Abstract
Organisms are perpetually facing noxious insults but exhibit surprising diverse reaction patterns. Depending on the strength, frequency and quality of the stress stimuli biological systems may react with increased vitality, future stress resistance or with injury and degeneration. Whereas a multitude of such specific stress responses has been observed in diverse biological systems the underlying molecular mechanisms are mainly unknown. These knowledge restrictions urge the exploration of specific molecular signaling reactions controlling the ambivalent responses of cells and organisms to noxious effects. The adaptive responses of signaling networks to defined stress stimuli need to be investigated in a time-and dose-resolved manner in cellular and organismic models. Anticipated results are expected to significantly advance the understanding of the molecular signatures of stress responses and may also promote ongoing efforts for the effective use of the organism's preventive and regenerative potentials in modern medicine.
INTRODUCTION
“Stress” has been defined as a complex response of biological systems to potentially harmful factors, so-called stressors. These typically include environmental factors such as toxins, irradiation, dietary restriction or infectious agents. In addition, secondary responses such as the release of inflammatory mediators induced by infections or trauma, or the increased production of reactive oxygen species after physical exercise belong to this category. Stress-dependent adaptive responses of biological systems characteristically display pronounced dose dependency. Thus, in many cases low doses of a potentially harmful environmental factor can even cause beneficial effects increasing the vitality of the affected organism. This dose-dependent phenomenon characterized by low dose-induced prevention and/or regeneration processes on the one hand and damage caused by high doses on the other hand has been termed “hormesis” (Calabrese 2008).
Hormetic reactions have been first mentioned by Paracelsus and were since then observed in multiple biological systems (Calabrese 2008). One particular type of beneficial adaptive responses is known as “(pre)conditioning” or “induced tolerance”. It is well established that when treated with a potentially noxious stimulus below a specific threshold, organisms are capable of developing resistance or at least marked robustness towards the same or mechanistically related stressors. Such (pre)conditioning effects of stressors are of extraordinary medical interest.
Whereas a multiplicity of hormetic (dose-dependent) and conditioning (time-related) responses of biological systems to stressors has been described, the molecular understanding of these phenomena is clearly rudimentary. The present overview seeks to summarize and to exemplify the current knowledge on adaptive stress responses and aims to develop some idea how to shed light on the underlying molecular signaling mechanisms.

Signaling topology of adaptive stress responses.
GENERAL IDEAS
To explore the molecular dimension of hormetic responses, signaling processes controlling the ambivalent reactions of cells and organisms to noxious factors (stressors) have to be analyzed. As illustrated in Fig. 1, cells (also organisms) are exposed to stressors either directly or indirectly via specific sensor molecules. In terms of a heuristic approach the subsequent signaling reactions may be structured in three layers. The interaction of stressor and sensor in the “input layer” does not necessarily implicate a definite response of the biological system. The final effect of the stressor on the vitality of the cell will be probably predisposed by signal transducers and lastly defined in the decision layer. As a major challenge for researchers in the field the topology of the complex signaling processes in the transducer region and in the decision layer are of special importance with respect to the molecular understanding of hormetic or adaptive responses of biological systems. The complex reaction patterns of signaling mediators and networks to defined stressors has to be investigated in a dose- and time-related manner in selected animal and plant models.

