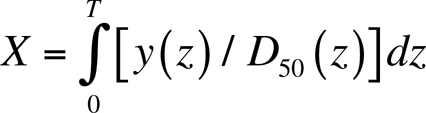Abstract
A radiological emergency exists at the Fukushima Daiichi (Fukushima I) nuclear power plant in Japan as a result of the March 11, 2011 magnitude 9.0 earthquake and the massive tsunami that arrived later. News media misinformation related to the emergency triggered enormous social fear worldwide of the radioactivity that is being released from damaged fuel rods. The heroic recovery workers are a major concern because they are being exposed to mostly gamma radiation during their work shifts and life-threatening damage to the radiosensitive bone marrow could occur over time. This paper presents a way in which the bone marrow equivalent dose (in millisieverts), as estimated per work shift, could be used along with the hazard function model previously developed for radiological risk assessment to repeatedly check for potential life-threatening harm (hematopoietic system damage) to workers. Three categories of radiation hazard indication are proposed: 1, life-threatening damage unlikely; 2, life-threatening damage possible; 3, life-threatening damage likely. Categories 2 and 3 would be avoided if the whole body effective dose did not exceed the annual effective dose limit of 250 mSv. For down-wind populations, hormetic effects (activated natural protective processes) are much more likely than are deleterious effects.
INTRODUCTION
On March 11, 2011, a magnitude 9.0 earthquake struck near Sendai, Japan and was followed one hour afterwards by a massive tsunami. The earthquake was the largest in the country's history, causing massive property damage (including the Fukushima nuclear power facility) and the loss of thousands of lives. Although the six nuclear reactor units at the Fukushima Daiichi (Fukushima I) power plant apparently withstood the 9.0 magnitude earthquake, the tsunami carried away the fuel tanks for the power plant's emergency diesel generators. The generators provided back-up power for the pumps used for removing heat from the radioactive fuel.
Three of the reactors (Units 1, 2, and 3) were operating and they automatically shut-down when the earthquake occurred, as designed. The other reactors (Units 4, 5, and 6) were already shut down for routine maintenance. Thus, after the earthquake all six reactors were in shut-down mode.
The earthquake disabled normal electrical power and the tsunami incapacitated backup electrical power. This created a problem for continued cooling of the fuel inside each reactor pressure vessel and in the associated used-fuel pool (water) located in its’ reactor building. The pressure vessels serve to prevent the release of fission-product radionuclides to the environment. Some of the water boiled away and some of the fuel was damaged. Unfortunately, the loss of electricity also led to production of hydrogen (due to the radiolysis of water and possibly the chemical reaction of water with the hot zirconium fuel cladding). Several of the reactor buildings were damaged when hydrogen ignited causing explosions inside and radioactivity (fission-product radionuclides) was released to the environment.
The events at Units 1, 2, and 3 were rated as Level 7 (major release of radioactive material with widespread health and environmental effects requiring implementation of planned and extended countermeasures) on the International Nuclear Event Scale and those at Unit 4 were rated as Level 3 (serious incident).
There were three casualties related to the earthquake and tsunami. A crane operator was killed during the earthquake and two workers were found dead in one of the turbine buildings after the tsunami. It is presumed that the two workers drowned during the tsunami. Of the approximate 300 people at the Fukushima Daiichi site, 28 have accumulated effective radiation doses of more than 100 millisieverts (mSv) as of April 15 (IAEA 2011). The workers wear protective clothing and equipment (including respirators) as their work continues to bring the reactors and used fuel pools toward a stable cooling condition which would help mitigate the release of radioactive materials.
A target of about three months was set on April 17, 2011 to reduce the radiation exposure rate (Step 1) at the Fukushima Daiichi plant. Step 2 involves controlling the release of radioactivity and significantly suppressing the radiation exposure level and is expected to be achieved three to six months after completion of Step 1.
During each work shift, the workers are exposed over their total bodies to penetrating gamma rays. Their effective doses are monitored and recorded in units of millisievert. The worker effective dose limit was recently raised to 250 mSv in one year.
Recent air measurements in the area to the east of the No. 3 reactor and building had levels up to 70 mGy per hour. The indicated building was damaged by a hydrogen explosion on March 14, 2011. Uniform exposure of the whole body for four hours to gamma rays at the indicated rate would exceed the 250 mSv limit (Katori and Tsuboya 2011).
