct the diagnostic evaluation and treatment of IBS patients. Certainly, subgrouping has been widely adopted by clinical investigators and pharmaceutical companies interested in developing novel therapies for IBS. Although subtyping is practical and useful for clinical trials, it is important to recognize that in a substantial subset of patients, IBS is a dynamic condition. There can be significant changes in the specific bowel symptoms and the severity of symptoms reported by IBS patients over time [5]. For instance, a recent observational study found that significant changes in bowel pattern (from that of diarrhea or constipation to alternating of bowel habits) occurred in 50% of 209 patients over a period of 3 months [6].
Diagnostic criteria for irritable bowel syndrome.
At least 12 weeks or more, which need not be consecutive, in the preceding 12 months of abdominal discomfort or pain that has two out of three features:
– Relieved with defecation
– Onset associated with a change in frequency of stools
– Onset associated with a change in form (appearance) of stool
Symptoms that cumulatively support the diagnosis of irritable bowel syndrome:
Abnormal stool frequency (for research purposes, abnormal may be defined as more than three bowel movements/day or less than three bowel movements/week)
Abnormal stool form (lumpy/hard or loose/watery stool)
Abnormal stool passage (straining, urgency, or feeling of incomplete evacuation)
Passage of mucus
Bloating or feeling of abdominal distention
At least 3 months of continuous or recurrent symptoms of the following:
– Abdominal pain or discomfort relieved with defecation, or
– Associated with a change in frequency of stool, or
– Associated with a change in consistency of stool
Plus two or more of the following, at least on a quarter of occasions or days:
Altered stool frequency (> three bowel movements a day or < three bowel movements/week), or
Altered stool form (lumpy/hard or loose/watery stool), or
Altered stool passage (straining, urgency, or feeling of incomplete evacuation), or
Passage of mucus, or
Bloating or feeling of abdominal distention
Pain relieved by defecation
Looser stools at onset of pain
More frequent bowel movements at onset of pain
Abdominal distention (visible)
Mucus per rectum
Feeling of incomplete rectal emptying
Impact of irritable bowel syndrome
Although there is no evidence of increased mortality in those with IBS, its high prevalence, chronicity and negative impact on affected individuals present a great burden to society. A recent systematic review found that the prevalence of IBS in North America ranged from 3–20%, with most studies reporting a prevalence of 10–15% [7]. This finding suggests that an estimated 30–45 million North American adults are affected during any given year [7]. Moreover, those with IBS are more inclined to seek healthcare, with as many as 3.5 million physician visits/year, accounting for 12% of primary care visits and 28% of gastroenterology visits [8]. The direct healthcare costs in the USA alone have been estimated at US$1.3–10 billion/year, with yearly indirect costs, largely due to work absenteeism and decreased productivity, ranging from US$205 million to US$20 billion [9].
Pathophysiology of irritable bowel syndrome
Although the precise pathophysiology of IBS remains elusive, multiple potential contributing factors have been proposed [10]. These include altered gut motility [11,12], visceral hypersensitivity [13], mucosal inflammation [14], genetic predisposition [15,16], food sensitivity [17], psychologic and emotional distress [18], and alterations in the brain–gut axis [19]. Various combinations of these factors are thought to interact in any specific patient, leading to the symptom complex currently referred to as IBS. An overview of this is found in Figure 1.
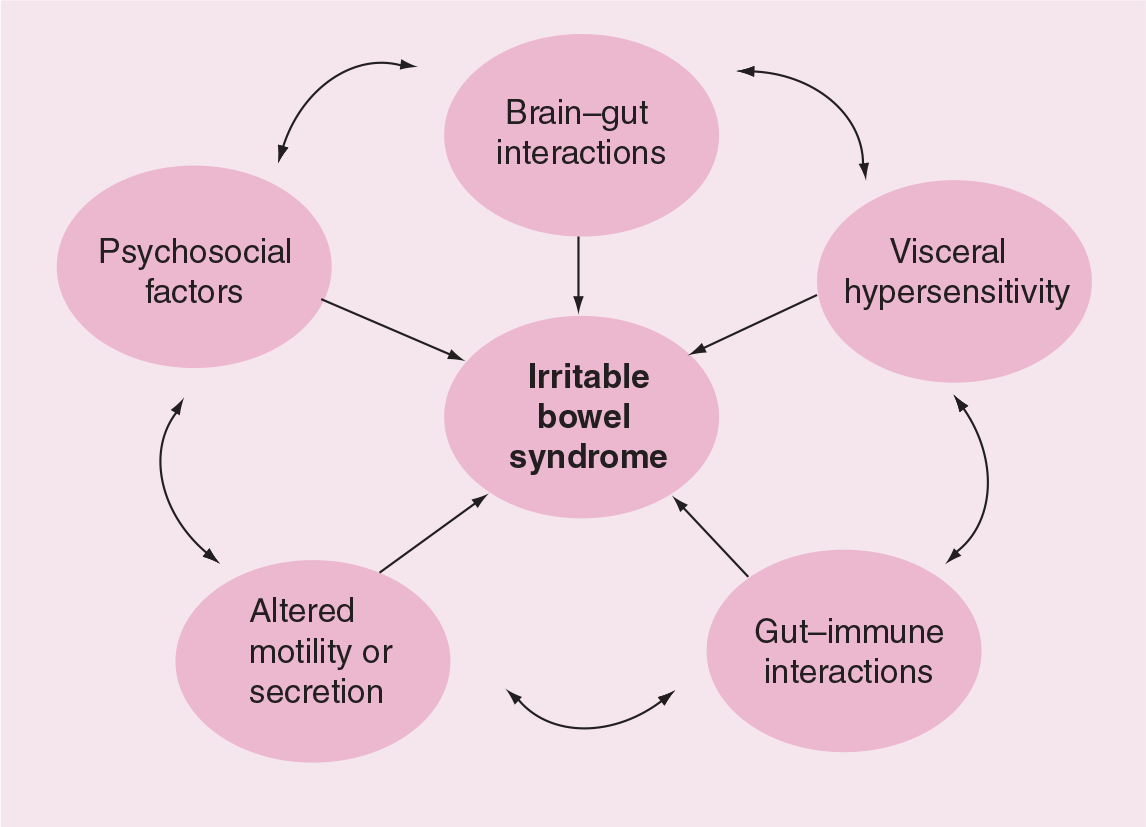
The pathophysiologic mechanisms of irritable bowel syndrome.
