Abstract
Yasminelle® is a newly developed combined oral contraceptive that contains ethinyl estradiol 20 μg and drospirenone 3 mg. Yasminelle is unlike other low-dose ethinyl estradiol combined oral contraceptives, since drospirenone produces antiandrogenic and antimineralocorticoid effects that offer various benefits including a reduced potential for estrogen-induced water retention. Pharmacological and pharmacokinetic data have been derived from clinical experience with ethinyl estradiol 30 μg and drospirenone 3 mg (Yasmin®). Clinical studies with Yasminelle have shown a high degree of contraceptive efficacy, similar to that of combined oral contraceptives that contain a low dose of ethinyl estradiol combined with other progestins. These studies have also demonstrated that Yasminelle produces excellent cycle control and has a good tolerability profile, similar to that of other low-dose ethinyl estradiol combined oral contraceptives.
Keywords
Combined oral contraceptives (COCs) are among the most effective forms of contraception [1,2], and are a popular method of fertility control in many countries [3,101]. Since their introduction in the early 1960s, the estrogen dose has been progressively reduced and newer progestins have been introduced. The reduction in estrogen dose became necessary as the earlier formulations, typically those that contained estrogen 50 μg, were associated with estrogen-related adverse effects and specifically venous thromboembolism (VTE). Moreover, the realization that the estrogen and progestin components act synergistically to inhibit the pituitary and subsequent ovulation led to the breakthrough that allowed the development of the current generation of low-dose COCs. Indeed, much of the focus of current COC development has been on combining low doses of estrogen with progestational agents that provide good cycle control with minimal adverse effects, as well as additional noncontraceptive benefits.
However, despite the undoubted efficacy, convenience and improved safety profile of COCs, a significant number of women who use this method for fertility regulation put themselves at risk of unintended pregnancy as they adhere poorly to their regimen or discontinue treatment completely [4,5]. Factors associated with poor tolerance with some COC preparations include unwanted side effects such as fluid retention-related symptoms (e.g., breast tenderness, bloating and weight gain), acne, intermenstrual bleeding and mood changes [4–6]. Consequently, as the progestin is usually the distinguishing component between formulations, newer progestins are being developed to resolve any tolerance issues and provide additional noncontraceptive benefits.
Until recently, the progestins used in COCs were generally derivatives of either 19-nortestosterone or 17α-hydroxyprogesterone. In this respect, drospirenone – a derivative of spirolactone – represents a new class of progestin. Drospirenone is an analog of spironolactone, which means that unlike any other progestin except natural progesterone, it has antimineralocorticoid properties [7]. In addition, drospirenone has antiandrogenic properties [6]. Clinical studies with a COC containing ethinyl estradiol 30 μg and drospirenone 3 mg (Yasmin®) have demonstrated that it provides effective contraceptive protection together with a good safety and tolerability profile [8–10], as well as ameliorating acne and other skin-related problems [11,12].
Consistent with the progressive development of COCs, a new contraceptive formulation in which the ethinyl estradiol dose has been reduced from 30 to 20 μg and combined with drospirenone 3 mg (Yasminelle®) has been developed. Each cycle of use of Yasminelle lasts 28 days, and comprises 21 days of active pill use followed by a 7-day treatment-free interval. The Yasminelle formulation has recently been approved in Europe, and this article focuses on its clinical properties and role in preventing pregnancy.
Pharmacology of Yasminelle
Formulation
Yasminelle comprises 21 tablets; each tablet contains ethinyl estradiol 20 μg and drospirenone 3 mg (Figure 1). To improve the stability of Yasminelle and to prolong its shelf life, the ethinyl estradiol component is encapsulated within a betadex clathrate (Figure 1) – one molecule of ethinyl estradiol within two molecules of betadex (β-cyclodextrin). This betadex complex dissociates rapidly in solution (the calculated dissociation half-life is 2.6 min) [Data on file, Berlex Laboratories].

Drospirenone, ethinyl estradiol and betadex clathrate.
