Abstract
Defined as the presence of functional endometrial tissue outside the uterine cavity, endometriosis is a chronic and recurrent disease that affects 7–10% of reproductive-age women, causing pain, infertility or both, resulting in serious life disruption, emotional and physical suffering, and decreased productivity. The observation that endometriosis is more common in women with early ménarche, polymenorrhea, outflow genital tract obstruction or a family history of disease suggests that its development depends on the complex interaction of genetic, immunological, environmental and hormonal factors. Current therapies for the management of endometriosis are medical, surgical or both. Medical therapies induce a hypoestrogenic state to reduce menstrual flow and apoptosis of endometriotic lesions. They include androgenic or progestational compounds and gonadotropin-releasing hormone analogs, which are comparably effective but have very different adverse-effect profiles. Consequently, the choice of which medical treatment to prescribe may not be based on differences in efficacy but on differences in tolerability, safety and, when everything else is equal, cost. Although surgery to remove endometriosis is effective in relieving pain and restoring fertility, recurrence of symptoms is common and repeated medical and surgical interventions are often needed. The need for additional surgery may be reduced by the thorough excision of all lesions and disease at the initial surgery, followed by the postoperative administration of hormonal suppressive therapy with progestins to induce hypomenorrhea or amenorrhea. Definitive surgery with hysterectomy and removal of ovaries is frequently curative. Future therapies should be directed at identifying women at risk for the disease and implementing interventions that will prevent disease development altogether. Ultimately, the goal is to make endometriosis a disease of the past.
Keywords
Endometriosis is a chronic and recurrent disease affecting 7–10% of reproductive-age women. Of the 7 million women affected by endometriosis in the USA, 5–6 million suffer from severe pain and 1–2 million suffer from infertility. The symptoms of endometriosis cause serious life disruption, emotional suffering, marital and social dysfunction, and decreased productivity [1]. Although the prevalence of endometriosis is highest during the third and fourth decades of life [2], it is not at all rare in adolescent girls. Endometriosis is diagnosed at laparoscopy in 45% of teenagers with chronic pelvic pain [3], and when their pain does not respond to either oral contraceptives or nonsteroidal anti-inflammatory drugs (NSAIDs), endometriosis will be found in 70% of these girls at laparoscopy [4].
Endometriosis is defined as the presence of functional endometrial tissue (glands and stroma) outside the uterine cavity. Similar normal eutopic endometrium, endometriosis responds to cyclic ovarian hormone fluctuations, proliferating and bleeding with each menstrual cycle. This leads to diffuse inflammation, adhesion formation and growth of endometriotic nodules and cysts. The symptoms of endometriosis, listed in Box 1, usually reflect the affected organs. Since the pelvic organs are most commonly involved, the most frequent symptoms are: dysmenorrhea, dyspareunia and infertility, often referred to as the classic triad of endometriosis symptoms. When the urinary bladder is involved, the patient frequently complains of urinary urgency, dysuria and sometimes hematuria. Rectal involvement is usually manifested by dyschezia, tenesmus and hematochezia. Occasionally, endometriosis involves distant sites such as the chest wall, lungs or diaphragm, and in such cases, the patient may complain of respiratory symptoms, hemoptysis, pneumothorax or shoulder pain. Catamenial symptoms are the hallmark of endometriosis; being usually cyclic and most severe around menses. Clinical signs of endometriosis include fibrosis and tenderness of the cul de sac and along the uterosacral ligaments, fixed retroverted or anteverted uterus, and adnexal cystic masses. Since none of these symptoms or signs is specific for endometriosis, the current standard for diagnosis is laparoscopy, which allows the surgeon to visualize the extent of the disease and confirm it histologically with directed excisional biopsies. The degree of pain does not always correlate with disease severity; however, it may better correlate with the inflammatory cytokines, focal bleeding and irritation of pelvic floor nerves.
Symptoms of endometriosis.
