Abstract
Women with epilepsy have many special considerations; one of the most important of these is the risk and management of pregnancy. Both recurrent seizures during pregnancy and antiepileptic medications may have adverse maternal–fetal outcomes. Multiple registries of pregnancy outcomes for women with epilepsy worldwide are underway and data from these studies are emerging. Converging evidence from the registries thus far indicates an increased risk of major fetal congenital malformations with valproate exposure and one registry has shown an increased risk with phenobarbital exposure; further supporting evidence of the phenobarbital risk is needed.
Approximately 3–5 births/1000 will be to women with epilepsy [1]. In the USA alone, approximately 1.1 million women with epilepsy are of childbearing age [2]. Epilepsy, by definition, is a chronic condition and as such the treatment is typically a daily, long-term regimen. People with epilepsy are often otherwise healthy, and women with well-controlled epilepsy are not restricted from participating fully in life, including childbearing. Since most women with epilepsy do take antiepileptic drugs (AEDs) while pregnant, the risks of fetal exposure to AED treatment during pregnancy are of concern. However, AED use during pregnancy minimizes the risk of recurrent seizures and their accompanying medical and neurological sequelae. The American Academy of Neurology endorses adequate treatment of seizures during pregnancy and recommends stopping AEDs preconceptionally only in clinical situations where many indicators support the likelihood that seizures will not recur when the patient is off medication [3]. Furthermore, most neurologists and obstetricians find it intuitively appropriate to prevent seizures during pregnancy. This review will discuss the current evidence for risks during pregnancy for women with epilepsy, as well as the management of women of childbearing potential.
Factors affecting choice of therapy
Major congenital malformations
Major congenital malformations (MCMs) have been the focus of most research on pregnancy outcomes for women with epilepsy and usually raise the greatest concern for prospective mothers with epilepsy and their care providers. MCMs in reports related to epilepsy have been defined as structural abnormalities with medical, surgical or cosmetic importance identified within the first 5 days of life [4]. This definition is used in some reports, but there is no universally accepted definition as to the exact timing of when birth defects are identified. These include cardiac malformations (e.g., ventricular septal defect, coarctation of the aorta, tetralogy of Fal-lot, aortic valve stenosis and hypoplasia of the mitral valve), neural tube defects (e.g., spina bifida, myelomeningocele and anencephaly), craniofacial defects (e.g., cleft lip and palate), microcephaly, congenital megacolon and uro-genital malformations [4]. Minor anomalies may be outgrown during the first several years of life, such as hypoplasia of the midface and fingers [5–7]. Organogenesis is complete by 13 weeks of gestation, indicating that AED-related MCMs occur with first-trimester exposure to certain drugs.
Currently used AEDs carry US FDA Category C (adverse effect in animal studies but no human studies) or D (adverse human fetal risk) classifications. Category-D AEDs include valproate, carbamazepine and phenytoin. Category-C AEDs include gabapentin, felbamate, lamotrigine, levetiracetem, oxcarbazepine, pregabalin, topiramate, tiagabine and zonisamide.
Recent evidence of the risks of AED-related MCMs, including comparative risks, has emerged from pregnancy registries. These registries aim to survey the outcomes of a large number of AED-exposed pregnancies in order to provide accurate information regarding the magnitude of teratogenic risks and early evidence of any increased risk associated with newer and older AEDs. The various pregnancy registries employ slightly different methodologies, specifically their inclusion criteria, but where results are available, all have revealed important information thus far.
The first report of the North American AED Pregnancy Registry (1997–2002) showed a significantly increased risk of MCMs in the offspring of mothers receiving phenobarbital monotherapy [8]. In this registry, mothers enroll themselves by telephone early in their pregnancy and only the outcomes of mothers who do not have knowledge of the fetal status (malformation present or not) at the time of enrollment are used in the statistical analysis. Information is released from this registry when the teratogenic risk is twice that of the control group. The control MCM rate in this study was 1.62% in the first 5 days of life, from the general population in the Active Malformations Surveillance Program at Brigham and Women's Hospital in Boston (MA, USA). After 77 birth outcomes, phenobarbital monotherapy used early in pregnancy was associated with MCMs in 6.5% of pregnancies, which indicates a significant relative risk of 4.2-times the expected rate. The second report from the North American AED Pregnancy Registry (1997–2003) reported an increased rate of MCMs with valproate [9]. Following the same methodology as the phenobarbital study above, valproate monotherapy used in the first trimester was associated with a risk of MCMs of 10.7%, indicating a significant relative risk of 7.3-times the expected rate. All other AEDs combined in this registry have a MCM rate of 2.9% (95% confidence interval [CI]: 2.0–4.1%) [9].