Pharmacodynamics
Drospirenone is directly progestogenic – it is not a prodrug that has to be metabolized to an active form.
Contraceptive effects
The main contraceptive efficacy of a COC derives primarily from the progestin component, which inhibits ovulation by inhibition of follicle-stimulating hormone (FSH) release and prevention of the midcycle luteinizing hormone (LH) surge required for release of the oocyte. Although inhibition of ovulation is sufficient to prevent pregnancy, other peripheral contraceptive effects such as thickening of cervical mucus, slowing of tubal motility and induction of endometrial atrophy contribute to the overall contraceptive efficacy. The estrogen component, which is included mainly to provide better cycle control, also acts synergistically with the progestin to enhance contraceptive efficacy [13,14].
Ovulation inhibition with drospirenone monotherapy was investigated in an open-label, randomized study in which 48 women were treated with three cycles of drospirenone 0.5, 1, 2 or 3 mg. The effects of drospirenone on ovulation and the inhibition of follicular maturation or growth were shown to be dose dependent, with the 3 mg dose of drospirenone producing inhibition of follicular maturation or growth in nearly all subjects (10/11) and suppression of ovulation in every subject (11/11) [15].
There are limited published pharmacological data relating to the use of Yasminelle. For this reason, the rest of this section includes data from studies that involved ethinyl estradiol 30 μg plus drospirenone 3 mg. The effect of ethinyl estradiol 30 μg plus drospirenone 2 or 3 mg on ovulation was studied in an open-label, randomized study that involved a total of 52 women who received three cycles of treatment. In the ethinyl estradiol 30 μg plus drospirenone 3 mg group, follicular maturation or growth was suppressed in 100, 78.3 and 91.3% of subjects in cycles 1, 2 and 3, respectively, and ovulation was suppressed in all subjects in each cycle. Decreases in the plasma concentrations of LH, FSH, 17β-estradiol and progesterone were also observed, confirming the anovulatory effects [15]. In addition, peripheral contraceptive effects, such as decreases in cervical score, spin-nbarkeit and the degree of crystallization of cervical mucus, were also observed with treatment [15]. In another open-label study that involved 40 women, treatment with ethinyl estradiol 30 μg plus drospirenone 3 mg had a marked antiprolifer-ative effect on the endometrium after three cycles of treatment, and after 13 cycles, 63% of subjects showed evidence of endometrial atrophy [16].
Mineralocorticoid & antimineralocorticoid effects
Progesterone exerts an antimineralocorticoid effect, and progesterone treatment has been shown to result in significant natriuresis [17]. To compensate for this, counter-regulatory increases occur in plasma renin activity (PRA) and the concentration of plasma aldosterone [17]. Estrogen produces an increase in plasma renin substrate (PRS) concentrations, which leads to an increase in plasma angiotensin I and II concentrations that then stimulate aldosterone production by the adrenal cortex [18,19]. Consequently, estrogens can induce retention of sodium, loss of potassium and the secondary retention of water. Unlike other synthetic progestins, drospirenone has consistently been shown to have antimineralocorticoid effects both in vitro and in vivo; thus, COCs that contain drospirenone have a reduced potential for causing water retention and associated weight increase.
Increases in plasma aldosterone concentrations and PRA have been demonstrated for COCs containing combinations of ethinyl estradiol plus drospirenone [18,20]. In a study in which groups of 20 women were treated with drospirenone 3 mg plus ethinyl estradiol 15, 20 or 30 μg, or levonorgestrel (LNG) 150 μg plus ethinyl estradiol 30 μg for six cycles, the concentrations of PRS (angiotensinogen) increased in all groups, as expected [20]. In contrast, the concentrations of plasma aldosterone and PRA increased significantly in the groups receiving drospirenone, but not in the group that received LNG. This was presumed to reflect a compensatory reaction to sodium loss caused by drospirenone [20]. In another study in which groups of 12 women were treated with drospirenone 0.5, 1, 2 or 3 mg for one cycle, there was a dose-dependent increase in plasma aldosterone concentrations and a dose-independent increase in PRA [18]. Moreover, when two groups of 30 women received ethinyl estradiol 30 μg plus either drospirenone 3 mg or desogestrel 150 μg for 13 treatment cycles in an open, randomized study, markedly greater increases in PRA and plasma aldosterone activity were observed at all time points in the ethinyl estradiol plus drospirenone group compared with the ethinyl estradiol plus desogestrel group [18].