Dysmenorrhea
Dyspareunia
Infertility
Pneumothorax
Irritable bowel syndrome
Dysuria
Urgency
Dyschezia
Dysfunctional uterine bleeding
Abdominal pain
Hematuria
Hematochezia
Hemoptisis
Pelvic pain
Several theories have been proposed to explain the pathogenesis of endometriosis but none fully explains the vagaries of the disease or why some women are affected and others are not. Sampson's theory of retrograde menstruation postulates that during menses, some endometrial detritus refluxes through the fallopian tubes and spills into the peritoneal cavity, where the endometrial cells may implant and proliferate. Halban's theory of vascular dissemination suggests that during menses, some of the endometrial cells enter the lymphatic or venous system and are transported to ectopic sites where they implant and grow. However, neither of these two theories explains the occasional development of endometriosis in women with Mullerian agenesis, who are born without a uterus, and hence do not menstruate. For these women, Myer offers his theory of metaplasia, which postulates that under the stimulating influence of ovarian estrogen, the pluripotential, coelomic epithelium can differentiate into functional endometrial tissue. Although these theories explain how endometrial cells reach ectopic sites, they do not explain why endometriosis develops in some women and not in others, since most, if not all, women experience retrograde menstruation and vascular dissemination and have the potential for coelomic metaplasia. A genetic predisposition to endometriosis has been suggested and supported by the finding that the endometrium of women with endometriosis differs from that of unaffected women by having aromatase enzyme activity and by expressing more vascular endothelial growth factor (VEGF) and more matrix metalloproteinase (MMP)-2. VEGF and MMP-2 promote proliferation, vascularization and invasiveness of ectopic endometrium in affected women. The observation that endometriosis is more common in women with early menarche, polymenorrhea, menorrhagia and outflow tract obstruction suggests that the development of endometriosis depends on the complex interaction of several factors, including genetic, immunological, environmental and hormonal factors.
That hormonal factors are of central importance is underscored by the clinical observations that endometriosis seldom occurs before menarche and usually improves after menopause. Endometriosis improves or stabilizes during prolonged periods of amenorrhea, as in pregnancy, hysterectomy or menopause. The condition is rarely observed in amenorrheic women but it is common in women with uninterrupted cyclic menses. Indeed, these observations form the basis for the medical management of endometriosis, which involves the induction of amenorrhea by inhibiting estrogen production with gonadotropin-releasing hormone (GnRH) analogs or by opposing estrogen action with either progestins or androgenic compounds. Other therapies involve surgical resection or ablation of endometriosis implants with or without conservation of reproductive function.
In this article, the various therapeutic options currently available for the management of endometriosis-related pain will be reviewed, drawing on the recent literature and the clinical experience of the authors. The pros and cons of each therapy will be discussed and, finally, recommendations regarding therapy will be offered, attempting to tailor the therapy to the needs of individual patients. Finally, the authors will indulge in speculative perspectives regarding future directions in the management of this debilitating and pervasive disease.
Current therapeutic options for endometriosis-related pain
The therapeutic goals in the management of symptomatic endometriosis are to relieve symptoms and prevent both progression and recurrence of disease. Unfortunately, none of the currently available conservative therapies, surgical, hormonal or both, has been associated with a cure or even long-lasting pain relief. As a result of the chronic nature of the disease and the predisposition to recurrence, unless definitive radical surgery is performed with removal of both ovaries and uterus, long-term or repeated courses of medical and surgical therapy are often required to control symptoms.
The medical therapies that have been used for the management of endometriosis-associated pain include NSAIDs and hormonal contraceptives (HCs) as the first-line therapies for mild and responsive symptoms, or androgenic agents, progestins and GnRH analogs for more severe and recalcitrant symptoms. Among the various different types of medical regimens, no significant difference in efficacy has yet been observed; however, the side effects and cost of the different regimens differ markedly.
Combined hormonal contraceptives
Combined HCs, containing both estrogen and progestin, are widely used as initial therapy for pelvic pain suspected to be related to endometriosis. These agents are readily available, generally well tolerated and relatively inexpensive. They inhibit ovulation and ovarian steroid production by decreasing gonadotropin levels, reducing menstrual flow and dysmenorrhea, and by inducing decidualization and apoptosis of endometriotic implants [5]. Although HCs have been used extensively in clinical practice, evidence for their efficacy in relieving endometriosis-related pain has been largely observational. When compared with GnRH-analog therapy, HCs were found to be significantly less effective in relieving dysmenorrhea and dyspareunia; and when compared with placebo for 6 months postoperatively, there was no difference in recurrence of symptoms [6]. However, in patients with dysmenorrhea who do not respond to cyclic HC, the administration of HCs continuously was successful in reducing menstrual pain as well as the frequency and duration of bleeding [7]. Although HC therapy has not been adequately studied and has not been found to be consistently effective, it may be appropriate as a first-line intervention, perhaps best administered continuously to effect amenorrhea, and decidualization and apoptosis of endometriotic implants. Women over the age of 35 years who smoke should not use HCs containing estrogen due to an unacceptably high risk of myocardial infarction.
