Abstract
According to the doctrine underlying the current radiation protection regulations each, no matter how small, exposure to ionizing radiation may be carcinogenic. However, numerous epidemiological observations demonstrate that cancer incidence and/or mortality are not elevated among inhabitants of the high- versus low-natural-background radiation areas and homes. Results of our own and other authors’ studies described in this paper bear testimony to the possibility that stimulation of the anti-neoplastic immune surveillance mediated by NK lymphocytes and activated macrophages explains, at least partially, the accumulating epidemiological and experimental evidence indicating that low-level exposures to the low-linear energy transfer (LET) radiation inhibit the development of spontaneous and artificial metastases in humans and laboratory animals, respectively. The results presented also suggest the possibility of using low-level X- and gamma-ray exposures to cure cancer and to prevent cancer metastases. For a broader perspective, the results presented may help towards relaxing the current radiation protection regulations, especially as they apply to diagnostic and therapeutic exposures of patients to the indicated forms of radiation.
I. INTRODUCTION
Humans have always been exposed to various natural sources of ionizing radiation emitted by the isotopes present in the earth's crust, air, water and biosphere, and also originating from the outer space. In some parts of the globe the level of this natural background radiation is significantly higher than the world average with no adverse health effects. Today, people can be additionally exposed to “man-made” radiation delivered at high doses (e.g., during radiotherapy and radiation accidents as well as after detonations of nuclear weapons) or low doses (e.g., during production and distribution of radioactive materials and use of radiation sources for industrial and medical purposes). The low-level environmental and occupational exposures are much more common and distributed over much larger populations than the high-level exposures.
Low doses and dose rates of ionizing radiation (low-level radiation) are defined as those below 0.1–0.2 Gy and below 0.05–0.1 mGy/min., respectively (UNSCEAR 1986, BEIR VII 2006). Absorption of low doses of ionizing radiation may stimulate cellular detoxification and repair mechanisms leading to reduction of the DNA damage even below the spontaneous level and decreasing the probability of neoplastic transformation (for review see: Azzam et al. 1996, Pollycove 2004, 2007, Mitchel 2007, Portess et al. 2007, Redpath and Elmore 2007, Feinendegen et al. 2008). Such exposures may also enhance immune reactions of the organism and attenuate harmful effects of higher doses of radiation (Liu et al. 1982, 1985, Tuschl et al. 1995, Safwat 2000b, Safwat et al. 2003; for review see: Liu 1989, 2004, Luckey 1980, 1999, Ju et al. 1995). These mechanisms may explain various epidemiological observations indicating that cancer incidence and mortality are not elevated among inhabitants of the high-versus low-background radiation areas (Ishii et al. 1996, Kesavan 1997, Jagger 1998; for review see: Luckey 1999, Wei and Sugahara 2000) as well as among tenants of homes with the elevated levels of radiation from 222Rn or 60Co (Cohen 1995, 1997, UNSCEAR 2000, Wang et al. 2002, Chen et al. 2004). Also, in many cohorts of nuclear workers and in the survivors of the Hiroshima and Nagasaki bombings whose absorbed doses did not exceed 0.25 Gy the incidence of leukemia and some solid tumors has been reported to be lower compared to the respective control groups (Matanoski et al. 1990, Cardis et al. 1995, Pierce et al. 1996, McKinney et al. 1998, Little et al. 1999, UNSCEAR 2000, Berrington et al. 2001, Katayama et al. 2002; for review see: Kondo 1993, Luckey 1999).
These results of epidemiological analyses encouraged many to perform experimental studies utilizing the low-level low-LET irradiations of cells and animals in strictly defined conditions. Such experiments have been providing data which have already contributed to the more detailed understanding of the mechanisms plausibly responsible for the decreased incidence of tumors among people exposed to the low-level ionizing radiation. More broadly, these data may be instrumental in testing the linear-no-threshold (LNT) hypothesis which is central to establishing radiation exposure limits for humans. The LNT hypothesis is based on the controversial assumption that the underlying biological processes triggered by low radiation doses are essentially the same as those that function after higher radiation doses (Mossman 2009, Tubiana 2008). Under the LNT hypothesis, any amount of radiation would be considered to cause cancer among some members of a very large population and cancer risk would increase linearly with increasing dose.