For assessing potential harm from radiation deterministic effects such as potential life-threatening damage to the radiosensitive bone marrow, estimates of the average radiation absorbed dose in units such as gray (Gy) to the target organ are needed. Because the radiation weighting factor for gamma rays is 1, an equivalent dose in millisieverts to the bone marrow is the same numerical value as the bone marrow absorbed dose in milligrays (mGy).
This paper provides risk-assessment tools for evaluating the potential for life-threatening harm to emergency workers at Fukushima Daiichi facility from the cumulative gamma-ray exposures over multiple work shifts. The tools are based on the hazard-function (HF) model (Scott et al. 1988, 1998; Scott and Hahn 1989; NRPB 1996; Scott 2004, 2005, 2009; IAEA 2005), which was developed for assessing the risk of harm from deterministic effects (i.e., threshold effects) of high-level radiation exposure and allows for taking into consideration variation in the average dose rate to bone marrow over different work shifts. The model is based on extensive review of available data (animals and humans) and has been tested in laboratory animal models and subjected to high-level peer review (Scott and Hahn 1989). The focus on bone marrow is because its severe damage (massive stem cell killing) poses the largest life-threatening risk for circumstances of total-body exposure to gamma rays. Avoidance of life-threatening damage to bone marrow would be expected to also avoid life-threatening damage to other more radioresistant organs.
HAZARD FUNCTION MODEL AND NORMALIZED DOSE
The hazard function (HF) model has been used to characterize risk for deterministic health effects from complex irradiation patterns that arise during radiological emergencies (IAEA 2005). Here the focus is on external whole-body exposure of humans to gamma rays during the current Japanese radiological emergency.
The simplified form of the HF model described below relates specifically to life-threatening damage to bone marrow. The more general form (Scott and Hahn 1989, NRPB 1996, IAEA 2005) addresses damage to multiple organs with a focus on evaluating the risk of lethal damage for competing modes of lethality (e.g., hematopoietic, gastrointestinal, and pulmonary).
With the form of the HF model as presented here, the risk Rhem (X) for life-threatening damage to bone marrow (hematopoietic damage) is related to the corresponding hazard function, Hhem (X), through the equation:
The independent variable X is called the normalized dose and has units of LD50 (i.e., median lethal dose). Thus, X=1 corresponds to an LD50 absorbed radiation dose. Hhem (X) is given by the following equation (Scott and Hahn, 1989):
The normalized dose X is given by the following:
The parameter T is the total exposure time. The variable y(z) is the average (spatially) dose rate at exposure time t=z, and V is the shape parameter. D 50(y(z)) as used here is the dose-rate-dependent LD50 expressed as a bone marrow absorbed dose (usually expressed in Gy). Although the integral in Equation 3 is evaluated over the total exposure time T, it can be partitioned to cover each daily work shift, with y(z) replaced by the average dose rate yj to bone marrow for the jth (j=1,2,…,n) work shift. In this case the cumulative normalized dose X is the sum of the respective, shift-specific normalized doses X 1, X 2, …, and X n (Scott et al. 1998). The indicated partitioning over work shifts is valid because normalized doses between work shifts are zero.
The dose-rate-dependent function D 50(y), evaluated separately for each work shift (i.e., y = yj ), is based on the following empirical equation with a fixed dose rate y (Scott and Hahn 1989; NRPB 1996):
As can be seen from the construct of Equation 4, D 50(y) decreases as y increases and approaches an asymptotic value θ ∞ at very high dose rate. This feature of the D 50(y) as it relates to continuous exposure has been called the Ainsworth phenomenon (Scott 2009), in honor of John Ainsworth who first reported the feature (Ainsworth et al. 1964). Prior to the realization of the Ainsworth phenomenon, it was widely thought that D 50(y) would approach zero at very high dose rates.
