Abstract
Hormetic morphogens are morphogens such as transforming growth factor beta (TGF-β) in mammals and auxin in plants that induce hormetic responses. For example, in vitro, TGF-β stimulates and inhibits cell proliferation at low and high concentrations respectively. I developed a model of hormetic morphogen gradient control of the morphogenesis of the fusion of bilateral aortic precursors (Anlagen) that form the aorta during development; and validated the model with findings obtained by Daucus Carota fusion experiments. Theoretically, radial concentration gradients of a hormetic morphogen can form hollow (vessels) or solid (Carota) tubular structures. In arteries, blood flow and pressure can shape mural gradients and determine wall curvature and thereby vessel diameter. As Anlagen grow they form a temporary common wall that is subsequently removed, which results in fusion of the Anlagen lumina and an aorta with a lumen diameter that accommodates the combined blood flow to the iliac arteries. Carota seedlings grown close together exhibited proximally fused root cones, serial cross-sections of which exhibited coaxial fusion patterns that closely resembled the predicted vascular fusion patterns, thus validating a role for hormesis and hormetic morphogens in the morphogenesis of the aorta and possibly the morphogenesis of other human midline structures.
INTRODUCTION
During development, left and right dorsal arteries (Anlagen) are formed by fusion of blood islands that arise in the embryonic mesenchyme. The two dorsal aortas then fuse to form the midline aorta, which in the lower abdomen bifurcates into the left and right iliac arteries. Importantly, the blood flow in the aorta above the point of iliac bifurcation must equal the combined blood flow of the two iliac arteries (Figure 1, left panel). Ozgüner and Sulak (2011) performed morphometry of the bifurcation, however, no report could be found to explain how the Anlagen fuse above the point of bifurcation and how the diameters of the abdominal aorta and the iliac arteries adjust to accommodate for changes in blood flow requirements during vascular development.
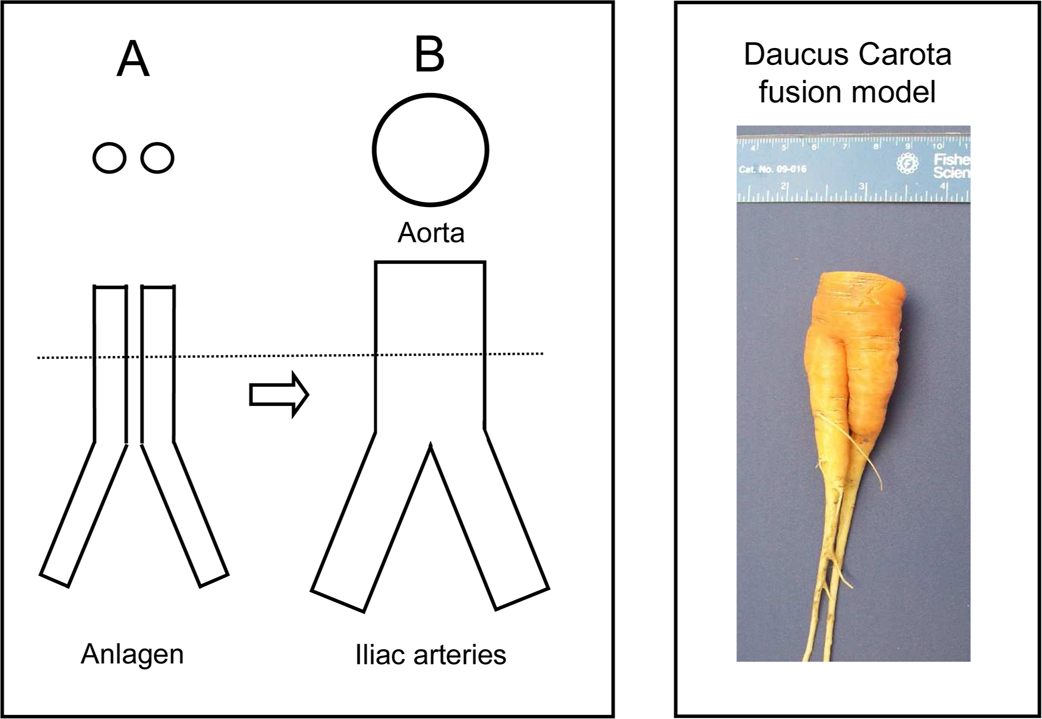
Illustration of study design; Left panel: During development, the abdominal aorta above the iliac bifurcation is formed by fusion of bilateral aortas (Anlagen). Dotted line: area of cross sections, shown above; A: cross sections of aortic Anlagen; B: Cross section of abdominal aorta above the iliac bifurcation. Right panel: C: Gross picture of Daucus Carota model of tubular fusion morphogenesis.