II. ANTI-TUMOR PROPERTIES OF THE LOW-LEVEL LOW-LET IRRADIATIONS
The recent evidence has demonstrated that in animals exposed to single or fractionated low total doses of X- or γ-rays the growth of primary and/or metastatic tumors is inhibited or retarded (Hosoi and Sakamoto 1993, Ishii et al. 1996, Caratero et al. 1998, Cai 1999, Hashimoto et al. 1999, Mitchel et al. 1999, 2003, Wang and Cai 2000, Sakai et al. 2003, Ina and Sakai 2004, for review see: Ju et al. 1995, Liu 2004, 2007). In many of these investigations, anti-tumor properties of the low-level exposures were detected when whole animals were irradiated prior to inoculation of neoplastic cells, indicating that the immune surveillance mechanisms might be involved (Hashimoto et al. 1999, for review see: Safwat 2000a). In their pioneering study, Hosoi and Sakamoto (1993) detected marked reductions in the numbers of both artificial and spontaneous pulmonary metastases after single whole body-irradiation (WBI) of mice with 0.15, 0.2, or 0.5 Gy X-rays. In that study the inhibitory effect was expressed when tumor cells were inoculated either a few hours before or after the exposure. Likewise, Ju et al. (1995) and Cai (1999) who irradiated mice with single doses of X-rays ranging from 0.05 to 0.15 Gy 24 hours before the intravenous (i.v.) injection of B16 melanoma or Lewis Lung Carcinoma (LLC) cells reported a significant retardation of the development of pulmonary tumor nodules. Moreover, a decreased incidence of the lung and lymph node metastases accompanied by the enhanced infiltration of the metastatic foci by lymphocytes was demonstrated by Hashimoto et al. (1999) who exposed rats to 0.2 Gy γ-rays 14 days after subcutaneous (s.c.) implantation of hepatoma cells. Importantly, a local irradiation of the developing tumors did not reduce the number of spontaneous metastases derived thereof. Likewise, Sakai et al. (2003) reported that protracted irradiation of mice with γ-rays at 1 mGy/h dose rate for over 250 days attenuated the growth of the 20-methylocholantrene-induced skin tumors. In our own experiments, two hours after cessation of both single (Fig. 1) and fractionated (Fig. 2) WBI of mice with 0.1 or 0.2 Gy X-rays the animals were i.v. injected with L1 sarcoma cells and 14 days later macroscopic tumor colonies were counted on the lungs’ surfaces (Cheda et al. 2004a, 2004b, 2006, Nowosielska et al. 2005, 2006b, 2008, Janiak et al. 2006). We showed that development of the induced tumor metastases was significantly inhibited after single WBI with the two low doses of X-rays (Fig. 3A and Table 1). Similarly, mice exposed to the total dose of 0.1 or 0.2 Gy applied in ten equal fractions tended to have less pulmonary tumor colonies than sham-exposed control animals (Fig. 3B) (Nowosielska et al. 2008).
Relative numbers (percentage of the control value) of the pulmonary L1 sarcoma colonies and cytotoxic activity of the NK cell-enriched splenocytes (NK cells) and IFN-γ- and LPS-stimulated peritoneal macrophages (Mφ) after single WBI (Fig. 1) of mice with 0.1 or 0.2 Gy X-rays and i.p. injection with anti-asialo GM1 antibody or CGN.

The parameters were examined on the 14th (pulmonary colonies), 2nd (NK cells), and 3rd (Mφ) days after single WBI. Control – sham-exposed mice; 0.1 Gy – mice exposed to a single WBI with 0.1 Gy X-rays; 0.2 Gy – mice exposed to a single WBI with 0.2 Gy X-rays; PBS – mice i.p. injected with phosphate buffered saline; Ab – mice i.p. injected with anti-asialo GM1 antibody; CGN – mice i.p. injected with CGN.
indicates statistically significant (p<0.05) difference from the control/PBS value.

Schematic outline of single exposures of BALB/c mice to X-rays (HS320 Pantak X-ray generator [230 kV, 20 mA] supplied with the Al and Cu filters, at 2.2 Gy/h dose rate) and times of the assessment of tumor lung colonies and activities of the NK cell-enriched splenocytes (NK cells) and peritoneal macrophages (Mφ).