Antiandrogenic effects
The antiandrogenic properties of drospirenone have been demonstrated in receptor assays, animal models and clinical studies [21,22]. The effect of ethinyl estradiol 30 μg plus drospirenone 3 mg on acne and seborrhea have been shown to be at least comparable with those of a COC containing ethinyl estradiol 35 μg and cyproterone acetate 2 mg [12]. Moreover, ethinyl estradiol 30 μg plus drospirenone 3 mg was shown to be superior to a triphasic ethinyl estradiol plus norgestimate formulation (Pramino®, Ortho Tri-Cyclen®) in reducing total lesion count and the investigators' assessment of therapeutic effect [11].
Pharmacokinetics
The pharmacokinetics of ethinyl estradiol following oral administration are well established [23]. Briefly, ethinyl estradiol is rapidly absorbed following oral administration –after a 30 μg dose, maximum serum concentrations (Cmax) of 90–130 pg/ml are reached within 1–2 h (time to Cmax [Tmax]). The mean oral bioavailability is approximately 45%, but interindividual variability ranges 20–60%. The mean area under the ethinyl estradiol concentration–time curve over 24 h after dosing following administration of the first pill of Yasminelle in cycle 1 was reported to be 108 and 97 pg/h/ml in Caucasian and Japanese women, respectively, which increased to 220 and 225 pg/h/ml by day 21, respectively, giving a mean accumulation ratio of approximately 2 for both groups [24].
Although ethinyl estradiol induces sex hormone-binding globulin (SHBG) synthesis, it does not bind to SHBG and is almost completely (95–98%) bound to albumin. Following daily dosing, steady-state ethinyl estradiol concentrations are reached within 5–10 days. Ethinyl estradiol is rapidly metabolized by cytochrome P (CYP)450-dependent mechanisms, and excreted mainly as glucuronide conjugates in feces and urine. The mean total clearance ranges between 2.3–7 ml/min/kg, with an elimination half-life of 6.8–26.1 h. Ethinyl estradiol accumulation ratios are approximately 1.23–1.77 with daily dosing.
The available evidence suggests that the pharmacokinetics and bioavailability of ethinyl estradiol are generally unaffected by its inclusion within a betadex clathrate. Indeed, an open study involving 18 postmenopausal women aged 45–75 years who received single oral doses of ethinyl estradiol 40 μg plus drospirenone 6 mg showed little variation in the relative bioavailability and pharmacokinetics of ethinyl estradiol and drospirenone when the doses were administered as an ethinyl estradiol–betadex clathrate plus drospirenone formulation, as an ethinyl estradiol plus drospirenone formulation that contained ethinyl estradiol as a free steroid, or as a microcrys-talline suspension containing ethinyl estradiol and drospirenone [Data on file, Berlex Laboratories].
Drospirenone is rapidly and extensively absorbed following oral administration. Serum drospirenone concentrations increase linearly following single oral doses in the range of 1–10 mg [7,25,102]. The mean absolute bioavailability of drospirenone is approximately 76% after a 2 mg oral dose [7,102]. Steady-state drospirenone concentrations are achieved within 7–10 days following daily administration of ethinyl estradiol 30 μg plus drospirenone 3 mg [7,25,102]. Following the first pill in the first treatment cycle, the Cmax of drospirenone was 36.9 ng/ml, increasing to 87.5 ng/ml by day 21 of the cycle, with similar values seen at the end of cycle 13 [26]. The Tmax of drospirenone at the beginning and end of treatment cycle 1 were 1.5–2 h. Furthermore, mean Tmax did not differ significantly between the first and last days of active treatment in any cycle, and was unchanged after 13 cycles [26]. The mean apparent volume of distribution of drospirenone is approximately 4 l/kg [25,102].
