Abstract
Nocturia, one of the most bothersome of all urological symptoms, has previously been both poorly classified and understood. Multiple factors may result in nocturia, among which are pathological conditions such as cardiovascular disease, diabetes mellitus, lower urinary tract obstruction, anxiety or primary sleep disorders, and behavioral and environmental factors. Nocturia may be attributed to nocturnal polyuria (nocturnal urine overproduction), diminished nocturnal bladder capacity, polyuria or a combination of the three. These conditions can be distinguished by a simple arithmetic analysis of the patients 24-h voiding diary. After reviewing the current state of knowledge, a scheme for rational diagnosis and care of patients suffering from loss of sleep due to nocturnal micturition is presented in this article.
One of the most common reasons for interrupted sleep in the general adult population is nocturia – waking during the night to urinate [1]. Many individuals with nocturia suffer from other lower urinary tract symptoms (LUTS) such as urinary frequency, weak stream, urgency and incontinence. In women, these symptoms are often considered to result from aging, a consequence of childbirth or just ‘being a woman’. The majority of elderly patients with nocturia are likely to be exposed to serious health risks since nocturia causes fatigue due to sleep deprivation, which increases the chance of traumatic injury through falling [2]. In one study comparing night-time falls in elderly people, those with nocturia were at a significantly greater risk of falling – their risk increasing from 10 to 21% with two or more micturitions/night [3]. Epidemiological data point toward the high prevalence of nocturia in women (63% of 479 healthy females) [4].
The International Continence Society's definition of nocturia is the condition of waking up to void one or more times during the night [5]. A stricter definition would be voiding during intended sleep time that is preceded and followed by sleep. This is an important distinction, since many in the general population are shift workers who sleep during the day. Nocturia is not necessarily troublesome; voiding less than twice per night is often considered to be normal. The underlying pathophysiology that accounts for nocturia in all of these conditions falls into four broad categories:
Nocturnal polyuria (NP)
Low nocturnal bladder capacity (NBC)
Mixed (a combination of NP and low NBC)
Global polyuria
These categories are derived from the 24-h voiding diary in which each voided volume and its corresponding time is tabulated.
In general, nocturia may be associated with medical causes, originating outside the lower
urinary tract, and/or urological causes. In general, the former will be related to NP or
global polyuria. These are usually best evaluated and treated by a primary care physician or
internist. The latter includes conditions requiring treatment by a urologist; these conditions
