Abstract
The aim of this study was to investigate the effect of pre-irradiation with microwaves on the induction of radioadaptive response. In the 1st phase of the study, 110 male mice were divided into 8 groups. The animals in these groups were exposed/sham-exposed to microwave, low dose rate gamma or both for 5 days. On day six, the animals were exposed to a lethal dose (LD). In the 2nd phase, 30 male rats were divided into 2 groups of 15 animals. The 1st group received microwave exposure. The 2nd group (controls) received the same LD but there was no treatment before the LD. On day 5, all animals were whole-body irradiated with the LD. Statistically significant differences between the survival rate of the mice only exposed to lethal dose of gamma radiation before irradiation with a lethal dose of gamma radiation with those of the animals pre-exposed to either microwave (p=0.02), low dose rate gamma (p=0.001) or both of these physical adapting doses (p=0.003) were observed. Likewise, a statistically significant difference between survival rates of the rats in control and test groups was observed. Altogether, these experiments showed that exposure to microwave radiation may induce a significant survival adaptive response.
INTRODUCTION
Radioadaptive response or radiation-induced adaptive response is defined as the acquisition of radiation resistance against irradiation with a high radiation dose in cultured cells or organisms that had been pre-treated with a priming low radiation dose. The priming low radiation dose is usually called “adapting dose” or “conditioning dose” while the high radiation dose is called “challenge dose”. The induction of radioadaptive response was first reported by Olivieri et al. (1984) who showed that the frequency of chromatid aberrations were down to 50% less than expected after exposure to 1.5 Gy of x-rays. Many articles have demonstrated radioadaptive response in plant cells (Cortes et al. 1990), insects (Fritz-Niggli and Schaeppi-Buechi 1991), Chinese hamster V79 cells (Ikushima 1987, 1989a, 1989b), cultured human lymphocytes (Shadley and Wolff 1987; Wolff et al. 1988; Sankaranarayanan et al. 1989; Shadley and Wiencke 1989; Ghiassi-nejad et al. 2002), embryonic and HeLa cells (Ishii and Watanabe 1996), occupationally exposed persons (Barquinero et al. 1995; Gourabi and Mozdarani 1998), cultured animal lymphocytes (Wojcik and Tuschl 1990), and in vivo studies on laboratory animals (Bosi and Olivieri 1989; Cai and Liu 1990; Liu et al. 1992; Farooqi and Kesavan 1993). However, there are reports indicating lack of radioadaptive response in cultured human lymphocytes (Mortazavi et al. 2000; Mortazavi et al. 2003b; Joksic and Petrovic 2004). Furthermore, long-term follow up studies indicate that lack of radioadaptive response is not a temporary effect and, in contrast with the early reports of Olivieri and Bosi (1990), does not depend on transient physiological factors (Ikushima and Mortazavi 2000).
Although there is substantial evidence about the induction of adaptive response with low doses of ionizing radiation, doubt still persists concerning whether it is possible to induce such a response after exposure to adapting doses of non-ionizing radiations such as microwaves. Mobile phones, as wireless communication devices with drastically widespread use and increased popularity, use electromagnetic radiation in the microwave range. Although mobile phones and base stations (towers) produce the same type of radiation, phones play a much more significant role in human exposures than towers (Slesin 2000). It is reported that the output power of towers is two to three orders of magnitude greater than that of mobile phones. However, the phones are held just a few centimeters from some sensitive parts of the body such as the brain and the eyes, which causes a dramatic concern regarding the health effects of the microwaves. It is estimated that approximately 50–70% of the power output of a phone is absorbed by the user (Slesin 2000).
The aim of this study was to investigate the effects of irradiation of mice and rats with microwaves, as a priming or conditioning dose, on the induction of adaptive response to a subsequent lethal dose (LD) of gamma rays.
MATERIALS & METHODS
Animals
In the 1st phase of the study, one hundred and ten male Balb/c mice (25–30 g) were randomly divided into 8 groups of 13–15 animals. In the 2nd phase, to investigate if exposure of another animal model to a challenge dose of a common LD 50/30 can induce the same survival adaptive response, thirty male adult Wistar rats (200–250 g) were randomly divided into 2 groups of 15 animals. The animals were kept in special cages with controlled temperature, humidity and lighting. All animal experiments were considered and approved by the Animal Experimentation Ethics Committee of Shiraz University of Medical Sciences prior to commencing work.