From our rapidly expanding understanding of the enteric nervous system and brain–gut interactions, it has become increasingly clear that the neurotransmitter serotonin (5-hydroxytryptamine [5-HT]) plays an important role in the pathogenesis of subsets of patients with IBS. It is known that approximately 95% of the body's 5-HT is synthesized in the enterochromaffin, mast and smooth muscle cells of the GI tract. 5-HT, acting through the intrinsic nervous system of the GI tract, is believed to play a critical role in GI motor, sensory and secretory functions [20]. To date, 14 5-HT-receptor subtypes have been identified, with 5-HT1p, 5-HT3 and 5-HT4 being the most clinically relevant in GI tract functioning [21]. Specific activation of the 5-HT4 receptor increases intestinal motor activity and secretion and may affect visceral sensation [20].
Treatment of irritable bowel syndrome
Traditionally, pharmacologic therapy for IBS has been directed at relieving specific symptoms, typically those that are the most frequent, severe or life altering for the patient [22]. Data from clinical trials suggest that traditional pharmacologic therapies such as fiber supplements, antispasmodics, antidiarrheals and laxatives are only modestly effective in the treatment of IBS [23,24]. Moreover, these agents tend to improve individual, but not necessarily global, IBS symptoms. Furthermore, side effects are not uncommon with these agents, limiting their usefulness in clinical practice. These points were recently underscored in a community-based survey study from Seattle (WA, USA), which found that just over half of patients reported satisfactory relief with traditional medical care for IBS [25].
In the Rome II consensus document on the functional bowel disorders, experts recommended that the optimal primary outcome for clinical trials should integrate the symptoms of a particular functional GI disorder [26]. This sentiment recognizes the importance of improving global symptoms in patients suffering from disorders such as IBS. As 5-HT appears to play an integral role in GI motility, secretion and visceral perception, it is logical to surmise that pharmaceuticals with effects at specific 5-HT receptors might be of benefit to IBS. In methodologically rigorous, randomized, placebo-controlled trials, serotonergic agents, including 5-HT3 receptor antagonists (e.g., alosetron and cilansetron) and 5-HT4 agonists (e.g., tegaserod), have been shown to improve global and individual symptoms in diarrhea- and constipation-predominant IBS patients, respectively [23]. Despite these encouraging findings, it is worth noting that no single medical therapy has been found to be universally effective in the treatment of IBS patients, which is probably related to the heterogeneous pathogenesis of this disorder. In spite of this, the remainder of this review will focus on the evidence supporting the efficacy of tegaserod in women with IBS and a predominant bowel complaint of constipation.
Tegaserod
Introduction
Tegaserod is a selective 5-HT4 receptor partial agonist, approved in the USA in April 2002 for the treatment of female IBS patients with constipation. It binds with high affinity to 5-HT4 receptors, with a potency that is a fifth of that of endogenous 5-HT. It has limited affinity for 5-HT1 receptors and no appreciable affinity for other 5-HT receptors, muscarinic, adrenergic, dopaminergic or opiate receptors [27]. Tegaserod's actions on 5-HT4 receptors lead to a variety of physiologic effects that are potentially advantageous in patients with IBS and constipation.
Chemistry
Tegaserod, an indole carboxyaldehyde derivative and member of the aminoguanidine drug class, is structurally similar to 5-HT. Tegaserod is commercially available as tegaserod maleate, a hydrogen maleate salt chemically designated as 3-(5-methoxy-1H-indol-3-ylmethylene)-N-pen-tylcarbazimidamide hydrogen maleate. Its chemcial structure is shown in Figure 2. It is available as an oral tablet containing either 2 or 6 mg of tegaserod, with crospovidine, glyceryl monostearate, hydroxypropyl methylcellulose, lactose monohydrate, poloxamer 188 and polyethylene glycol 4000 as inactive ingredients.

