Abstract
Many farmers in developing countries protect their crops with oxytetracycline and fertilize their farmlands with manure from animals that received this drug as growth promoter. In this study, a tropical soil was exposed to 0.1 mg kg−1, 1 mg kg−1, and 10 mg kg−1 of oxytetracycline for 22 days to evaluate whether this antibiotic alters the capacity of a soil microbial community to metabolize 31 carbon sources. The communities exposed to 1 and 10 mg kg−1 of oxytetracycline exhibited reduced catabolic activities for 3 and 6 substrates, respectively. In contrast, the communities exposed to 0.1 mg kg−1 of oxytetracycline showed higher catabolic activities than the controls and the other two treatments for 19 substrates. These data reveal a hormetic response at the community level not previously described for soil bacteria and oxytetracycline.
INTRODUCTION
The microorganisms of the soil contribute to maintaining life by catalyzing reactions that globally impact carbon, nitrogen and sulfur cycles (Chen et al. 2003). In addition, this abundant group of living beings plays an important role in agriculture by influencing soil structure, the formation and breakdown of humus, the availability of nutrients, the breakdown of organic waste and the activity of pathogens (van Elsas et al. 2006; Steinberg et al. 2008; Berg 2009).
Over the past several years, there has been much documentation on the damaging effect of excessive urbanization, (Doichinova et al. 2006), drought (Sardans et al. 2008), soil erosion (Lal 1998), deforestation (Nourbakhsh 2007), heavy metal contamination (Oliveira and Pampulha 2006) or pesticide use (Spyrou et al. 2009), among others, on soil quality. Nevertheless, little is known about the environmental consequences of applying tetracyclines to crops or of preparing farmlands with manure of animals that received these growth-promoting antibiotics in their diets. These practices are widely accepted in tropical countries (Vidaver 2002).
Bearing in mind that massive amounts of tetracyclines are imported to Costa Rica for agricultural use every year (Rodríguez-Sánchez 2008a), as well as the utility of the community level physiological profiling technique (Garland and Mills 1991) to evaluate the ecotoxicology of disinfectants (Liu et al. 2009a) and antibiotics in terrestrial environments (Kong et al., 2006; Demoling et al. 2009), we used EcoPlates (Biolog®) to clarify whether oxytetracycline has the capacity to alter the catabolic activity of the microbial community from a tropical soil.
MATERIALS AND METHODS
Sample collection and preparation of soil microcosms
Twelve microcosms of 100 g each were prepared with soil that was collected from the top layer (5 cm) of a regenerating secondary forest floor that has never been deliberately exposed to antibiotics (Leonel Oviedo Forest, Ciudad Universitaria Rodrigo Facio). This soil was sieved in order to remove rocks and organic material and then homogenized to neutralize potential spatial differences in the composition of the microbial community.
Controlled exposure of the microcosms to increasing concentrations of oxytetracycline
The microcosms were exposed to 0.1 mg kg−1, 1 mg kg−1 and 10 mg kg−1 of oxytetracycline. This antibiotic was added in a final volume of 10 ml of culture medium in order to stimulate the growth of the microbial community and thus demonstrate the bacteriostatic effect of oxytetracycline (trypticase soy broth diluted 1:10; CTS). All of the treatments, including a negative control to which 10 ml of CTS were added, were independently conducted in triplicate. At time zero, the microcosms were covered with a plastic lid and their weigh was recorded. A number of crops that may be protected with oxytetracycline have productive cycles of 3–4 weeks. Thus, the microscosms were exposed for 22 days at room temperature and away from the sunlight to avoid photolysis of the antibiotic. To reduce the possibility of functional changes occurring due to dryness, the weight of the microcosms was adjusted every 48 hours by adding the volume of CTS required to reach their weight at time zero.
Extraction of bacteria from the microcosms and standardization of inocula
On the 22nd day of exposure, 10 g from each microcosm were taken and mixed with 100 ml of a phosphate buffer (100 mM, pH=7) in a paddle homogenizer. These suspensions were centrifuged at 350 g for 10 minutes and the supernatants were cryopreserved at −70°C. The amount of bacteria in these supernatants was determined by cultivation using traditional microbiological procedures and trypticase-soy plates.
Community level physiological profiling (CLPP)
The capacity of each extracted microbial community to catabolize β-methyl-D-glucoside, D-galactonic acid γ-lactone, L-arginine, pyruvic acid methyl ester, D-xylose, D-galacturonic acid, L-asparagine, Tween-40, i-erythritol, 2-hydroxybenzoic acid, L-phenylalanine, Tween-80, D-mannitol, 4-hidroxibenzoic acid, L-serine, α-cyclodextrin, N-acetyl-D-glucosamine, γ-hydroxybutyric acid, L-threonine, glycogen, D-glucosaminic acid, itaconic acid, glycyl-L-glutamic acid, D-cellobiose, glucose-1-phosphate, α-ketobutyric acid, phenylethylamine, α-D-lactose, D,L-α-glycerol phosphate, D-malic acid and putrescine was determined using Eco-plate plates (Biolog). In order to avoid differences due to the amount of bacteria added to each plate, each well was inoculated with 20 μL of bacterial suspensions adjusted to 106 UFC ml−1. The development of color in each well due to the reduction of the tetrazolium violet redox indicator was measured every 24 h at 590 nm and the plates were incubated in darkness and at room temperature until the optical density of the substrates stopped increasing (approx. 10 days). The absorbance of wells containing a carbon source was corrected by subtracting the absorbance of the control well containing water.