The mean area under the concentration–time curve over 24 h for drospirenone following the first pill of Yasminelle in cycle 1 was reported to be 268 and 271 ng/h/ml in Caucasian and Japanese women, respectively, which increased to 763 and 803 ng/h/ml by day 21, giving a mean accumulation ratio of 3 for both groups [24].
Drospirenone is metabolized extensively – the two main metabolites are the acid form of drospirenone and 4,5-dihydrospirenone-3-sulphate, both of which are pharmacologically inactive and are produced without the involvement of the CYP450 system [7,25]. In vitro studies have demonstrated that drospirenone is metabolized only to a small extent by CYP3A4 and that drospirenone has an inhibitory influence on the turnover of substrates of CYP1A1, CYP2C9, CYP2C19 and CYP3A4, with the greatest effects observed with CYP2C19.26 The absorption of drospirenone is slowed by food, and although Cmax is reduced by approximately 40% [102], this is not considered to be clinically relevant, and ethinyl estradiol plus drospirenone does not need to be taken with food [25].
Approximately 95–97% of drospirenone is bound to serum proteins, but drospirenone does not bind to SHBG or corticosteroid-binding globulin (CBG) and thus does not affect the ethinyl estradiol-induced increase in these serum proteins [7,102]. The elimination of drospirenone is biphasic, with a mean distribution phase half-life of approximately 2 h and a terminal disposition phase half-life of approximately 30 h [102]. Only trace amounts of unmetabolized drospirenone are excreted in urine or feces, and at least 20 metabolites of drospirenone have been identified. In urine, approximately 38–47% of excreted drospirenone metabolites are glucuronide and sulfate conjugates, and the proportion of these excreted in feces is approximately 17–20% [102].
Clinical characteristics of Yasminelle
The clinical profile of Yasminelle has been established in two large trials: a multicenter, randomized, open-label trial in which Yasminelle (n = 220) was compared with a COC containing ethinyl estradiol 20 μg and desogestrel 150 μg (n = 221 over seven treatment cycles; and a multicenter, noncomparative study over 26 cycles (n = 516). The main exclusion criteria for both studies were typical for COC trials and included substantial obesity (body mass index > 30), metabolic disease, liver or vascular disease, specific organ dysfunction, current or history of clinically significant depression, tumors, concomitant use of other contraceptive methods (apart from condoms to protect against sexually transmitted diseases) and the use of laxatives. Pregnant or lactating women or smokers older than 30 years of age were excluded. The primary objective of the noncomparative trial over 26 cycles was to assess contraceptive efficacy, with cycle control, treatment satisfaction and tolerability as secondary objectives. The seven-cycle comparative trial evaluated cycle control and safety as well as contraceptive efficacy, without distinction between primary and secondary target variables.
Contraceptive efficacy
In the 26-cycle noncomparative study, two pregnancies occurred during 11,165 cycles of treatment, giving a Pearl Index (PI) of 0.23 (upper limit of 97.5% confidence interval [CI]: 0.84) for typical use. Over 2 years, the cumulative pregnancy rate was 0.44% (95% CI: 0–1.05%), which corresponds to 99.56% contraceptive protection [27]. However, one of the two pregnancies was attributed to noncompliance, giving an adjusted PI of 0.12 (upper 97.5% CI: 0.67) for perfect use based upon 10,827/compliant cycles. In the seven-cycle comparative trial, two pregnancies occurred during treatment with ethinyl estradiol plus drospirenone treatment and three occurred during treatment with ethinyl estradiol plus desogestrel treatment, giving a PI of 0.95 (upper 97.5% CI: 5.3) for typical use in both groups [28]. The contraceptive efficacy rates observed with Yasminelle are comparable with those for other low-dose COCs.