Danazol
Danazol is an isoxazol derivative of methyltestosterone with impeded androgenic, strong antiestrogenic and moderate progestastional activity. It induces amenorrhea by suppressing the hypothalamic–pituitary–ovarian axis and inducing a hypoestrogenic and hyperandrogenic milieu. Consequently, most women receiving danazol will experience amenorrhea, improvement of pelvic pain, regression of endometriotic implants and mild androgenic side effects, including acne, lowering of the voice and, occasionally, mild hirsutism. As with other androgens, danazol therapy is associated with a significant reduction in high-density lipoprotein (HDL)-cholesterol levels and elevation of low-density lipoprotein (LDL)-cholesterol levels, which may be atherogenic. In a prospectively randomized study, danazol and a GnRH analog were compared for efficacy and safety in endometriosis-related pain. Both drugs were equally effective in relieving most symptoms of endometriosis and in reducing the endometriosis severity scores, assessed with the revised American Fertility Society (AFS) endometriosis classification. However, danazol was not as well tolerated as the GnRH analog, with a dropout rate of 18.5 versus 5.5% [8]. Due to poor tolerability and the adverse metabolic effects of danazol, its use in endometriosis has been progressively declining during the past several decades. Danazol should not be used in women with liver disease or hyperlipidemia.
GnRH analogs
These are currently considered the gold standard in the medical treatment of endometriosis-associated symptoms. GnRH analogs induce medical menopause by downregulating pituitary GnRH receptors, causing hypogonadotropic hypogonadism, a hypoestrogenic state associated with low serum levels of both luteinizing hormone (LH) and follicle-stimulating hormone (FSH). Several GnRH-analog preparations are currently available for the management of endometriosis, including leuprolide, nafarelin, buserelin, goserelin, histrelin and triptorelin, which may be administered daily by injections or the nasal route, or as monthly or trimonthly depot injections intramuscularly. They have been studied in numerous prospectively randomized studies, comparing their efficacy and safety with placebo and other agents, such as danazol, medroxyprogesterone acetate (MPA) and HCs. GnRH-analog therapy has been found to be at least as effective as other agents in relieving symptoms of endometriosis, but with a different side-effect profile.
As expected from their mechanism of action, GnRH-analog therapy is associated with hypoestrogenic symptoms similar to menopause, including: hot flashes, vaginal dryness, decreased libido and emotional lability. Therapy for 6 months or more is associated with a significant reduction in bone mineral density (BMD), both at the lumbar spine and hip, thus limiting therapy duration to a maximum of 6 months. To reduce these adverse effects, add-back therapy with progestins, with or without estrogen, or biphosphonates has been studied with good results. In a prospectively randomized, placebo-controlled, double-blind trial, the addition of add-back therapy in the form of estradiol–norethindrone acetate to a GnRH analog for 6 months significantly reduced menopausal symptoms and eliminated the 5.02% bone loss experienced by the placebo group without compromizing the therapeutic efficacy in controlling endometriosis-associated pain [9]. In a similar study by Zupi and colleagues, symptomatic women with endometriosis were randomly treated with an oral HC or a GnRH analog with or without estrogen–progestin add-back therapy for 12 months. Patients treated with a GnRH analog, either with or without add-back therapy, experienced a significantly greater reduction of pelvic pain, dysmenorrhea and dyspareunia than patients treated with HCs. Compared with the patients treated with the GnRH analog alone, those treated with add-back therapy experienced fewer adverse effects, better quality of life (QoL) scores and a significantly lower reduction in BMD [10]. It appears from these studies that a GnRH analog plus add-back therapy is successful in relieving endometriosis-associated pain with minimal side effects, so that the duration of therapy may be extended beyond the recommended 6-month course. Indeed, the US FDA recommends add-back therapy if a GnRH analog is to be used for more than 6 months. Some authors feel that add-back therapy should always be used in women undergoing GnRH-analog therapy for symptomatic endometriosis. However, it should be noted that add-back therapy with estrogen and progestin is associated with its own side effects (e.g., breakthrough bleeding) besides the additional cost to the already expensive GnRH-analog therapy, which ranges between US$400 and 500/month in the USA.