The objective of this study is to elucidate the process of tubular fusion morphogenesis. The approach is to apply the hormetic morphogen theory of curvature to predict the pattern of tubular fusion morphogenesis and tubular diameter adjustments during developmental formation of the abdominal aorta above the iliac bifurcation and to establish an experimental model using Daucus Carota fusion growth to simulate Anlagen fusion above the iliac bifurcation (Figure 1, right panel) to validate the theoretical predictions.
PREDICTED FUSION PATTERNS
The predicted patterns of luminal fusion morphogenesis of the aorta Anlagen above the iliac bifurcation presented in this paper are based on the proposed role of biphasic morphogens in tubulogenesis, on the idea that mitochondria play a role in the morphogen-induced growth regulation, and on the hormetic morphogen theory of curvature in which I proposed the term hormetic morphogens for biphasic morphogens such as transforming growth factor-beta (TGF-β) that induce cell hormesis in vitro (Fosslien 2002, 2008, 2009). The theory is based on reports of in vitro hormetic growth responses of human vascular type mural cells supplemented with a range of concentrations of hormetic morphogens (Battegay et al. 1990; Qiu 1995; Fosslien et al. 1997).
Retinoic acid is a hormetic morphogen that like TGF-β causes apoptosis at very high concentrations. Retinoic acid diffusing from pellets soaked in retinoic acid and placed on cultured explants of embryonic chicken skin can cause apoptosis of adjacent cells and form rings of cell disintegration and complete lack of cell growth around the pellets; the diameter of the cleared area increases with increasing concentration of the morphogen in the pellet (Chuong et al. 1992). Based upon findings by the Chuong group on the effects of retinoid acid and on the reported in vitro hormetic responses to TGF-β noted above (Battegay et al. 1990; Qiu 1995; Fosslien et al. 1997), it seems reasonable to generalize that a hormetic morphogen diffusing from a central pellet would form three circles around the pellet, a central circle of apoptosis surrounded by a ring of growth inhibition followed by a ring of growth stimulation (Figure 2, top panel).

In vitro diffusion and in vivo tubulogenesis models. Top panel: In vitro, radial gradients of hormetic morphogen diffusing from central pellet soaked in morphogen on a surface of cultured cells (A, blue) induce formation of rings of decreasing growth inhibition around pellet (B); Yellow: apoptosis, red: growth inhibition; green: growth stimulation. Bottom panel: C; Proposed tubulogenesis effects of linear growth modulation (ΔG, y-axis) along in vivo transmural hormetic morphogen gradients (x-axis) radiating from the luminal side; γ: Vascular wall thickness; D: Differential growth through vessel wall induces wall curvature; Red: Area of growth inhibition; Green: Area of growth stimulation; Vertical dotted line: Equidyne point along transmural gradient where growth inhibition equals growth stimulation resulting in no observable change in cell growth rate; Horizontal dotted lines: Hormetic morphogen concentration at equidyne point (hormetic morphogen equidyne concentration).
The expected in vivo effects of hormetic morphogen radial gradients during tubulogenesis are summarized in Figure 2, bottom panel: Transmural differential growth along the mural radial gradient of a hormetic morphogen induces a transmural linear growth gradient; it follows, that along such gradients, starting from the luminal side, mural cell growth is increasingly less inhibited until points along the radial gradients form a circle, where cell growth is not visibly affected. I refer to the morphogen concentration at the circle as the morphogen equidyne concentration, signifying that inhibiting and stimulating effects of the morphogen at the circle are balanced. From the equidyne circle towards the outside, the mural cell growth is gradually stimulated. This yin-yang effect of inhibition of mural cell growth inside the equidyne circle and stimulation of mural cell growth outside the circle causes the tissue to curve, so that radial morphogen gradients can determine mural curvature and thereby the tubular diameter.
