Abstract
The assessment and then treatment of a change in libido, or a change in the desire to partake in sexual activity, during the menopausal transition and beyond has been a challenging and elusive area of clinical research. This is partly due to the multidimensional nature of female sexuality, the difficulties of measuring testosterone in women in a reliable and accurate manner, and the complexity of the neurobiology and neurobehavior of female sexual desire. In addition, there is a lack of evidence for diagnostic specificity of low free testosterone levels for the symptom of low libido in women for whom there are no confounding interpersonal or psychological factors; although, in the symptomatic population of surgically or naturally menopausal women, a low level of free testosterone often accompanies a complaint of reduced desire/libido. The randomized clinical trial research on testosterone replacement for naturally and/or surgically menopausal women with sexual dysfunction has been criticized for a high placebo response rate, supraphysiological replacement levels of testosterone, the perception of modest clinical outcome when measuring objective data such as the frequency of sexual intercourse relative to placebo, and the unknown safety of long-term testosterone replacement in the estrogen-replete surgically or naturally menopausal woman. A careful review of current evidence from randomized, controlled trials lends support to the value of the replacement of testosterone in the estrogen-replete menopausal woman for whom libido and desire has declined. The issue of long-term safety remains to be answered.
Multifactorial nature of female sexuality
The assessment and then treatment of a change in libido, or a change in the desire to partake in sexual activity, during the menopausal transition and beyond has been a challenging and elusive area of clinical research. There is a lack of evidence for the diagnostic specificity of low free testosterone levels for the symptom of low libido in the women for whom there are no confounding interpersonal or psychological factors; although, in the symptomatic population of surgically or naturally menopausal women, a low level of free testosterone often accompanies a complaint of reduced desire/libido [1–6].
Female sexuality is dependent on biological, psychosexual, sociocultural and context-related factors [7–9]. As a consequence, any movement or change in any of these realms may increase or decrease a woman's perception of her drive or motivation to participate in sexual activity. The presence of any chronic medical illness such as diabetes, pulmonary or cardiovascular problems, or depression will in many instances impact a woman's sexuality concurrent with changes related to age [10,11]. The length of the relationship with a partner, as well as aging, has been demonstrated to impact sexual interest and frequency of sexual activity [12,13]. The menopause has been shown to have an incremental effect on a woman's sexuality, separate from the change brought about by aging [14]. The quality of her intimate relationship and the degree to which she feels empowered in it have been demonstrated to affect sexual desire [15]. The culture in which she lives has been shown to affect frequency of sexual intercourse [16]. The degree of stress she is under, as well as her general wellbeing, has also been shown to affect her libido. A recent longitudinal study demonstrated that higher stress lowers well-being, resulting in a decrease in sexual arousal, enjoyment, orgasm and desire [17]. In addition, the balance between sexual inhibition and sexual excitement may be unique to each woman and may change according to her circumstances, along with any other change she may have with regard to these opposing forces [18]. Thus, the treatment of any woman distressed by a change in desire at midlife and beyond is driven by many factors. The clinician's task is to elucidate which one or more of these many variables changed in her life, concurrent with her change in sexual desire, and then to determine whether or not these changes are related to her change in sexual desire.
Prevalence of sexual problems in the population
Estimates for female sexual dysfunction in the population range from 25 to 63% [19]. This range is dependent on many variables including whether or not the patient is distressed by the dysfunction, the nature of the sampling and the definitions of dysfunction. Epidemiological studies have shown a decline in coital rate [12,13,20,21], sexual interest [20–22], orgasm [13,20,22], arousal [20] and enjoyment with age. Most studies find an incremental effect of menopausal status over aging on sexuality [14]. Estimates for a decline in desire or hypoactive sexual desire disorder (HSDD) vary according to the group studied, such as women who have undergone hysterectomy with bilateral oophorectomy, or naturally menopausal women. Few studies address whether or not the woman is distressed by her lack of desire. The Women's International Study of Health and Sexuality (WISHeS), using a European sample drawn from 2467 women, found the prevalence of low desire in reproductively intact women aged 20–49 years to be 16%, but only 7% had HSDD (low desire and distress). Women in the same age group who had undergone hysterectomy and bilateral oophorectomy were significantly more affected. A total of 29% of these women reported low desire and 16% met criteria for HSDD [23]. WISHeS, using an American sample drawn from 2671 women, found the prevalence of low desire ranged from 9% in naturally postmenopausal women to 26% in younger surgically postmenopausal women. The prevalence of HSDD for younger surgically postmenopausal women was statistically greater than that found for age-matched premenopausal women. The percentage of the American sample with HSDD who were distressed by their HSDD symptoms was greater in the surgically menopausal younger group (aged 20–49 years) than in the age-matched premenopausal group with HSDD (72 vs 59%). No statistically significant difference was found in the percentage of surgically menopausal women with HSDD and naturally menopausal women with HSDD, aged 50–70 years, who were distressed by their HSDD (44 vs 33%) [24].
Definition of libido & arousal
The classifications described below were arrived at by expert opinion and consensus and have not as yet been incorporated into either the
Sexual interest/desire
Libido has been referred to as desire, appetite, drive and/or the motivation to seek out sexual satisfaction. The first and second female sexual function consensus conferences attempted to combine biological and psychological factors, a balance felt to be lacking in previous classifications [25,26]. These classifications distinguish between generalized and situational, lifelong or recent, and whether or not the woman is distressed by the symptom.
Arousal
The definition of arousal has evolved from a purely physical definition to definitions of arousal in categories reflecting the subjective and physical aspects of arousal [26]. The definitions below (Box 1) are taken directly from the work of Basson and colleagues during a consensus conference on female sexuality [26]. In addition, arousal problems can be further categorized by generalized and situational, lifelong or recent, and whether or not the woman is distressed by the symptom.