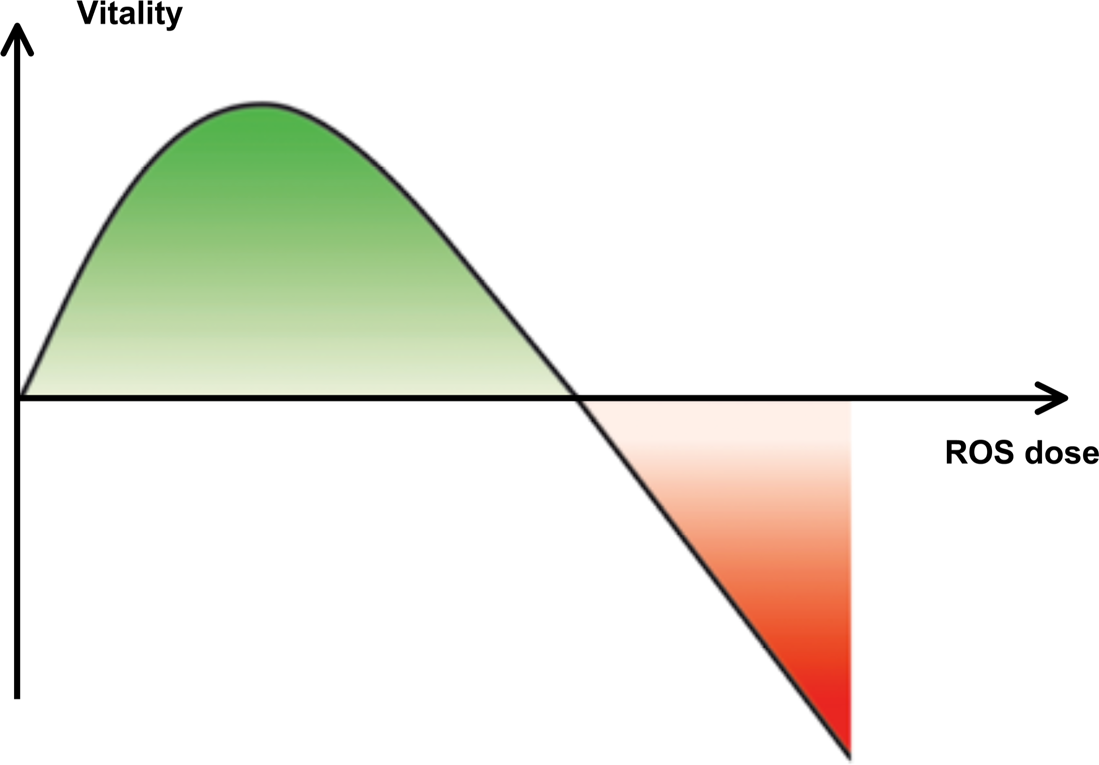
Ambivalent effects of reactive oxygene species (ROS) on the vitality of cells, organs and organisms.
Three goals seem of major relevance for the analysis of stress related signaling processes:
Identification of signaling reactions involved in adaptive stress responses. Selected animal and plant models should be used to identify and to characterize signaling mediators and signaling pathways conveying specific stress responses of cells and organisms. The current knowledge on candidate stress mediators and pathways needs to be ameliorated and deepened.
Dissection of the dose-dependent and time-related effects of stressors. Detailed investigations of hormetic (dose-dependent) and conditioning (time-related) effects of stressors on selected signaling pathways represent the main challenge for researchers of signaling processes controlling adaptive stress reactions. Typically, the bell-shaped response of cells and organisms to increasing doses of stressors has to be analyzed concerning its underlying signaling patterns. The adaptive response pattern of cells to the stressor ROS has been exemplified in Fig. 2. At low doses ROS induced signaling reactions prevail, which are able to increase the vitality of cells. At high doses damaging effects of the mediator are dominating. The molecular signature
corresponding to the maximum vitality in the ∩-shaped curve may be used as a mark for directed manipulations of the signaling mediators and pathways involved (reverse engineering). These investigations may add novel prophylactic and therapeutical concepts in medicine.
Integrative analysis of adaptive stress responses. The variety of molecular signatures of hormetic and conditioning effects investigated in developmentally diverse cellular and organismic models has to be merged in order to identify common codes of adaptive stress responses. These efforts may contribute to the global understanding of the stress induced molecular reaction patterns of living beings.
Overall it seems possible to build a hypothesis-driven mechanistic basis for the understanding of preventive ('pre-damage') and regenerative ('post-damage') responses in eukaryotic organisms. The results of these investigations may advance the current knowledge on the molecular dimension of stress responses and also promote ongoing efforts of modern medicine to exploit the organism's preventive and regenerative potentials.
Notwithstanding these promises the current view on molecular processes comprising adaptive stress responses is fragmentary, mostly restricted to selected signaling reactions induced by specific noxious factors. A small selection of such stress stimuli, which have been shown to express hormetic effects on biological systems has been summarized in Table 1. Most of these stress inducing mediators have been identified and characterized by genetic or pharmacological approaches but the molecular mechanisms governing the dose-dependent stress reactions of cells and organisms are only partially understood. As a typical example the biological effects of the stressor ROS (reactive oxygen species) seem appropriate to describe the ongoing efforts to explore the ambivalent functions of candidate mediators of adaptive stress responses.