Cycle control
In the seven-cycle comparative trial, the effects of Yasminelle on withdrawal bleeding were at least comparable with an ethinyl estradiol 20 μg plus desogestrel 150 μg COC [28]. Withdrawal bleeding, which reassures COC users that they are not pregnant, was experienced by more than 90% of women in each group and remained reasonably uniform in duration throughout the seven-cycle study (Figure 2). The proportion of women with an absence of withdrawal bleeding was highest during the first cycle (8 and 9.3% in the Yasminelle and ethinyl estradiol 20 μg plus desogestrel 150 μg groups, respectively), but decreased thereafter to less than 2.5% in both groups during cycles 3–7. In addition, the proportion of women who experienced unexpected bleeding (intracyclic bleeding) was highest during the first cycle in both groups. Intracyclic bleeding was classified more often as spotting in both groups (Figures 3a & 3b). Similarly, in the 26-cycle noncomparative study, intracyclic bleeding was restricted mainly to the first cycle of use, decreasing to a low incidence by cycle 2 and remaining at a low incidence thereafter with continued treatment duration [27]. In addition, the vast majority of women (93%) experienced withdrawal bleeding in the first treatment cycle, increasing to 96.6–99.1% throughout the rest of the study. Overall, only 0.69% of women discontinued study medication due to intracyclic bleeding over the 2 years of treatment, suggesting a highly acceptable bleeding pattern.
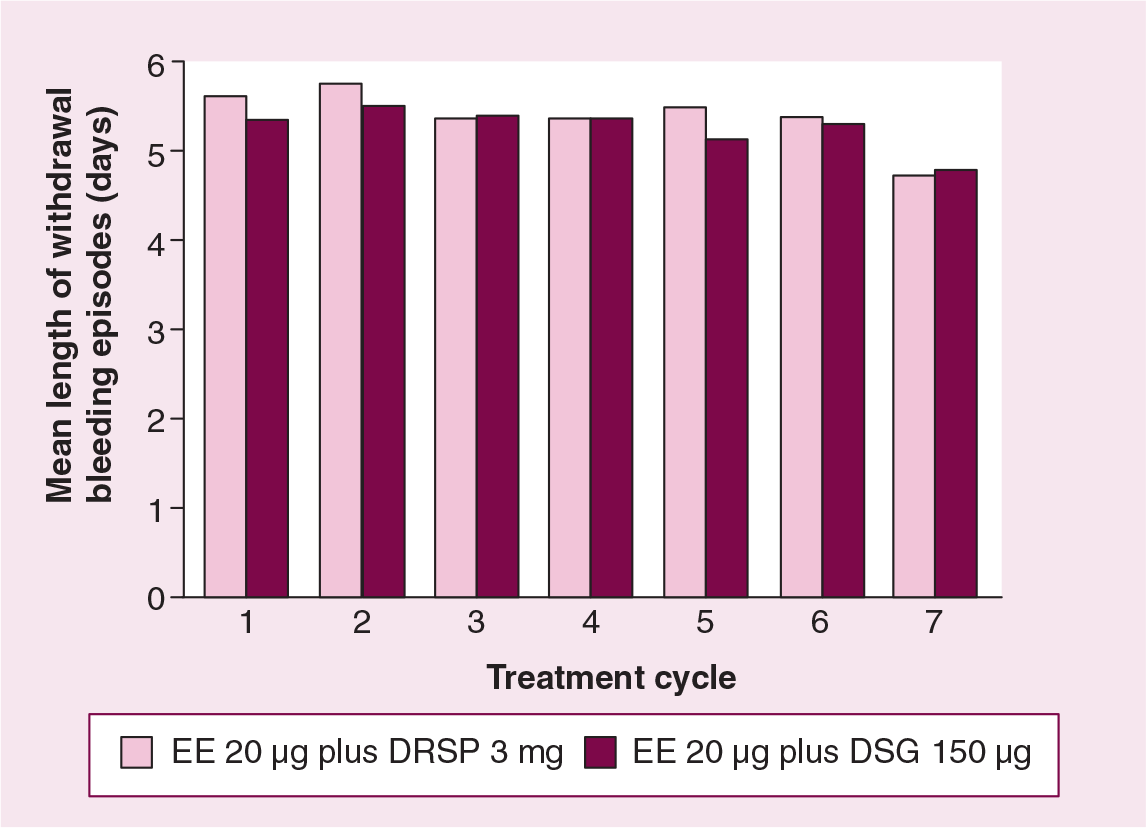
Consistency in the duration of withdrawal bleeding (days) during seven treatment cycles.