As a logical extension of such effects of biphasic growth responses by vascular wall cells along radial transmural concentration gradients of a hormetic morphogen I suggest that modulation of the slope of such gradient by luminal blood pressure can adapt the curvature of vessel diameters to the local blood flow (Figure 3). Thus, as an example, during vascular development, an increase in blood flow and an increase in blood pressure would reduce the slope of the transmural morphogen perfusion gradients, resulting in reduced curvature. Reduced vascular wall curvature would increase the vessel diameter and lower the blood pressure until a new balance is reached (Figure 3, grey gradients).

Proposed regulation of vessel diameter. A: Hormetic morphogen concentration (y-axis) gradient (blue curve); x-axis: transmural distance from hormetic morphogen gradient origin (HMG, blue hexagon); γ: Vascular wall thickness; Grey curve: increased blood pressure reduces slope of transmural perfusion gradients. B: Transmural differential growth (ΔG) induces vessel wall curvature (C); Red: Area of growth inhibition; Green: Area of growth stimulation; Vertical dotted line: Equidyne point; Horizontal dotted lines: Hormetic morphogen equidyne concentration. D: reduced slope of transmural growth gradient reduces vascular curvature and increases vessel diameter (E). C: Flattened transmural growth gradient (ΔG1, grey); Illustration of vessel wall: Blue arrows: Radial transmural hormetic morphogen gradients; Red: Ring of growth inhibition; Green: Ring of growth stimulation; δ: vessel wall thickness.
Based upon these background considerations, it can be predicted that during the process of fusion of the aorta Anlagen above the iliac bifurcation (below the iliac bifurcation the Anlagen are to far apart to fuse), the combining parts of the vessel walls will be exposed to opposing transmural radial gradients of hormetic morphogen as illustrated in Figure 4. As the morphogen concentrations of the low ends of the opposing gradients combine (Figure 4, orange dots), the slope of the combined growth gradient in the common structure is reduced, which lessens vessel wall curvature in this area (Figure 4, orange area). However, as the Anlagen grow and expand further into each other, the combined morphogen concentration in the common wall reaches apoptotic (luminal) levels, it begins to erase the common wall between the two Anlagen; During further vascular remodeling, the Anlagen lumina completely fuse into one larger aortic lumen the diameter of which adjusts to carry the combined blood flow of the iliac arteries as outlined above (Figure 5). The sequential, temporal, developmental cross-sectional patterns of tubular fusion above the iliac bifurcation predicted by the hormetic morphogen theory of curvature are summarized in Figure 6 A-E.

Predicted hormetic fusion morphogenesis: A: Cross sections of bilateral aorta Anlagen. B: Cross sections of bilateral adjoining Anlagen; B1: opposing radial transmural gradients of hormetic morphogen (blue); C: Tubular fusion has been initiated: Orange: Approximate area of intervascular structural reorganization; C1: Overlapping gradients, combined morphogen concentrations illustrated by orange dots.
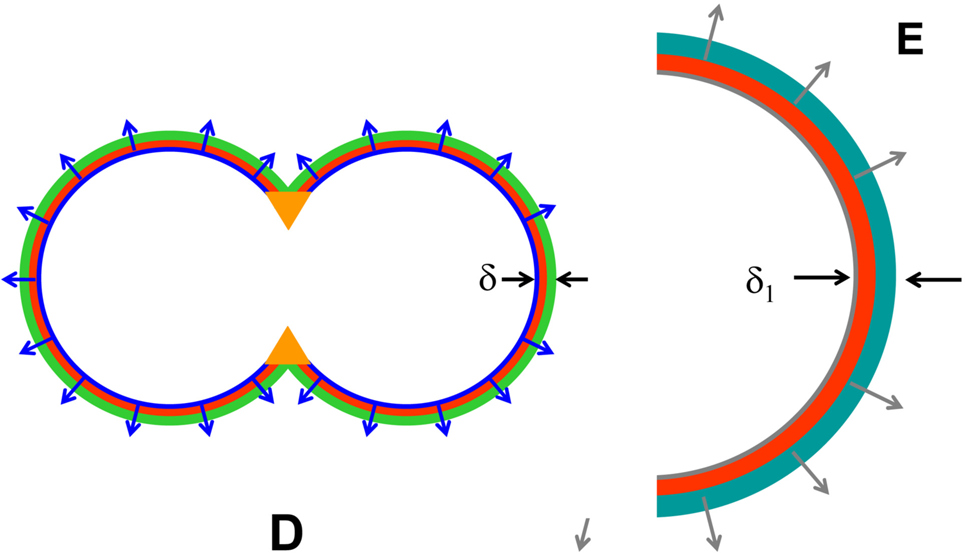
Illustration of partial (D) and complete (E) tubular fusion. D: Part of the combined wall between the Anlagen has been removed. Orange: Approximate area of further vascular reorganization. The complete reorganization of the Anlagen results in a diameter of the lumen of the aorta (E, half-cross section shown) that accommodates the combined flow to the iliac arteries below the iliac bifurcation.