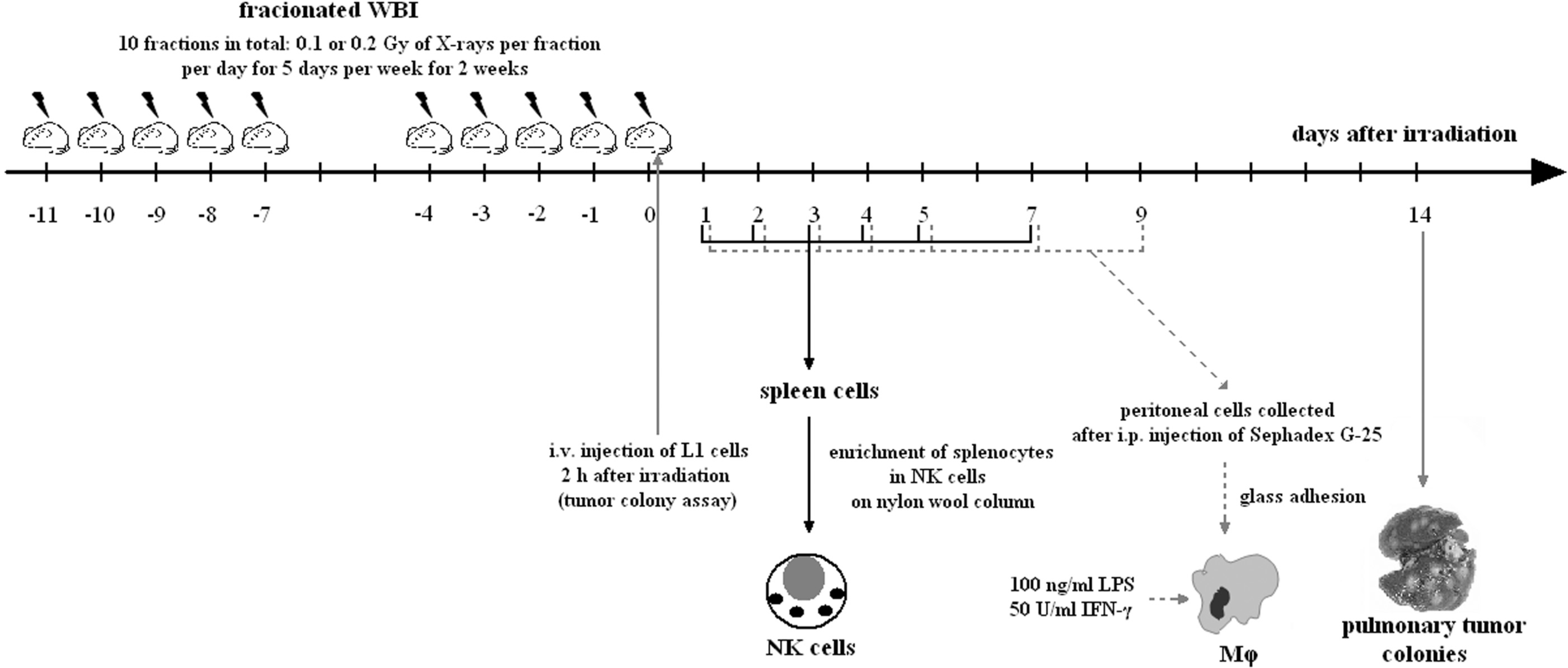
Schematic outline of fractionated exposures of BALB/c mice to X-rays (ANDREX X-ray generator [150 kV, 3 mA], at 2.16 Gy/h dose rate) and times of the assessment of tumor lung colonies and activities of the NK cell-enriched splenocytes (NK cells) and peritoneal macrophages (Mφ).