are generally associated with diminished NBC. An algorithm for the analysis of nocturia is
presented in
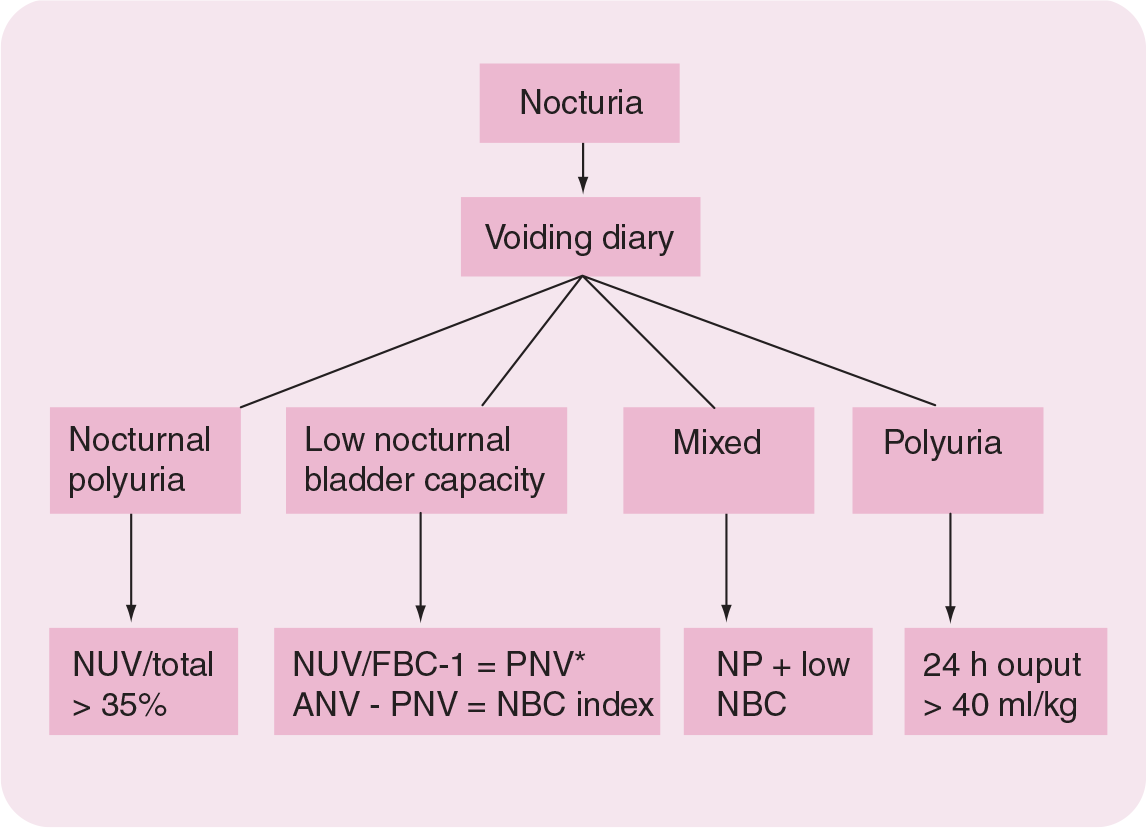
Algorithm for voiding diary analysis of the etiology of nocturia.
Diminished nocturnal bladder capacity
Nocturia due to diminished NBC is of two general types – decreased functional bladder capacity (FBC) and decreased NBC. FBC is the maximum voided volume (MVV [now preferred International Continence Society nomenclature]) recorded on a bladder diary. In both conditions, nocturnal urinary volume (NUV) exceeds bladder capacity and the patient is awakened by the need to void because the bladder does not hold enough. If the FBC is less than the NUV, nocturia ensues [6].
In order to quantify NBC, several terms need to be defined – the nocturia index (Ni) and the NBC index (NBCi). The Ni is NUV/FBC, remembering to include the first morning void in the NUV. The Ni minus one (rounded up to the nearest integer) equals the predicted number of nightly voids (PNV). The NBCi is defined as the difference between the PNV and the actual nightly voids (ANV). The significance of this is that the greater the NBCi, the more nocturia may be attributed to diminished NBC and sensory urge disorders. For example, if NUV is 750 ml and the FBC is 250 ml, the Ni is 750/250 = 3. This patient would have a PNV of 2 (Ni −1 = 2) and would be expected to void twice per night – the first 500 ml during sleep hours in two voids and then awaken and void the remaining 250 ml. In the illustration above, if the patient actually arose seven times to void the same 750 ml, she would have a NBCi of 5 (7[ANV]-2[PNV]). This woman, for whatever reason, has significantly diminished bladder capacity during sleep hours. Thus, the higher the NBCi the lower the NBC (compared with the FBC) [7]. The causes of decreased NBC are listed in Box 1.
Treatment of diminished nocturnal bladder capacity
An overview of Box 1 indicates that low NBC is primarily a urological as opposed to medical diagnosis. Thus, infravesical obstruction, infection and cancer may all require specific treatment by a urologist as opposed to, for example, diabetes mellitus (DM) or obstructive sleep apnea (OSA), which may cause nocturia/NP and require treatment by their respective medical specialists. Interestingly, low NBC is not related to detrusor instability, at least not that can be diagnosed through awake urodynamic studies [8].
The role of antimuscarinics in the treatment of nocturia in patients with overactive bladder (OAB) is currently being investigated. Episodes of nocturia were statistically significantly decreased in patients treated with solifenacin 10 mg (−0.71, −38.5%) versus placebo (−0.52, −16.4%; p = 0.036) [9]. Tolterodine extended-release caused a 59% median reduction in OAB-specific nocturnal voids compared with a 43% reduction by placebo (p = 0.02), although overall benefit for all types of nocturic micturitions was not seen [10].