Dynamic interplay between arousal & sexual desire/libido
There is some controversy in the field as to the dynamic interplay and nature of arousal and desire/libido. The patient often does not distinguish between the two or does not understand the difference between physical arousal, subjective arousal and desire/sexual interest [18]. In addition, the order in which she experiences these may vary [18]. For purposes of research, sexual desire is often viewed as distinct, a drive to seek out or respond to sexual activities (or anticipation) and considered different from genital arousal. There is some early evidence that diminished desire may impact and diminish arousal [27]. Others, however, view sexual interest as a subcategory of arousal and, along these lines, view arousal and sexual desire/libido as a combination of sexual arousal (a state), sexual arousability (varies in time and by person), and sexual interest as an aspect of sexual arousal [28]. These differences are made more complex by the use of different scales to measure sexuality in randomized clinical trials examining hormone therapy and its impact on sexuality [9]. An 8-year prospective, population-based study of middle-aged women was unable to separate items denoting sexual interest from those denoting arousal (responsiveness) [29].
Definitions of libido and arousal disorders.
Absence of or markedly diminished feelings of sexual arousal (sexual excitement and sexual pleasure) from any type of sexual stimulation. Vaginal lubrication or other signs of physical response still occur.
Complaints of absent or impaired genital sexual arousal. Self-report may include minimal swelling or vaginal lubrication from any type of sexual stimulation and reduced sexual sensations from caressing genitalia. Subjective sexual excitement still occurs from nongenital sexual stimuli.
Absence of or markedly diminished feelings of sexual arousal (sexual excitement and sexual pleasure) from any type of sexual stimulation as well as complaints of absent or impaired genital sexual arousal (e.g., vulval swelling and lubrication)
Spontaneous, intrusive and unwanted genital arousal (e.g., tingling, throbbing and pulsating) in the absence of sexual interest and desire. Any awareness of subjective arousal is typically but not invariably unpleasant. The arousal is unrelieved by one or more orgasms, and the feelings of arousal persist for hours or days.
There are absent or diminished feelings of sexual interest or desire, absent sexual thoughts or fantasies and a lack of responsive desire. Motivations (here defined as reasons/incentives) for attempting to become sexually aroused are scarce or absent. The lack of interest is considered to be more than that due to a normative lessening with the life cycle and length of a relationship.
The perimenopausal transition & beyond
During the natural menopausal transition, estradiol levels decline significantly [30]. The decline in estradiol occurs during the late menopausal transition phase, which coincides with reports of at least two skipped menstrual cycles [30]. Although estrone production also decreases, it is the predominant estrogen postmenopausally. The postmenopausal ovary is believed to be an ongoing source of androgen production [31]. Following surgical menopause in younger women, there is an approximately 50% fall in circulating levels of testosterone and androstendione [32]. Estradiol levels also decline abruptly following bilateral oophorectomy, and the fall in estrogen is clearly much larger in the premenopausal woman [32]. Observational studies of young premenopausal women who have undergone bilateral oophorectomy provide an opportunity to explore the effects of sudden reduction of both androgen and estrogen on sexual functioning. A number of observational studies, both cross-sectional and longitudinal, have suggested that surgical menopause has a more negative impact on sexual functioning than hysterectomy alone [33–35]. The cross-sectional WISHeS demonstrated that the major impact of surgical menopause was on younger women [23].
Davison and colleagues found that women aged 55 years or older who had a bilateral oophorectomy had lower androgen levels than those who had not had a bilateral oophorectomy [36]. Androgen levels in this study were found to decline more rapidly in younger premenopausal women and then continue to decline with age with no significant changes across the menopausal transition [36]. Randomized, controlled trials of hormone therapies have focused on the evaluation of estrogen (± progestogens) alone or combined with different forms of delivery of testosterone for naturally postmenopausal women, surgically menopausal women, and a combination of both groups. One unique randomized clinical trial has examined testosterone replacement with and without an aromatase inhibitor, thus attempting to determine whether the effects of testosterone on mood and sexuality require aromatization to estrogen.
Difficulties & controversy around testosterone levels & measurement
The study of normative values for androgens per decade of life has been impacted by the number of women studied, the timing of sampling, their reproductive status and the sensitivity of the assay [36]. Furthermore, there has yet to be a study that has shown a relationship between serum androgen levels and complaints of sexual function, specifically desire or libido [37,38]. However, a low level of free testosterone often accompanies a complaint of reduced desire/libido, as illustrated in a number of the randomized, controlled trials [1–3,6]. Davis and colleagues, in a community-based, cross-sectional study, examined a sample of 1423 normal women aged 18–75 years. A total of 1021 women were included in the final analysis. There was no relationship between serum total or free testosterone or androstenedione levels with any domain score for the Profile of Female Sexual Function (PFSF), irrespective of age [38]. Although there was some association of low domain scores of arousal, sexual responsiveness and desire with low dehydroepiandrosterone sulfate (DHEAS) levels in women aged 18–44 years, there was nonetheless no association in the majority of women between low DHEAS levels and domain scores of the PFSF.
Female testosterone levels require a highly sensitive test, as their total and free levels are much lower than males. Many of the newer research studies have used a highly sensitive radioimmunoassay (RIA) that combines a solvent step with a chromatographic step to separate the testosterone from other steroids. Davison and colleagues relate that this test would be time consuming and expensive for clinical practice [39]. Other methods of measurement such as analog technique have not been as accurate, with poor consistency [39–41]. Davison and colleagues have recently demonstrated that a direct RIA method can be used in clinical practice with high validity for the low testosterone range compared with the conventional RIA method used in research. It was less accurate at higher testosterone levels [39].
Testosterone levels are a combination of the free testosterone level and the total testosterone bound to sex hormone-binding globulin (SHBG), as well as the portion loosely bound to albumin. Of circulating testosterone, 55–60% is bound to SHBG, 35–40% to albumin, and the remainder, the free testosterone, is nonbound [42]. The effect of SHBG on free and total testosterone is significant. This becomes important in those women receiving oral estrogen replacement preceding the exogenous replacement of testosterone. Oral estrogen artificially increases the woman's SHBG to ‘supraphysiological levels’ as well as artificially lowering her natural free testosterone level [43].