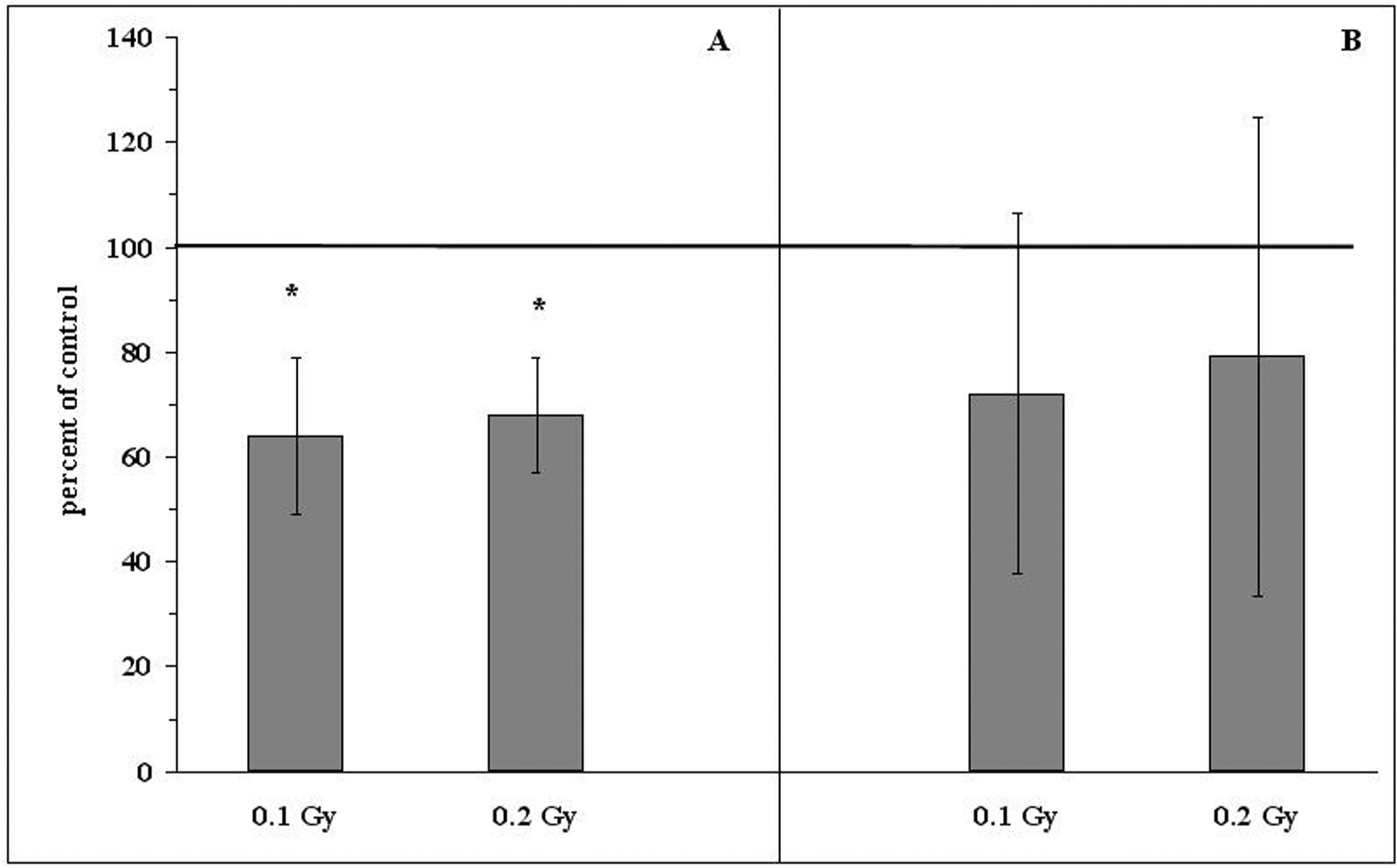
Relative numbers (percentages of the control values indicated as solid line at 100%) of the induced tumor colonies in the lungs of BALB/c mice exposed to single (A) or fractionated (B) total doses of 0.1 or 0.2 Gy X-rays and two hours later i.v. injected with L1 sarcoma cells. Mean values ± SD (bars) are shown. ∗indicates statistically significant (p<0.05) difference from the control (100%) value.
The above results suggest that the inhibitory effects of the WBI with low doses of X- or γ-rays on the development of both primary and secondary tumor foci may result from stimulation by such exposures of anticancer immune mechanisms rather than from a direct impairment of the viability and/or function of the neoplastic cells.