Simulated values for D 50 vs. fixed dose rate for different mammalian species are presented in Figure 1, based on a previous publication in this journal (Scott 2009). The term (θ 1/y) accounts for the steep rise in D 50(y) above θ ∞ as dose rate decreases to very low values. For life-threatening damage to the bone marrow under circumstances where no medical countermeasures (mitigating medical procedures) are employed, the central estimate of the indicated HF model parameters for humans are θ ∞ = 3 Gy and θ 1 = 0.07 Gy2/h when the dose rate is in Gy/h (Scott 2004). Subjective lower and upper bounds for θ ∞ are 2.5 and 3.5 Gy, respectively. Subjective lower and upper bounds for θ 1 are 0.06 and 0.08 Gy2/h, respectively (Scott and Hahn 1989).

Predicted median lethal dose (D 50(y)) for the hematopoietic mode of death for different mammals (Sprague-Dawley rats, white non-inbred mice, dogs, swine, goats, sheep, and humans) as a function of the external low-LET radiation dose rate y to bone marrow in Gy/h. Dose rate is presented as a categorical variable and is not aligned to tick marks.
The indicated value of 3 Gy for θ ∞ is less than the implied corresponding values of 4 and 5 Sv sometimes stated in the news media as the LD50 for humans. Firstly, the value of 3 Gy (for bone marrow) is based on extensive research (Scott and Hahn 1989) and is consistent with the value found for people residing inside reinforced concrete buildings in Nagasaki, Japan after the atomic bombing (Levin et al., 1992). Secondly, the LD50 should not be stated in units of sievert, especially when derived from data for mixed high- and low-LET radiations or high-LET radiation only because the radiation weighting factors used to obtain equivalent dose in sieverts are inappropriate for deterministic effects (NRPB, 1996; IAEA 2005).
The work-shift-specific normalized doses X 1, X 2, …, and X n for a given worker can be evaluated based on estimates of the shift-specific average radiation dose to bone marrow and the length of the work shift in hours. If Y 1, Y 2, …, and Y n are respective estimates of the gamma-ray doses to bone marrow (which are here approximated by corresponding bone marrow equivalent doses evaluated with a radiation weighting factor of 1), then dividing bone marrow doses by the total time on each corresponding shift gives estimates of the average dose rates y 1, y 2, …, and yn to bone marrow over each shift for a given individual. The normalized dose Xj to bone marrow for the jth shift can then be evaluated using the following:
Table 1 gives central estimates of Hhem (X) and Rhem (X) as a function of the cumulative normalized dose X for life-threatening injury to the hematopoietic system after whole-body gamma-ray exposure. Column 4 of the table gives radiation hazard (RADHAZ) indications for gamma-irradiated individuals that fall into the following three RADHAZ categories that depend on the value for Hhem (X):
0 ≤ Hhem (X) < 0.003: life-threatening damage unlikely (RADHAZ low);
0.003 ≤ Hhem (X) < 0.1: life-threatening damage possible (RADHAZ moderate);
Hhem (X) ≥ 0.1: life-threatening damage likely (RADHAZ high).
RADHAZ indication for monitoring for potential life-threatening hematopoietic damage caused by whole-body, gamma-ray exposure. a

X, cumulative normalized dose; Hhem (X), HF for life-threatening hematopoietic damage; Rhem (X), risk of life-threatening hematopoietic damage. The lower bound on the threshold is X = 0.4 based on 10,000 Monte Carlo calculations with triangular distributions (lower bound, central estimate, upper bound) for the threshold absorbed dose (1.2 Gy, 1.5 Gy, 1.8 Gy) and for the LD50 (2.5 Gy, 3 Gy, 3.5 Gy) for high-rate exposure to gamma rays (Scott and Hahn 1989).
The subjective RADHAZ indications apply irrespective of whether a person received medical countermeasures (i.e., medical support) or not. However, application of medical countermeasures for external gamma-ray exposure will impact on the value for the cumulative normalized dose X (Scott 2005). This can be addressed by dividing X by the appropriate protection factor (PF) (Scott 2005) associated with the countermeasures employed [e.g., administration of a radioprotector and/or providing supportive medical treatment (IAEA 1988; AFRRI 1997)]. The new value of X would then be used to evaluate Hhem (X) and Rhem (X). This approach is equivalent to increasing the D 50 (Equation 4) by a fixed PF.
Table 2 shows the potential significant impact that application of medical countermeasures could have on the resultant RADHAZ indication that is assigned. Results are for a hypothetical set of countermeasures leading to a PF of 2. Each value of X in Table 1 was divided by 2 and Hhem (X) was recalculated (Table 2). As implied, providing medical support to heavily irradiated emergency workers would be expected to have significant life-saving benefits, as is already well known.