Tegaserod.
Pharmacodynamics & pharmacokinetics
Tegaserod is rapidly absorbed following oral administration in the fasting state, with an absolute bioavailability of 10% [28]. Its median time to peak plasma concentration is 1–1.3 h and its mean elimination half-life is 7.5 h following ingestion [28], with a steady state obtained within 8 days of initiating therapy [29]. Tegaserod's bioavailability is reduced by 40–65% if administered with food, largely due to its basic and lipophilic properties. It should therefore be taken prior to a meal, although the exact timing prior to the meal does not appear to be important. Tegaserod is 98% bound to plasma proteins, predominantly α1-glycoprotein, and does not cross the blood–brain barrier [30]. Tegasearod is primarily metabolized through two pathways, one presystemic and the other systemic. Approximately 20% of the drug is metabolized via the presystemic pathway through hydrolytic breakdown in the stomach, followed by oxidation and glucuronidation, producing the main metabolite of tegaserod, which has no prokinetic activity. The second metabolic pathway is through direct glucuronidation leading to the generation of three isomeric N-glucuronides [30]. Approximately two-thirds of tegaserod is excreted unchanged in the feces, with the remainder excreted in the urine as metabolites [30]. Age, race and gender do not appear to influence the pharmacokinetics of tegaserod [30]. There is no need for dosage adjustment based on body weight given the intersubject variability of the pharmacokinetic properties and the wide therapeutic margin of tegaserod [30]. No dosage adjustment is required in individuals with mild-to-moderate hepatic or renal impairment; however, tegaserod has not been studied in patients with severe hepatic or renal impairment (creatinine clearance of <15 ml/min). Therefore, until safety data become available, tegaserod should be avoided in these patient populations [30]. There appears to be a dose–response relationship with regard to its prokinetic actions on colonic transit, although decreased effects compared with placebo are observed at low- (1 mg) and high-dose (25–100 mg) ends of the dosing range [31].
Preclinical trials
Preclinical studies with tegaserod, utilizing various animal models, have demonstrated clear prokinetic effects. By binding to 5-HT4 receptors on intrinsic primary afferent neurons, tegaserod stimulates the release of neurotransmitters including calcitonin gene-related peptide, substance P and vasoactive intestinal peptide [32]. This in turn results in augmentation of the peristaltic reflex, leading to accelerated gastric, small bowel and regional colonic transit [27,30,33]. Augmentation of the peristaltic reflex by tegaserod has been demonstrated in the human jejunum as well as in the rat and guinea pig colon [34]. Tegaserod was initially shown to accelerate colonic transit time in vivo in a canine model [35]. Later, tegaserod was shown to accelerate gastric, intestinal and colonic transit in healthy human males [36], as well as orocecal and colonic transit in patients with IBS and constipation [37]. In addition, tegaserod increases water and sodium secretion from colonocytes [38] and exerts effects on visceral sensation in animal and human models [39–42].
Phase III clinical trials
The efficacy and tolerability of tegaserod was initially evaluated in three Phase III clinical trials involving more than 3000 patients from the Western hemisphere [43–45]. All were multicenter, double-blind, placebo-controlled trials designed to evaluate a 12-week course of therapy in patients with constipation-predominant IBS based on the Rome I or II criteria. The primary outcome in these trials was the subject's global assessment (SGA) of relief of overall GI symptoms in the last 4 weeks of the treatment trial. The specific question asked of patients was: ‘Please consider how you felt this past week in regard to your IBS, in particular your overall wellbeing and symptoms of abdominal discomfort, pain and altered bowel habit. Compared with the way you usually felt before entering the study, how would you rate your relief of symptoms during the past week?’ The possible responses to this question included: completely relieved, considerably relieved, somewhat relieved, unchanged or worsened. A positive response to treatment was defined as ‘complete or considerable relief more than 50% of the time’ or ‘somewhat relief 100% of the time’. The vast majority of patients enrolled in these trials were Caucasian women (>85%) with a longstanding history of IBS and constipation (mean duration of symptoms of 14.4 years). Overall, these were adequately powered studies that largely con-formed to the guidelines recommended by the Rome committee for treatment trials in patients with functional GI disorders [26]. However, these trials have been criticized for a variety of methodologic issues, including a lack of uniform criteria for study enrollment and treatment response, differences in study design and a high placebo response rate. While acknowledging these issues, these studies consistently demonstrated improvement in global IBS symptoms and individual symptoms, such as abdominal pain, stool frequency, stool consistency, straining and bloating with tegaserod versus placebo. For the primary end point, the therapeutic gain with tegaserod ranged from 5 to 13% over placebo, with an initial response occurring within 1 week of initiating treatment (Table 1). In a study authored by Muller-Lissner and colleagues, the only side effect occurring with greater frequency than with placebo was diarrhea (9.6 vs 2.5%) [43]. In a study by Novick and colleagues, diarrhea, headache, nausea and flatulence occurred with greater frequency in those treated with tegaserod compared with placebo [44]; however, only 1.6% of patients treated with tegaserod discontinued therapy due to side effects. The results of these studies ultimately led to the US Food and Drug Administration's (FDA) approval of tegaserod in 2002 for short-term use in women suffering from IBS and constipation. The exact mechanism by which tegaserod provides relief of the global symptoms of IBS is not clear. Tegaserod clearly promotes motor and secretory effects in the small and large intestine and may exert an effect on visceral sensation. Unfortunately, the available trials were not designed to address the specifics regarding responder characteristics.