Progestins
The observation that endometriosis commonly improves during and after pregnancy has led many investigators to mimic the hormonal milieu of pregnancy, either with combined estrogen–progestin preparations or with progestin alone, to treat endometriosis and associated symptoms. Progestins inhibit the proliferation of endometriotic implants by inducing a hypoestrogenic state with elimination or reduction of menstrual flow, and by inducing decidualization, atrophy and regression of endometriotic implants [11]. These factors, along with inhibition of angiogenesis and peritoneal inflammation, are most likely responsible for the beneficial effects of progestin therapy in the relief of endometriosis-associated pain.
Progestins are available in multiple preparations and may be administered as pills, intramuscular and subcutaneous depot injections, or intrauterine devices (IUDs), several of which have been evaluated and used for the treatment of endometriosis during the past 40 years. Orally administered MPA was perhaps the first progestin to be evaluated and found to be effective at daily doses of 30, 50 and 100 mg. In a placebo-controlled, prospectively randomized study, oral MPA (100 mg/day) was found to be as effective as danazol (600 mg/day) and significantly more effective than placebo in reducing the severity of endometriosis objectively assessed at second-look laparoscopy, and in decreasing the symptoms of pelvic pain, back pain and dyschezia. The side effects attributed to MPA, edema and spotting, were significantly less severe that those of danazol, which included acne, muscle cramps, weight gain, edema and spotting [12].
A review by Vercellini and colleagues pooled the results of several studies evaluating the efficacy of oral or injectable progestin in the management of endometriosis-associated pain, and the results indicated that progestins reduced or eliminated pain symptoms in 70–100% of women, similar to the rates reported with danazol or GnRH analogs [13]. Progestin administration via IUD has also been reported to effectively reduce endometriosis pain. In a recent randomized clinical trial comparing the relative efficacy of a levonorgestrel-releasing IUD and a GnRH analog in the management of chronic pelvic pain in women with endometriosis, Petta and colleagues found both therapies to be equally effective and comparably tolerated. The authors appropriately concluded, “because the levonorgestrel intrauterine system does not induce hypoestrogenism and is effective for up to 5 years with only a single procedure, it could be one of the treatments of choice for chronic pelvic pain associated with endometriosis in women who do not wish to conceive” [14].
The intramuscular formulation of depoMPA (DMPA) 150 mg administered every 3 months has been used for many years for contraception and for the management of endometriosis. A new formulation of DMPA-subcutaneous 104, administered as a single subcutaneous injection of 104 mg/0.65 ml every 3 months, has recently been approved by the FDA for both contraception and the management of endometriosis-associated symptoms. DMPA-subcutaneous 104 delivers a 30% lower dose than the 150 mg intramuscular preparation, and may have a better safety and tolerability profile. Its safety, tolerability and efficacy in the management of endometriosis-associated symptoms has been demonstrated in two large, 18-month, multinational, randomized, evaluator-blinded comparative trials. The two trials randomized a total of 573 symptomatic women with laparoscopically proven endometriosis to 6 months of treatment with either DMPA-sub-cutaneous 104 or leuprolide, followed by 12 months of post-treatment evaluation. As expected, the tolerability and safety profile of the two treatment groups differed in vasomotor symptoms, changes in BMD of the hip and lumbar spine, as well as bleeding patterns [15]. The DMPA-subcutaneous 104 group experienced more spotting but significantly lower BMD losses compared with the leuprolide group and no vasomotor symptoms. After 6 months of treatment, the median percentage changes in BMD for DMPA-subcutaneous 104 and leuprolide at the lumbar spine were −1.1 and −3.95%, respectively, and at the hip were −0.3 and −1.65%, respectively (p < 0.001). At 12 months post-treatment, the BMD returned to baseline in the DMPA-subcutaneous 104 group but remained at −1.7 and −1.3% at the lumbar and hip, respectively, in the leuprolide group (p < 0.001). The overall incidence of adverse events, other than hypoestrogenic symptoms, was similar between groups, including weight gain, which averaged a less than 1 kg increase after 6 months of treatment in both studies [16]. The reduced impact of DMPA-sub-cutaneous 104 on BMD compared with leuprolide is particularly important, given the frequent need for extended and repeated courses of medical therapy to control symptom recurrences of endometriosis.