Expected impact of medical countermeasures on results in Table 1 when PF=2.

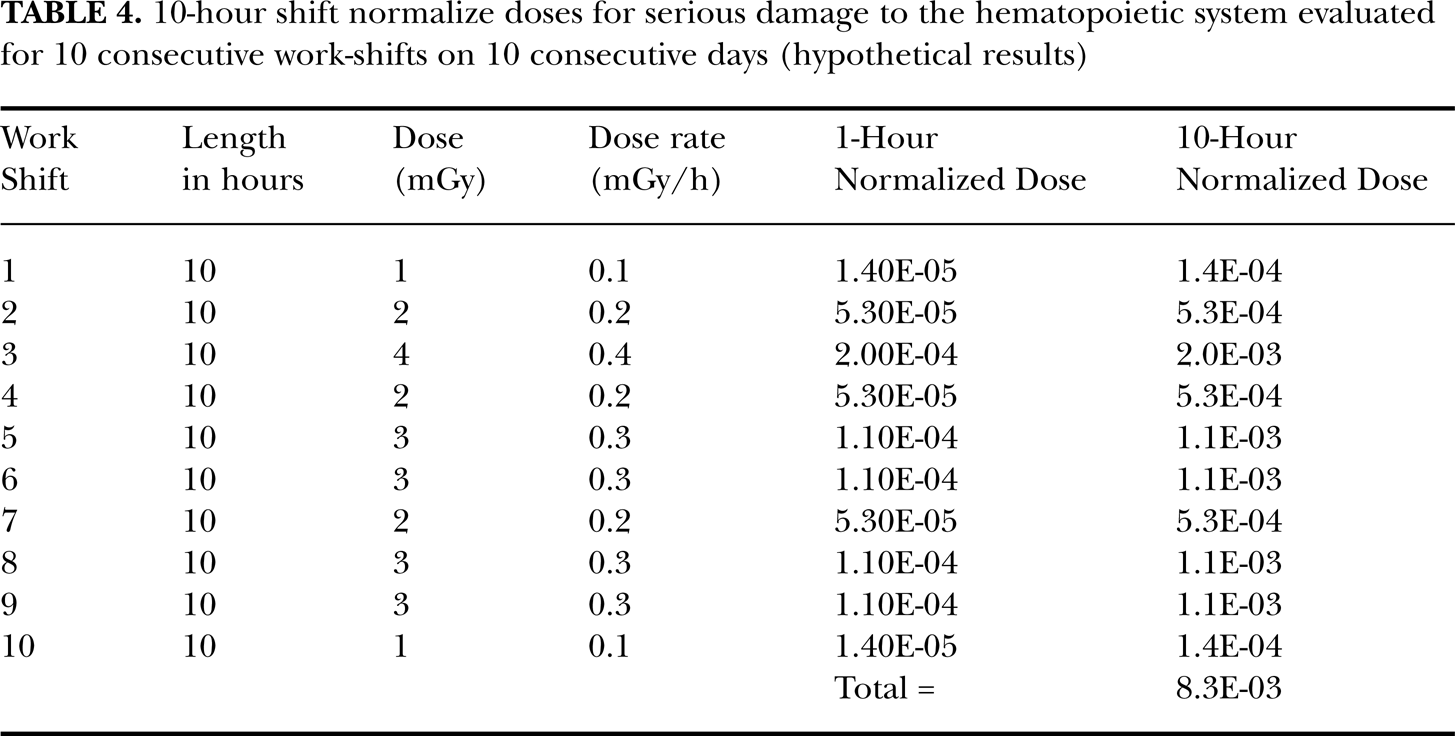
Table 3 gives estimates (central) of Xj for a given value of yj for a 1-hour-exposure shift, which facilitates application of the risk assessment framework presented here. The values of yj range from 1 mGy/h to100 mGy/h. As an example of application of Table 3, consider the hypothetical case where a worker has undergone 10 consecutive days of work at the Fukushima Daiichi plant, working 10-h shifts per day. For this example the 10-shift, bone marrow equivalent doses (based on radiation weighting factor =1) are given by Y 1 * = 1 mSv, Y 2 * = 2 mSv, Y 3 * = 4 mSv, Y 4 *= 2 mSv, Y 5 * = 3 mSv, Y 6 * = 3 mSv, Y 7 * = 2 mSv, Y 8 * = 3 mSv, Y 9 * = 3 mSv, Y 10 * = 1 (total = 24 mSv). These equivalent doses Yj * are used as estimates of the average absorbed radiation doses Yj to bone marrow. Thus, changing the unit to mGy and dividing by 10 h (length of each work shift) yields the corresponding values for the work-shift-specific average dose rates to bone marrow: y 1 = 0.1 mGy/h, y 2 = 0.2 mGy/h, y 3 = 0.4 mGy/h, y 4 = 0.2 mGy/h, y 5 = 0.3 mGy/h, y 6 = 0.3 mGy/h, y 7 = 0.2 mGy/h, y 8 = 0.3 mGy/h, y 9 = 0.3 mGy/h, and y 10 = 0.1 mGy/h.