III. LOW DOSE-INDUCED STIMULATION OF THE ANTI-NEOPLASTIC IMMUNITY
Numerous experimental investigations have demonstrated that exposures to low doses of ionizing radiation spur functions of the anti-neoplastic immune surveillance system. For example, Ina and coworkers (Ina and Sakai 2005a, Ina et al. 2005) showed that chronic irradiation of C57BL/6 mice with γ-rays at 1.2 mGy/h dose rate resulted in the activation of T and B lymphocytes, including the plaque forming cells (PFC), as well as in the total depletion of the abnormal T CD3−CD4+ lymphocytes. In another study, the same authors demonstrated that chronic low-dose-rate (0.35 or 1.2 mGy/h) irradiation with γ-rays enhanced survival of the MRL-lpr/lpr mice carrying a deletion in the apoptosis-regulating Fas gene leading to a severe autoimmune disease and reduced life-span. This effect was accompanied by significant elevations in the number of CD4+CD8+ T cells in the thymus and CD8+ T cells in the spleen as well as by a marked down-regulation of the abnormal CD3+↑CD45R/B220+ and CD45R/B220+↓CD40+ splenocytes accompanied by a drastic attenuation of the total-body lymphadenopathy, splenomegaly, proteinuria, and kidney and brain disorders (Ina and Sakai 2004, 2005b).
1. NK cell-mediated activity
Primary cellular effectors of the non-specific anti-tumor surveillance system are natural killer (NK) lymphocytes and activated macrophages (Mφ) (Nathan 1991, Farias-Eisner et al. 1994, Liu et al. 1994a,b, Moretta et al. 1994; for review see: Barao and Ascensao 1998, Al-Sarireh and Eremin 2000). Stimulation of the NK cell-mediated cytotoxicity after a single irradiation of mice was described by Liu et al. (1994b) and by Kojima et al. (2002, 2004). The former group detected the effect 24 hours after exposure to 0.075 Gy X-rays and the latter – between the fourth and sixth hours post irradiation with 0.5 Gy γ-rays. Other authors (Ju et al. 1995) reviewed the enhanced cytocidal function of murine NK-type lymphocytes 2–6 days after a single exposure of the animals to 0.075 Gy X-rays. These findings were corroborated by the results of our own studies demonstrating that single WBI (Fig. 1) of mice with 0.1 or 0.2 Gy X-rays increased the cytolytic function of NK lymphocytes obtained from the spleen (NK cell-enriched splenocytes), as measured by the classic 51Cr release from the YAC-1 tumor target cells; this effect was detectable between the first and third days post-irradiation and was most pronounced on the second day after the exposure (Table 1 and 2) (Cheda et al. 2004a, 2004b, 2006, Nowosielska et al. 2005, 2006a, 2008, Janiak et al. 2006). We also showed that fractionated WBI of mice (Fig. 2) with either of the above two low doses of X-rays led to the significant upregulation of the cytotoxic function of the NK cell-enriched splenocytes (Table 3); in this case the enhanced cytotoxicity was most pronounced between the first and fourth days post-exposure and declined to the baseline level on the seventh day (Nowosielska et al. 2008). When mice were intraperitoneally (i.p.) injected with anti-asialo GM1 antibody (a classical blocker of the NK cells activity) 24 hours before single WBI, the NK-type activity tested two days later was totally suppressed and this inhibition could not be reversed by a single WBI with 0.1 or 0.2 Gy X-rays (Table 1) (Cheda et al. 2004b, 2006, Nowosielska et al. 2005, Janiak et al. 2006). Moreover, i.p. injection of the anti-asialo GM1 antibody resulted in the significant increase in the numbers of the tumor colonies developing in the lungs of both irradiated and sham-exposed mice (Table 1).
Immunological parameters in mice exposed to single WBI (Fig. 1) with 0.1 or 0.2 Gy X-rays.

The parameters were examined on the 2nd (NK cell-enriched splenocytes – NK-cells) and 3rd (PHA-stimulated splenocytes and IFN-γ and LPS-stimulated peritoneal macrophages – Mφ) days after single WBI. Control – sham-exposed mice; 0.1 Gy – mice exposed to a single WBI with 0.1 Gy X-rays; 0.2 Gy – mice exposed to a single WBI with 0.2 Gy X-rays.
indicates statistically significant (p<0.05) difference from the results obtained in the control group.