GSM mobile phone simulator
A GSM mobile phone simulator (designed and produced at the School of Electrical and Computer Engineering, Shiraz University in collaboration with the Center for Research in Radition Sciences, Shiraz University of Medical Sciences) was used for microwave irradiation. The frequency was adjustable from 800 to 1000 MHz but in this experiment 900 MHz radiation (a wavelength of about 33.4 cm) that is similar to GSM mobile phone was used. Amplitude was digitally modulated with a 217Hz frequency and the duty cycle was 0.125 (the signal was switched on for 463 micro seconds periodically with a rate of 217 Hz. The power was adjustable from zero to over 3 Watts but we fixed it at 2 W during exposure. The signal bandwidth was 200kHz (similar to GSM mobile phone channels).
Adapting Dose (Microwave and Low Dose Rate Gamma Irradiation)
During the MW exposure, the animals were immobilized by placing their body through plastic restrainers. The distance between the antenna of the mobile simulator and animal's head was 10 cm. To expose the animals to LDRG radiation, a Cs-137 source with activity of 10 mCi was used. The dose rate for animals located at a distance of 10 cm was 2.6 mSv/h.
As summarized in Table 1, the 8 groups in the 1st phase of the study were as follows. The 1st group or controls received neither an adapting nor a challenge dose. The animals in the 2nd, 3rd and 4th groups were exposed only to adapting doses of microwave (MW) radiation, low dose rate gama radiation (LDRG) or both (MW+LDRG), respectively. The animals in the 5th, 6th and 7th groups were exposed to adapting doses of MW, LDRG or both (MW+LDRG), respectively, and then irradiated with a challenge lethal dose of gamma radiation. LDRG irradiation was scheduled for 18 hours per day for 5 days (a subtotal dose of 46.8 mSv per day and a total dose of 234 mSv). Finally, animals in the 8th group received no adapting dose but were exposed to a challenge dose of gamma radiation. The animals in the appropriate groups were exposed to microwave radiation 6 hours per day for either 5 days (1st phase of the study), or six days (2nd phase) before exposure to the lethal dose of gamma radiation. In the 2nd phase, the animals in the 1st group received microwave exposure 6 hours per day for 4 days. The animals in the 2nd group received no microwave irradiation.
Survival rates in different groups of mice 6 and 12 days after exposure/sham exposure to the lethal dose (LD 50/6) of gamma radiation in the 1st phase of the study.

Challenge Lethal Dose (LD)
On day 6, exposure of the Balb/c mice to a previously reported (Hanson et al. 1987) lethal dose (LD 50/6) of 8.8 Gy of gamma rays was performed using a radiotherapy Cobalt-60 unit (35_35 cm2 field, 48.54 cGy/min dose rate, 18.13 min irradiation time). In the 2nd phase, on day 5, all Wistar rats were whole-body irradiated with a previously reported (Lee et al. 1998) LD 50/30 of 8 Gy gamma radiation emitted by a radiotherapy Cobalt-60 source (35_35 cm2 field, 54.68 cGy/min dose rate, 14.63 min irradiation time).
Assessment of Adaptive Response
Our basic measurement was the DR (Death Rate), calculated as (1 − survival fraction). In the next step, the expected DRs in animals that were pretreated with an adapting dose of MW, LDRG, or both (MW+LDRG) were calculated as:

where, DRRF, LDRG, (MW+LDRG) is inverse survival in animals only exposed to adapting doses of MW, LDRG, or both (MW+LDRG), without subsequent irradiation with lethal dose (LD50). DRLD50 is inverse survival in animals only exposed to a challenge lethal dose without pre-irradiation with adapting doses of MW, LDRG, or both (MW+LDRG).
The coefficient of induced adaptive response, k, in each experiment was calculated as follows:

where, DRObserved is inverse survival in animals not only exposed to adapting doses of MW, low dose rate gamma (LDRG), or both (MW+LDRG), but also subsequently irradiated with a lethal dose. A k value of less than one indicates a radioadaptive response. If k = 1, it means that there is no radioadaptive (a simple additivity) effect. Finally, if k is significantly greater than 1, it means that a synergistic effect 1 was induced.
Survival Study
After gamma irradiation, the animals were monitored either for 12 days (1st phase, animals that were exposed to LD 50/6) or 30 days (2nd phase, animals that were exposed to LD 50/30). During these two intervals, the general clinical state of the living animals as well as death events were controlled and recorded daily by an expert group.
Statistical Analysis
Kaplan-Meier's survival analysis was used for assessing the survival rate in each group. A dead animal was counted as 0, whereas alive animals were defined as 1. The difference among the survival rates of the groups was evaluated by the log-rank (Mantel-Cox) test. The statistical significance of the log-rank test was considered if p < 0.05 according to chi-square test.