Central estimates of normalized dose hourly increment Xj for life-threatening injury from the hematopoietic mode as a function of the hourly-averaged gamma-ray dose rate to bone marrow in mSv/h when no medical countermeasures are applied. a

For hourly dose rates < 1 mGy/h, Xj can be calculated based on Equation 5.
Results for this hypothetical exposure scenario where no medical countermeasures are provided are presented in Table 4. Both 1-h and 10-h normalized doses X are presented with the 10-h results being 10 times the 1-h results. Thus, for a 5-h shift, the results would have been 5 times the 1-h results. The calculated cumulative normalized dose X for the ten 10-h shifts was 8.3×10−3, which is below the threshold lower bound of 0.4 (Table 1) for life-threatening damage. The bound corresponds to the 10th percentile of the uncertainty distribution for the threshold obtained via Monte Carlo calculations (10,000 realizations) based on triangular distributions for the threshold and LD50 doses (see footnote in Table 1). For X = 8.3×10−3, the corresponding value for Hhem (X) is therefore 0 (Table 1), although a meaningless value of ln(2)[8.3×10−3]6 which is 2.3×10−13 (rounded) can be calculated. For the indicated value for X, the RADHAZ indication is “life-threatening damage unlikely.” Should this same individual work additional shifts that bring the cumulative normalize dose up to a value of X = 0.75, then Hhem (X) would be ln(2)[0.756] which equals 0.12 (rounded). The RADHAZ indication would then be “life-threatening damage likely.” Such a worker should not be expected to continue to work in the gamma-ray field and would likely need some form of medical support. Normally a worker would not accumulate a normalized dose as high as 0.75 in a given year (due to the effective dose limit of 250 mSv) as is the case at the Fukushima Daiichi facility. This is because exposure at the current already raised annual effective dose limit of 250 mSv (or a somewhat higher new limit) would guarantee that such a large normalized dose would not occur.
10-hour shift normalize doses for serious damage to the hematopoietic system evaluated for 10 consecutive work-shifts on 10 consecutive days (hypothetical results)
Should medical support (i.e., medical countermeasures) with a PF=2 be given to a worker with X=0.75 before the medical support is provided, then X would need to be reduced by a factor of 2, leading to X = 0.375. In this case, Hhem (X) would be calculated to be 0 since X is below the threshold lower bound of 0.4 (Table 1) for life-threatening damage. The RADHAZ indication would then be modified to be “life-threatening damage unlikely.”
An additional example involves one work shift for 10 hours when the average equivalent gamma-ray dose rate to the bone marrow is 25 mSv/h. In this case assuming uniform whole-body exposure the worker would have received their current annual effective dose limit in the single work shift. This is because a uniform whole-body dose of 250 mGy would also represent an effective dose of 250 mSv, a consequence of the definition of the effective dose (i.e., the dose which equates the cancer risk for the actual exposure to the same risk had the total body been uniformly irradiated). The normalized dose X for 1-h exposure from Table 3 is 4.3×10−3, which is below the threshold lower bound for life-threatening damage. For a 10-h exposure it would be 10-fold larger at 4.3×10−2, which is also below the threshold lower bound. The corresponding value for Hhem (X) is therefore 0. The RADHAZ evaluation indication would be “life-threatening damage unlikely”. Thus, even with the raised annual effective dose limit of 250 mSv, one would not expect any life-threatening damage to bone marrow at the indicated limit. With an additional 9 such exposures (same average equivalent dose rate), the value for X would increase to 0.43 and Hhem (X) would be expected to climb to 4.4×10−3 and in which case the RADHAZ indication would be “life-threatening damage possible”. With medical countermeasures applied and providing a PF of 2, the value for X would decrease to 0.215 in which case the RADHAZ indication would again be “life-threatening damage unlikely.”