Immunological parameters in mice exposed to fractionated WBI (10 fractions in total: 0.01 or 0.02 Gy per fraction per day for 5 days per week for 2 weeks; Fig. 2) with total doses of 0.1 or 0.2 Gy X-rays.
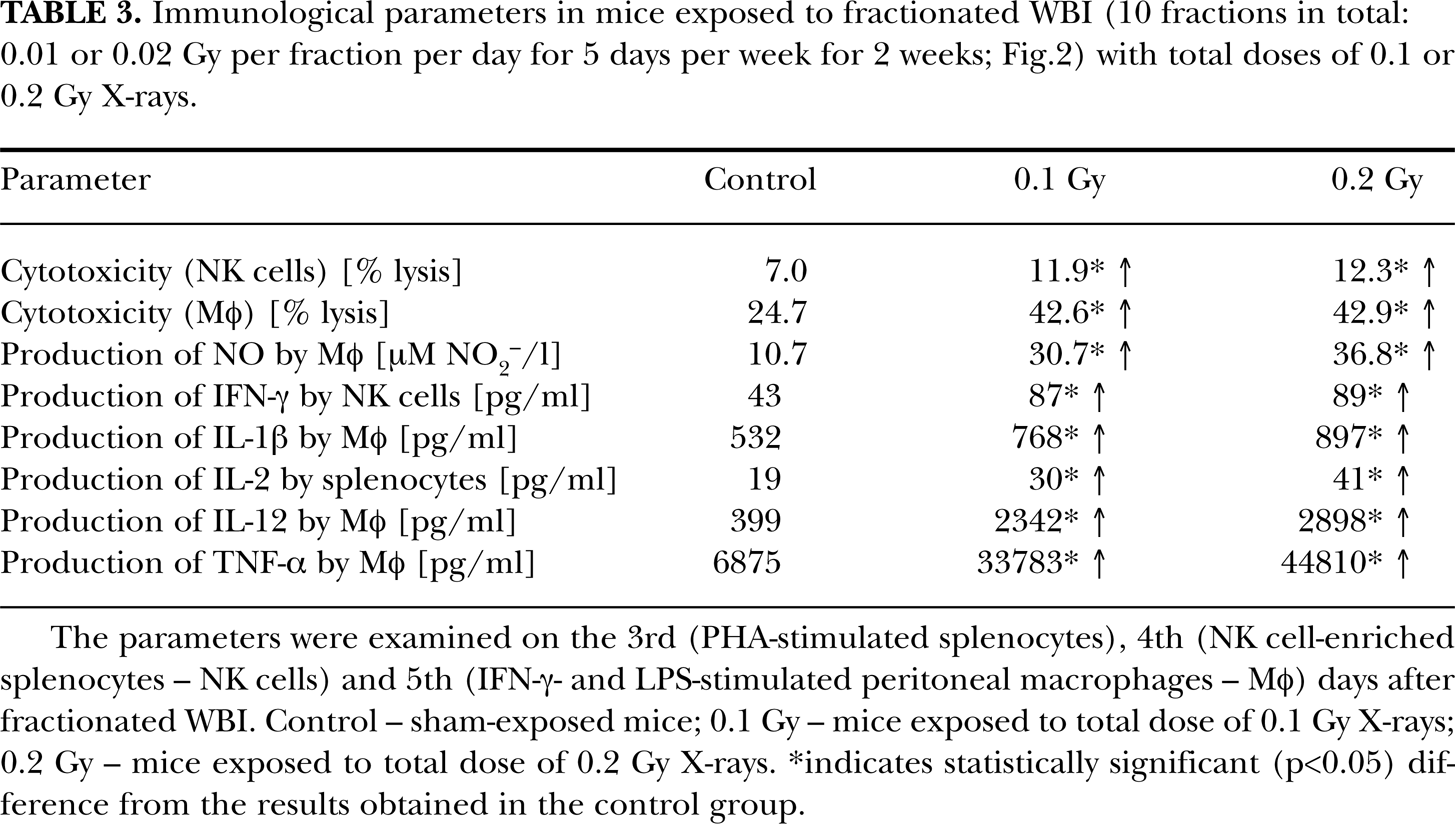
The parameters were examined on the 3rd (PHA-stimulated splenocytes), 4th (NK cell-enriched splenocytes – NK cells) and 5th (IFN-γ- and LPS-stimulated peritoneal macrophages – Mφ) days after fractionated WBI. Control – sham-exposed mice; 0.1 Gy – mice exposed to total dose of 0.1 Gy X-rays; 0.2 Gy – mice exposed to total dose of 0.2 Gy X-rays.
indicates statistically significant (p<0.05) difference from the results obtained in the control group.