Several studies have evaluated and compared the relative efficacy and safety of various medical treatments for the management of endometriosis-associated pain. The results from these studies have been compiled, normalized and schematically represented in Figure 1. As the figure shows, the relative efficacy of these various therapies is very similar in the reduction of symptoms, not only during therapy but also at follow-up. Consequently, the choice of which medical treatment to prescribe may not be based on differences in efficacy but on differences in tolerability, safety and, when everything else is equal, cost. A study by Telimaa and colleagues, comparing oral MPA versus danazol, found them to be similar in efficacy, but MPA was much better tolerated and associated with fewer adverse effects than danazol. A study by Henzl and colleagues, comparing a GnRH analog with danazol, also reported similar efficacy between the two drugs, but GnRH-analog therapy was better tolerated, with a lower dropout rate, and was associated with a much better safety profile, especially in terms of the lipid and lipoprotein changes in serum. Similarly, a study by Crosignani and colleagues, comparing leuprolide and the new preparation of DMPA-subcutaneous 104, demonstrated comparable efficacy in relieving pain symptoms and objective signs of endometriosis but reported very different safety profiles. Given the chronic and recurrent nature of endometriosis, affected women usually require prolonged and frequent courses of medical treatment. Medications that can be used for only 6–12 months, such as danazol and GnRH analogs, have a limited application in the long-term management of endometriosis. Progestins, oral or injectable, alone or with estrogen, comprise a viable alternative option for the long-term management of endometriosis pain in women not currently desiring pregnancy. They are readily available, generally well tolerated, relatively inexpensive and associated with less metabolic impact than other more expensive agents.

Percentage of endometriosis patients whose symptoms improved after 6 months of indicated therapy.
Investigational agents for the management of endometriosis
Aromatase inhibitors
Aromatase P450 is the key enzyme for estrogen synthesis, converting testosterone and androstenedione to estradiol and estrone, respectively, either by the granulosa cells in the ovary or peripherally by adipose tissues. Although the normal endometrium does not have any aromatase activity, endometriotic tissues contain significant aromatase activity, which increases the local production of estrogen within the implant, stimulating its growth and proliferation. The inflammatory mediator prostaglandin E2, which is abounds in the peritoneal fluid of patients with endometriosis, stimulates the activity of aromatase enzymes in endometriosis implants, further stimulating proliferation of implants, inflammatory reactions and progressive growth of disease. Aromatase inhibitors specifically target this enzyme, decreasing local estrogen production and inhibiting the growth of endometriotic implants. In the first case report of the use of the aromatase inhibitor anastrazole, a 57-year-old women with severe symptoms, which had not responded to either medical or surgical therapy, experienced complete relief of symptoms after 2 months of anastrozole therapy [17]. In a recent small study of ten reproductive-aged women with treatment-resistant endometriosis, the aromatase inhibitor letrozole was administered at a daily dose of 2.5 mg together with norethindrone acetate 2.5 mg/day and calcium with vitamin D for 6 months. The treatment reduced pelvic pain scores in nine of ten patients and decreased laparoscopically visible lesions in all ten women without significant changes in bone loss [18]. In a more recent study, 15 women with treatment-refractory pelvic pain associated with endometriosis were treated for 6 months with anastrazole 1 mg plus ethinyl estradiol 20 μg and levonorgestrel 0.1 mg/day; 14 of these patients reported improvement in their pain [19]. Although promising, these results require further confirmation in randomized studies.
Selective estrogen-receptor modulators
Selective estrogen-receptor modulators (SERMs) are nonsteroidal compounds that bind to estrogen receptors and act either as estrogen agonists or antagonists, depending on the target tissue. Raloxifene is one such SERM with estrogen agonistic effects on bone and circulating lipoprotein, but antagonistic effects on the breast and uterus, including endometrium. Small pilot studies have evaluated and are currently evaluating the role of SERMs in the management of endometriosis pain. The effects of these compounds in human clinical studies are currently unknown.
Progesterone-receptor modulators
Similar to SERMs, selective progesterone-receptor modulators (SPRMs) can act either as progesterone agonists or antagonists, depending on the target tissue, dose, and presence or absence of progesterone receptors. Some of these agents have the potential to selectively suppress endometrial proliferation despite normal levels of estrogen. The ability to suppress endometrial proliferation and endometriosis without inducing a hypoestrogenic state with its adverse effects would be a great advance in the management of endometriosis pain. Although some small studies have been successfully conducted, clinical trials are still needed to assess the role of these agents in the treatment of endometriosis [20].
