Abstract
Paclitaxel, a chemotherapeutic drug isolated from the Pacific yew, was approved for the treatment of metastatic breast cancer in 1994 and remains one of the most important agents in the treatment of patients with this disease. It is currently approved for the adjuvant treatment of node-positive breast cancer, administered sequentially after a standard doxorubicin-containing regimen, and for metastatic disease after failure of combination chemotherapy or relapse within 6 months of adjuvant chemotherapy. In this article, data on the pharmacology, clinical efficacy and safety of paclitaxel for the treatment of breast cancer will be reviewed.
Breast cancer remains the most common cancer among women in the USA. Worldwide, more than a million cases will be diagnosed in 2005, with more than 50% of those patients dying of their disease [1]. Despite the increasing incidence of breast cancer, mortality rates have been decreasing due to heightened awareness, early detection and improved treatment strategies. In part, this is most likely due to the increasing use and effectiveness of adjuvant and neoadjuvant chemotherapy. Although there are many agents that have contributed to this success, the taxanes are among the most useful tools in the armamentarium to combat breast cancer. At present, the taxanes have a role in metastatic breast cancer, locally advanced breast cancer and adjuvant therapy of node-positive disease.
Taxanes in breast cancer
Although the scope of this article is the role of paclitaxel in breast cancer, this drug has many other applications. Paclitaxel is approved for use in lung cancer, ovarian cancer and Kaposi's sarcoma, and is under investigation for the treatment of a wide variety of tumors. The first report of the clinical activity of paclitaxel in metastatic breast cancer was published in 1991 [2], the benefit of paclitaxel in the adjuvant setting for node-positive breast cancer was established in the late 1990s [3,4], and the role of paclitaxel in the adjuvant treatment of lymph node-negative tumors remains under investigation. Aside from paclitaxel, there are other drugs in the taxane class that have proven efficacy in breast cancer. Docetaxel, a semisynthetic taxane from the European yew (Taxus bacatta), has demonstrated a survival benefit in the adjuvant treatment of breast cancer, as well as in the metastatic setting. In the adjuvant setting, the combination of docetaxel with doxorubicin and cyclophosphamide (TAC) improved survival when compared with fluorouracil, doxorubicin and cyclophosphamide (FAC) [5]. When compared directly with paclitaxel in patients with advanced breast cancer who had progressed after treatment with an anthracycline, docetaxel had a superior response rate, time to progression and overall survival (OS) [6]. However, as paclitaxel is more effective when scheduled weekly, which is the superior taxane remains unclear. In addition, ABI-007, a nanoparticle albumin-bound (nab) paclitaxel, is a novel, Cremophor-free paclitaxel preparation with a high therapeutic index, which does not require premedication [7]. ABI-007 has demonstrated superior efficacy to paclitaxel with fewer hematologic side effects in the metastatic setting. The role of ABI-007 in the adjuvant setting has not yet been established.
Introduction to the compound
Paclitaxel, first isolated from the bark of the Pacific yew, Taxus brevifolia, was the product of a drug-development program at the National Cancer Institute in the 1960s to evaluate the antitumor activity of natural products. It was first isolated and characterized in 1971 by Wani and colleagues in North Carolina (NC, USA) [8]. In 1977, Susan Horowitz at Albert Einstein College of Medicine (NY, USA) began to study paclitaxel's mechanism of action [9]. Although it was found to have a unique mechanism of cell-cycle arrest, the compound had many problems that delayed development. First, the compound was scarce in quantity in T. brevifolia, making isolation of large quantities difficult. Second, it had aqueous instability, leading to formulation difficulties. Finally, it was found that exposure to the Cremophor diluent frequently led to anaphylactic reactions in patients [10]. It was not until 21 years after paclitaxel's initial discovery that it was first approved by the US Food and Drug Administration (FDA) for use in ovarian cancer, and later in breast cancer. Over time, paclitaxel has proven to be one of the most effective chemotherapies in the treatment of breast cancer.
Chemistry
Paclitaxel is a diterpenoid compound with the empirical formula C47H51NO14. The chemical structure of paclitaxel is shown in Figure 1. The side chain off carbon 13 is crucial to paclitaxel's antitumor activity, and modification of this side chain produces docetaxel. Paclitaxel is a highly lipophilic drug and is therefore insoluble in water. Due to its limited solubility, paclitaxel must be administered in a diluent of ethanol and polyethoxylated castor oil (Cremophor). The diluent is responsible for the high rate of hypersensitivity reactions observed with paclitaxel administration [11]. While the compound was originally isolated from the bark of the Pacific yew tree, paclitaxel is currently produced through a semisynthetic process, chemically altering 10-deacetylbaccatin-III to form paclitaxel in the laboratory [12].