There are currently two opposing theories in the field: the ‘free-hormone hypothesis’ and the ‘megalin-dependent mode of steroid entry hypothesis’. These theories have opposite conclusions as to whether a supraphysiological level of total testosterone in excess of a normal pre-menopausal level is of concern or not, as long as the free testosterone is in the normal range for a premenopausal woman. The free-hormone hypothesis proposes that the free testosterone is the biologically active component, its level defines exogenous testosterone replacement as physiological or supraphysiological, and that the SHBG-bound testosterone or the loosely albumin-bound testosterone are not biologically active [44]. Since these latter two bound portions have been generally regarded as nonbiologically active by those who support the free-hormone hypothesis, then it would not be of concern if exogenous testosterone replacement results in supraphysiological levels of bound testosterone. This hypothesis is based on the observation that the small size and lipid solubility of steroid hormones, in this case testosterone, means that the free molcule can traverse the plasma membrane of cells, whereas bound to SHBG or albumin it cannot. Hammes and colleagues have proposed that there are two mechanisms for entry: free and protein bound. The latter is referred to as the megalin-dependent mode of steroid entry hypothesis [45]. They also argue that cell-surface receptors for SHBG in steroid target cells as well as passive diffusion both play a role on steroid target cells [46]. For a full discussion of this alternative view, the reader is referred to the review by Adams [44] as well as the commentary on this by Willnow and Nykjaer [47] and Rosner [46]. The importance of this new alternative view to to the meaning of a supraphysiological level of total testosterone and its implications for the exogenous replacement of testosterone is significant. If the megalin-dependent mode of steroid entry hypothesis is correct, then it is important that the pharmacokinetics of testosterone replacement in women be fully understood with regard to premenopausal physiological ranges for free, albumin-bound and SHBG-bound testosterone, rather than just being concerned with the level of free testosterone after exogenous replacement, as indicated by the free-hormone hypothesis.
Notably, Davis and colleagues carried out the only randomized, controlled trials in which physiological levels for both free and total testosterone were achieved along with statistically significant changes in mood and sexual function [4]. However, the placebo group was treated with testosterone rather than transdermal estrogen alone. Due to the use of oral estrogens in the testosterone-patch studies [1,3,6] and the consequent high SHBG levels prior to exogenous replacement, the results are skewed towards higher-dose testosterone replacement patches with consequent supraphysiological total testosterone, along with physiological free testosterone levels. The fact that Davis and colleagues were able to achieve these results while maintaining physiological levels in both free and total testosterone points to the need for further pharmacokinetic studies of testosterone delivery [4].
In this review, any exogenous replacement of testosterone in surgically or naturally menopausal women will be referred to as supraphysiological if it exceeds levels to be expected in a premenopausal woman either for free or total testosterone.
The placebo effect
Sexual desire or libido is a complex behavior whose etiology is often a combination of physiological and psychosocial mechanisms. This is particularly so in the patient with previously good sexual desire who experiences an insidious
Patients may achieve a beneficial effect by being in a therapeutic trial despite receiving placebo. Many explanations have been put forward to explain this phenomenon. They include:
Subjects'/patients' expectations of the therapeutic intervention [50]
Nonspecific effects of the study [51]
Social support
Attention
Education
Other treatments that have a beneficial effect
Characteristics of outcome measures in the trials
Measures with poor test–retest reliability, inter-rater reliability or response to change will increase artifactual placebo responses [52,53]
Length of study
May or may not improve validity of treatment response [51]
Some feel the idea that the placebo response is not treatment is a misnomer; the placebo response results from nonspecific treatment [54]. In psychiatric research, particularly clinical trials for the treatment of depression, the placebo response has been reported to be 30–70% [49,52,55–57]. One meta-analysis of randomized clinical trials found that in recent years the placebo response has increased for reasons that were not discernable [57]. Attempts to reduce the placebo response by increasing the severity of symptoms required for those entering a study did not result in a lower placebo response [53]. Fava and colleagues, among others, have theorized that the placebo response is not sustained, therefore the longer the study the greater the chance that the treatment will discriminate from placebo [53]. Dworkin and colleagues argue that the longer the trial the more opportunity there is for the nonspecific effects of participating in the study to impact outcome for the nontreated group, for example, reassurance and social support [51]. The heterogeneity of the pathophysiological mechanisms for both chronic pain and depression may be the etiology of the difficulties in discriminating between placebo and treatment [51]. A similar issue may exist for randomized, controlled studies examining the efficacy of different agents in improving libido or desire. Sexual desire is as complex and heterogeneous as depression. It is believed to be a consequence of a change in a woman's hormonal milieu as well as the many factors discussed in this article.
Commenting on antidepressants, Paul Leber, former director of the FDA's Division of Neuropharmacology Drugs Products states: “In my view, the reason why efforts to develop psychotropic drugs so often flounder has little, if anything, to do with our methods, techniques or development strategies. Given a modestly effective drug and a reasonably homogeneous sample of patients biologically capable of responding to that drug, it is not especially difficult, using the crudest of clinical methods currently available (e.g., a 10 cm line scale), to demonstrate that the drug (e.g., an analgesic) provides a clinical benefit. Admittedly, some progress is being made, but the field still has a long, long way to go. One must wonder just how anyone can possibly claim to be surprised when an attempt at rational drug discovery fails or when samples of patients recruited using identical selection criteria vary in their average behavior from one clinical study to the next. Indeed, given how little we actually understand about the behaviors and affects we seek to manage through pharmacological interventions, it has long seemed to this commentator that we are exceedingly fortunate to possess the number of modestly effective drugs that we do” [58].
Pharmacokinetics
The pharmacokinetics of testosterone replacement have not been fully elucidated and appear to vary by delivery mechanism. The testosterone patch, testosterone gel by different manufacturers, orally inhaled testosterone, methyltestosterone with esterified estrogens, testosterone undecanoate, as well as other novel forms of delivery, have different pharmacokinetics. This has been preliminarily examined in HIV-infected women, as well as in healthy women. These studies will help establish dosing regimens that lead to physiological rather than supraphysiological replacement for either or both free and total testosterone. There are many variables that may impact levels. These include body mass index (BMI), SHBG levels, rapid or slow metabolizers, impact of concomitant drugs for comorbid illnesses on liver metabolism, quality of absorption and depot effects. For more discussion of these very important issues the reader is referred to Singh and colleagues [59], Davison and colleagues [60] and Javanbakht and colleagues [61], among others.