Pivotal clinical trials.
International clinical trials
Following tegaserod's approval by the FDA, several additional clinical trials have been completed that have increased our understanding of this drug's role in the treatment of IBS with constipation. A summary is shown in Table 2. A large, multicenter, double-blind, placebo-controlled trial (Zelmac in Asia Pacific [ZAP] study), involving 520 IBS patients (88% female with a mean age of 36 years) from the Asia–Pacific region, demonstrated the superiority of tegaserod over placebo in providing satisfactory relief of overall IBS symptoms, reducing the frequency of abdominal pain and bloating, and improving the consistency of stools [46]. Unlike the pivotal trials discussed earlier, this study used a dichotomous primary outcome measure for satisfactory relief. Furthermore, this study evaluated a nondiarrhea-predominant IBS population. Diarrhea, headache, abdominal pain and nausea occurred more commonly with tegaserod than with placebo, but only 2.3% of patients discontinued tegaserod therapy due to side effects. Thus, this study not only demonstrated tegaserod's efficacy and tolerability in an Asian–Pacific population, but also suggested that tegaserod may be beneficial in IBS patients with either constipation predominance or a mixed bowel pattern. Unfortunately, this study was not designed or powered to specifically address the efficacy of tegaserod in patients with IBS and a mixed bowel pattern. The efficacy of tegaserod in Asian populations was recently confirmed in a study by Lin and colleagues, which enrolled 510 Chinese patients (87% female with a mean age of 39 years) in a multicenter, randomized, double-blind, placebo-controlled, 4-week treatment trial [47]. Similar to the ZAP study, tegaserod 6 mg twice daily was significantly more effective than placebo in relieving subjects' overall IBS symptoms, as well as constipation-related symptoms, abdominal pain and bloating.
International randomized, controlled trials.
OR: Odds ratio.
Recent studies from Europe provide further validation of the efficacy of tegaserod in patients with nondiarrhea-predominant IBS. A multicenter, randomized, controlled trial evaluating 647 IBS patients (86% female with a mean age of 44 years) from the Nordic countries enrolled patients with nondiarrhea-predominant IBS [48]. Patients enrolled in this study fulfilled the Rome II criteria but did not have diarrhea as their predominant bowel symptom. Within the first 4 weeks of therapy, those receiving tegaserod were 54% more likely to experience overall satisfactory relief of IBS symptoms than those receiving placebo, increasing to 78% over the entire 12-week treatment period. Diarrhea, headache and abodminal pain were more commonly experienced with tegaserod therapy, although only 2.8% withdrew from the trial due to side effects. A recent secondary analysis from this trial reported that tegaserod led to significant improvements in quality of life (QoL) as measured by the EuroQoL health state description (EQ-5D) questionnaire, a validated, generic QoL assessment tool [49]. In a recent large, 8-week, open-label, prospective, multicenter trial from Switzerland, 66% of nondiarrhea-predominant IBS patients experienced global symptom improvement [50]. Diarrhea was again the most common side effect, occuring in 13% of subjects in the first week and 7% thereafter. Diarrhea led to discontinuation from the trial at a rate of 8%.