Decreased proportion of women experiencing intracyclic spotting vs bleeding (light/normal/heavy) during seven treatment cycles.
A higher incidence of intracyclic bleeding during the first treatment cycle is commonly seen with COCs [29,30], and the level of cycle control observed in the Yasminelle group was generally similar to that found with other COCs containing a low dose of ethinyl estradiol combined with a progestin [29,30].
Tolerability
The two clinical studies to date have demonstrated that Yasminelle is well tolerated and that the adverse events associated with its use were typical of COCs. Moreover, the adverse events reported during treatment were mainly mild-to-moderate in intensity. In the 26-cycle, noncomparative study, adverse events that were considered treatment-related (possibly, probably or definitely) occurred in 24.2% of subjects, with the most frequent (headache) occurring in less than 10% of subjects (Table 1) [27]. In the seven-cycle comparative study, 24.3% of women in both groups experienced adverse events that were considered treatment related [28]. The frequency of discontinuations due to adverse events with Yasminelle was low in both the noncomparative (7.2% over 2 years of treatment) and comparative (6.8% over seven cycles of treatment) studies. The general safety profile of Yasminelle during both clinical studies was good, with no cause for concern.
The most frequently reported treatment-related adverse events* in a long-term (2-year) study of EE 20 μg plus drospirenone 3 mg.
>1% of women (n = 516).
EE: Ethinyl estradiol; HARTS: Hoechst Adverse Reactions Terminology System.
Adapted from [27].
In general, the use of COCs is associated with a slightly increased risk of VTE [31], although there is no evidence of any increase in long-term mortality in COC users [14]. Available data indicate that the use of a COC containing ethinyl estradiol 30 μg and drospirenone 3 mg is not associated with any additional risk of VTEs compared with other COCs. The EUropean Active Surveillance (EURAS) study, a large, multinational, ongoing postmarketing surveillance study, has found no evidence of increased incidence of VTEs in women who had received a COC containing ethinyl estradiol 30 μg plus drospirenone 3 mg compared with COCs containing LNG or other oral contraceptives [32]. Moreover, the risk of VTE with ethinyl estradiol 30 μg and drospirenone 3 mg or other low-dose COCs is at least tenfold lower than that associated with pregnancy and delivery.
Conclusions
Yasminelle is an effective contraceptive with a highly acceptable bleeding pattern and tolerability profile. Drospirenone is a spirolactone derivative that, in addition to its progestogenic effects, provides antiandrogenic and antimineralocorticoid benefits that include effects on acne and other related skin conditions, as well as prevention of water retention-related symptoms. Moreover, Yasminelle has all the benefits inherent in a COC containing drospirenone.
Future perspective
Safe, reversible COCs will continue to be a major requirement for women who need to regulate their fertility. Contraceptive efficacy is no longer an issue with modern COC preparations – all are very effective in preventing unintended pregnancies if used correctly. Furthermore, women today have a greatly expanded array of COC formulations from which to choose one that is appropriate for their needs and lifestyle. Nonetheless, adverse effects associated with COC use, whether perceived or real, need to be addressed with current formulations as well as with those in development. Concerns regarding estrogen-related adverse effects such as breast cancer and VTEs have driven and will continue to drive the development of lower-dose estrogen formulations.