Does testosterone or aromatization of testosterone impact sexual function?
Testosterone binds to androgen receptors. This binding results in the transcription of genes and consequent production of different effector proteins [62]. In most tissues, testosterone acts via the androgen receptor or is converted to the biologically active androgen dihydrotestosterone, or is converted to 17β-estradiol [63]. A recurrent question posed in the field of sexual medicine is whether or not the impact of testosterone on sexual behavior in women is through its aromatization to estrogen. This question has been made more difficult by the finding that aromatization to estrogen is the case in mice [64]. Davis and colleagues conducted a randomized, double-blind, controlled study comparing two groups of postmenopausal women with complaints of low desire [4]. One group was replaced with testosterone (n = 30) while the other group was replaced with testosterone and an aromatase inhibitor (n = 30). The women entering the study reported low sexual satisfaction, Sabbatsberg Sexual Self-rating Scale (SSS) score under 42, and had a total testosterone value of less than 1.2 nmol/l [65]. They were treated with 400 μl of a 0.5% testosterone gel, giving a total dose of 2 mg. The aromatase inhibitor group received 2.5 mg of letrozole daily. Both groups were followed with the SSS and the psychological general wellbeing index (PGWB) [66], as well as the Beck Depression Inventory at entry and at 16 weeks [67]. After 16 weeks of estradiol, levels of SHBG, fasting lipids, lipoprotein (a) and C-reactive protein did not differ, either at baseline or between groups. The SSS and the PGWB significantly increased along with a reduction of the Beck Depression Inventory in both groups. Free and total testosterone were in the physiological range for a premenopausal woman in both groups. Sexual satisfaction, wellbeing and mood improved. These results seemed to indicate that aromatization was not necessary for the beneficial response to testosterone. Shifren, in her commentary on this study, discusses whether or not the testosterone-only group can be considered a placebo group, while Davis and colleagues have argued that the comparison of the two groups is valid [4,62].
Randomized, placebo-controlled trials
A literature search for relevant studies was performed on PubMed. Reference lists from these articles were also examined. For this review, double-blind, randomized, controlled trials of postmenopausal hormone therapies, in samples of women with natural or surgical menopause, which included specific and comprehensive measures of sexual function, have been included. One single-blind study by Davis and colleagues was included [68]. Trials must have included a placebo or drug comparison (for example, in studies of the addition of testosterone to estrogen therapy, the placebo group received estrogen only, while in another study, testosterone was compared with testosterone combined with an aromatase inhibitor, and the testosterone-only group was the placebo). Studies that only compared treatment outcomes to baseline levels of function were not included. For example, a much-cited study by Sarrel and colleagues was not included [69], which compared all outcomes to baseline rather than to a control group (Boxes 2 & 3).
List of randomized, controlled trials: natural, combined or surgically menopausal.
Nathorst-Boos and colleagues, 2006 [5]
Lobo and colleagues, 2003 [71]
Myers and colleagues, 1990 [72]
Dobs and colleagues, 2002 [73]
Three of these four crossover studies were felt to not have a sufficient washout period to prevent carry-over effect by the Cochrane Review [78]. Somboonporn and colleagues felt that the lack of a washout period for Floter and colleagues and Shifren and colleagues may have contributed to a reduced treatment effect [1,75,78].
Design of studies.
Davis and colleagues, 2006 [4]
Braunstein and colleagues, 2005 [3]
Buster and colleagues, 2005 [2]
Simon and colleagues, 2005 [6]
Warnock and colleagues, 2005 [74]
Lobo and colleagues, 2003 [71]
Dobs and colleagues, 2002 [73]
Myers and colleagues, 1990 [72]
Impact of estrogen replacement in naturally menopausal women
In naturally menopausal women there is support for the beneficial effect of estrogen on different aspects of sexual function. A decline in estrogen has been shown to have the most profound impact on vulvar/vaginal tissues, leading to delayed or absent orgasm, pain with sexual relations (dyspareunia), decreased lubrication, among many symptoms associated with atrophy [69]. There was a statistically significant improvement over placebo for a number of parameters measured on the McCoy Female Sexuality Scale, including the total score, satisfaction with sexual activity, increase in the feeling of sexual attractiveness, improvement in sex life [79], sexual fantasies, degree of sexual enjoyment, vaginal lubrication and frequency of sexual activity [80], with estrogen replacement in the naturally menopausal woman. Sherwin found that oral conjugated equine estrogens had a significant beneficial effect on sexual desire and arousal compared with the nonhormonal phase of cyclical administration [81]. Cross-sectional studies as well as longitudinal studies of menopausal women have been supportive of these findings. The lack of lubrication, change in quality of orgasm, increased pain with sexual relations, among other contemporaneous psychosocial factors such as education, emotional closeness with partner, stress and health problems impact sexual satisfaction and rate of intercourse [7,82,83].
Entry criteria: symptomatic and/or low free or bioavailable testosterone
The trials sought out symptomatic postmenopausal women who would also be considered to have low levels of free testosterone for a premenopausal woman [1–6]. Although Warnock and colleagues [74] and Lobo and colleagues [71] sought out women who were symptomatic, their mean free and bioavailable testosterone at entry were not in the lowest quartile. Floter and colleagues [75] and Dobs and colleagues [73] did not seek out symptomatic women, and for Floter and colleagues [75] the mean free testosterone was not in the lowest quartile. Of the trials that met both the symptomatic and low free testosterone criteria, Davis and colleagues, Brauntein and colleagues, Buster and colleagues, Simon and colleagues and Shifren and colleagues [1–4,6] all had replacement median or mean free testosterone in the physiological range [1]. All of these trials demonstrated an increase in sexual function with testosterone treatment. A summary can be found in Table 1.
Low entry free testosterone for baseline and/or placebo group (from 2000)

Highest median and highest spread for the four groups, placebo and three patch groups at baseline
Median for placebo and baseline for treatment group was the same in these two studies, for range the lowest and highest level for each group are reported here. E/A: Estrogen/androgen; SD: Standard deviation.