Paclitaxel.
Mechanisms of action & resistance
The main antitumor effect of paclitaxel is derived from the compound binding the β-sub-unit of tubulin, causing polymerization of microtubules, preventing normal mitotic spindle formation, leading to cell-cycle arrest in the G2/M phase and triggering the mitochondrial activation of the pathways of apotosis. Paclitaxel, docetaxel and ABI-007 have been deemed the microtubule-targeted tubulin-polymerizing agents (MTPAs). By binding β-tubulin and causing microtubule polymerization, they prohibit the normal dynamic function of microtubules during mitosis [13].
The best described mechanism of resistance is the increased expression of the multidrug resistance (MDR) gene [14]. Overexpression of this gene, belonging to the ATP-binding cassette (ABC) superfamily, results in increased synthesis of membrane P-glycoprotein, which acts as an efflux pump and prevents the action of a variety of chemotherapy agents, including vinka alkaloids, anthracyclines and taxanes [15]. The family of ABC multidrug transporters, which consists of nine members, has many important roles in paclitaxel resistance. Generally, they may act by increasing drug efflux, decreasing drug uptake, recruiting drug-processing and -metabolizing enzymes, changing the activity of conjugating enzymes, altering DNA repair activity and reducing cell susceptibility to apoptosis [16]. P-glycoprotein (ABCB1), motility-related protein (MRP)-1 (ABCC1) and breast cancer-resistance protein (ABCG2) can make tumor cells resistant to various chemotherapies and decrease their oral absorption. MRP-2 (ABCC2) also limits oral availability and has been found to increase hepatobiliary and intestinal excretion [17]. Multidrug MDR protein 7 (ABCC10) has also recently been shown to serve as an efflux pump for paclitaxel [18]. Overcoming the actions of the ABC transporters is an area of ongoing research. Tubulin mutations may also play a role in drug resistance [19].
Pharmacokinetics & metabolism
Paclitaxel distribution and elimination is largely dependent on the diluent used in administration, and different formulations of paclitaxel therefore exhibit different pharmacokinetic properties. For example, the total-body clearance and volume of distribution of ABI-007 were 50% higher than paclitaxel suspended in Cremophor, suggesting that the Cremophor prevents the distribution of paclitaxel out of the circulation and into tissues [20]. The pharmacokinetic properties of paclitaxel suspended in Cremophor are outlined in Box 1. The plasma concentrations of paclitaxel decline rapidly after intravenous infusion is complete [21]. The metabolism of paclitaxel is predominantly hepatic, utilizing the cytochrome (CYP)450 enzymes, CYP2C8 and CYP3A4. Elimination is via hepatic detoxification and biliary excretion, with only a fraction of the drug in its unchanged form cleared by renal or fecal excretion [22]. Dose adjustment in patients with hepatic dysfunction is important [23]. Paclitaxel has drug interactions with anthracyclines and cisplatin, and other formulations of paclitaxel can interact with drugs that compete with its metabolism or activate a MDR pump. The drug interaction with anthracyclines causes cardiotoxicity, an important consideration, as they are often given together. When paclitaxel precedes doxorubicin, the clearance of doxorubicin is reduced and an increase in cardiotoxicity is seen. Cardiotoxicity can be minimized by administering doxorubicin first and separating the drugs by more than 1 h [24]. When paclitaxel is administered following cisplatin, the clearance of paclitaxel is decreased by 33%.
Pharmacokinetic properties of paclitaxel.