Surgical therapy
Although endometriosis has long been considered a surgical disease, only two randomized trials have been published to evaluate the effect of surgery on the pain associated with the disease. The results of these two studies will be presented to illustrate the important role of surgery in the management of endometriosis.
In the first study, published by Sutton and colleagues, 74 symptomatic women with Stage I, II or III disease were randomized to either diagnostic laparoscopy or operative laparoscopy with ablation of implants, resection of adhesions and uterosacral nerve ablation. Pain improved significantly in 75% of the treated women and only 22% of the untreated group. Moreover, at 1 year after surgery, 65% of the treated group continued to experience pain relief, as opposed to only 18% of the untreated group [21].
In the second study, 39 symptomatic women with endometriosis were randomized to either diagnostic or operative laparoscopy with excision of endometriotic implants and adhesions. Symptomatic improvement was observed in 80% of the treated and 32% of the untreated group. QoL was also evaluated and showed a greater improvement in the treated than nontreated group. The authors concluded that laparoscopic surgery is more effective than placebo in reducing pain and improving QoL. Moreover, surgery is associated with a 30% placebo response rate that is not dependent on the severity of disease [22].
The surgical management of endometriosis-associated pain may be conservative or radical, and may be performed by laparoscopy or laparotomy. The conservative management preserves fertility potential and involves the resection and/or ablation of endometriosis implants, resection of associated adhesions, aspiration with resection or ablation of endometriosis cysts, and restoration of normal pelvic anatomy. The goals are to restore fertility and reduce recurrence of symptoms. To date, there is no consensus as to whether to ablate or resect implants, whether to coagulate or resect endometriotic cysts, or whether to perform adjunctive nerve ablation procedures such as uterosacral ligament resection or presacral neurectomy [23]. Most advanced surgeons prefer resection over ablation, feeling that complete removal of the lesions may result in better relief of symptoms and reduced risk of recurrence of disease. Ablation or dessication may not completely devitalize or remove the lesions, predisposing patients to persistence of symptoms and a higher risk of disease recurrence. The nerve ablative procedure of uterosacral ligament resection adds little to the complete resection of the implants when the ligaments are involved with disease. Presacral neurectomy has been reported to be of therapeutic value only in women suffering from central pain and dysmenorrhea. Localized lateral pain does not improve with presacral neurectomy.
The surgical approach to the conservative management of endometriosis may be performed by laparoscopy or laparotomy. There has not been a single randomized study to address the issue as to which of these two approaches yields better results. Given the minimally invasive approach of laparoscopy, the associated shorter periods of hospitalization and postoperative convalescence, and reduced costs, as illustrated in Table 1 [24], it seems that whenever feasible the laparoscopic approach should be preferred, provided that the surgeon has the proper endoscopic surgical skills to adequately treat the disease. If not, it is better to perform a safe and complete surgery by laparotomy than an incomplete or unsafe operative laparoscopy.
Therapeutic, economic and social benefits of the laparoscopic versus laparotomy treatment of endometriosis.
Adapted from [23].
Conservative surgery has been shown to be of therapeutic benefit in reducing pelvic pain and improving QoL, but it is seldom curative. Most women will experience recurrent symptoms requiring additional therapies. In patients who are interested in imminent pregnancy, postoperative therapy with progestins has been reported to reduce recurrence of pain and to lengthen the symptom-free period much more effectively than placebo [25]. Indeed, following conservative surgical therapy for endometriosis in patients not interested in pregnancy, the author strongly recommends medical therapy with either HC continuously or progestin to induce a state of amenorrhea or hypomenorrhea until such time as the woman wishes to conceive. This approach is likely to reduce the frequency of surgical interventions or the need for more aggressive and expensive repeated therapies with a GnRH analog. The authors' recommendation for the conservative management of endometriosis-associated pelvic pain is illustrated in Figure 2.