Other recently published international studies provide insight into the natural history of IBS in patients who discontinue therapy with tegaserod. An open-label, multicenter trial from Latin America in 678 IBS patients with constipation (92% female and 48% Hispanic) reported overall symptom relief in 82% of patients after 4 weeks of tegaserod therapy [51]. Following the 4-week, open-label period, patients were randomized to continued tegaserod for an additional 8 weeks or withdrawal of therapy. Recurrence of IBS symptoms occurred in 10% of those maintained on tegaserod versus 67% of those who were withdrawn from therapy. Another randomized, open-label trial involving predominantly female IBS patients from the UK assessed the effects of treatment withdrawal in tegaserod responders (based on an overall relief assessment) [52]. In this trial, patients responding to tegaserod at 4 weeks were randomized to continued therapy or withdrawal of therapy for the next 8 weeks. Therapy with tegaserod was reinitiated in those experiencing early symptom recurrence. Those maintained on therapy for the entire 12 weeks were significantly less likely to experience symptom recurrence compared with those withdrawn from therapy (p < 0.0001). An additional multicenter trial from Germany evaluated the efficacy of retreatment with tegaserod in 307 predominantly female patients with IBS and constipation who suffered with symptom recurrence after an initial 12-week, open-label treatment period [53]. Nearly 90% of patients with recurrent IBS symptoms who were retreated with tegaserod responded to the second 4-week course of therapy. A recently published trial involving 2660 female IBS patients from 267 centers in 24 countries demonstrated the superiority of tegaserod over placebo for global IBS symptoms, abdominal discomfort, bloating, stool frequency and stool consistency with initial treatment, as well as with repeated treatment in those experiencing symptom recurrence [54]. In this prospective, double-blind, placebo-controlled, randomized study, female patients meeting the Rome II criteria for IBS with constipation received a 4-week course of therapy, with 34% achieving relief of IBS symptoms with tegaserod compared with 24% of those receiving placebo. Patients were then followed for symptom recurrence over the ensuing 12 weeks, with more than 90% developing recurrent symptoms. Of those with recurrent symptoms, 1191 were again randomized to treatment with a 4-week course of tegaserod or placebo, with a 45% response rate in the tegaserod group versus 29% in the placebo group. Response to therapy was rapid, occurring within the first week of therapy both with the initial and repeated treatment. These trials provide strong evidence that the majority of patients with IBS and constipation will develop recurrent symptoms upon withdrawal of tegaserod therapy. Furthermore, these studies suggest that retreatment with tegaserod for symptomatic relapses is efficacious and opens the door to the possibility of intermittent therapy. This is a potentially important observation given the transient nature of symptoms in many IBS patients and the tendency of patients to prefer intermittent or on-demand, as opposed to chronic, therapy.
A number of recently completed reviews have attempted to summarize the efficacy and safety data on tegaserod. The pivotal trials and ZAP study were critically reviewed by the American College of Gastroenterology functional GI disorders task force as part of a systematic review on the management of IBS [55]. This group concluded that, ‘the 5-HT4 receptor agonist (tegaserod) is more effective than placebo at relieving the global IBS symptom in female IBS patients with constipation’. Of the drugs reviewed, only alosetron for IBS and diarrhea and tegaserod for IBS and constipation received a grade A recommendation (evidence from more than two high-quality, randomized, controlled trials without conflicting evidence from other high-quality trials). It is important to note that other more traditional drugs may still be effective in the treatment of IBS. Certainly, the lack of credible evidence neither supports nor rejects the efficacy of a particular drug. However, until adequately powered, appropriately designed clinical trials are available, clinicians will be forced to make decisions based upon poor-quality data and/or personal experience. A Cochrane review addressing tegaserod in IBS and constipation was also published in 2004 [56]. This review included eight randomized, controlled trials including 5320 patients, of whom 90% were women. The analysis revealed a greater likelihood of global relief of GI symptoms with either tegaserod 6 or 2 mg twice daily compared with placebo, with a number needed to treat of 14 and 20, respectively. The authors concluded that tegaserod improved overall IBS symptoms, stool frequency and the number of days without a bowel movement. On the other hand, this review concluded that benefits for abdominal pain, bloating, stool consistency and straining were not clearly demonstrated. This may be related to methodologic differences between studies or a lack of a clear benefit for symptoms unrelated to the prokinetic effects of tegaserod.
Postmarketing surveillance
Tegaserod has been globally available for more than 3 years and has been used in over 3 million patients. As of April 30, 2005, patient exposure was estimated to be 640,000 patient years worldwide. Postmarketing surveillance has largely centered around side effects, particularly severe diarrhea and reports of colonic ischemia.
Harms data & tolerability
Overall, tegaserod has been shown to be safe and well tolerated in multiple, large, 12-week clinical trials [43,44,46,48,57–59,101] and a 12-month safety trial [60]. A summary of these trials is shown in Table 3. In these studies, diarrhea has consistently been the most commonly reported adverse event with tegaserod. A pooled analysis of the Phase III clinical trials, involving 2632 patients, revealed a frequency of diarrhea of 8.8% in those receiving tegaserod versus 3.8% in those receiving placebo. Tegaserod-associated diarrhea tended to occur soon after initiating therapy and was often transient. Serious diarrhea complicated by the development of hypovolemia, hypotension, syncope and the need for hospitalization occurred in 0.04% of those enrolled in randomized, controlled trials [61]. In the recent Cochrane analysis, tegaserod 6 mg twice daily led to a significantly greater likelihood of diarrhea than placebo (relative risk 2.75; 95% confidence interval [CI]: 1.9–3.97), with a number needed to harm of 20. Related to these observations, tegaserod's label was amended in the USA to include a warning for diarrhea. Coleski and colleagues described a possible mechanism for transient diarrhea associated with tegaserod in a study involving ten healthy adults [62]. Acute administration was associated with increased colon motor activity, experimentally induced peristalsis and the gastro-colonic response. Increases in colon motor activity observed with the acute use of tegaserod were significantly reduced after 1 week of treatment. The authors concluded that differential desensitization of segmental colonic propulsion may provide a possible mechanism for the transient diarrhea that can occur with tegaserod therapy.
Tegaserod-associated diarrhea (12 mg daily dose).