Nonlinear
95–98%
Hepatic via cytochrome P (CYP)450 enzymes CYP2C8 and CYP3A4
Primarily hepatic detoxification and biliary excretion, 5% renal
Doxorubicin and others
Clinical efficacy
Phase I studies
Phase I trials of paclitaxel demonstrated many tolerable doses and administration schedules (Table 1) [25]. The maximum tolerated dose for paclitaxel given over 3 h was 250 mg/m2, over 24 h it was 225 mg/m2, and for 96-h administration it was 140 mg/m2 [26–28]. Although the Phase I studies of paclitaxel did not clarify an optimal treatment dose, they did elucidate important knowledge regarding its administration. In early trials, hypersensitivity reactions, which could be life-threatening, were common, occurring in 18% of patients receiving paclitaxel at 15–160 mg/m2 over a 3-h infusion and 60% of patients receiving higher doses (190–230 mg/m2) over a 3-h infusion [29]. Hypersensitivity reactions were later understood to be largely a function of the Cremophor diluent and preventable with longer infusion times and premedication with steroids and histamine blockade. As the diluent is largely responsible for these reactions, it is reasonable to expect that hypersensitivity reactions would be minimized with other formulations of the drug. For example, studies of ABI-007 have shown no severe hypersensitivity reactions [30,31]. However, mild hypersensitivity reactions have been observed with ABI-007, suggesting that some of these reactions may be mediated by paclitaxel and are not entirely explainable by the Cremophor solvent.
Phase I studies of paclitaxel.
Phase II studies
Due to the lack of optimal dose to pursue in Phase II studies, paclitaxel doses varied largely in subsequent trials, with considerable controversy over the optimal dose and scheduling (Table 2). When a 3-h infusion time was compared with a 24-h infusion time, both treatment arms had similar time to progression, but more toxicity was observed with longer infusions [32]. In the registration trial, paclitaxel 135 mg/m2 was compared with 175 mg/m2, with response rates of 22 and 29%, respectively (p = not significant). These studies led to the approval of paclitaxel 175 mg/m2 over 3 h for breast cancer treatment [33]. Although subsequent studies with higher doses or longer infusion schedules suggested higher response rates, no survival benefit was apparent and toxicity was substantially increased [34–36]. A randomized Phase III study has since confirmed that increasing doses of paclitaxel are not associated with improved response rates or survival in patients with metastatic breast cancer [34]. More recent studies have evaluated the role of weekly paclitaxel, where an optimal dose appears to be 80 mg/m2 [37]. In a relatively heavily pretreated population of patients with metastatic breast cancer, 21.5% of patients responded and 41.8% had disease stabilization [37]. Therapy was well tolerated, with the most significant toxicities being a 15% incidence of grade 3 or 4 hematologic toxicity and a 9% incidence of grade 3 neurotoxicity.
Phase II studies of paclitaxel.
NA: Not available; NS: Not significant; ORR: Overall response rate; OS: Overall survival; TTP: Time to progression.
Phase III studies in metastatic breast cancer
Paclitaxel was then studied in the first-line metastatic setting against the most active current regimens. When compared with cyclophosphamide, methotrexate, 5-fluorouracil and prednisone (CMFP), paclitaxel resulted in similar response rates and survival, although this study was underpowered to detect survival differences [38]. Two studies compared paclitaxel with doxorubicin, with somewhat conflicting results. The American Intergroup study showed comparable efficacy between doxorubicin 60 mg/m2 and paclitaxel 175 mg/m2, but the European Organisation for Research and Treatment of Cancer (EORTC) study reported that doxorubicin 75 mg/m2 had higher response rates and longer time to progression than paclitaxel 200 mg/m2 [39,40]. Patient selection or unknown biases may account for these differences. A summary of Phase III studies can be seen in Table 3.
Phase III studies of paclitaxel for metastatic breast cancer.
CMFP: Cyclophosphamide, methotrexate, 5-fluorouracil and prednisone; NS: Not significant; ORR: Overall response rate; OS: Overall survival; TTP: Time to progression.
Considerable effort has continued to be invested in finding the optimal scheduling of paclitaxel, with weekly administration holding particular promise. The Cancer And Leukemia Group B (CALGB) 9840 trial compared weekly paclitaxel 80 mg/m2 over a 1-h infusion with 3-weekly paclitaxel 175mg/m2 over a 3-h infusion. The weekly administration schedule demonstrated a significant benefit in overall response rate (40 vs 28%, p = 0.017), time to progression (9 vs 5 months, p = 0.0008) and had a nonstatistically significant trend toward superior OS (24 vs 16 months, p = 017) [41]. This study suggests that paclitaxel is most effective when given weekly. However, the validity of this trial has been questioned as, in an effort to gain statistical power, it included patients treated in another CALGB study and thus was not truly randomized.