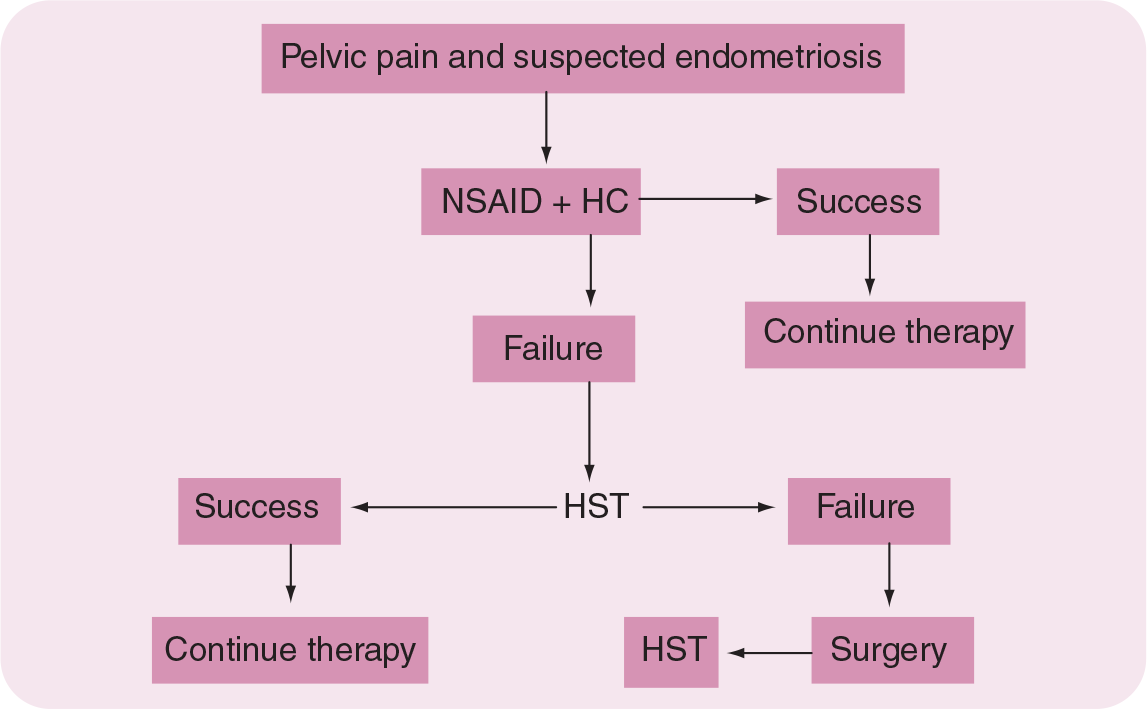
Algorithm for the treatment of pelvic pain associated with endometriosis.
Definitive surgery for endometriosis should be offered to symptomatic women in whom conservative therapies have failed and who do not desire further childbearing. It involves performing hysterectomy with bilateral salpingoophorectomy and resection of endometriosis and associated adhesions. Since most of these women are young, the removal of the ovaries has been questioned due to the consequences of surgical castration, including vasomotor symptoms, deleterious impact on sexual health and QoL, and increased risk of osteoporosis. However, preservation of ovarian function may be associated with a major risk of recurrence of symptoms and endometriosis. A cohort study of 138 women with endometriosis whose ovaries were retained at the time of hysterectomy experienced a recurrence rate of pain that was six-times higher, and a re-operation rate that was eight-times higher than comparable women who had hysterectomy and their ovaries removed [26]. However, it is unclear whether or not the endometriotic implants were completely removed at the time of hysterectomy. In most cases involving relatively young women, perhaps younger than 40 years of age, the ovaries may be preserved if the disease can be adequately removed at the time of hysterectomy. The consequences of surgical castration in these young women can be devastating, especially if hormone replacement therapy is poorly tolerated or contraindicated.
Symptomatic women with dyspareunia associated with deep rectovaginal endometriosis and obliteration of the posterior cul de sac respond poorly to medical therapy and seldom obtain pain relief even with surgery, unless the rectovaginal space is dissected and the endometriosis removed from this area. Since the rectum is frequently involved, this type of surgery is very difficult and may require perirectal dissection or segmental rectal resection. This complex surgery should be performed only by the most expert gynecological and colorectal surgeons [27].