Nocturnal polyuria
The increased production of urine at night experienced in NP is offset by lowered daytime urine production such that the 24-h urine volume remains normal [11]. NUV is defined as the total urinary output during the hours of sleep, to which is added the volume of the first morning void within 30 min of arising. It is assumed the patient voids just prior to retiring so that all urine voided during sleep time plus the first morning void belongs with the tally of NUV. There are several definitions of NP including NUV greater than 6.4 ml/kg; nocturnal diuresis greater than 0.9 ml/min; and NUV/24-h urine volume of more than 35% [12]. The authors favor the latter definition. Data supporting the latter definition are found in a study including elderly (aged > 70 years) women in whom NUV/24-h volume was 45% in nocturics as compared with 31% in non-nocturics [13]. In the same study, nocturics excreted a greater NUV than non-nocturics (700 and 417 ml, respectively) and had smaller FBCs (325 vs 400 ml). However, the most dramatic parametric distinction between nocturics and non-nocturics lies in analysis of the Ni, where Ni = 2.1 for nocturics and 1.0 for non-nocturics, implying that the most significant reason for nocturia in the elderly is a mismatch between volume of urine excreted and the bladder's capacity to hold adequate urine volumes during the hours of sleep [13].
The etiology of NP is diverse and includes congestive heart failure, DM, diabetes insipidus (DI), cerebrovascular accident (via effect on hypothalamic–pituitary axis and resultant loss in circadian rhythmicity of antidiuretic hormone (ADH) secretion [14]), third space fluid resorption (venous stasis, nephrotic syndrome or hepatic failure) and late evening diuretic/fluid intake (Box 2).
Common causes of low nocturnal bladder capacity.
Infravesical obstruction (e.g., pelvic organ prolapse or postsurgical)
Idiopathic nocturnal detrusor overactivity
Neurogenic bladder
Cystitis: bacterial, interstitial, tuberculous or radiation
Cancer of bladder or urethra
Acquired voiding dysfunction
Anxiety disorders
Pharmacological: xanthines (theophylline, caffeine); β-blockers
Bladder calculi
Ureteral calculi
Abnormalities of actual nightly void secretion
Urine production assumes a standard circadian pattern, which is age-dependent. In young people (aged < 25 years), mean NUV is 14% as compared with those over the age of 65 years whose mean NUV is 34% [15]. This circadian pattern appears to be closely related to a corresponding increase in secretion of ADH during the hours of sleep, a change in which may cause NP in the elderly [12]. One reason for this diurnal change is thought to be a disruption of the diurnal variation in secretion of ADH. ADH is synthesized in the supraoptic and paraventricular nuclei of the hypothalamus and is released by the posterior pituitary in response to various stimuli such as increased plasma osmolality [12]. ADH, normally secreted in a diurnal pattern, is partly responsible for the regulation of urine production [16]. Since ADH increases the resorption of water from the renal tubule, higher concentrations of ADH occurring at night result in the production of lower volumes of concentrated urine. Plasma ADH levels are often undetectable during the night in elderly subjects with nocturia, thus implying a cause-and-effect relationship between ADH secretion and NP [17].
Causes of nocturnal polyuria.
Congestive heart failure
Diabetes mellitus
Obstructive sleep apnea
Peripheral edema
Excessive night-time fluid intake
Loss in circadian rhythmicity of arginine vasopressin secretion
In the renal inner medullary collecting duct, ADH regulates two key transporters: aquaporin-2 (AQP2) and vasopressin (ADH)-regulated urea transporter (VRUT). AQP2 is present in intracellular vesicles as well as the apical plasma membrane. Short-term regulation of AQP2 (but not VRUT) occurs by ADH-induced trafficking of AQP2-containing vesicles to the apical plasma membrane [18]. Long-term regulation is such that a prolonged (>24 h) increase in circulating ADH leads in turn to increased AQP2 production [19]. Reduced secretion of ADH and absorptive hypercalciuria are independently associated with decreases in nocturnal urinary AQP2 and resulting NP [19]. ADH causes water reabsorption in the collecting duct via the V2 receptor, which promotes a cascade of events leading to activation of G-protein. This causes adenylate cyclase to convert ATP to cAMP [20]. cAMP then activates ‘protein kinase A’, which stimulates intracellular vesicles containing AQP2 water channels. The latter translocate to the apical membrane by exocytosis of intracellular vesicles, leading to water transport from urine into the circulation. When ADH levels are low, water permeability is decreased by the shift of AQP2 channels back into the intracellular vesicles [18].