As taxanes demonstrate their efficacy in the metastatic setting, questions of the superior taxane are often raised, and a comparative efficacy and toxicity profile is shown in Table 4. Paclitaxel and docetaxel have been directly compared in the metastatic breast cancer setting among patients treated with prior anthracycline therapy [6]. In the Phase III study, paclitaxel 175 mg/m2 was compared with docetaxel 100 mg/m2 every 21 days. Overall response rates were 25% in the paclitaxel arm versus 32% in the docetaxel arm (p = 0.1), with time to progression of 3.6 versus 5.7 months (p = 0.001) and an OS of 12.7 versus 15.4 months (p = 0.03), respectively. However, the relative dose of docetaxel was higher than that of paclitaxel, and the hematologic and nonhematologic toxicities were substantially higher in the docetaxel arm.
Paclitaxel every 3 weeks compared with docetaxel, ABI-007 and weekly paclitaxel.
p: NS
Grade 4 only
Infection.
NS: Not significant; ORR: Overall response rate; OS: Overall survival; TTP: Time to progression.
ABI-007 has also demonstrated superior efficacy to paclitaxel in a Phase III study. In this study, 454 patients were assigned to ABI-007 260 mg/m2 every 3 weeks or paclitaxel 175 mg/m2 every 3 weeks. Patients in the ABI-007 arm had a superior response rate and time to tumor progression (33 vs 19%, p = 0.001; 23 vs 16.9 weeks, p = 0.006), with less neutropenia. Grade 3 sensory neuropathy occurred in 10% of patients treated with ABI-007 versus 2% of patients treated with paclitaxel. However, the neuropathy recovered quickly, in a median of 22 days, so the clinical significance is unclear [31]. Given that Phase III studies have not directly compared weekly paclitaxel with docetaxel or ABI-007, the optimal taxane remains unclear, and each remains a reasonable therapeutic option for women with metastatic breast cancer.
Another important issue in the treatment of metastatic breast cancer with paclitaxel is the role of combination versus sequential chemotherapy. In a study led by the Eastern Cooperative Oncology Group (ECOG), paclitaxel alone was compared with doxorubicin alone and with the combination of paclitaxel and doxorubicin [39]. When patients treated with single agents progressed, they crossed over to the alternate single agent. Although the patients treated with combination therapy had higher response rates, OS did not differ between the three study arms. In addition, concerns over the potential cardiotoxicity of paclitaxel and anthracycline combinations have limited the use of this therapy. More recent studies have studied paclitaxel in combination with other chemotherapeutic agents, such as gemcitabine, but whether sequential or combination therapy is superior has not been established [42–44].
Paclitaxel combinations with newer biologic agents have shown more promise. These agents used in combination with traditional chemotherapy have an application in the metastatic setting and warrant investigation in the adjuvant setting. In the pivotal trastuzumab trial for metastatic breast cancer, the combination of paclitaxel and trastuzumab produced superior response rates and OS than paclitaxel alone [45]. A recent study showed that bevacizumab, a monoclonal antibody against vascular endothelial growth factor, improves response rate, time to progression and trend towards OS in patients with metastatic or locally recurrent breast cancer when given with paclitaxel [46]. These studies suggest an important future role for paclitaxel in combination with biologic agents.
Phase III adjuvant studies
After the efficacy of paclitaxel was demonstrated in the metastatic setting, trials were initiated to determine its efficacy in the adjuvant setting. CALGB 9344 was the first randomized trial to evaluate the impact of paclitaxel on the survival of women with lymph node-positive breast cancer [4]. This study randomized patients with node-positive breast cancer to receive one of three doses of doxorubicin as part of four cycles of doxorubicin and cyclophosphamide (AC), followed by either no further therapy or four cycles of paclitaxel 175 mg/m2. The hazard reductions with the addition of paclitaxel were 17% for recurrence (p = 0.0023) and 18% for death (p = 0.0064). At 5 years, the disease-free survival (DFS) was 70% in the paclitaxel-containing arm versus 65% in the nonpaclitaxel-containing arm, and OS was 80 and 77%, respectively. The results of CALGB 9344 were first presented in May 1998 and led to the approval of paclitaxel for the adjuvant treatment of node-positive breast cancer in 1999 (Figure 2). Of note, in an unplanned subset analysis, the benefit of paclitaxel appeared to be primarily in patients with estrogen receptor (ER)-negative breast cancer. This finding was further evaluated in an analysis of over 6000 patients treated over the past 20 years in CALGB trials, which showed that adjuvant chemotherapy had a consistently greater impact on patients with ER-negative disease [47].