The first report of the Australian Registry of Antiepileptic Drugs in Pregnancy, in which women with epilepsy enroll voluntarily both prospectively and retrospectively, demonstrated a MCM rate for valproate monotherapy of 16% [10]. Data gathered over 30 months are now available [11]; the authors report on the first 334 births, most of which were enrolled prospectively. The incidence of MCMs in offspring not exposed to AEDs was 4.3%, in offspring exposed to valproate the rate was 16.7%, with phenytoin the rate was 10.5%, with carbamazepine the rate was 3.3%, and with lamotrigine monotherapy the rate was 7.7%. In both reports from this registry, a dose effect was found for valproate; doses above 1100 mg/day were associated with a markedly increased risk. Although this collection of data is valuable, there are still a small number of evaluable outcomes so far, and therefore these results should be followed over time as the registry records more outcomes.
The UK Epilepsy and Pregnancy Register recently published outcome data on 3607 pregnancies; 239 women had epilepsy but did not receive AEDs [12]. The registry is prospective, and women must register before any abnormal prenatal tests have been found. The MCM rate includes those found within the first 3 months of life. The overall MCM rate for all AED-exposed cases was 4.2% and was significantly higher for offspring exposed to polytherapy at 6.0% (n = 770), compared with monotherapy at 3.7% (n = 2598) (adjusted odds ratio [OR]: 1.83; p = 0.002). The MCM rate for women with epilepsy who had not received AEDs during pregnancy (n = 239) was 3.5%. The MCM rate was significantly greater for pregnancies exposed only to valproate (6.2%) compared with those exposed only to carbamazepine (2.2%) (OR: 2.97; p < 0.001). The MCM rate for lamotrigine was 3.2% (n = 647) and it was 3.7% for phenytoin (n = 82). Gabapentin, topiramate and levetiracetam were also reported upon, but had 31 or less outcomes for each AED. A positive dose response for MCMs was noted for lamotrigine (p = 0.006), with a MCM rate of 5.4% for total daily doses of more than 200 mg. Of polytherapy combinations, those containing valproate had a significantly higher risk of MCM than combinations without valproate (OR: 2.49). The authors emphasize that 96% of live births born to women with epilepsy did not have a MCM.
The GlaxoSmithKline International Lamotrigine Pregnancy Registry (11-year study) reported a MCM rate of 2.9% detected within the first month of life (12/414 births) with first-trimester monotherapy exposure [13]. Subjects were enrolled by healthcare professionals worldwide, prior to knowledge of fetal tests.
Data from the worldwide Novartis safety database and pregnancy registries or study centers in six countries revealed a total of 248 pregnancies involving maternal exposure to oxcarbazepine monotherapy and 61 involving adjunctive therapy. The MCM rate in the monotherapy group was 2.4% (6/248) and the MCM rate in the adjunctive therapy group was 6.6% (4/61) [15]. The Gabapentin Pregnancy Registry reported no major malformations in 19 infants exposed to gabapentin monotherapy in early pregnancy [16]. Three cases of levetiracetam monotherapy during pregnancy have demonstrated normal outcomes 12 months postnatally [17]. Although the results of these last three registries appear to be promising, the numbers are too small to extrapolate definite conclusions.
Although the prospective pregnancy registries provide new and accurate information, they often confirm what has been found in retrospective surveys. A recent retrospective study evaluated the pregnancy outcomes of the National Medical Birth Registry, cross-referenced with individuals eligible for AED reimbursement from the Social Insurance Institution of Finland [18]. MCMs were significantly more frequent in offspring of patients with epilepsy who received AEDs during the first trimester (65/1411; 4.6%) than in offspring of patients who were untreated (26/939; 2.8%; p = 0.02). Valproate monotherapy was associated with a fourfold increased risk of MCMs compared with the epilepsy group not exposed to AEDs. Only one MCM was found in 99 oxcarbazepine-exposed offspring (uro-genital); therefore, the MCM rate for oxcarbazepine was less than the untreated epilepsy group. Overall, the authors concluded that the risk of monotherapy and polytherapy, excluding valproate, was not associated with an increased risk of MCMs.