RESULTS
In the 1st phase of the study, as shown in Table 1, six days after exposing the mice to the LD 50/6, the survival rate in the 1st group (controls) that received neither adapting nor challenge dose was 95%. This rate in the animals of the 2nd, 3rd and 4th groups that were exposed only to adapting doses of microwave radiation, LDRG or both (MW+LDRG), was 95%, 85% and 100%, respectively. Survival rates in the animals of the 5th, 6th and 7th groups that were first exposed to conditioning doses of microwave radiation, LDRG or both (MW+LDRG) and then irradiated with an LD 50/6 dose of gamma radiation were 80%, 90% and 90%, respectively. Finally, the survival rate in the animals of the 8th group that received no adapting dose but were exposed to the LD 50/6 dose was 60%.
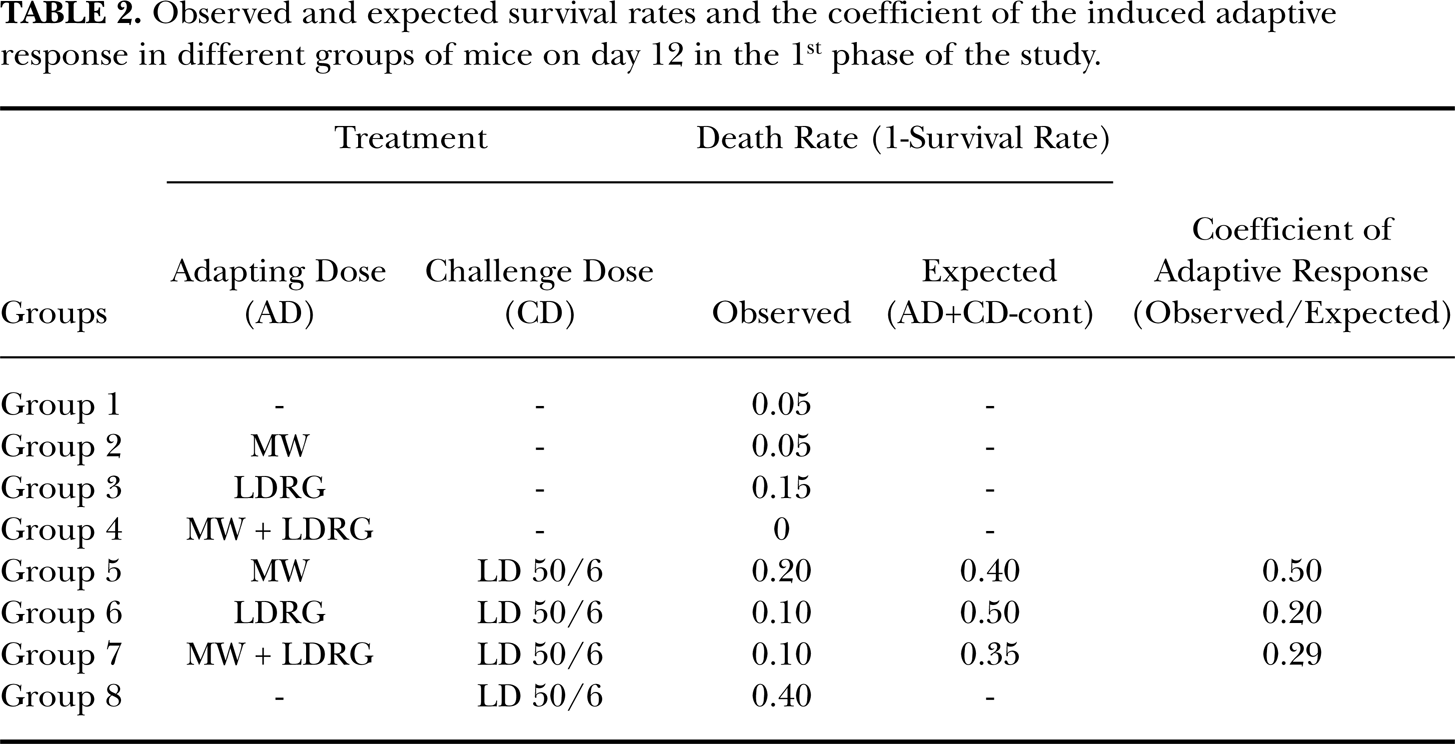
As shown in Table 2, twelve days after exposing/sham exposing the mice to the LD 50/6 dose, the survival rate in the 1st group (controls) was still 95%. This rate in the animals of the 2nd, 3rd and 4th groups was 95%, 85% and 100%, respectively. Survival rate in the animals of the 5th, 6th and 7th groups was 60%, 90% and 80%, respectively. Finally, the survival rate in animals of the 8th group was only 10%. When overall animal survival data were evaluated using the Kaplan-Meier test, a statistically significant difference among survival rates of the mice in different groups was observed. Furthermore, statistically significant differences between the survival rate of the mice only exposed to lethal dose of gamma radiation before irradiation with a lethal dose of gamma radiation with those of the animals pre-exposed to either microwave (p=0.02), low dose rate gamma (p=0.001) or both of these physical adapting doses (p=0.003) were observed. Kaplan-Meier survival plots of the mice pre-exposed/sham-exposed to microwave, low dose rate gamma or both of these adapting doses before irradiation with a lethal dose (LD) of gamma radiation is shown in Figure 1.