Obstructive sleep apnea
Sleep apnea, a form of sleep-disordered breathing, is defined as sudden cessation of respiration due to airway obstruction during sleep. Respiratory diseases associated with increased airway resistance such as OSA are associated with increased renal sodium and water excretion mediated by plasma atrial natriuretic peptide (ANP) levels [12,21]. The prevalence of OSA is approximately 2% in women [22]. The mechanism for elevated ANP release associated with OSA has been demonstrated to be increased right atrial transmural pressure resulting from hypoxia-induced pulmonary vasoconstriction [21]. Hence, a population of patients with nocturia may result from OSA and secondary NP [23]. Polysomnographic sleep studies including measurement of plasma oxygen saturation and respiratory patterns are therefore recommended in patients with nocturia with a suspected relation to OSA. Patient selection is based upon increased risk as follows: patients with N P, morbid obesity, acromegaly, asthma, hypertension, adult-onset DM and craniofacial abnormalities may be submitted for sleep studies owing to their 30–40% chances of having OSA [22,23]. Treatment of OSA includes nasal continuous positive airway pressure during sleep, as well as uvulectomy/tonsillectomy when these structures are causing obstruction.
Treatment of nocturnal polyuria
Remediable medical causes of NP should be identified and treated but in some instances nocturia persists and, in most patients, clearly identifiable remediable conditions are not found. Empiric treatment options include evening fluid restriction (a form of behavior modification), timed diuretics [24,25], afternoon naps and/or elevation of the legs, application of compressive stockings in patients with edema or varicosities and ADH administration [26,27]. The latter should be avoided or used judiciously at best in patients with congestive heart failure.
In the elderly, simple fluid restriction is rarely effective in reducing NP that is due to the mobilization of gravitational-induced third space of interstitial fluid residing in the lower extremities upon achieving the recumbent position during the hours of sleep. Compressive devices may prevent this fluid accumulation and help to diminish nocturnal urinary output. In addition, late afternoon naps with elevation of the legs simulate sleep hours during the day and may diminish the burden of fluid excretion otherwise inevitable during normal sleep time. If edema is present either in the legs or presacral area, diuretics may be helpful to diminish this third spacing. Virtually all diuretics act within 2 h of administration and, therefore, may be given in mid-to-late afternoon or early evening, as they are least needed just after arising and may exacerbate NP if given later in the evening. Bumetanide has been shown to reduce the number of weekly night-time voids by four in patients without obstructive symptoms [25]. In addition, furosemide has been shown to be effective at reducing both nocturia and NP [24]. Doses of the diuretic taken 6 h before bedtime were shown to reduce the number of night-time voids by 0.5, and to lower the percentage night-time voided volume by 18%. Evidence has been presented for the treatment of NP with imipramine [28]. The mechanism for its antidiuretic effect is thought to result from imipramine-mediated α-adrenergic stimulation in the proximal nephron along with increased urea and water reabsorption in the distal nephron. Nonsteroidal anti-inflammatory drugs have been proposed as treatment for NP mediated by inhibition of prostaglandin E2-mediated diuresis [29].
Exogenous administration of antidiuretic treatment is effective in preventing nocturia in women [30]. Desmopressin (1-deamino-8-d-arginine-vasopressin [DDAVP]) is an ADH analog that has been proven to be effective and well tolerated in the treatment of neurogenic DI [31] and enuresis, both in children [32] and adults [33]. DDAVP has also been shown to reduce or eliminate nocturnal voiding in patients with autonomic dysfunction and Parkinson's disease [34]. Patients with nocturia due to multiple sclerosis have been successfully treated with desmopressin, experiencing a decrease in the number of voids/night and a corresponding increase in nights free from voiding and hours of uninterrupted sleep [35]. In 142 women receiving desmopressin or placebo, the proportion of women who showed at least a 50% reduction in the number of nightly voids during the double-blind period was 46% in the desmopression group as compared with 7% in the placebo group (primary end point; p < 0.0001) [30]. In the latter study, nocturnal urine output decreased significantly in the desmopressin group (from 1.51 to 0.82 ml/min) compared with the placebo group (secondary end point, from 1.44 to 1.35 ml/min) [30].