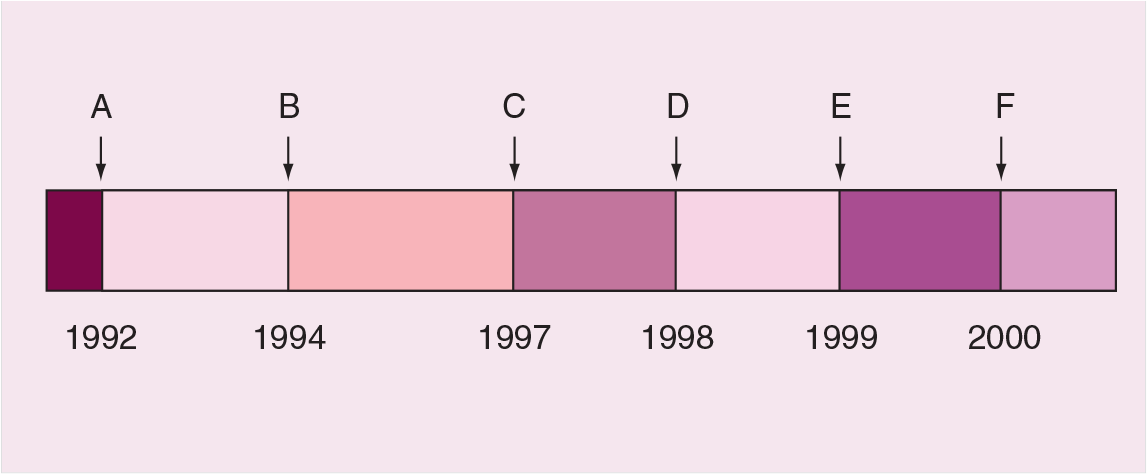
FDA approved indications for paclitaxel.
In 2000, the results of the National Surgical Adjuvant Breast and bowel Project (NSABP)-B28 trial were initially presented, which also showed a benefit for paclitaxel after a standard anthracycline regimen [3]. NSABP B-28 enrolled 3060 women with node-positive breast cancer and randomized them to treatment with four cycles of AC versus four cycles of AC followed by followed by four cycles of paclitaxel. This trial demonstrated a significant improvement in DFS: 76% in the paclitaxel arm versus 72% in the control arm. However, at 5 years follow-up, there was no significant improvement in OS. These discrepant findings may be due to a higher proportion of ER-positive patients in the NSABP study, concomitant use of tamoxifen with chemotherapy or chance. It should be noted that both the CALGB and the NSABP study had an important limitation, in that they compared patients treated with unequal duration of chemotherapy (eight vs four cycles). Therefore, the benefits observed could be due to either the addition of paclitaxel or longer duration of chemotherapy.
Weekly paclitaxel has been studied in a smaller Phase III neoadjuvant study. Patients were randomized to weekly paclitaxel for 12 weeks versus paclitaxel given every 3 weeks for four cycles [48]. In both treatment arms, paclitaxel was followed by four cycles of FAC. Patients treated on the weekly schedule had a pathologic complete response (pCR) rate of 28.2% compared with 15.7% of patients treated on the 3-weekly schedule (p = 0.02) [48]. In addition, patients treated on the weekly paclitaxel arm had increased rates of breast conservation.
There are many ongoing studies designed to answer questions regarding the optimal taxane, dosing schedule and role of combination therapies. ECOG 1199 is an ongoing adjuvant study to compare weekly versus 3-weekly paclitaxel and docetaxel in node-positive and -negative breast cancer. This study randomized patients to one of four arms after four courses of AC: weekly paclitaxel for 12 weeks, weekly docetaxel for 12 weeks, 3-weekly paclitaxel for four cycles, or 3-weekly docetaxel for four cycles. The NSABP B-28 is an ongoing trial for node-positive patients that compares three adjuvant therapies: TAC for six cycles, dose-dense AC for six cycles followed by dose-dense paclitaxel, and dose-dense AC for six cycles followed by dose-dense paclitaxel plus gemcitabine. Other studies are investigating the role of paclitaxel in node-negative breast cancer. CALGB 40101 randomized high-risk, node-negative patients to dose-dense AC for four cycles, dose-dense AC for six cycles, paclitaxel every 2 weeks for four cycles and paclitaxel every 2 weeks for six cycles. These and other ongoing studies will help to clarify the optimal use of adjuvant taxanes.