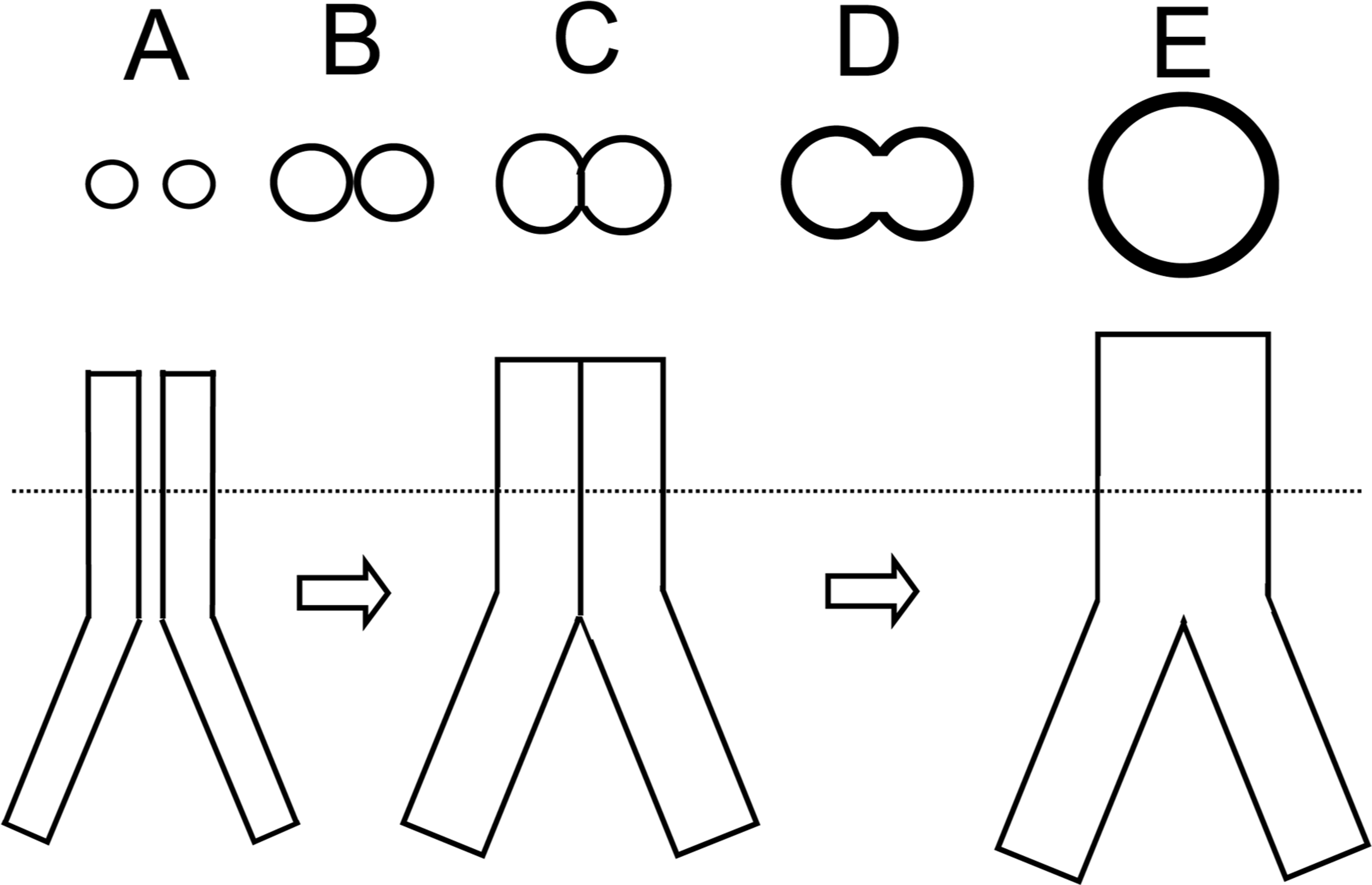
Predicted patterns of temporal cross-sections during developmental fusion of bilateral aorta Anlagen to form the abdominal aorta above the iliac bifurcation; the patterns were derived from application of the hormetic morphogen theory of curvature to tubulogenesis as shown in Figure 2 and to fusion tubulogenesis as illustrated in Figures 3–5. A-E: The letters indicating the pattern of gradual change of tubular (A-B) via partial (C-D) to complete (E) tubular fusion patterns correspond approximately to the lettering shown in Figures 3–5. Dotted line: level of cross sections.