A body of previous evidence has shown similar results, especially with regard to valproate. For example, an international survey of malformations (Malformations And DRug Exposure [MADRE] study) of 299 patients with AED exposure revealed oral clefts associated with phenobarbital and methylphenobarbital. Cardiac malformations were associated with phenobarbital, methylphenobarbital, valproate and carbamazepine [19]. Valproate was associated with spina bifida, hypospadias, porencephaly and other brain anomalies, and limb-reduction defects [19]. Numerous other studies have confirmed the increased risk of fetal major malformations with the use of Category-D AEDs [20–23]. In addition, a dose-related increase in MCMs has been found with valproate; doses greater than 1000 mg/day were associated with an increased risk of congenital malformations [23,24]. Therefore, phenobarbital and, most convincingly, valproate have clearly emerged as imparting a risk of malformations of at least double that of the general population. Polytherapy with more than one AED has also been repeatedly associated with increased risk. For example, in addition to the above information, polytherapy increased the overall risk of congenital malformations up to four-times compared with monotherapy in separate studies by Holmes and colleagues [4] and Kaaja and colleagues [25].
Maternal seizures & major malformations
Although there is no debate surrounding the importance of prevention of congenital malformations in women with epilepsy, the separate contributions of maternal seizures and AED treatment to the occurrence of congenital malformations are not so clear. The independent effect of maternal seizures is difficult to determine due to such confounding factors as superimposed AED effect(s).
In a prospective study, convulsive seizures during the first trimester, which is the type and timing of seizures thought to potentially have the most effect, were associated with malformations in 7.4% of pregnancies [4]. The rates of malformation in offspring of women with simple partial seizures was 7.8%, while in women with AED monotherapy the rate was 5.7%, and in women with AED polytherapy the rate was 8.6% [4]. Although the association of seizures and malformations is not significantly higher than that found with AEDs, these results suggest that seizures during pregnancy at least add to the risk of major malformations imparted by AED treatment.
A historical population-based study demonstrated that seizures during pregnancy were associated with an increased risk of MCMs of 3.8-times greater than the general population [21]. However, this study did not report the timing of seizure occurrence and seizure type. Information regarding seizures was only available for 40% of the study population, which also limits the strength of this association. Another prospective population-based study in Rotterdam (The Netherlands) found an increased risk of malformation (12.3%) associated with first-trimester seizure occurrence, versus a 4% rate of malformation in the seizure-free group [22].
Conversely, two large, prospective studies have shown no increased risk of malformations in women with seizures in pregnancy. An international, collaborative, prospective study of 983 pregnancies in Japan, Italy and Canada found no association between first-trimester seizures and malformations [24]. Another prospective study of 970 pregnancies found no associated risk between first-trimester seizures and malformations [25]. A retrospective study from the Mayo clinic (MN, USA) showed no increased risk of malformations linked with seizures during pregnancy [26].
Based on the previous discussion of the aforementioned six studies, the evidence on the effect of maternal seizures upon fetal malformations is mixed. Two studies indicate an association, one study suggests an association, and three studies show no association between seizures during pregnancy and the risk of malformations. Since none of these studies included a large proportion of women with epilepsy not receiving AEDs, the independent risk of seizures is difficult to establish. Perhaps the best interpretation of these data is that seizures can add to the risk imparted by AEDs, providing further support for the goal of maintaining seizure freedom during pregnancy.
Maternal epilepsy & major malformations
There is little evidence to indicate that maternal epilepsy as a chronic condition (without seizures during pregnancy) is associated with any major malformations [4,20,21,25]. In a case-controlled study of 57 children born to mothers with epilepsy not receiving AEDs compared with 57 controls, no statistically significant difference in major malformation rate was observed; 8.7% of the study group versus 5.3% of the control group [27]. No subject had convulsive seizures during pregnancy but 11/57 mothers had simple partial seizures during pregnancy. However, the sample size in this study was not determined to detect a difference in malformation rates; the study was aimed at finding a difference in intellectual outcome. Therefore, the question remains as to whether a larger study would have revealed a difference based on epilepsy alone, since AED use was not a factor in this study.