Was replacement in excess of levels expected for premenopausal woman?
To determine whether or not the trials had significant numbers of women receiving supraphysiological levels of testosterone, it is necessary to review the reference ranges for each study for free and total testosterone and then to compare the levels found in the treatment arm of the study at completion with the reference ranges for the study. Note that the placebo arm and the entry treatment arm of these studies had postmenopausal levels of free and total testosterone. The studies in which estrogen replacement was largely oral, with some having less than a quarter of patients receiving transdermal estrogen replacement [2,6], had supraphysiological levels of SHBG for all groups at baseline (all testosterone-patch studies as well as Warnock and colleagues, Lobo and colleagues and Floter and colleagues) [71,74,75]. The high SHBG levels resulted in a higher total testosterone relative to free testosterone (the active portion) at baseline and at treatment end point for the respective testosterone replacement doses than would have occurred if these women had their own unaltered physiological levels of SHBG [84–87]. Consequently, both the placebo group and the treatment arm had artificially reduced serum levels of free testosterone at baseline, and for the testosterone-patch studies supraphysiological total testosterone levels at the study end point with nonetheless physiological free testosterone levels. For the methyltestosterone studies, no meaningful concentrations can be determined. In terms of SHBG, for Nathorst Boos and colleagues [5] the type of estrogen replacement was not reported, and for Davis and colleagues [4] this was not an issue as all of the patients were receiving transdermal estrogen.
Whether or not the patients were replaced physiologically or supraphysiologically by type of testosterone delivery will now be reviewed, with a focus on the biologically active testosterone, free testosterone. Reference ranges and whether or not the reference range is pre- or postmenopausal will be indicated.
The recent testosterone gel studies include Nathorst Boos and colleagues [5] and Davis and colleagues [4]. Nathorst-Boos and colleagues, in their 12-week, double blind, randomized, crossover design examined 53 naturally postmenopausal women, aged 50–65 years, with serum free testosterone levels below 54 ng/l (2 nmol/l), receiving estrogen replacement and treated with testosterone gel 10 mg/day. By the end of the study, total testosterone levels were supraphysiological, having increased by tenfold compared with placebo [5]. This study measured dihydrotestosterone at baseline and at 12 weeks, the treatment group doubled compared with the placebo group. This supraphysiological level was not observed in their previous, shorter-duration study using this testosterone gel [42]. They surmised that this resulted from an accumulation of testosterone in the skin over the 3-month period. This highlights the importance of extended-length studies to determine the pharmacokinetics of different forms of replacement over time.
Davis and colleagues, in a 16-week, randomized, placebo-controlled trial, enrolled 80 postmenopausal women aged 55 years or older, or women who had undergone bilateral oophorectomy, receiving a transdermal estrogen patch equivalent to 50 μgm of 17β-estradiol by matrix patch or 1 mg/day by transdermal gel for 8 weeks. The patients were assigned randomly to either 400 μl of a 0.5% testosterone gel (total dose 2 mg) plus a placebo tablet or 400 μl of a 0.5% testosterone gel plus 2.5 mg of letrozole, an aromatase inhibitor, for 16 weeks [4]. The free and total testosterone levels were physiological as compared with the reference ranges for premenopausal women. This study is the only study to date using the gold standard for testosterone measurement that has demonstrated positive mood and sexual function results and that has achieved physiological replacement of free and total testosterone. One could surmise based on the placebo response of other studies that if the study had included a transdermal estrogen replacement only arm, there would have been improvement in this arm, although not as significant as the testosterone-only and testosterone and letrozole arms.
Floter and colleagues compared the effects of 2 mg oral estradiol valerate with estradiol valerate plus 40 mg testosterone undecanoate in surgically menopausal women aged 45–60 years [75]. The mean level of free testosterone and total testosterone in the group receiving testosterone therapy was supraphysiological for the postmenopausal woman reference range provided, but the range of levels was wide, in keeping with the known erratic absorption of testosterone undecanoate. Free and total testosterone levels would be considered supraphysiological for a premenopausal woman by other reference ranges for total and free testosterone [1].
Three recent studies examined replacement of methyltestosterone in estrogen-replete women receiving esterified estrogens [71,73,74]. Dobs and colleagues did not provide reference ranges for their testosterone measurements [73]. Warnock and colleagues assessed 102 women aged 32–62 years who were in a 2-week lead-in phase receiving 1.25 mg esterified estrogens [74]. They found that the addition of 2.5 mg of methyltestosterone resulted in the patients being in the postmenopausal physiological reference range at study completion for free testosterone, bioavailable testosterone, and just above the postmenopausal reference range for total testosterone. Lobo and colleagues assessed 218 women aged 40–65 years who were already receiving 0.625 mg of esterified estrogens [71]. They found that the addition of 1.25 mg of methyltestosterone to esterified estrogens was associated with total testosterone levels in the low range, while the bioavailable testosterone levels were just above the normal range. Menopausal status for the reference ranges in this study were not indicated by the authors. For all the methyltestosterone studies, it is unclear what role the unmeasured levels of methyltestosterone played in terms of androgenic effect.