The authors' group's protocol involves beginning with 10 μg of intranasally or 0.1 mg orally administered desmopressin increased by increments of 10 μg or 0.1 mg, respectively, every third night until the desired effect is reached, to a maximum dose of 40 μg or 0.4 mg, respectively, before going to sleep. The patient is seen again 7 days after the first dose, since if electrolyte abnormalities or fluid overload occur, they are most likely to commence during this initial time interval. After each dose increment, the patient should be questioned regarding headache, nausea, vomiting and light-headedness. If any of these symptoms occur, the medication should be discontinued until electrolyte values are known. The patient or a caregiver should be taught to monitor the legs and presacral area for edema and daily weights for early identification of excess fluid retention induced by desmopressin in susceptible patients. Significant weight increases, new onset or worsening of edema, or symptoms of hyponatremia such as headache or visual disturbance should be reported immediately to the physician and the medication discontinued. The above protocol may be prescribed to appropriately selected patients with nocturia owing to nocturnal urine overproduction who are aged 21 years or over.
Nocturia case study.
– Recent body computed tomography scan unremarkable.
– Usual voided volume = 300 ml.
– Functional bladder capacity = 1220 ml. Awake hours: number of voids = 14. Sleep hours: number of voids = 3.
– Nocturnal urine volume (NUV) = 1530 ml.
– Nocturia index = 1.2.
– NUV/total (nocturnal polyuria index) = 29%.
– Nocturnal bladder capacity index = 2.
In the opinion of the authors, claims of lack of side effects from antidiuretic treatment apply to pediatric patients with enuresis and cannot be expected in the elderly, especially those with a history of congestive heart failure. It is important to remind patients receiving desmopressin that they should sharply curtail their evening water intake in order to minimize the occurrence of fluid retention. Recent research suggests that there is a relationship between gender, plasma level of desmopressin and the incidence of adverse events. Specifically, women seem to have a significantly higher plasma desmopressin concentration and more adverse events than do men [36]. Elderly individuals with congestive heart failure have high, sometimes very high, levels of circulating ADH. Desmopressin should not be used in these cases as it is not very probable that it would be effective. Individuals with NP due to lack of nocturnal rise in circulating ADH, in contrast, show in general no symptoms of cardiac failure.
Mannucci and coworkers were the first to demonstrate the therapeutic value of desmopressin in the treatment and prevention of bleeding complications in patients with mild forms of hemophilia A and von Willebrand's disease [37]. However, the doses of desmopressin used in the treatment of coagulation disorders are approximately ten-times higher than those recommended for antidiuretic therapy. Thus, it is unlikely that the dosage levels of desmopressin used in treatment of NP would be associated with thrombotic complications.
The efficacy of any combination of the above treatment methods for NP may be easily determined using repeated voiding diaries.
Polyuria, disorders of thirst & diabetes insipidus
Polyuria (defined as 24-h urine output in excess of 40 ml/kg/24 h) is related to increased intake so that polyuria and polydipsia (at least in the steady state) are closely related [15]. Polyuria thus results in both day and night urinary frequency due to global urine overproduction in excess of bladder capacity. Causes of polyuria include:
DM
DI
Primary thirst disorders
While polyuria causes increased NUV similar to the situation for NP, treatment is directed at a reduction in both water intake and its resultant output through specific measures such as insulin replacement, voluntary restriction of water intake or supplementary administration of ADH analogs, where appropriate.
The causes of DI are generally classified into two categories: central (neurogenic) and renal (nephrogenic); polydipsia (either dipsogenic or psychogenic) may cause polyuria but is not considered a form of DI. In central DI, there is a lack of production of ADH from the posterior pituitary, for example, due to the loss of neurosecretory neurons in the hypothalamus as a result of surgery, infection (e.g., tuberculosis or meningitis), or primary (craniopharyngioma) or metastatic (e.g., lung or breast) tumor or head trauma. Infiltrative disease, such as sarcoid or Wegener's granulomatosis, plus idiopathic conditions, account for the remainder of cases of central DI. Renal (nephrogenic) DI is caused by defective renal responsiveness to adequate circulating levels of ADH so that the renal tubules are incapable of water reabsorption, leading to dehydration and excessive thirst with secondary polydipsia. Etiologies of nephrogenic DI include hypercalcemia, hypokalemia, lithium, tetracyclines and abnormal secretion of ANP. Psychogenic polydipsia presents as DI due to compulsive water drinking. There may be an associated secondary nephrogenic component to DI in these patients owing to washout of the countercurrent multiplier gradient responsible for renal concentration of urine. This defect may reverse with psychiatric treatment of the underlying compulsion to drink excessive volumes of water. Patients with primary thirst disorders (dipsogenic polydipsia) may present with global polyuria and secondary N P. Dipsogenic polydipsia usually occurs in the setting of central neurological disorders related to brain tumors, trauma or radiation injury.