Analysis of catabolic profiles
Dose-response curves were generated with the set of maximum absorbance values obtained and the base-ten logarithm of the concentrations of oxytetracycline added to the microcosms. A variance analysis was carried out to reveal the substrates that significantly contributed to the discrimination of the four treatments (0 mg kg1, 0.1 mg kg1, 1 mg kg−1 and 10 mg kg−1 of oxytetracycline) and a multivariate analysis of multidimensional scaling was employed to graphically depict differences in the catabolic profiles.
RESULTS
Most substrate utilization curves exhibited an initial phase in which the necessary enzymes for transport and degradation of the compounds were hypothetically induced, another phase in which the absorbance increased exponentially due to the oxidation of the compounds, and a final phase in which the absorbance did not change because all of the substrates had been consumed. The length of each phase (data not shown), the slope of the curve in the exponential phase (data not shown), and the maximum absorbance values reached were characteristic for each substrate and treatment.
Catabolic profiles of the microbial community of a tropical soil exposed to different concentrations of oxytetracycline for 22 days

The treatments with 1 and 10 mg kg−1 of oxytetracycline diminished the capacity of the communities to oxidize all of the substrates except L-phenylalanine, L-threonine, 2-OH-benzoic acid and γ-hydroxybutyric acid (Table 1 and Fig. 1). Contrarily, the communities exposed to 0.1 mg kg−1 of oxytetracycline showed higher catabolic activities than the controls and the other two treatments. This was true for all of the substrates except i-erythritol and L-threonine (Table 1 and Fig 1).

Utilization of 4-hydroxybenzoic acid by the microbial community of a tropical soil exposed to different concentrations of oxytetracycline for 22 days. This figure exemplifies the hormetic response observed for most carbon sources.
Differences in the absorbance values between the control and treatment groups were more often significant for the microcosms exposed to 0.1 mg kg−1 (n=19) than for the microcosms exposed to 1 mg kg−1 (n=3) or 10 mg kg−1 (n=6) (Table 2). This finding was confirmed by the multi-variable analysis, since the catabolic profile of the communities exposed to 0.1 mg kg−1 was very different than that of the communities exposed to 0 mg kg−1, 1 mg kg−1, and 10 mg kg−1 of oxytetracycline (Fig. 2). We thus conclude that the stimulatory effect exerted by the lowest concentration of oxytetracycline was more pronounced than the inhibitory effect exerted by higher concentrations of this antibiotic.
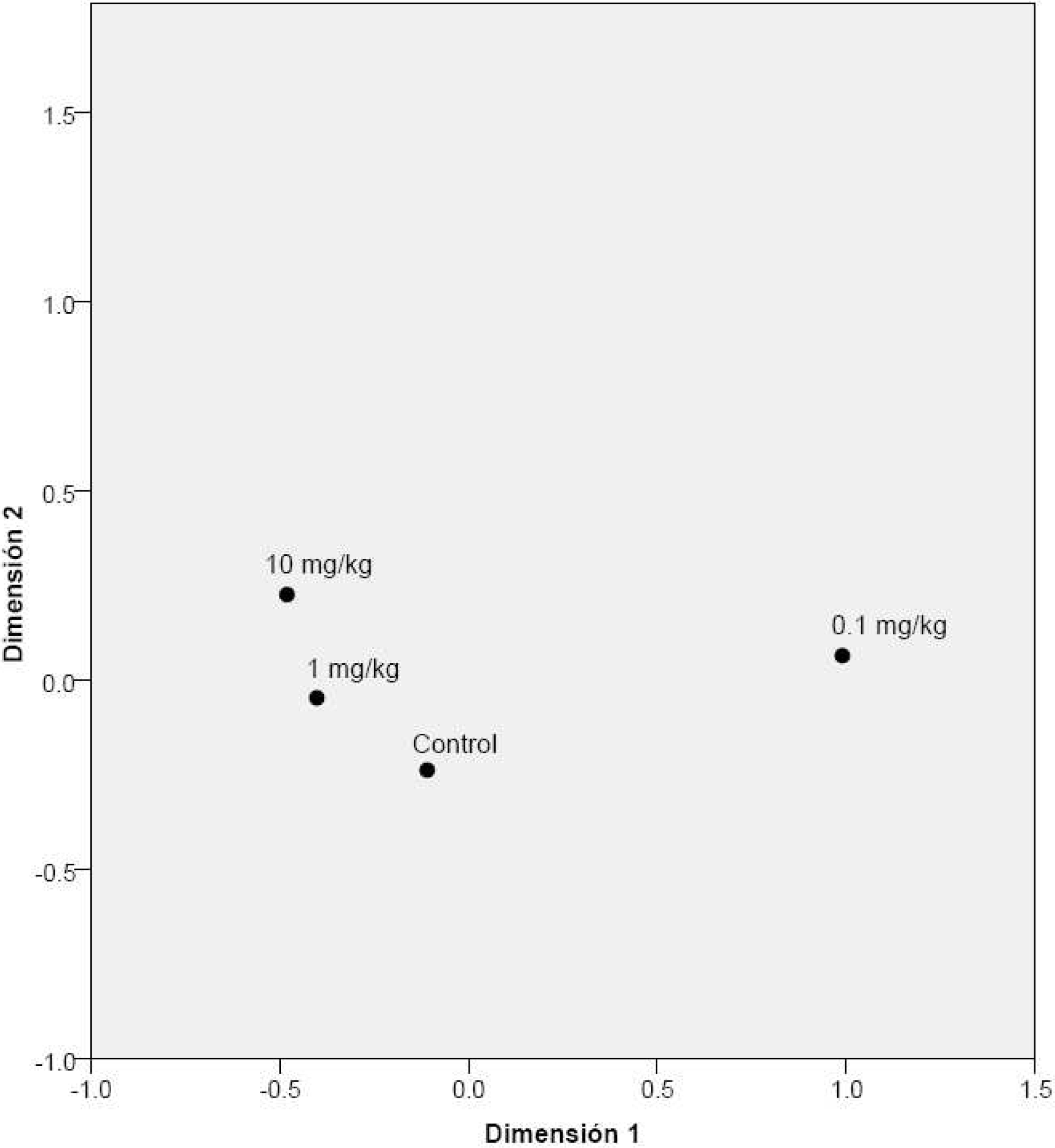
Multidimensional scaling of the catabolic profiles of the microbial community of a tropical soil exposed to different concentrations of oxytetracycline for 22 days
Carbon sources that significantly contributed to the differentiation of the catabolic profiles of the microbial community of a tropical soil exposed to different concentrations of oxytetracycline for 22 days (