Four of the studies reviewed testosterone replacement with a testosterone patch. Simon and colleagues and Buster and colleagues used a single dose of 300 μg, Braunstein and colleagues used three doses of 150, 300 and 450 μg in different arms of their study [3], and Shifren and colleagues used either one 150 or one 300 μg dose. Buster and colleagues, Braunstein and colleagues and Simon and colleagues [2,3,6] used the same laboratory [88], with a study duration of 24 weeks and 447 patients; Shifren and colleagues used a different laboratory with a study duration of 12 weeks and 75 participants [1]. Table 2 illustrates and compares these studies. The Shifren and colleagues study was distinguished by being a crossover design. The women had undergone oophorectomy and hysterectomy and were aged from early twenties to 70 years. The women's median (mean for [1]) free and total testosterone levels pretreatment were in the low range for postmenopausal women in all treatment groups. The testosterone-replacement serum levels at the study end point for biologically active free testosterone were physiological compared with a reference range for premenopausal women in all four studies [1–3,6]. There were a small number of patients who were modestly above a premenopausal physiological free testosterone level, as illustrated by the ranges and standard deviations for the 300 μg patches, and in one study for the 450 μg patch it was also above the range for a small proportion of the patients. There was also a group of women receiving both the 300 and 450 μg patch who remained in the lowest range for a premenopausal woman. The existence of outliers – extremely low or high scores by one or a small number of the subjects – exists in most currently prescribed hormone treatments approved by the FDA and is supported by a number of factors impacting hormone levels. These include variability of metabolism, route of administration, individual differences in liver function, skin absorption, body composition (fat mass/lean mass), body size, medication interactions and levels of different circulating binding proteins such as SHBG and albumin [89]. Not surprisingly, due to the baseline supraphysiological SHBG levels, the total testosterone values were supraphysiological, except for Braunstein and colleagues' 150 μg patch [3]. In the latter case, the median was low premenopausal for free testosterone and physiological premenopausal for total testosterone, and this group failed to show a treatment effect.
Testosterone patch replacement studies.

Testosterone 1 ng/dl = 0.0347 nmol/l. SI conversion factors: to convert total testosterone and bioavailable.
Testosterone to nm/l, multiply by 0.0347. To convert nmol/l to ng/dl, pmol/l or pg/dl, divide by 0.0347.
BISF: Brief index of sexual functioning for women; CEE: Conjugated equine estrogen; EOE: Equivalent oral estrogen; PDS: Personal distress scale
PFSF: Profile of female sexual function; PGWB: Psychological general well being index: RR: Reference range; SAL: Sexual activity log
SHBG: Sex hormone-binding globulin.
Sexual function outcome: improvement in sexual function
The sexual function outcome measures for the randomized, controlled trials examining add-back testosterone in the estrogen-replete naturally or surgically menopausal woman that met the review criteria are listed below. A full discussion of the difficulties in analyzing the older randomized, controlled trials can be found in a review by Alexander and colleagues [9]. This would include Sherwin & Gelfand, Burger and colleagues and Myers and colleagues [70,72,76]. The studies from 2000 onward [1–6,71,73–75], Shifren and colleagues, Floter and colleagues and Lobo and colleagues [1,71,75], were also reviewed by Alexander and colleagues [9].
These trials used different validated scales for measuring sexuality. Consequently, in describing the results it is necessary to refer to the domain or descriptor separately. A full discussion of these validated scales is beyond the scope of this review. All of the trials found some improvements or positive change in sexual symptoms, and some of the trials found improvements in psychological symptoms, as illustrated in Boxes 3 & 4. Davis and colleagues found that the use of multiple questions in a single study to measure one effect, such as sexual desire, may be problematic [90]. This may have resulted in conflicting results for Warnock and colleagues and Lobo and colleagues for desire, where the first questionnaire had statistically positive results and the other did not [71,74]. Psychological symptoms were not the primary end point in many of these trials and, as such, have fewer data collectively. Table 3 shows a summary of these studies. Some studies were inadequately powered for psychological symptoms and, in those instances, their negative results were not listed [9]. Due to the nature of randomized clinical trials, these improvements may have been mild, moderate or significant in the different women in these trials. The statistical analysis simply demonstrates that the treatment arm was superior to the placebo arm for outcome on the variables studied. The question of whether or not the clinical improvement is as remarkable as the outcome measures has been raised in other reviews [90–94], and will be addressed later in this review.
Improvement in psychological variables with add-back testosterone in the estrogen-replete surgically or naturally postmenopausal woman.
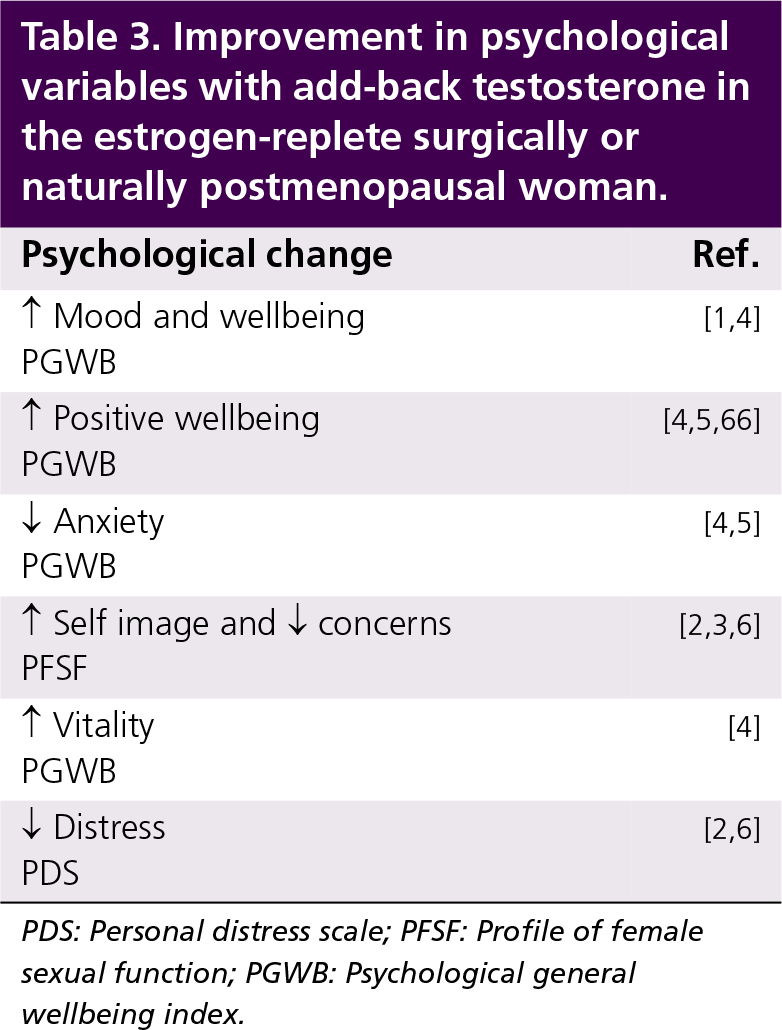
PDS: Personal distress scale; PFSF: Profile of female sexual function; PGWB: Psychological general wellbeing index.