Adapted from [56].
In the Phase III trials, the only other adverse event that was reported more frequently with tegaserod than placebo was headache. The incidence of headache was found to be 15% with tegaserod versus 12.3% with placebo (p < 0.05) in the pooled analysis of the Phase III trials [61]. These headaches were typically mild and rarely led to discontinuation of therapy.
The long-term safety and tolerability of tegaserod was assessed in a multicenter, open-label trial involving 579 IBS patients with constipation (90% female, with a mean age of 44 years). Enrolled patients received tegaserod for a period of 12 months [60]. All patients were initiated on tegaserod 2 mg twice daily and the dose was maintained or increased to 6 mg twice daily as indicated by their clinical response. The most commonly reported adverse events included diarrhea (10%), headache (8%), abdominal pain (7%) and flatulence (6%). Overall, 11% of patients discontinued therapy over the course of the study due to adverse events. Standard laboratory evaluations, vital signs and electrocardiograms collected throughout the trial demonstrated no abnormalities.
Given the association between benzamide prokinetic agents such as cisapride and cardiac arrhythmias and tegaserod's effects on the delayed rectifier potassium current in vitro [1], the effects of this drug on cardiac conduction and rhythm have been the subject of intense scrutiny. In contrast to the prokinetic agents cisapride and erythromycin, tegaserod was shown in an animal model to lack such pro-arrhythmic effects on cardiac repolarization [63]. An initial study in 24 healthy volunteers who received doses of tegaserod ranging 50–200 mg/day demonstrated no clinically relevant changes in the QRS or QTc intervals on electrocardiography [29]. Furthermore, tegaserod was not associated with QTc interval prolongation or other electrocardiographic abnormalities in a study including 36 healthy men and an analysis of data from three randomized, double-blind, placebo-controlled trials including 2516 IBS patients with constipation [64]. In fact, no electrocardiographic parameter changes have been observed in healthy male subjects with serum concentrations of tegaserod up to 100-times greater than expected with therapeutic dosing. Finally, due to tegaserod's effects on the cytochrome P (CYP)450 system, inhibiting both the CYP1A2 and CYP2D6 isoenzymes and its prokinetic effects, five drug–drug interaction studies have been reported. Available evidence suggests that coadministration with theophylline, the prototype substrate for the CYP1A2 isoenzyme, dextromethorphan, the prototype substrate for the CYP2D6, digoxin, warfarin and oral contraceptives requires no dosage adjustment for tegaserod [30].
Other safety concerns that have been raised with tegaserod include the risk of abdominal/pelvic surgery and pregnancy-related risks. An analysis of the Phase II/III and postmarketing randomized, controlled trials involving 6197 patients demonstrated no significant difference in the rate of abdominal/pelvic surgery in those treated with tegaserod compared with those receiving placebo [65]. A total of 31 pregnancies have occurred during clinical trials with a miscarriage rate of 17% (4 out of 23) in women receiving tegaserod versus 12% (1 out of 8) in the placebo group (not significant). No other pregnancy-related complications have been reported in the clinical trials. Animal studies have found no adverse effects on fertility or evidence of teratogenicity; however, as no human studies have been performed to specifically address these issues, tegaserod has been designated as a pregnancy category B drug by the FDA.
The 5-HT3 receptor antagonists, such as alosetron and cilansetron, have been associated with an increased risk of colonic ischemia [66,67]. Concerns have recently been raised regarding the possibility of a similar association between colonic ischemia and tegaserod [67]. Although a detailed discussion of how ischemic colitis might be associated with IBS is beyond the scope of this manuscript, potential explanations include the misdiagnosis of ischemic colitis patients as suffering from IBS, the effects of altered 5-HT metabolism and signaling on visceral blood flow in IBS patients [68] and transient ischemia produced by exaggerated high-amplitude propagating colonic contractions in IBS patients [69]. Randomized, controlled trials involving 11,640 patients, resulting in 3456 patient years of tegaserod exposure, have produced no reports of colonic ischemia [61]. During postmarketing surveillance of tegaserod, 26 cases of possible colonic ischemia have been reported worldwide. This provides an estimated incidence of 7–8 cases of colonic ischemia per 100,000 patient years of tegaserod use [61]. The event rate for possible colonic ischemia with tegaserod can be better put into perspective when one considers the respective event rates for the general population and patients with IBS. Based upon recent epidemiologic data, IBS in and of itself may be associated with a three- to fourfold increase in the likelihood of colonic ischemia compared with the general population (general population: 4–47/100,000 person years vs IBS: 43–179 cases/100,000 patient years) [70,71]. Unfortunately, this matter may never be definitively resolved as an adequately powered, randomized, controlled clinical trial is unlikely to ever be performed. As a result of the recently reported cases of possible colonic ischemia, a precaution was added to the label of tegaserod in the USA. A precaution is intended to increase awareness that an adverse event has been reported in patients using a drug but does not imply a cause and effect relationship between the drug and the event.