The realization that ethinyl estradiol and progestin act synergistically to inhibit ovulation inhibition offers several developmental directions for future COCs. One possibility would be the development of oral contraceptives that contain less than ethinyl estradiol 20 μg. Indeed, one COC formulation containing ethinyl estradiol 15 μg is already on the market and their numbers are likely to increase [33]. Another approach would be to substitute ethinyl estradiol with a less potent estrogenic compound (e.g., 17β-estradiol), provided adequate cycle control could be achieved. Another possibility would be the continual development of new progestins such as drospirenone, which have short-term additional noncontraceptive benefits (e.g., improving acne, and reducing water retention and premenstrual symptoms), as well as possible long-term protective benefits (e.g., cardioprotective). Indeed, since the vast majority of COCs contain ethinyl estradiol, unless newer estrogen formulations are developed, the main distinguishing component between the various COC formulations would be the pharmacological profile of the progestin.
Another issue that relates to current COC use pertains to the cyclical regimen (conventionally, 21 days of active treatment followed by 7 hormone-free days), which was initially designed to mimic the natural menstrual cycle. However, the conventional treatment cycle recreates some of the problems that occur with spontaneous menstruation, such as headaches, cramps, breast tenderness and bloating/swelling, during the hormone-free interval [34]. Possible solutions for ameliorating and managing these symptoms include extended administration of COCs and reducing the number of hormone-free days per cycle [34–36]. Moreover, increasing the number of active treatment days per cycle to 24 and decreasing the hormone-free interval to 4 days with Yaz® has been demonstrated to provide effective contraception [37], as well as improve emotional and physical symptoms associated with premenstrual dysphoric disorder compared with placebo treatment [38,39].
The use of off-label, extended-treatment regimens in clinical practice is recognized, although the extent to which this occurs is unknown. Moreover, a 91-day oral contraceptive regimen (12 weeks [84 days] of active treatment followed by 1 hormone-free week) with a COC containing ethinyl estradiol 20 μg plus LNG 100 μg has been approved; however, there were issues such as the high dropout rate and high incidence of breakthrough bleeding (particularly in the first few months of treatment) compared with the conventional regimen in a large study [40]. It is possible that these issues may be resolved by a move towards ‘patient defined’ treatment regimens, with the proviso that active treatment duration should not be less than 21 days and the hormone-free interval does not exceed 7 days.
Finally, it should be noted that since some women find taking a pill every day to be inconvenient, research into combined hormonal contraceptive methods with a lower dosing frequency and/or different route of administration will continue to be an area of interest.
Executive summary
Ethinyl estradiol (EE) 20 μg plus drospirenone 3 mg prevents pregnancy through the combined actions of a low dose of estradiol and the unique progestin, drospirenone.
Drospirenone provides both antimineralocorticoid and antiandrogenic benefits.
Drospirenone produces dose-dependent inhibition of follicular maturation or growth, with the 3 mg dose preventing ovulation in all subjects.
The antimineralocorticoid activity of drospirenone results in natriuresis, which opposes the water retention and weight gain associated with the estradiol-stimulated production of plasma renin substrate and the consequent retention of sodium.
Drospirenone is rapidly absorbed and steady-state concentrations are achieved within 7–10days without accumulation problems.
EE 20 μg plus drospirenone 3 mg does not need to be taken with food.
Clinical studies show that EE 20 μg plus drospirenone 3 mg has a Pearl Index of 0.23 for typical use, which is consistent with other low-dose combined oral contraceptives (COCs).
Cycle control with EE 20 μg plus drospirenone 3 mg is as good, and is similar to that seen with a COC containing EE 20 μg plus desogestrel 150 μg.
The most common adverse events observed with EE 20 μg plus drospirenone 3 mg are typical of those found with COCs.
The majority of adverse events observed with EE 20 μg plus drospirenone 3 mg were mild-to-moderate in intensity and the rate of discontinuations due to adverse events was low.
Footnotes
Acknowledgements
Editorial assistance for the development of this manuscript was provided by Phil Jones, with financial support from Schering AG (Germany).