Conclusion
Endometriosis is a chronic recurrent disease with multiple manifestations of cyclic or chronic pain, usually reflecting the affected organs. Since pelvic organs are mostly affected, pelvic symptoms of dysmenorrhea, dyspareunia and infertility are most common. Endometriosis can also affect distant sites such as the lungs and upper abdomen, as well as the small and large bowel, with catamenial symptoms referred to these organs. Although surgery to remove endometriosis is effective in relieving pain, recurrence of symptoms is common and repeated medical and surgical interventions are often needed. Due to its invasiveness and inherent risks, surgical intervention should involve minimally invasive approaches by laparoscopy and should be kept to a minimum. Following the initial resective surgery, the physician should be cognizant of the high recurrence rate and should consider interventions that prolong the disease-free interval and delay recurrence of symptoms. A number of medical treatment options are currently available to inhibit the growth and activity of endometriotic implants, including HCs, danazol, GnRH analogs and progestins. Due to its metabolic adverse effects, danazol is currently seldom used. GnRH-analog therapies are highly effective but their use alone is limited to 6 months and with add-back therapy has not been approved beyond 12 months. For the long-term management of endometriosis pain, progestins, oral or injectable, alone or combined with estrogen, are currently the safest and perhaps the most cost-effective option.
At times these medical and surgical treatments may not ‘cure’ the pelvic pain and other therapeutic modalities may need to be explored. Although outside the scope of this article, alternative and complementary therapies ranging from acupuncture to herbal medicines could be helpful to the patient and should be offered as suggestions. Often, help from pain clinics and clinical psychologists can also be useful. Endometriosis is a chronic and difficult condition for gynecologists to treat as it affects many aspects of patients' lives. Expanding treatment options by incorporating this multidisciplinary approach may help gynecologists treat this complex disease.
Future perspective
Future therapeutic options, not currently available but under investigation, include aromatase inhibitors, SERMs and SPRMs. They offer the promise of more specific targeting of the endometriotic implants without inducing a general hypoestrogenic state that can adversely affect both health and QoL. The goal of future research should be directed at developing therapies that specifically target endometriosis cells to prevent their proliferation and, more importantly, their initial implantation at ectopic sites. All currently available therapies for symptomatic endometriosis are directed at treating or managing the disease. However, research efforts should be focused on preventing the disease. Since we have learned that the endometrium of women affected by endometriosis differs from the endometrium of unaffected women by having aromatase enzyme activity, and by expressing more VGEF and MMP-2, research should be directed toward understanding the mechanisms for such diversity in the endometrium of affected women and ultimately correcting it. The active research that is currently being conducted to better understand the pathogenesis and pathophysiology of endometriosis, and the current development of noninvasive diagnostic blood tests to diagnose the disease or, better yet, to identify women at risk before they develop the disease, will ultimately allow us to conquer it and make endometriosis a disease of the past.
Executive summary
Catamenial symptoms are the hallmark of endometriosis, being usually cyclic and most severe around menses. The most frequent symptoms are dysmenorrhea, dyspareunia and infertility.
Combined hormonal contraception is a first-line intervention and should be administered continuously to effect amenorrhea and promote decidualization and apoptosis of endometriotic implants.
Danazol causes amenorrhea, improvement of pelvic pain and regression of endometriosis implants but has poorly tolerated androgenic and hypoestrogenic side effects, including potentially serious adverse metabolic effects. Its use in endometriosis has been progressively declining during the past several years. Danazol should not be used in women with liver disease or hyperlipidemia.
Gonadotropin-releasing hormone (GnRH) analogs have long been considered the gold-standard treatment for endometriosis. The associated hypoestrogenic symptoms of hot flashes, vaginal dryness, decreased libido and bone loss may be reduced or avoided with add-back therapy. But its use even with add-back therapy has not been approved beyond 12 months.
Progestins, administered orally, systemically or in intrauterine devices, inhibit proliferation of endometriosis implants and offer symptom relief that is comparable to GnRH analogs, but with fewer hypoestrogenic side effects and better bone preservation. Progestins are associated with weight gain, depressed mood and irregular bleeding.
The relative efficacies of the various medical therapies are very similar in the reduction of symptoms during treatment and on follow-up, but their adverse effects and tolerabilities are different. Consequently, the choice of which medical treatment to prescribe should be based on tolerability, safety and, when everything else is equal, cost.
Surgery has been proven effective in the management of endometriosis-related symptoms, whether it is performed by laparoscopy or laparotomy. Whenever possible, excision rather than ablation or dessication of lesions should be performed to achieve better results and reduce recurrence.
Recurrence following conservative surgery is common and medical therapy with progestins, hormonal contraceptives, GnRH analogs or danazol is effective in prolonging disease-free intervals.