Neuropsychological effects of antiepileptic drug exposure
The neuropsychological effects of intrauterine exposure to AEDs have long been a concern. Evidence for these effects is difficult to obtain, since multiple influences on intellectual outcome must be taken into consideration; primarily, the maternal intelligence quotient (IQ) and the social environment. Furthermore, the children must be followed and evaluated years after birth. However, several carefully performed studies have shown both reassuring and cautionary results.
The first report emerged in 1995 from a cohort of adult men in Denmark who were exposed to phenobarbital in utero [28]. The indication for phenobarbital use was not epilepsy; in fact, maternal epilepsy was an exclusion criteria and phenobarbital was used generally for hypertension or sedation. The cohort was studied at the age of approximately 30 years and verbal intelligence was significantly decreased in phenobarbital-exposed men compared with matched controls. Interestingly, exposure that included the third trimester was a risk factor, which suggests that the risk of negative neurocognitive effects has a different timing of exposure than MCMs.
Two other studies reported a risk of significantly lower verbal IQ with valproate exposure compared with other AED exposure or nonexposed children [29,30]; one of these studies showed no risk of impaired intelligence with carbamazepine use [30]. Therefore, valproate has again emerged as having particular risk during pregnancy for both MCMs and adverse neurocognitive outcome.
Maternal & fetal risks associated with seizures
Seizures are known to cause fetal heart rate depression, fetal hypoxia with resultant acidosis, and fetal intracranial hemorrhage [31–33]. Intrauterine deaths related to fetal intracranial hemorrhage, which probably occurred after a single seizure, have been reported [31]. Spontaneous abortion, fetal hypoxia, bradycardia and antenatal death have been reported with both partial and generalized convulsive status epilepticus, possibly related to maternal trauma and placental hypoperfusion [34,35]. Furthermore, status epilepticus during pregnancy was extremely risky; it was associated with both a high fetal (48%) and maternal (33%) mortality rate [32]. Although exact statistical risks to the pregnancy from seizure-related complications are difficult to determine, it is clear that seizures impart an increased risk of fetal loss and maternal death. Status epilepticus in pregnant women may be precipitated by abrupt changes in or withdrawal of AEDs. Therefore, careful monitoring and counseling of patients needs to occur when a pregnant woman's AEDs are titrated or changed. The neurocognitive-out-come study cited previously found that five or more convulsive seizures were associated with a significantly lower verbal IQ in the offspring [29]. This new information provides another compelling reason to prevent seizures during pregnancy.
Management of women with epilepsy of childbearing potential
Monotherapy instead of polytherapy if possible
The foregoing information indicates that optimal seizure control, ideally seizure freedom, using the lowest-possible AED dose as monotherapy if possible, will be associated with the least risk of teratogenesis, lowest medical and obstetrical risk, and reduced cognitive risk to the offspring. Some women will not be able to achieve AED monotherapy and still have well-controlled seizures; in these situations, seizure control should not be sacrificed in order to reduce AEDs due to the significant risks of seizures to fetal and maternal survival.
Avoid phenobarbital & valproate if possible
Emerging data indicate that phenobarbital and valproate pose risks of malformations that are at least twice as great than that found in the general population, and that valproate carries an additional risk of adverse neurocognitive outcome. Therefore, the use of these AEDs during pregnancy deserves careful consideration as to the risk:benefit balance. The evidence for risk with phenobarbital is from the North American Pregnancy Registry; the rigorous approach of the methods employed in this registry make it difficult to refute, although this finding would be even more convincing if the other prospective registries also confirmed the risk. Clearly, further information regarding the teratogenic risk of phenobarbital is needed since it is a frequently used epilepsy treatement in the developing world. The risk for valproate was found in the North American registry, as well as in the Australian and UK registries and three other well-designed retrospective surveys. Definitive risks with other AEDs, including the newer AEDs, will be forthcoming from the excellent pregnancy registries underway worldwide.