The above results relate to radiation deterministic effects. Radiogenic cancer and reproductive effects are other possible risks that need to be assessed for persons receiving moderate to high radiation doses. However, a recent study revealed large thresholds for radiogenic cancer (Tanooka 2011). The threshold also increases as dose rate decreases. For low doses and dose rates, hormetic (beneficial) effects may occur for doses up to and exceeding 250 mSv (Scott and Di Palma 2007) and are expected to be orders of magnitude more likely than radiogenetic cancer (Scott 2008). This may be especially relevant for down-wind populations relative to the Fukushima Daiichi power plant, including persons residing in Hawaii and on the West Coast of the United States. For a point of reference, each second of our lives we received more than 1 million natural radiation hits to our body (Feinendegen et al. 2011). By age 20 (years), the cumulative number of natural radiation hits each person receives exceeds 630 trillion and there is no evidence of a positive association between natural radiation exposure and cancer. In fact the reported associations are negative (i.e., radiation hormesis) implicating likely benefit rather than harm (Scott and Di Palma 2007).
Alarmists claiming that a single radiation hit to a citizen in the U.S. from a stray radioactive atom coming across the Pacific Ocean from Japan could cause cancer have no credibility. Even so, many persons across the U.S. rushed to purchase potassium iodide tablets to supposedly protect from phantom risks related to the Fukushima incident. Unfortunately, side effects from potassium iodide are similar to symptoms from large radiation doses (e.g., nausea, vomiting, darkened stools). Potassium iodide can also cause an irregular heart beat. Thus, radiation phobia related casualties (e.g., potassium-iodide-related) are far more likely than any casualty directly related to radiation exposure from a stray radioactive atom coming from Japan. Following the Chernobyl accident in 1986, more than 100,000 wanted pregnancies were aborted at the advice of misinformed physicians (Ketchum 1987). Hopefully such a tragedy will not be repeated in Japan and elsewhere as a result of the Fukushima Daiichi radiological situation.
The current framework for evaluating the potential health consequences to down-wind populations of the Fukushima Daiichi plant is based on the invalid linear-no-threshold (LNT) hypothesis: any amount of radiation can cause cancer no matter how small. Very different implications for managing the Fukushima emergency arise from the LNT hypothesis and proven low-dose radiation hormesis (Scott 2011):
Different choices of the evacuation zone radius around plant.
Different policies about when to use bottle water.
Different policies about when to ban use of radionuclide contaminated foods and milk.
Different focus of news media reporting of the emergency (e.g., local health risks [hormetic model] vs. global health risks [LNT hypothesis])
Different ways of controlling radiation-phobia-related casualties (e.g., abortions, potassium-iodide-usage-related harm, stress-related psychological effects).
CONCLUSIONS
Recovery workers at the Fukushima Daiichi nuclear facility that are exposed to radiation doses up to the annual effective dose limit of 250 mSv and somewhat beyond are not likely to have any life-threatening damage to their bone marrow. Much higher doses would be expected to be needed to pose a serious threat from radiation-induced deterministic effects to the radiosensitive bone marrow. If a risk of harm exists, it would be expected to relate to possible cancer induction and/or reproductive effects. For effective doses up to and somewhat exceeding 250 mSv, hormetic effects are orders of magnitude more likely than cancer induction.
Footnotes
ACKNOWLEDGMENTS
This research was supported by the Office of Science (BER), U.S. Department of Energy, Grant No. DE-FG02-09ER64783. This paper is dedicated to the heroic and courageous recovery workers at the Fukushima Daiichi nuclear power facility in Japan. I am grateful to the journal reviewers for their helpful comments and to Steven Shinnick for timely editorial assistance.