DISCUSSION AND CONCLUSIONS
Among other harmful effects, it has been shown that tetracycline in the mg kg−1 range reduces the population density (Yang et al. 2009a), phosphatase (Yang et al. 2009a) and Fe (III) reduction activities (Thiele-Bruhn and Beck 2005) and, to a lesser extent, the respiration of soil microbial communities (Liu et al. 2009b).
Our results revealed two types of responses: a notable increase in the capacity of the community to metabolize a large variety of substrates in the treatment with 0.1 mg kg−1, and a slight decrease in the catabolic activities of the communities exposed to higher concentrations. This hormetic response (Calabrese and Baldwin 2002) can be explained by the capacity of antibiotics in sub-inhibitory concentrations to provoke global changes in the transcription of genes unrelated to their cell targets (Goh et al. 2002; Davies et al. 2006). Based on this argument, we hypothesize that the catabolic stimulation elicited by low concentrations represents a physiological adaptation unrelated to changes in the structure of the community and that, on the other hand, the decrease in the catabolic activity in the communities exposed to 1 and 10 mg kg−1 was due to the extinction of sensitive organisms. In this last case, the high metabolic redundancy expected for tropical soils (Kent and Triplett 2002) explains the slight magnitude of the deleterious effects provoked by the two highest concentrations tested.
Oxytetracycline is usually detected in concentrations of parts per billion (g kg−1) in agricultural soils fertilized with the excrement of animals that received this drug (De Liguoro et al. 2003; Aust et al. 2008). This antibiotic has a half-life of 29 to 56 days in soil under aerobic conditions (Yang et al. 2009b), nevertheless, its bioavailability is low because it is highly adsorbed in clay (Avisar et al. 2009) and also because it forms complexes with divalent cations, iron and aluminium oxides, and humic acids (Jones et al. 2005). Keeping this environmental behavior in mind, and that the concentrations chosen for our study are greater in at least three orders of magnitude than those usually detected in the field, we anticipate that the use of oxytetracycline in traditional agricultural activities should not compromise the catabolic capacity of tropical microbial communities. This idea is supported by the absence of effects documented in previous investigations related to the structure of the microbial community and to the prevalence of
Our results indicate that oxytetracycline, in concentrations not likely used in standard agronomic practices, alters the ability of a tropical soil microbial community to metabolize multiple carbon sources in a way that harmonizes with the hormetic dose-response model.
Footnotes
ACKNOWLEDGMENTS
This work was supported by the National Council of Rectors (CONARE) through a FEES grant and forms part of the DAGRANAC project. Heike Schmitt and María de Jesús Arias are acknowledged for fruitful discussions on the CLPP technique and its interpretation.