Cytotoxic activity of NK lymphocytes is mediated by the extra-cellularly secreted perforin which creates ‘pores’ in the target cell's membrane whereby the concomitantly released granzymes enter and kill the target (Lord et al. 2003, Smyth et al. 2005). Another potential cytotoxic mechanism consists in the activation of the Fas receptor on the surface of tumor cells upon binding of the specific ligand (FasL) whose expression is upregulated on the activated NK lymphocytes (Reyburn et al. 1997, for review see: Barao and Ascensao 1998). In our experiments suppression of the perforin activity by concanamicin A (CMA) significantly inhibited cytotoxic function of the NK cell-enriched splenocytes collected from both irradiated and non-irradiated mice; the suppression was lower but still significantly expressed when the anti-FasL antibody was added to the incubation medium (Table 4) (unpublished data). Also, surface expression of FasL was significantly increased on the NK-type splenocytes obtained from mice two days after a single WBI (Fig. 1) with 0.1 and 0.2 Gy X-rays, i.e. at the time when the cytotoxic function of these cells was maximally stimulated (Table 2) (Nowosielska et al. 2005, 2006a, Janiak et al. 2006). These results clearly indicate that both secretion of perforin by and expression of FasL on the surface of the effector cells were responsible for the demonstrated by us enhanced cytotoxic function of the NK cell-enriched splenocytes obtained from mice exposed to single irradiations with low doses of X-rays.
Inhibition of the cytotoxic activity [expressed in % lysis] of the NK cell-enriched splenocytes (NK cells – inhibited by CMA and anti-FasL Ab) and the IFN-γ- and LPS-stimulated peritoneal macrophages (Mφ – inhibited by CGN and AG) on the 2nd and 3rd days after single WBI (Fig. 1) with X-rays, respectively.

Control – sham-exposed mice; 0.1 Gy – mice exposed to a single WBI with 0.1 Gy X-rays; 0.2 Gy – mice exposed to a single WBI with 0.2 Gy X-rays.
CM – NK cells incubated in culture medium without blockers; CMA – NK cells incubated with CMA; anti-FasL Ab – NK cells incubated with anti-FasL antibody; CMA + anti-FasL Ab – NK cells incubated with CMA and anti-FasL antibody.
indicates statistically significant p<0.05) difference between NK cells collected from irradiated mice and the respective NK cells obtained from non-irradiated mice.
indicates statistically significant (p<0.05) difference within each sham-irradiated or irradiated group between NK cells incubated with CMA and/or anti-FasL antibody and NK cells incubated without blockers (CM group).
PBS – Mφ obtained from mice pretreated with PBS; CGN – Mφ obtained from mice pretreated with CGN; AG – Mφ obtained from mice pretreated with PBS and incubated in vitro in the presence of AG; CGN + AG – Mφ obtained from mice pretreated with CGN and incubated in vitro in the presence of AG.
indicates statistically significant (p<0.05) difference within sham-irradiated or irradiated groups between Mφ incubated with AG and/or obtained from mice pretreated with CGN and Mφ collected from mice pretreated with PBS and incubated without AG.
indicates statistically significant (p<0.05) difference between Mφ collected from irradiated mice and the respective Mφ obtained from non-irradiated mice.