EXPERIMENTAL FUSION
Hormesis is a general phenomenon in mammals and plants (Calabrese and Baldwin 2001a; Mattson 2007; Calabrese 2008; Calabrese and Blain 2009; Calabrese and Mattson 2011; Calabrese and Blain 2011). Calabrese and Baldwin (2001b) have suggested that concentration gradients of hormetic agents can form generalized regulatory systems in biology. This general concept is also expressed in the hormetic morphogen theory of curvature and is supported by observations that in vitro, auxin (indolacetic acid) in plants elicits hormetic growth responses (Foster et al. 1952; Pennazio 2002) and the findings that auxin gradients can induce and regulate the radius of gross curvature in plants (Bonner 1933). Guided by these reports, I selected to develop and experiment with a simple plant model of fusion morphology to validate the predicted patterns of hormetic fusion morphogenesis of the human aorta above the iliac bifurcation.
I planted Daucus Carota seeds spaced .2 – 1 cm apart, and by sampling at different times, I observed that closely spaced seedlings grown naturally gradually fused. Mature root cones exhibited proximal fusion and reached maximal diameters of up to 3 cm whereas the distal cones remained separate; serial cross-sections of a mature cone are shown in Figure 7. As illustrated in the figure, the Carota cross-sections exhibit discrete coaxial tubular structures, and significantly, their tubular fusion patterns closely resemble the patterns of vascular luminal fusion of the Anlagen to form the aorta above the iliac bifurcations as predicted by the hormetic morphogen theory of curvature (Figure 8).

Experimental tubular fusion morphogenesis (Daucus Carota model): Bottom panel: Multiple cross sections of separate (a-b), partially fused (c-d), and completely fused (e-f) Carota root cones; Top panel (a-f): Tracing of tubular geometries of the Carota sections below.

Comparison of predicted and experimental patterns of tubular fusion morphogenesis: Top panel (A-D): Predicted patterns; Bottom panel (a-f): experimental patterns.
DISCUSSION
Curvature: An association of transmural radial gradients with curved structures during vascular development was first proposed by Burke et al. (1994) who in their studies of developmental cell differentiation observed the sequential formation of mural ring structures during the ontogeny of the chick outflow tract. The investigators noted that the formation of the rings appeared to be related to radial gradients of fibronectin with highest expression near the endothelium; however, the researchers may not have considered involvement of hormetic growth responses of the mural cells as a cause of ring formation.
By comparison, the theoretical predictions in this study focus on the role of radial gradients of hormetic morphogens in inducing tissue curvature and their involvement in developmental tubular fusion. The predictions are based on the effects of transforming growth factor-β as an example of a hormetic morphogen. TGF-β is an extraordinarily versatile signaling molecule (Zhang 2011) and an essential morphogen in human vascular development (Dickson et al. 1995; Agah 2000; Goumans 2003). It can inhibit tubulogenesis in explanted heart tissue (Holifield et al. 2004). It can stimulate in vitro tubulogenesis by mammary epithelial cells forming spherical cysts or branching tubes depending upon culture conditions (Montesano et al. 2007). In addition, the hormetic responses of cultured cells supplemented with TGF-β are typical for hormetic morphogens (Fosslien 2009); they show a biphasic response of cell proliferation that within a certain range of morphogen concentration is linearly related to the log of the morphogen concentration, stimulating proliferation below and inhibiting proliferation above a morphogen concentration level, at which no change in cell proliferation is observable as compared with cells not supplemented with the morphogen (Battegay et al. 1990; Qiu 1995; Fosslien et al. 1997). Thus, within this morphogen concentration range, a transmural concentration gradient of TGF-β with high concentration on the luminal side that exponentially declines towards the abluminal side can be expected to induce a transmural linear gradient of growth modulation (Fosslien 2002).