Sexual dysfunction as a criteria for entry.
Floter and colleagues, 2002 [75]
Dobs and colleagues, 2002 [73]
Davis and colleagues, 1995 [68]
Myers and colleagues, 1990 [72]
Sherwin & Gelfand, 1985 [76]
Placebo response: context & meaning
The placebo response varied from study to study, as well as from sexual function domain to sexual function domain, for the respective studies. It was consistent with established placebo responses for this type of study as well as for other psychometric behavioral outcome studies for conditions that have behavioral heterogeneity. Given the good placebo response for the nonspecific treatment aspects of being in these studies as well as the greater responses to testosterone treatment, the question remains for clinical practice as to what the clinical significance is of treatment, and whether or not the benefits to sexual function over placebo are worth the risk of treatment.
To better inform healthcare practitioners regarding the clinical significance of any of these results, the relevance of any improvements in sexual function could be measured, for example, in terms of what percentage of women are satisfied with a particular result [9,90], their reduction in personal distress or concerns, their increase in wellbeing, and their improved quality of orgasm/arousal/desire/interest. Although frequency of sexual activity has received much attention and has improved in these studies, it may or may not be as clinically important to the practitioner and the patient as these other factors. The degree of improvement in frequency of sexual activity relative to placebo has been criticized for being unimpressive per month [93]. However, the authors of this article believe that the percentage increase in frequency over baseline and/or placebo may be a more valuable way to access clinical success than the actual number of sexual intercourse events. The average midlife woman in a relationship has one sexual interaction/week, with this number rapidly declining with age [105]. These testosterone-replacement studies have examined a very broad range of ages, from the early twenties up to the age of 70 years. For the mid- to late-life woman, the percentage increase in sexual frequency achieved over placebo with these treatments may be greatly appreciated by herself and her partner relative to their own baseline. Female sexuality is multifactorial and more elusive than the sexuality of males, whose treatment end points such as quality and duration of erection, ejaculation, frequency of intercourse, and fantasies have led to faster progress in the treatment of male sexual dysfunction. Relative to placebo, the studies with a mean or median physiological replacement range for a premenopausal woman since 2000 found improvement in sexual function and psychological indices including wellbeing [1,4], decreased concerns and distress [2,6], desire [2,3,6], interest [4,71,74], fantasies [4], arousal [2,3], orgasm [2,4,6], sexual responsiveness [2,71], satisfaction [2,4,6], relevancy [4] and frequency [1–4,6]. The one study of transdermal estrogen replacement with add-back testosterone with or without an aromatase inhibitor, by Davis and colleagues, had physiological free and total testosterone with improvement in wellbeing, interest, fantasies, orgasm, satisfaction, relevancy and frequency [4].
The fact that the placebo group had a lesser but improved response relative to treatment is also a very valuable result from these trials. It indicates the need for further research on nonhormonal, behavioral interventions that may give positive treatment results for patients distressed by their sexual function who cannot have hormone replacement, choose not to have hormone replacement, or do not require hormone replacement due to the positive outcome from receiving a behavioral intervention. Studies on the concomitant use of different behavioral interventions in the symptomatic naturally or surgically postmenopausal woman without significant confounding interpersonal or psychiatric etiologies indicate added benefit to the treatment outcome for patients who receive hormone replacement with add-back testosterone. This would be consistent with the findings in psychiatry for the efficacy of behavioral interventions for depression with or without antidepressant treatment [106].
In future, randomized, controlled trials of testosterone replacement, having a sufficiently high number in the treatment as well as the placebo group so that the study is adequately powered to report on the percentages of women who improve and to what degree they improve in the treatment and placebo groups, would also be helpful. The recent testosterone-patch studies comment on the percentage improvement in the domains of sexual function for the treatment group as well as the placebo group, but do not comment on the percentage of women in each group who improve and to what degree [2,3,6]. Commenting on the percentage of patients who improve and to what degree has been a critical methodology for psychiatric studies that examine antidepressant treatment of depression. It has been possible to further elucidate the percentage of patients who remit, partially remit and have treatment-resistant depression with antidepressant treatment, as well as examining the impact of concomitant cognitive behavioral therapy with pharmacotherapy using this approach [91,107,108]. Interestingly, for depressive illness, Fava and colleagues, in an excellent review on the issue of efficacy of antidepressant drugs to achieve remission, report antidepressant responses as follows: depression remission in 51–69%, partial response in 12–15% and nonresponse in 19–35% of patients [109]. This methodological and theoretical approach to the study of depression and depression treatments has led to breakthroughs in the understanding of the heterogeneity of depression, its neurobiology, improved methodology for treatment efficacy studies, and improved treatment strategies using pharmacological, psychological and psychosocial approaches, individually or collectively, depending on the patient's characteristics. It has also helped to establish a better understanding of pharamacokinetics, dose response and etiology for variation in dose response.
Conclusions
This review of the randomized, controlled studies, both recent and older, illustrates a positive benefit of add-back testosterone in some surgically or naturally postmenopausal women. It also illustrates that a behavioral intervention in the form of a randomized, controlled trial will lead to improvement in sexual function, although not as dramatically as testosterone replacement. A summary of the improvements in sexual response of women taking part in randomized, controlled trials is shown in Table 4. With these randomized, controlled trials, it is not possible to ascertain the cohort of women who have a significant improvement from testosterone replacement versus the women who have modest or no improvement. The pharmacokinetics of the different delivery systems is important in establishing dosing guidelines and achieving physiological levels of free and total testosterone. The implications of whether free and/or protein-bound testosterone acts at the cellular level, on which targets, and to what degree, will impact clinical treatment. Further exploration of the nonaromatization of testosterone has enormous implications for neurobiology as well as the clinical treatment of women for whom estrogen replacement is contraindicated, such as breast cancer patients receiving aromatase inhibitors [4]. This latter group has many of the same quality of life (QoL) issues as surgically and naturally menopausal women and does not have the option of estrogen replacement. Whether or not testosterone replacement would be safe in this group regardless of whether it aromatizes to estrogen will require further study.