One of the most exciting developments in the adjuvant treatment of breast cancer has been the recent reports of the dramatic benefits of adjuvant trastuzumab, given concurrently with or after paclitaxel, among women with human epidermal growth factor receptor type 2 (HER2)-overexpressing breast cancer [49]. Two large, multicenter trials compared patients treated with chemotherapy with patients treated with chemotherapy plus 1 year of trastuzumab. These trials were based on the preclinical studies that demonstrated synergistic effects between taxanes and trastuzumab, as well as previous studies in the metastatic setting [45,50]. The NSABP B-31 trial randomized patients to AC followed by 3-weekly paclitaxel or the same regimen plus 52 weeks of trastuzumab. Trastuzumab was initiated with the first dose of paclitaxel. The North Central Cancer Treatment Group (NCCTG) N9831 trial compared three regimens: AC followed by weekly paclitaxel, the same regimen plus 52 weeks of trastuzumab initiated after completion of paclitaxel, and the same regimen plus 52 weeks of trastuzumab initiated concomitantly with paclitaxel. The studies were combined for analysis, with the control arm consisting of the nontrastuzumab regimens and the experimental arm combining the two regimens in which trastuzumab was administered concurrently with paclitaxel. Although these results are early, the findings were dramatic. Patients treated with trastuzumab had a 12% absolute benefit in DFS at 3 years. Trastuzumab therapy was associated with a 33% reduction in the risk of death (p = 0.015). A second analysis was performed on the patients in the N9831 study, comparing those patients treated concurrently with paclitaxel and trastuzumab with those treated sequentially. Those patients treated with concurrent paclitaxel and trastuzumab had a 36% lower risk of recurrence than those treated sequentially (p = 0.011). These studies demonstrate that adjuvant trastuzumab with chemotherapy is the new standard for women with HER2-overexpressing breast cancer and suggest that concurrent paclitaxel and trastuzumab may the the most effective regimen.
Safety & tolerability
Hypersensitivity reactions
In early clinical trials, one third of patients developed major hypersensitivity reactions to paclitaxel characterized by urticaria, hypotension, dyspnea and angioedema. Eventually, it was determined that these reactions were primarily due to the Cremophor suspension [10]. However, with the administration of steroids and histamine blockade, these hypersensitivity reactions can be prevented and now only occur in 2–5% of patients [31,48,51]. Premedication with histamine blockade and steroids is now considered the standard of care.
Neutropenia
Neutropenia is a serious dose-limiting toxicity of paclitaxel administration and appears to be directly related to dose and the length of infusion time. Patients begin to have falling white blood cell counts on days 5–7, nadir between days 7–14, and generally recover by days 15–21. Many trials have supported patients with granulocyte colony-stimulating factor (G-CSF) to prevent infectious complications related to neutropenia. The maximal tolerated dose without G-CSF support is 175–200 mg/m2 [10]. Granulocytopenia is more common with a 3-weekly dosing schedule.
Neurotoxicity
Neurosensory toxicity is another common side effect of paclitaxel, typically characterized by a burning pain or numbness and tingling of the extremities in a stocking–glove distribution [52]. Symptoms typically appear as the cumulative dose increases and are directly related to dose. These symptoms often do not resolve after paclitaxel therapy is stopped and can become chronic for months or years. Paclitaxel-induced neuropathy is difficult to treat and resistant to opiods. Ethosuximide has demonstrated reversal of mechanical allodynia/hyperalgesia and may have a role in treatment [53]. Neuromotor toxicity also occurs. Neurotoxicity is more common with the weekly dosing schedule. Generalized myalgia is also common.
Cardiotoxicity
The cardiotoxicity of paclitaxel manifests as conduction disturbances, typically effecting the Purkinje system or extracardiac autonomic control [54]. Transient asymptomatic bradycardia is reported in up to 29% of patients. Heart block is an uncommon side effect.
Others
Other side effects include reversible alopecia, asthenia/fatigue, rash, gastrointestinal toxicity, extravasation injury and radiation recall. Radiation recall is a phenomenon seen with both paclitaxel and docetaxel.