Observed and expected survival rates and the coefficient of the induced adaptive response in different groups of mice on day 12 in the 1st phase of the study.

Kaplan-Meier survival curves of the mice pre-exposed/sham-exposed to microwave (MW), low dose rate gamma (LDRG) or both (MW+LDRG) before irradiation with a lethal dose (LD) of gamma radiation.
In the 2nd phase of the study, 30 days after irradiation of the rats, the survival fractions for the control group (no pretreatment before LD), was 53.3%, while there was no death event (survival rate of 100%) in the animals receiving microwave radiation before the LD 50/30 (Table 3). Alteration of the survival rates of the rats pre-exposed to microwave (MW) before irradiation with a lethal dose (LD) of gamma radiation compared to those only exposed to lethal dose (No microwave pre-irradiation) is shown in Figure 2.
Survival rates in different groups of rats 10, 20 and 30 days after exposure/sham exposure to the lethal dose (LD 50/30) of gamma radiation in the 2nd phase of the study.

MW 12:3 = Microwave irradiation for 3 hours every 12 hours (6 h/day)

Alteration of the survival rates of the rats pre-exposed to microwave (MW) before irradiation with a lethal dose (LD) of gamma radiation compared to those only exposed to lethal dose (No microwave pre-irradiation).
DISCUSSION
An earlier exposure of the cells or tissues to a small dose of ionizing radiation usually increases their resistance to toxicity caused by a high dose radiation; a phenomenon that is usually called adaptive or radioadaptive response. Results obtained in both phases of this study clearly indicate the possibility of the induction of survival adaptive response as increased resistance to lethal doses of gamma rays by pre-exposing the animals to microwave radiation.
In the 1st phase of the study, 6 days after exposing the mice to the LD 50/6, the survival rate in the controls (no pretreatment, only LD) was 60% (a reasonably good level of agreement between our results with the previously published values for LD 50/6), while the survival rates in the animals pretreated with LDRG, microwave radiation and both LDRG and microwave radiation, were 90%, 80% and 90%, respectively. In the 2nd phase of the study, survival fractions, 30 days after irradiation of the animals with LD 50/30, was 100% for the animals pre-treated with microwave radiation, while it was 53.3% for the animals that had no pre-treatment before LD 50/30 (control group).
These findings are in line with the very limited recently published reports that indicated the possibility of the induction of adaptive response after pre-treatment with microwave radiation (Sannino et al. 2009; Cao et al. 2011; Sannino et al. 2011; Jiang et al. 2012; Zeni et al. 2012). Sannino et al. (2009) have previously reported that pre-exposure of peripheral blood lymphocytes collected from human volunteers to non-ionizing RF radiation (900 MHz, at a peak specific absorption rate of 10 W/kg for 20 h) increases their resistance to a challenge dose of mitomycin C (100 ng/ml at 48 h). Later, they confirmed their previous results and showed that the timing of adapting dose exposure of radiofrequency plays an important role in the process of adaptive response induction (Sannino et al. 2011). On the other hand, Chinese researchers have recently shown that pre-exposure of mice to non-ionizing 900 MHz RF induced adaptive response and thus reduced the hematopoietic tissue damage from a subsequent challenge dose of ionizing radiation (Cao et al. 2011).