Stopping antiepileptic drugs
Guidelines for considering discontinuing AEDs prior to conception in women with prolonged seizure freedom are put forth in the Practice Parameters [3] and, obviously, this is a clinical decision based primarily on risk factors for recurrent seizures, such as the presence of a structural brain lesion, history of frequent seizures and persistently abnormal electroencephalograms. For women with very mild epilepsy and rare seizures, a frequent point of discussion among practitioners is whether AEDs can be stopped during the first trimester, the period of organogenesis. Although this may seem reasonable for some patients, there is no supporting evidence for the safety of such an approach. Furthermore, in general, pregnancy is not a time to change AEDs to an agent the patient has not tried before, due to the risks of adverse effects such as allergy.
Folic acid
An ongoing tenet of management for all women of reproductive potential, including women with epilepsy, is to ensure adequate folic acid intake. This important vitamin supplementation clearly reduces the risk of neural tube defects and ventricular septal defects in the general population at a dose of 0.8 mg/day [36]. Since many AEDs have effects on folic-acid absorption or metabolism, it seems even more important for women receiving AEDs to receive folic-acid supplementation. However, a recommendation for the exact dose of folic acid for women receiving AEDs cannot be derived from available evidence. Thus, according to the Practice Parameter [3], women receiving AEDs should all be receiving folate supplementation at a dose of 0.4–4 mg/day [3]; usual doses are 1–2 mg/day.
Management of antiepileptic drug dosing during pregnancy
Seizure frequency during pregnancy can vary but usually remains the same as that of the pre-pregnancy months [37]. Importantly therefore, when seizures are well controlled with AEDs, they usually remain so during pregnancy [37]. However, seizure increase, decrease, or no change in frequency has been reported during pregnancy [38]. The mechanisms for increased seizure activity are multifactorial, including physiological, hormonal and metabolic changes, which can alter the pharmacokinetics of antiepileptic medications [39]. Decreased AED levels can be observed due to decreased plasma protein binding, decreased albumin concentration and increased drug clearance. Other precipitants include stress, sleep deprivation and noncompliance [21]. However, clinicians can be encouraged and somewhat reassured by the finding that women who are seizure-free on medication before pregnancy are likely to remain seizure-free while they are pregnant [37].
Due to the changes in the pharmacokinetics of antiepileptic medication as described above, it is imperative that clinicians carefully monitor seizure frequency as well as AED levels during pregnancy, and adjust the AED dose to achieve a serum level appropriate for the individual to maintain seizure freedom. Table 1 shows ranges of expected increases in clearance and decreases in both total and free (unbound) levels of the older AEDs, where available. As this table demonstrates, carbamazepine has the least alteration in metabolism during pregnancy of the AEDs listed. Levels of all AEDs, including the newer AEDs, should be checked prior to conception and during the pregnancy. Although complete information regarding changes in levels of the newer AEDs during pregnancy is not available, it can be assumed the levels will decline somewhat during pregnancy, due to the mechanisms described previously. A level at which the patient is seizure-free, if known through preconception testing, can be used as a target level upon which to base dosing.
Antiepileptic drug alterations during pregnancy.
Adapted from [44].
With some important exceptions, a reasonable approach for women with well-controlled seizures is to check AED levels at baseline (before conception) and monthly, with dose adjustments to maintain an effective level. Levels should be performed more frequently and as needed for women with recurrent seizures [34], or perhaps less frequently for women who have not had breakththrough seizures. Since phenytoin is 90–95% protein bound and the free fraction greatly increases during pregnancy, free levels of phenytoin should be followed during pregnancy, when available, rather than just total levels. Valproate is also highly protein bound, and free levels should also be obtained during pregnancy, if possible.
Lamotrigine dosing during pregnancy
More frequent monitoring of AED levels may be required if the patient is receiving lamotrigine, since serum levels have been shown to decrease by 60–90% during pregnancy [40,41]. The increased clearance of lamotrigine can occur within the first several weeks of the pregnancy, and return to baseline within 2 weeks postpartum (Figure 1). Therefore, dose adjustment of lamotrigine may be required early in the pregnancy, and dramatic dose increases of several multiples of the preconception dose may be required during the pregnancy, with a rapid downward escalation after delivery. However, as Figure 1 illustrates, some women have very little change in their lamotrigine level during pregnancy. This is likely due to the genetic polymorphism of the UDP-glucuronosyltransferase (UGT) enzymes, which are the main metabolic enzymes for lamotrigine and are induced by reproductive hormones during pregnancy. Individualized degrees of UGT induction during pregnancy result from genetic variation in UGT isoenzymes and this, in turn, has a widely disparate effect on lamotrigine metabolism. Lamotrigine levels may need to be checked every 2–4 weeks during pregnancy for many women, while for others it will be quite stable during the pregnancy.