Regulatory affairs
Tegaserod is presently approved for use in more than 55 countries and marketed in more than 30 countries under the trade names Zelmac® or Zelnorm®. Tegaserod is currently approved by the FDA in the USA for use in the treatment of women with IBS and constipation, and women and men under the age of 65 years with chronic idiopathic constipation. The manufacturer of tegaserod, Novartis Pharmaceuticals, has submitted an application to the European Union for an indication in patients with IBS; however, the final word regarding this application is not expected until some time in 2006.
Future perspective
Although it is now clear that tegaserod offers benefits for global symptoms and constipation-related complaints in a subset of patients with IBS and constipation, a number of important clinical questions remain. First, whether tegaserod possesses clinically important visceral analgesic properties remains to be adequately elucidated. As has already been discussed, there are animal and emerging human data to support the effects of tegaserod on visceral sensation [39–42]. However, the benefits of tegaserod for abdominal pain in patients with IBS have been conflicting [56]. Furthermore, human data to better understand the mechanisms by which tegaserod affects visceral sensation and the magnitude of any such effect are eagerly awaited.
Bloating is a common and bothersome symptom in patients with IBS. The current understanding of the pathogenesis of bloating in IBS is quite limited. Perhaps more importantly, our therapeutic armamentarium for bloating is virtually nonexistent. Several studies suggest that tegaserod offers benefits for bloating in IBS patients. This was confirmed by a recent pooled analysis presented at Digestive Diseases Week 2004 (IL, USA) [72]. Whether the suggested benefits of tegaserod for bloating are clinically meaningful requires validation. Furthermore, the precise mechanism for the benefits of tegaserod in bloating remain to be determined, although preliminary evidence suggests that the prokinetic properties of this drug provide the most likely explanation [73,74].
The efficacy of tegaserod in men with IBS and constipation requires further evaluation. Available randomized, controlled clinical trials have not reported any significant benefits of this drug in men with IBS. It is possible that the lack of benefit may represent a Type II error, as the overwhelming majority of patients enrolled in the IBS clinical trials were women. However, it is also possible that differences in the pathogenesis of IBS in females and males account for the difference in efficacy of tegaserod. The recent reports of benefits from tegaserod in men with chronic idiopathic constipation at least raises the possibility of treatment in men with IBS [75,76].
Two linked issues that require further study include the long-term efficacy of tegaserod and whether a subset of tegaserod responders develop tolerance with extended treatment. The first 12-month efficacy data for tegaserod were recently presented in abstract form at Digestive Diseases Week 2005 (IL, USA). This study found that 59% of initial responders to tegaserod remained responders at 12 months. A total of 14% of patients became nonresponders and 27% discontinued for other reasons [77]. These data suggests that tolerance may develop in a small subset of patients receiving tegaserod for extended periods. If tolerance to tegaserod occurs in a subset of IBS patients, further studies are necessary to better understand the mechanism of action, incidence, time of onset and strategies to avert this clinically important problem.
In addition to improvements in global and individual symptoms, an ideal therapy for IBS would improve disease-specific and generic QoL, decrease health resource utilization and improve work productivity. A recent double-blind, placebo-controlled, multinational trial involving 2660 women with IBS and constipation demonstrated that tegaserod decreased work productivity loss and activity impairment to a significantly greater degree than placebo [78]. A recent retrospective, longitudinal, parallel cohort study comparing 3365 tegaserod users with 3364 nonusers from a large, geographically diverse, managed care organization, demonstrated decreased GI-related resource utilization in tegaserod users compared with nonusers [79]. An abstract recently presented at an international meeting provides further evidence that tegaserod may improve other QoL outcomes [49]. An additional issue that requires formal assessment is the cost effectiveness of tegaserod in IBS patients. To date, this issue has not been adequately examined. The ability to utilize tegaserod in an on-demand or as-needed fashion would greatly enhance its potential cost-effectiveness. As has been discussed, a recent study demonstrated the effectiveness of retreatment with tegaserod. A study specifically designed to assess the efficacy of tegaserod when used in an on-demand or as-needed fashion would be of great interest to clinicians managing IBS patients.
Tegaserod's physiologic properties are likely to provide clinical applications beyond IBS. Tegaserod has been shown in large, randomized, controlled trials to be safe, well tolerated and effective in the treatment of idiopathic chronic constipation [75,76]. A number needed to treat for chronic constipation was calculated at 7.3 for tegaserod 6 mg twice daily and 11.1 for tegaserod 2 mg twice daily. Tegaserod was well tolerated, with the most common side effects including headache and diarrhea, of which only diarrhea occurred more commonly than with placebo. A recent abstract reported the long-term safety and tolerability of tegaserod in chronic constipation for up to 16 months [80].