Regulatory affairs
Paclitaxel was approved for use in metastatic breast cancer in the USA in 1994, and an FDA approval timeline is represented in Figure 2. Currently, paclitaxel is approved in the USA for metastatic breast cancer, disease that has relapsed within 6 months of adjuvant therapy and adjuvant therapy of lymph node-positive breast cancer. In addition to its role in breast cancer, paclitaxel is FDA-approved for the treatment of AIDS-related Kaposi's sarcoma (second-line), non-small cell lung cancer in combination with cisplatin in patients who are not candidates for surgery or radiation, and in the treatment of ovarian cancer. Paclitaxel is now widely used throughout the world in the treatment of these tumors. Less commonly, paclitaxel is used in the treatment of other tumor types. Paclitaxel also has a role in paclitaxel-eluting stents for use in the prevention of restenosis in coronary artery disease [55].
Conclusion
Paclitaxel has demonstrated antitumor activity in metastatic breast cancer. Paclitaxel decreases the risk of cancer recurrence and improves OS in the adjuvant setting for patients with node-positive disease. While initial Phase I–II studies suggested that paclitaxel 175 mg/m2 every 3 weeks was an optimal dose, more recent studies suggest that weekly dosing at 80 mg/m2 is more effective. Other taxane formulations, such as docetaxel and ABI-007, have also demonstrated efficacy in the metastatic setting. Newer combinations of drugs with paclitaxel are improving the treatment of breast cancer further.
Future perspective
The last few decades have led to much improvement in the treatment and survival of breast cancer patients. The movement of taxanes in the treatment of breast cancer from the metastatic to the adjuvant and neoadjuvant settings has improved outcomes. Many new formulations of paclitaxel are under development, with the hope of increasing efficacy and reducing toxicity. These include an oral paclitaxel, polyglutamated paclitaxel and formulations using emulsions, stabilized emulsions with soluble polymers, liposomes and diblock copolymer micelles. ABI-007 has recently been approved in the USA, with superior response rates and decreased risk of hypersensitivity reactions. An additional class of drugs, the epothilones, is under development. The epothilones are microtubule-stabilizing agents that have activity in taxane-sensitive and -resistant disease and are currently being tested in Phase III trials for metastatic breast cancer [56]. These agents may provide an additional option for women with taxane-resistant breast cancer. Paclitaxel in combination with biologic agents, such as antibodies and small molecules, is one of the most promising areas of development. In addition, as we learn that breast cancer is not one but many diseases that each has a unique signature, patient-specific therapies may maximize benefit for susceptible patients while limiting toxicity in those who would not benefit.
Executive summary
Paclitaxel binds the β-subunit of tubulin, causing polymerization of microtubules.
It prevents normal mitotic spindle formation.
It causes cell cycle arrest in the G2/M phase.
It induces apoptosis.
Pharmakokinetic properties regarding volume of distribution and clearance are highly variable and largely dependent on the diluent used in administration.
Paclitaxel is highly lipophilic.
Its metabolism is predominantly hepatic, utilizing cytochrome (CYP)450 enzymes, CYP2C8 and CYP3A4.
The renal excretion of the unchanged drug also plays a role in the elimination of paclitaxel.
In patients with hepatic insufficiency, dose reductions are recommended.
Paclitaxel has demonstrated efficacy at 175 mg/m2 in metastatic breast cancer, recurrent breast cancer and node-positive breast cancer.
Paclitaxel has demonstrated superior efficacy using a weekly administration schedule of 80 mg/m2 in these settings, but is not approved by the US Food and Drug Administration for this dose.
Paclitaxel is associated with hypersensitivity reactions in patients without premedication, which can largely be avoided with the use of steroids and histamine blockade.
Neutropenia is a dose-limiting toxicity at the approved dose and can be minimized with shorter infusion times and granulocyte colony-stimulating factor support.
Peripheral neuropathy is a common cumulative toxicity, particularly with weekly scheduling.
Interactions with cisplatin and doxorubicin are well documented. Concerns of interactions with drugs that are similarly metabolized or activate the multidrug resistance pump exist.
Paclitaxel is approved at 175 mg/m2 every 3 weeks.
It has superior efficacy at 80 mg/m2 weekly dosing.
Premedication with steroids and histamine-blocking agents is considered the standard of care when administering paclitaxel, to decrease the incidence of hypersensitivity reactions.