The differential diagnosis of DI may be made by careful diary evaluation of urinary output in addition to use of both the overnight dehydration test and the renal concentrating capacity test (RCCT). Overnight dehydration normally results in transformation of initially dilute urine to urine concentrated to an osmolality of over 600 mOsm/kg. A normal overnight dehydration test assures that both central production of and peripheral renal responsiveness to ADH is normal, thus excluding all forms of DI. These patients with polyuria have a primary thirst disorder, either psychogenic or dipsogenic. Patients with psychogenic polydipsia usually have low to low–normal serum sodium levels. If polyuric women continue to have dilute urine after overnight dehydration, a test of renal responsiveness to desmopressin is in order. In adults, 40 μg desmopressin is administered intranasally owing to a greater reliability of absorption as compared with the enteric route of administration. The bladder is emptied and a urine sample for osmolality obtained 3–5 h later. Water intake is restricted for the first 12 h after drug administration (so as to avoid hyponatremia from desmopressin in patients with polydipsia). The reference level for normal urine osmolality after desmopressin administration is 800 mOsm/kg, although polyurics rarely attain this degree of urine concentration; a more realistic target is more than 600 mOsm/kg. Following desmopressin administration, urine osmolality of under 550 mOsm/kg suggests nephrogenic (or chronic central) DI, whereas urine osmolality of 600–800 mOsm/kg is consistent with psychogenic or dipsogenic polydipsia. However, after several days of desmopressin administration, patients with central DI develop normal concentrating capacity.
Treatment of polyuria is in accordance with etiological diagnosis: reduced water intake for those without DI; specific treatment of DM; ADH analogs for central DI; and psychotherapy for compulsive water drinkers.
Nocturia & aging
In a study of 850 patients (of whom approximately half were women) with symptoms of OAB, younger patients (<50 years) were more likely to have diminished NBC (higher NBCi) and older patients (progressively for those aged 50–70 and >70 years) were more likely to have nocturnal urine overproduction [38]. In a cohort of elderly patients, nocturics had significantly greater nocturnal urine output (mean 700 ml) and diminished NBC (mean 325 ml) than their counterparts without nocturia (417 and 400 ml, respectively). The combination of increased output and decreased bladder capacity at night appears to account for the increasing incidence of nocturia that accompanies aging [13].
Mixed causes for nocturia
Many patients with nocturia are found to have a combination of NP and low NBC (‘mixed’ etiology of nocturia). The authors have reviewed 194 patients with nocturia, of whom 66% were women, and found that 13 (7%) had pure N P, 111 (57%) had diminished NBC and 70 (36%) had a combination of the two. Of these 194, 45 (23%) also had global polyuria [39]. Specific treatment of these patients should be directed at both disorders.
Nocturia case study
See Box 3. Urodynamic studies (performed to determine whether underlying vesicourethral function was normal in view of the cramping symptoms) revealed large capacity bladder, normal vesicourethral function and mild cystocele (Figures 2 & 3).

Urodynamic tracing showing large capacity bladder and normal vesicourethral function.

Cystogram demonstrating mild cystocele.
The patient has nocturia and suprapubic cramping associated with polyuria (>40 ml/kg/24 h) and no relevant endoscopic or urodynamic abnormalities. In the authors' view, the global polyuria accounted for her LUTS. Thus, testing was directed at identifying the cause for her polyuria. The results of an overnight dehydration test are shown in Table 1.
Results of overnight dehydration test.
Subsequent voiding diary after behavior modification for psychogenic polydipsia.
The patient was unable to fully concentrate her urine overnight, although this is a common problem in patients who are chronically overhydrated. A fasting serum ADH level was normal, excluding central DI. Therefore, a renal concentrating capacity test was subsequently carried out, comparing baseline serum and urine osmolality with the same parameters obtained 4 h following administration of desmopressin 40 μg intranasally. Baseline serum sodium was 140 mmol/l.