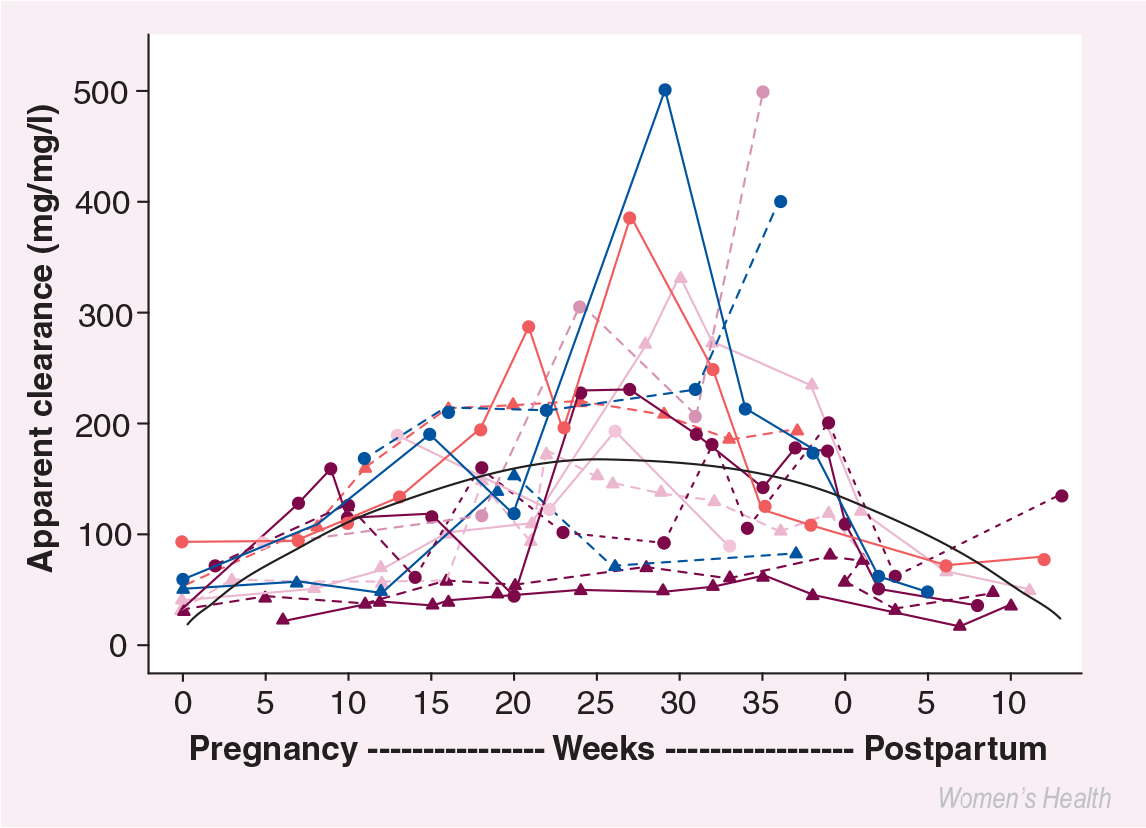
Maternal lamotrigine clearance during pregnancy.
Executive summary
Seizure control during pregnancy remains the primary goal of neurological treatment.
Worldwide evidence now indicates a clear risk of major congenital malformations (MCMs) in the offspring of women who receive valproate early in pregnancy that is at least 2-4-times the expected rate, and this antiepileptic drug (AED) should be avoided in women with epilepsy of childbearing potential whenever possible.
Evidence from the North American Pregnancy Registry indicates an increased risk of major malformations with phenobarbital that is at least twice the expected rate, confirming previous retrospective survey results, and this AED should be avoided in women of childbearing potential with epilepsy whenever possible.
Available evidence indicates that the risk of MCMs is related to AED exposure and not to seizures or epilepsy itself.