Mitochondrial energetics: The earliest report on hormetic morphogen-induced curvature appears to be the classical study by Bonner (1933) who found that gradients of the plant hormone auxin (indolacetic acid) can induce curvature in plants. Auxin can induce dose-responses that are characteristic of hormesis, stimulating and inhibiting Avena coleoptile elongation at low and high concentrations respectively (Foster et al. 1952; Pennazio 2002), thus it meets the proposed criteria of a hormetic morphogen. Significantly, auxin at low concentrations stimulates Avena coleoptile respiration by almost 30% whereas at higher concentration it greatly inhibits respiration (Bonner 1933). More recently, it has been reported that TGF-β is a mitochondrial coupling regulator that can regulate the synthesis of adenosine triphosphate (ATP) (Law et al. 2004). Thus, the hormetic morphogen theory of curvature proposes that in vivo, gradients of hormetic morphogens such as auxin in plants and TGF-β in mammals can produce curvature via modulation of mitochondrial synthesis of adenosine triphosphate (ATP) along radial gradients of hormetic morphogens. Accordingly, I have suggested that hormetic morphogen gradient-induced transmural ATP gradients can establish transmural growth gradients that lower the rate of mural cell growth on the luminal side and increase the rate of growth on the abluminal side, which regulates the vascular wall curvature (Fosslien 2009).
Hormetic responses involved in vascular morphogenesis are not limited to cell growth; for instance, Calabrese (2001) demonstrated biphasic dose responses involved in chemotaxis, and TGF-β-induced vascular cell chemotaxis plays an important role in the vasculature (Toma and McCaffrey 2012). Yamaguchi et al. (2011) demonstrated that fibronectin concentration gradients can guide cultured peritoneal myofibroblast-like cells to migrate and enter collagen gels. It follows that the in vitro chemotaxis effect of fibronectin may explain the in vivo formation of the vascular ring structures observed by the Burke group of investigators (Burke et al. 1994). Moreover, Yang et al. (2011) found that supplementing interstitial fibroblast with 10ng/ml of TGF-β1 significantly increased fibronectin synthesis; transmural gradients of TGF-β could therefore have controlled mural cell migration via TGF-β-induced fibronectin gradients to form the mural ring structures observed by Burke et al. (1994). Also, fibronectin can modulate the effects of TGF-β by binding the morphogen and regulate the expression of TGF-β receptors, which points to fibronectin participation in TGF-β feedback signaling circuits (Kawelke et al. 2011).
Chemical and electrical cues can interact to control cell behavior (McCaig et al. 2009). I have proposed that radial transmural ATP gradients can induce transmural ionic currents and transmural electrical fields that play a role in the development of the typical coaxial arrangement of vascular mural cells (Fosslien 2010). Involvement of electrical fields in vessel wall development is supported by reports that electrical fields can induce curvature of cells (Rajnicek et al. 1994), align elongated cells perpendicular to the fields (Chuong et al. 1992), and modulate bidirectional cell migration (Sato et al. 2009). A vital role for electrical fields in curvature formation in plants has been demonstrated as well: Schrank (1950) found that shunting of inherent electrical fields using an electrolyte completely abolished curvature responses of Avena sativa coleoptiles.
As the walls of the dorsal aortas begin to fuse, the opposing transmural electrical fields of the merging walls gradually mingle. The resulting abnormal ionic currents and altered electrical fields in the common wall are likely to participate in the reorganization of the common wall during the fusion process. Thus dissolution of the common wall during fusion of the dorsal aortas to form the resulting aortic lumen probably involves reversal of cell elongation, loss of cell orientation, and disruption of cell attachments.
As noted above, in presenting my theoretical prediction about the morphogenesis of the aortic fusion above the iliac bifurcation, I believe that the shape of vascular radial transmural gradients of a hormetic morphogen is determined by the maximal morphogen concentration on the luminal side and by the slope of the mural perfusion gradients. Whether the morphogen originates in blood cells such as platelets, and is carried in the blood stream or originates in the endothelium is unclear. Still, it seems logical to expect that the slope of mural perfusion gradients will depend upon the blood pressure. Thus one may surmise that the mural curvature and thereby the diameter of the aorta that forms by fusion of the Anlagen can adjust to accommodate the combined flow to the iliac arteries: an increase in blood pressure would reduce the slope of the transmural morphogen gradients and thereby the slope of the transmural growth gradients, which would reduce mural curvature and increase the vessel diameter, thus accommodating a lager blood flow rate and normalizing blood pressure. Such a feedback loop could explain the adaptation of the size of the aortic lumen to accommodate the combined flow of the two iliac arteries; however, while it seems to be a valid mechanism, experimental validation for this view is lacking.