Our results are in line with those reported recently by Zeni et al. (2012) who showed that when lymphocytes were pre-exposed to RF at 0.3W/kg SAR and then treated with mitomycin C, these cells showed a significant reduction in the frequency of micronuclei, compared with the cells treated with MMC alone. Jiang et al. (2012) also recently used a relatively similar method as we did previously (using the gamma radiation as the challenge dose) and showed that mice pre-exposed to RF for 3, 5, 7 and 14 days showed progressively decreased damage and were significantly different from those exposed to gamma-radiation alone. In this light, in spite of the fact that there were some basic differences between our study and the above-mentioned investigation, both studies confirm the potential of RF radiation for induction of adaptive response. The cardinal differences between our study and that conducted by Sannino et al. (2009) include the type of the challenge dose; i.e. lethal dose of gamma radiation in our study versus chemical agent of mitomycin C in the study by Sannino et al. (2009) and the endpoint; i.e. survival rate, 30 days after LD 50/30 in our study versus induction of micronuclei in the study by Sannino et al. (2009).
Some recent studies showed that radiofrequency radiation causes oxidative injury in different tissues mediated by lipid peroxidation, increased level of nitric oxide (NO) and suppression of antioxidant defense mechanism (Ozgur et al. 2010; Esmekaya et al. 2011). Feinendegen et al in their previous studies (1996, 1999), proposed that adaptive response could be induced by reactive oxygen species (ROS). Increased levels of ROS or NO have been usually observed in adapted cells (Tapio and Jacob 2007). The ROS refers to a group of molecules including peroxides and free radicals which are derived from oxygen and are highly reactive toward biomolecules (Maynard et al. 2009). ROS react with critical biomolecules such as DNA and induce oxidative stress (imbalance of prooxidants versus antioxidants) and damage in these macromolecules, multiple localized lesions such as base damage, single strand breaks (SSBs) and double strand breaks (DSBs), DNA-DNA cross links and DNA-protein cross links (Goldberg and Lehnert 2002; Marnett et al. 2003; Cejas et al. 2004; Murray et al. 2005). As indicated by some investigators (Bose Girigoswami and Ghosh 2005; Yan et al. 2006; Dimova et al. 2008), we believe that induction of adaptive response by pre-expo-sure to ionizing radiation needs a minimum level of damage that triggers this phenomenon and increases the resistance of living organisms (in vivo) or cells (in vitro) to higher levels of the same or of other sources of stress.
Our findings also confirmed that pre-irradiation of the animals with dose rates of gamma rays as low as 2.6 mSv/h (46.8 mSv/day, total dose of 234 mSv) as priming or conditioning dose significantly increases the radioresistance of animals. This finding is in line with the results reported by Elmore et al. (2008) who showed that a dose of 100 mGy at dose rates of 1–4 mGy/day was able to induce an adaptive response, while at dose rates below about 1 mGy/day the adaptive response was lost. This suggests that the lower dose threshold for adaption depends on the presence of a minimum number of lesions per unit time.
It should be noted that in the case of ionizing radiation, there are reports indicating lack of radioadaptive response in cultured lymphocytes of some individuals (Bosi and Olivieri 1989; Olivieri and Bosi 1990; Hain et al., 1992). Specially when long-term follow up studies of Mortazavi et al., which indicate that lack of radioadaptive response is not a temporary effect and does not depend on transient physiological factors (Ikushima and Mortazavi 2000; Mortazavi et al. 2000, 2003a), are taken into account, further studies are needed to clarify if the same pattern of variability exists in non-ionizing, radiation-induced adaptive response. Extending these data to humans would suggest that pre-exposure to electromagnetic radiation in the microwave region may decrease the therapeutic outcome of radiation therapy. On the other hand, this phenomenon can save the lives of humans who are exposed to life threatening doses of ionizing radiation, eg, astronauts who are selected for deep space missions.
CONCLUSIONS
The results obtained in this study clearly show that exposure of laboratory animals to microwave irradiation can induce a survival adaptive response as increased survival fraction at a specific time after exposure to a lethal dose of radiation. Although this phenomenon may have a wide variety of applications in radiation protection or radiation biology, the radioresistance induced by microwave radiations emitted by common sources of microwave radiation such as mobile phones may interfere with the outcome of any subsequent therapeutic application of radiation in radiotherapy. In this light, further studies are needed to clarify the extent of this effect and its persistence.
Footnotes
1
Synergism means the observed effect of exposure to a combination of substances is greater than the sum of the effects of each substance administered individually (e.g. 2+2 = 5).
None
ACKNOWLEDGEMENTS
This study was supported by the Center for Research in Radition Sciences (CRRS), SUMS. The authors express their sincere thanks to Dr. Imanieh, the Chancellor of Shiraz University of Medical Sciences for his critical support.