Comment
The patient's baseline urine osmolalities are low, but the RCCT excludes nephrogenic DI as the cause (post-DDAVP urine osmolality < 500 mOsm/kg is necessary for a diagnosis of nephrogenic DI) (Table 2). This leaves psychogenic versus dipsogenic polydipsia in the differential diagnosis. Magnetic resonance imaging (MRI) scanning of the head, carried out to exclude a pituitary tumor, was normal.
Results of renal concentrating capacity test.
DDAVP: 1-deamino-8-
As the patient denied water drinking to result from abnormal thirst and in view of the lack of a history of head trauma, surgery or radiation (along with a normal brain MRI) the diagnosis of psychogenic polydipsia was established. The patient was thus treated with behavior modification. The outcome is shown in Box 4.
Symptoms of both nocturia and suprapubic cramping have now resolved with near resolution of the patient's polyuria. Serum sodium levels have remained normal throughout treatment. If she had evidence of central DI, the authors would expect ADH replacement therapy with desmopressin to have equally beneficial results. While it is routine to perform urodynamic studies in most patients with voiding dysfunction, in retrospect this patient could have been managed without them [40,41].
Future perspective
The key to progress in the treatment of nocturia will involve education of healthcare providers in gaining an understanding of nocturia as a syndrome as opposed to a symptom, principally through widespread employment of micturitional diaries.
Safer, more specific strategies for the treatment of nocturnal urine overproduction will be developed in appropriate women.
Over the next 5–10 years, the author anticipates improvements in the treatment of diminished bladder capacity through agents that work centrally, as antimuscarinics have been shown to have minimal effects on NBC and nocturia in general.
The implications of menopause, aging, obesity, pregnancy, smoking and various dietary factors have not been rigorously studied with respect to their impact on nocturia in women. Nocturia as a career research topic is available at present for those interested in academic medicine, obstetrics/gynecology and urology.
Executive summary
Nocturia due to diminished nocturnal bladder capacity (NBC) is owing to a urological abnormality such as nocturnal detrusor overactivity, lower urinary tract obstruction or primary bladder disease.
Treatment of diminished NBC involves correction of the specific urological abnormality relevant to the symptomatology. For example, nocturnal detrusor overactivity may be treated with antimuscarinics at bedtime while pelvic floor dysfunction may require behavior modification with or without biofeedback.
The increased production of urine at night experienced in nocturnal polyuria (NP) is offset by lowered daytime urine production such that the 24-h urine volume remains normal.
The definition of NP is age-dependent; for example, in young people (aged <25 years), mean nocturnal urinary volume is 14% as compared with those over the age of 65 years, whose mean nocturnal urinary volume is 34%.
Congestive heart failure.
Diabetes mellitus (DM).
Obstructive sleep apnea.
Peripheral edema.
Excessive night-time fluid intake.
Loss in circadian rhythmicity of arginine vasopressin secretion.
Correction of the specific etiology defined above.
Simple and universally safe treatments (e.g., compressive stockings and afternoon naps for peripheral edema) should precede more complex therapy such as late-afternoon diuretics or bedtime antidiuretics.
Polyuria is defined as 24-h urine output in excess of 40 ml/kg/24 h.
Polyuria thus results in both day and night urinary frequency due to global urine overproduction in excess of bladder capacity.
Causes of polyuria include DM and diabetes insipidus (DI), and primary thirst disorders.
Treatment of polyuria is in accordance with etiological diagnosis: reduced water intake for those without DI; specific treatment (e.g., dietary, insulin or oral hypoglycemics) of DM; antidiuretic hormone analogs for central DI and psychotherapy for compulsive water drinkers.
Younger patients (aged <50 years) are more likely to have diminished NBC and older patients (progressively for ages 50–70 and >70 years) are more likely to have nocturnal urine overproduction as the etiology of nocturia.
The combination of increased output and decreased bladder capacity at night appears to account for the increasing incidence of nocturia that accompanies aging.
In patients with a combination of nocturnal polyuria and decreased bladder capacity, any serious disorders such as bladder cancer, diabetes or heart failure should be corrected promptly.
Initial treatment for mixed nocturia etiologies should be the safest, least invasive therapies followed by progressively more invasive therapies, such as bladder neck resection (for obstruction) or neuromodulation (for overactive bladder).