As briefly noted above, the bilateral dorsal aortas develop from bilateral strings of blood islands forming in the early embryonic mesenchyme. I propose the following hypothetical scenario of how hormesis and hormetic morphogen gradients may be involved in both the establishment of blood islands and their fusion to form the dorsal aortas.
First, it seems logical to assume that the locations where blood islands arise during development in the early mesenchyme are determined by bilateral location gradients of a morphogen diffusing out to the left and right from the central embryonic axis. Along these locating gradients there will be certain points in the mesenchyme where the locating morphogen provides just the right concentration to bilaterally trigger random mesenchymal cells to differentiate into blood cell precursors. These points will form bilateral lines that in part run rather parallel to the central embryonic axis, thus bilateral strings of foci of early blood precursor cell emerge in the mesenchyme.
Next, let us assume that a cluster or emerging blood cell precursor cells begin to secrete a different, hormetic morphogen that at high concentration dissolves mesenchymal cells but not the blood precursor cells. As the hormetic morphogen diffuses into the surrounding undifferentiated mesenchyme, it disintegrates the immediate adjacent mesenchymal cells, which in response release their plasma that form the early blood island plasma.
Then, the secreted hormetic morphogen forms short radial diffusion gradients that at certain points along the gradients differentiate mesenchymal cells to form endothelial cells. Because of their exposure to the hormetic morphogen radial gradients, these cells elongate perpendicular to the radial gradients and curve towards the cluster of blood cell precursors, thus forming blood islands consisting of a centrally located cluster of blood cell precursors surrounded by plasma and confined by a coaxial arrangement of endothelial cells. As adjacent blood islands expand and make contact, hormetic fusion mechanisms similar to those outlined above may then remove the cells between the blood islands in each bilateral string of islands and thus form the left and right dorsal aortas by hormetic tubular fusion.
Before blood island fusion there is no blood flow, so it seems reasonable to expect that the hormetic morphogen gradients would be formed mainly by diffusion and be relatively short and initially only reach across the endothelial cells that surround the blood precursor cells in the blood island lumen, but gradually extending further and organizing the maturing wall structure as developmental vascular morphogenesis proceeds.
Furthermore, hormetic fusion morphogenesis principles similar to those outlined in this paper may be involved in the development of other mammalian structures formed by partial or complete tubular fusion such as fusion of blood islands during vasculogenesis and morphogenesis of midline structures like the uterus and prostate. Arrested incomplete fusion may possibly results in tubular structures separated by septae or result in tissues having tubular or spherical lumina that connect to adjacent lumina by interluminal foramina.
Experimental fusion: The Daucus Carota is easy to grow and the root cones readily fuse during growth provided that the seeds are placed relatively close to each other. However, whereas in this study the Carota model corroborates the theoretical predictions of vascular fusion morphology, the model has limitations. For example, the Carota root cones lack a gross lumen. Even so, as noted above, the cross sections of the cones display discrete coaxial tubular structures and the morphology of the fusion of these tubular structures closely resembles the patterns of vascular fusion predicted by the hormetic morphogen theory of curvature. Another limitation of the experimental results in this study is that they did not include a demonstration of radial gradients of any hormetic morphogens in the Carota cross-sections. However, since Ribnicky et al. (2002) have reported that the Carota can synthesize auxin, it seems reasonable to expect that in future experiments one may be able to further validate the predicted fusion patterns by visualizing gradients of a plant hormetic morphogen such as auxin on the surfaces of the Carota cross-sections.
CONCLUSION
The morphological patterns of tubular fusion observed in the Daucus Carota experiments significantly replicate the tubular fusion patterns predicted by application of the hormetic morphogen theory of curvature to the formation of the abdominal aorta by fusion of the left and right dorsal aortas above the iliac bifurcation. These findings imply that mural cell hormesis and radial transmural gradients of hormetic morphogens can play important roles during the development of the aorta by vascular fusion of dorsal aortas above the iliac bifurcation.