Improvement in sexual function variables with add-back testosterone in the estrogen-replete surgically or naturally postmenopausal woman.

BISF-W: Brief Index of Sexual Function; CFSQ-F-C: Changes in Sexual Functioning Questionnaire; MSIQ or SIQ: Menopausal Sexual Interest Questionnaire; MSFS: McCoy Sexual Function Scale; PDS: Personal Distress Scale; PFSF: Profile of Female Sexual Function; SAL: Sexual Activity Log; SIS: Sexual inhibition scale; SSS: Sabbatsberg Sexual Self-Rating Scale.
Executive summary
Female sexuality is dependent on biological, psychosexual, sociocultural and context-related factors.
The length of the relationship with a partner as well as aging have been demonstrated to impact on sexual interest and frequency of sexual activity
The menopause has been shown to have an incremental effect on a woman's sexuality separate from the change brought about by aging.
The clinician's task is to elucidate which one or more of these many variables changed in the patient's life, concurrent with her change in sexual desire, and then to determine whether or not these changes are related to her change in sexual desire.
Estimates for female sexual dysfunction in the population range from 25 to 63%.
Few studies address whether or not the woman is distressed by her lack of desire.
The prevalence of hypoactive sexual desire disorder (HSDD) for younger, surgically postmenopausal women was statistically greater than that found for premenopausal women of the same age.
New classifications were developed by expert opinion and consensus but have not as yet been incorporated into either the
The patient often does not distinguish between the two or does not understand the difference between physical arousal, subjective arousal and desire/sexual interest. In addition, the order in which she experiences these may vary.
There is some early evidence that diminished desire may impact and diminish arousal.
During the natural menopausal transition, estradiol levels decline significantly.
The postmenopausal ovary is believed to be an ongoing source of androgen production.
Following surgical menopause in younger women, there is an approximately 50% fall in circulating levels of testosterone and androstendione.
Estradiol levels also decline abruptly following bilateral oophorectomy. The fall in estrogen is clearly much larger in the premenopausal woman.
The study of normative values for androgens per decade of life has been impacted by the number of women studied, timing of sampling, their reproductive status and the sensitivity of the assay.
There has yet to be a study that has shown a relationship between serum androgen levels and complaints of sexual function, specifically desire or libido.
Of circulating testosterone, 55–60% is bound to sex hormone-binding globulin (SHBG), 35–40% to albumin, and the remainder, the free testosterone, is nonbound.
Studies of sexual function in women have had placebo response rates of approximately 30%.
Studies of older individuals with depression have had placebo response rates of 32.5%.
Patients may achieve a beneficial effect of being in a therapeutic trial despite receiving placebo.
The testosterone patch, testosterone gel by different manufacturers, orally inhaled testosterone, methyltestosterone with esterified estrogens, testosterone undecanoate, as well as other novel forms of delivery, have different pharmacokinetics.
Variables impacting testosterone levels include body mass index, SHBG, rapid or slow metabolizers, impact of concomitant drugs for comorbid illnesses on liver metabolism, quality of absorption and depot effects.
Testosterone binds to androgen receptors.
A recurrent question posed in the field of sexual medicine is whether or not the impact of testosterone on sexual behavior in women is through its aromatization to estrogen.
Executive summary
A decline in estrogen has been shown to have the most profound impact on vulvar/vaginal tissues, leading to delayed or absent orgasm, pain with sexual relations (dyspareunia), decreased lubrication, among many symptoms associated with atrophy.
There was a statistically significant improvement over placebo for a number of parameters measured on the McCoy Female Sexuality Scale with estrogen replacement in the naturally menopausal woman.
Of the trials that met both the symptomatic and low free-testosterone criteria, all had replacement median or free testosterone in the physiological range. All of these trials demonstrated an increase in sexual function with testosterone treatment.
Estrogen replacement was largely oral in these studies, with some having less than a quarter of their patients receiving transdermal estrogen replacement.
This review of randomized, controlled studies, both recent and older, illustrates a positive benefit to add-back testosterone in some surgically or naturally postmenopausal women.
It also illustrates that a behavioral intervention in the form of a randomized, controlled trial will lead to improvement in sexual function, although not as dramatically as testosterone replacement.
With these randomized, controlled trials, it is not possible to ascertain the cohort of women who have a significant improvement from testosterone replacement versus the women who have modest or no improvement.
The pharmacokinetics of the different delivery systems are important in establishing dosing guidelines and achieving physiological levels of free and total testosterone.
The implications of whether free and/or protein bound testosterone act at the cellular level, on which targets, and to what degree, will impact clinical treatment. The controversies that remain are associated with the long-term safety of estrogen plus testosterone replacement, a topic beyond the scope of this review.
The role of behavioral interventions for women who are distressed by the change in their sexual function subsequent to surgical or natural menopause, with or without testosterone replacement, warrants further exploration.
The controversies that remain are associated with the long-term safety of estrogen plus testosterone replacement, a topic beyond the scope of this review. The role of behavioral interventions for women who are distressed by the change in their sexual function subsequent to surgical or natural menopause, with or without testosterone replacement, warrants further exploration.
Future perspective
This is a very exciting time for the study of psychoneuroendocrinology. Only a portion of the very interesting data on the impact of estrogen and testosterone on the brain was presented in this article. For further reading on this topic, several excellent review articles include Janowsky [110], Trainor and colleagues [111], Hogervorst and colleagues [112], Williamson and colleagues [113], Tuiten and colleagues [114] and Hermans and colleagues [115]. With novel studies such as that by Davis and colleagues [4], the excellent recent randomized, controlled trials, along with basic scientific research on the brain, our knowledge of these important hormones will exponentially increase in the coming years. The understanding of the actions of estrogens and androgens on behavior, wellness and neuropsychological functioning in general, and sexual function specifically, in the surgically, iatrogenically or naturally postmenopausal woman, holds hope for improved QoL in many women who are living longer and healthier lives.
Footnotes