Other potential applications for tegaserod include gastroparesis, functional dyspepsia and fuctional esophageal disorders. A recent study utilizing an animal model demonstrated tegaserod's ability to improve gastric emptying in the setting of diabetic gastroparesis [81]. There is evidence to support tegaserod's ability to normalize iatrogenically induced delayed gastric emptying [82]. Two recent, small studies have demonstrated the superiority of tegaserod over placebo in improving delayed gastric emptying in dyspeptic patients with gastroparesis [83,84]. A recent study demonstrated an increase in meal-induced gastric accomodation with the use of tegaserod in a small number of patients with functional dyspepsia and normal gastric emptying [85]. Large studies in patients with functional dyspepsia are currently underway. One final area of anticipated future research may include the therapeutic potential of tegaserod in the treatment of functional esophageal disorders. A recent study evaluating 42 patients with functional heartburn demonstrated significant improvements in a number of symptom parameters with tegaserod over placebo [86].
Conclusion
Tegaserod, a high-affinity 5-HT4 partial agonist, has become one of the accepted pharmacologic agents for the treatment of women with IBS and constipation. Unlike many other pharmacologic agents directed at the treatment of individual IBS symptoms, tegaserod has been shown to provide relief of global IBS symptoms. Furthermore, tegaserod has proven to be safe and well tolerated in multiple large, randomized, controlled trials around the world. The most commonly encountered side effects include diarrhea and headache. Although cause and effect has not been established, a small number of cases of possible colonic ischemia have been reported in patients receiving tegaserod during postmarketing surveillance. Important questions that remain to be more clearly answered include the role of tegaserod in the treatment of pain and bloating in IBS, whether tegaserod has a role in male IBS patients, the long-term efficacy of tegaserod, whether tolerance develops in a subset of patients with extended therapy, and whether tegaserod is beneficial for the treatment of other functional GI disorders.
Executive summary
Tegaserod is a selective serotonin (5-hydroxytryptamine [5-HT])4 receptor partial agonist.
It binds with high affinity to 5-HT4 receptors, with a potency that is a fifth of endogenous 5-HT.
Tegaserod's actions on 5-HT4 receptors trigger the release of other neurotransmitters, including calcitonin gene-related peptide, substance P and vasoactive intestinal peptide.
This in turn augments peristalsis, increases colonic propulsion, increases gastric and small intestinal motility, increases intestinal secretion, and exerts effects on visceral sensation.
It has limited affinity for 5-HT1 receptors and no appreciable affinity for other 5-HT receptors, muscarinic, adrenergic, dopaminergic or opiate receptors.
When ingested in the fasting state, tegaserod is rapidly absorbed, with a bioavailability of 10%.
Absorption is independent of dosing, with a daily dose range of 2–12 mg.
Median time to peak concentration is 1–1.3 h. It has a mean elimination half-life of 7.5 h following oral administration with a steady state obtained in 8 days.
It is 98% bound to plasma proteins, predominantly to α1-glycoproetein.
Tegaserod is not known to cross the blood-brain barrier.
20% of the drug is metabolized via hydrolytic breakdown in the stomach followed by oxidation and glucouronidation.
A second metabolic pathway involves direct glucuronidation, leading to the generation of three isomeric N-glucuronides.
Two-thirds of tegaserod is excreted unchanged in the feces, with the remainder excreted in the urine, primarily as the main metabolite.
There is no need for dosage adjustment based on body weight, age, race or gender.
No dosage adjustment is required with mild-to-moderate hepatic or renal impairment. It is not recommended in patients with severe hepatic or renal impairment (creatinine clearance of <15 ml/min).
The efficacy of tegaserod has been demonstrated in numerous randomized, placebo-controlled trials involving more than 5600 patients (>85% female) with irritable bowel syndrome (IBS) and constipation.
The effects are typically experienced within 1 week of starting treatment and persist over 12 weeks in the majority of patients.
Effects of therapy include an improvement in global IBS symptoms, increase in daily bowel movements and reduction in the number of days without a bowel movement.
There may also be an improvement in abdominal pain, bloating, stool consistency and straining, although these effects are less consistent in the clinical trials.
The effects of tegaserod in male patients with IBS and constipation remain unclear.
There is evidence to suggest efficacy in IBS patients whose predominant bowel complaint is not diarrhea. To date, the clinical trials have not specifically addressed the efficacy of tegaserod in patients with IBS and a mixed bowel pattern.
There are preliminary data to suggest efficacy with long-term therapy (greater than 12 weeks) and for the retreatment of recurrent IBS symptoms.
The most commonly reported side effects with tegaserod include diarrhea and headache.
There have been isolated cases of possible colonic ischemia reported in patients receiving tegaserod; however, a cause and effect relationship between tegaserod and colonic ischemia has not been established.
Animal studies have revealed no association between tegaserod and infertility or fetal harm. Formal testing has not been performed in humans. The US Food and Drug Administration has assigned tegaserod to pregnancy category B.
Although tegaserod has an inhibitory effect on the isoenzymes cytochrome P (CYP)1A2 and CYP2D6 of the CYP450 system, there is currently no evidence of clinically significant drug-drug interactions.
Tegaserod is available as 2 and 6 mg tablets.
Tegaserod's bioavailability is reduced by 40–65% if taken with food; therefore, it should be taken prior to a meal.
Footnotes
Dr Chey is on the speakers bureau for Novartis.