Neurocognitive outcome of the AED-exposed offspring, specifically decreased verbal intelligent quotient (IQ), is associated with in utero valproate exposure.
Women with epilepsy who become pregnant should be advised not to abruptly discontinue their AEDs, as this action may precipitate status epilepticus. Likewise, careful monitoring of seizure frequency needs to occur if there are any adjustments made in AED doses.
Five or more convulsive seizures during pregnancy are associated with decreased verbal IQ in offspring of women with epilepsy.
Monotherapy with AEDs during pregnancy poses less risk for MCMs than polytherapy.
A body of information strongly suggests that carbamazepine, lamotrigine and phenytoin have lower MCM rates than valproate and phenobarbital. Although there is some reassuring data regarding oxcarbazepine and MCM rate, the data is not as conclusive, and there is even less information available regarding the other, newer AEDs.
Folic acid of at least 1 mg/day should be given to all women with epilepsy of childbearing potential and continued throughout pregnancy.
Levels of AEDs should be obtained before pregnancy to be used as a guide for dosing adjustment during pregnancy; known therapeutic levels for an individual should be maintained.
Levels of AEDs should be obtained approximately monthly, except for lamotrigine, when levels should be followed more frequently, every 2–4 weeks.
Lamotrigine dosing may need to be increased several-fold during pregnancy to maintain a therapeutic level for the patient, and it should be adjusted downward soon after delivery; this is due to induction of lamotrigine metabolism during pregnancy.
Due to the incredible power and integrity of the human organism, despite seizures and AEDs exposure, over 90% of babies born to women with epilepsy are normal.
Vitamin K
An intramuscular injection of vitamin K 1 mg is generally given to all newborns to prevent hemorrhagic disease. Since the AEDs that induce the hepatic cytochrome P (CYP) enzymes can also induce vitamin K metabolism and therefore reduce the effectiveness of vitamin K-dependent clotting factors, oral augmentation of vitamin K by giving the mother phyton-adione 10 mg/day from 36 weeks gestation until birth has been suggested as a practice option to prevent hemorrhagic disease of the newborn [3]. Whether only women who are receiving CYP enzyme-inducing AEDs, all women receiving any AEDs or no women receiving AEDs should be prescribed oral vitamin K supplementation in the last few weeks of pregnancy is unclear, and this remains a varied practice. However, oral vitamin K supplementation is a safe intervention and, on this basis, it is often given, although the preventive benefit is unproven. There have been two studies that showed no difference in the incidence of hemorrhagic disease of the newborn in children born to mothers with epilepsy who were not supplemented with vitamin K in the last month of pregnancy.
The maternal serum testing of α-fetoprotein and ultrasound evaluations are in the purview of the obstetrician, but the neurologist should be aware of the timing and the outcome of these important milestones during the pregnancy. A partnering and open communication must occur between the neurologist, obstetrician and patient in this clinical situation.
Summary & conclusion
Although there are considerable risks related to seizures and AED therapy, it is important to be reminded that over 90% of women with epilepsy have normal, healthy children. It is imperative to counsel women in the preconception period regarding the underlying issues and risks. By instituting a few measures, such as striving for the simplest AED regimen that provides seizure control, vigilant testing of AED levels and supplementing folate, the chances of a good pregnancy outcome are maximized.
Future perspective
Mechanisms are already in place to provide answers to some of the questions raised herein; specifically, what are the risks to the fetus of AEDs? The pregnancy registries are underway throughout the world; therefore, we will have information regarding the risks of MCMs with the newer AEDs in a much shorter period of time since they were marketed than for the older AEDs. Although phenytoin has been used in the USA since 1938, reports of teratogenicity only emerged in the 1960s. There is now an ongoing study entitled the Neurocognitive Effects of Antiepileptic Drugs, which will provide information in the next 1–5 years on this aspect of AED exposure. More information needs to be obtained regarding levels of newer AEDs during pregnancy. The dramatic and unexpected effect of pregnancy on lamotrigine levels has raised the awareness of this potential safety issue for women with epilepsy who are pregnant. As the contribution of the UGT system to drug metabolism becomes more widely appreciated, the effects of UGT induction on AEDs and other drugs during pregnancy must be more fully explored.