Stress stimuli exhibiting hormetic dose-response relationships

EXAMPLE ROS
A contemporary case for ongoing efforts and interpretation problems in the molecular dissection of adaptive stress responses is the actual discussion regarding the role of ROS in the prevention versus induction of disease(s). ROS are inevitable metabolic by-products, mainly of oxidative phosphorylation processes in the respiratory chain within the mitochondria, but are also produced by NADPH-dependent oxidases in various cell types. Cellular ROS levels strongly increase during environmental challenges including continuous physical exercise or infections. Most of the current literatures in the field describe ROS and oxidative stress, as generally harmful mediators of degenerative processes (Serrano and Blasco 2007, Kadenbach et al. 2009). Specifically, ROS are considered as a major cause of cancer, type 2 diabetes, neurodegenerative processes and lastly ageing via their proposed detrimental effects on nucleic acids, proteins and lipids.
However, recent evidence suggests that ROS may also act as useful or even essential signaling molecules that promote health and extend organism lifespan. ROS may act as essential signaling mediators transducing external health-promoting stimuli to molecular mechanisms known to increase stress resistance and endogenous ROS defense. This has led to the “mitohormesis” concept (Tapia 2006, Schwartz and Sack 2008), which essentially proposes a marked beneficial effect of temporarily and/or intermittently increased mitochondrial ROS levels. The viewpoint may explain why preventive antioxidant supplementation in rodents and humans appears to even increase mortality, and why physical exercise leading to substantial increase of ROS is the most effective and attainable intervention for the promotion of human health and longevity.
Along this line a significant extension of C.elegans life span by increasing oxidative stress caused by activation of mitochondrial metabolism in states of calorie restriction has been recently observed (Schulz et al. 2007). A beneficial function of ROS has been further substantiated by work showing that health-promoting effects of physical exercise on glucose metabolism in humans are fully abolished by antioxidants (Ristow et al. 2009). Clearly low doses of ROS express ability to induce increased vitality of cells and organisms affected.
Taken together, these apparently paradoxical observations and interpretations unveil ROS as model mediators to characterize the signaling principles of ambivalent stress responses. As has been illustrated in Fig. 2 low ROS doses stimulate a series of signaling reactions, which alter the expression pattern leading to an increased vitality of the cells and organs under investigation. As shown in many biological systems the ability of ROS to inactivate protein tyrosine phosphatases leading to a net increase of tyrosine kinase activities significantly contributes to the vitalizing signaling effects of low doses of ROS (den Hertog et al. 2008). At higher ROS doses their deteriorating effects on nucleic acids, proteins and other bio-molecules predominate. Under these conditions ROS has been shown to induce apoptosis, autophagy or other cellular processes characteristically for decreased vitality of the attended organs and organisms.
CONCLUSIONS AND OUTLOOK
How may escalating doses of stressors induce alternative reactions of the cellular or organismic target? At least two principal suppositions allow mechanistical interpretation of ambivalent regulatory effects of stressors:
First, it seems possible that binding of a stressor to one specific receptor in the target cell(s) may mediate different outcomes at different doses by acting solely through that receptor. This may be true for receptors, which have been shown to be involved in regeneration as well as degeneration in the target cell. For example, the PI3K/Akt signaling pathway is known to suppress apoptosis and induce proliferation (Vanhaesebroeck et al. 2010) or, contrary, to induce apoptosis (Schulthess et al. 2009). Context-dependent feed back loops as suggested for the alternative regulatory functions of MAPK following EGF or NGF stimulation (Santos et al. 2007) could contribute to such alternative functions of signaling networks.
Second, a stressor may bind to two or more receptors, with one (high affinity) receptor mediating regenerative effects and the other (low-affinity) receptor mediating degenerative effects.
Hypothetical scheme describing two receptors exhibiting different affinity to one stressor and inducing differential effects on the vitality of the cellular target via different signaling pathways.
The co-existence of two putative receptors connected downstream to two different signaling pathways is illustrated in Fig. 3. In the example of ambivalent ROS effects described above different signaling pathways may permit the vitality stimulating effects of the stressor. The damaging effects on biomolecules of high doses of ROS have been described to induce vitality decreasing cell functions like apoptosis, autophagy or senescence.
It seems time to perform a detailed analysis of the adaptive responses of signaling networks to quantitative changes of noxious stimuli. In-depth investigations about the function, regulation and interconnection of central signaling pathways of adaptive stress responses will expand our current knowledge about the molecular dimension of these processes.
As a central focus these studies may intend to unveil novel mediators of regenerative effects of stressors or of stress resistance. Corresponding molecular insights in hormetic signaling processes will promote engineering attempts of adaptive stress responses from the red to the green area of the hormesis curve.
These directed manipulations of specific signaling pathways may contribute to future strategies of prevention or therapy of many diseases.