2. Macrophage-mediated activity
Activated macrophages (Mφ) kill susceptible tumor cells by means of a number of cytotoxic factors of which nitric oxide (NO) plays a prominent role (Nathan 1991, Cui et al. 1994, Farias-Eisner et al. 1994, Jenkins et al. 1995, Xie and Fidler 1998). Ibuki and Goto (1995) were among the first to demonstrate that irradiation of mice with 0.04 Gy γ-rays stimulated production of NO by the IFN-γ- and LPS-treated peritoneal Mφ collected on the day of exposure. This effect was associated with the significant enhancement of the cytotoxic function of Mφ against the P815 tumor cells. Up-regulated secretion of NO was also described by Pandey et al. (2005) who five times in a row irradiated C57BL/6 mice with 0.04 Gy γ-rays (with the 24-hour intervals between the irradiations) and assessed production of this cytocidal factor by the ConA-stimulated adherent splenocytes on the third day after cessation of the exposures; this effect was associated with the increased phagocytic function of peritoneal exudate cells obtained from the irradiated animals.
The above results were corroborated and extended by us in experiments demonstrating that single exposures (Fig. 1) of mice to 0.1 or 0.2 Gy X-rays led to the significant up-regulation of the cytolytic activity (measured in the 3H-thymidine release assay) of the untreated and IFN-γ and LPS-stimulated peritoneal Mφ against both the L1 (Table 1 and 2) and P815 tumor cells; the effect was most pronounced on the third day and sustained until the ninth day post-irradiation (Cheda et al. 2005, 2006, Janiak et al. 2006, Nowosielska et al. 2006a, 2006b, 2008). Moreover, fractionated WBI of mice (Fig. 2) with either of the two low total doses of X-rays resulted in the significant enhancement of the cytotoxic function of peritoneal Mφ (Table 3) against the L1 cells; the effect was most pronounced between the second and fifth days post-irradiation and declined thereafter (Nowosielska et al. 2008). The up-regulated cytotoxicity was accompanied by the elevated production of NO (Table 2 and 3) (Cheda et al. 2004a, 2005, 2006, Janiak et al. 2006, Nowosielska et al. 2006a, 2006b, 2008) and reactive superoxide anions in the IFN-γ and LPS-treated (NO) or untreated (superoxide anions) peritoneal Mφ (Cheda et al. 2005, Janiak et al. 2006). Interestingly, in these experiments the kinetics of the NO production closely followed the changes in the cytolytic activity of the peritoneal Mφ.
Notably, i.p. injection of mice with carrageenan (CGN – a lysosome-disrupting and phagocyte-damaging compound) (Frank et al. 2003) 24 hours before single WBI resulted in the almost total abrogation of the synthesis of NO in the collected peritoneal Mφ regardless of whether the cells were obtained from the irradiated or sham-exposed mice (Cheda et al. 2006, Janiak et al. 2006, Nowosielska et al. 2006b, 2008). This finding supports a possible involvement of NO in the Mφ-mediated anti-tumor effect of the low-level exposures to X-rays. However, peritoneal Mφ collected from the CGN-treated animals still exhibited cytotoxic activity in vitro, even in the absence of IFN-γ and LPS in the incubation medium. This observation can be explained by triggering of the synthesis of NO in the effector peritoneal Mφ by the target cells (Nowosielska et al. 2006b) and/or involvement of other stimulatory mechanisms in the Mφ-mediated function. Indeed, suppression of the activity of the inducible nitric oxide synthase (iNOS) by aminoguanidine (AG – a classical inhibitor of iNOS) markedly reduced the cytolytic function of the effector peritoneal Mφ obtained from both the sham-exposed and irradiated mice (Table 4) (Nowosielska et al. 2006b).
We also demonstrated that injection of mice with CGN, in addition to the submaximal abrogation of the synthesis of NO in the collected Mφ, totally abolished the inhibitory effect of the irradiations with both 0.1 and 0.2 Gy X-rays on the growth of the induced tumor metastases: the number of the pulmonary colonies developing in the irradiated mice pretreated with CGN did not differ from that obtained in the control animals (Table 1). Interestingly, CGN appeared to be a more potent suppressor of the anti-neoplastic effect of the low-level exposure to X-rays than the anti-asialo GM1 antibody (Cheda et al. 2004b, 2006, Nowosielska et al. 2005, 2008, Janiak et al. 2006): the suppressive effect of the former was several-fold greater than that of the latter. This observation may be explained by a possible shut-down or reduction by CGN of the cytotoxic functions of Mφ and, indirectly, NK cells. In fact, it has been shown that several cytokines produced by Mφ, such as IL-12 and IL-18, are potent modulators of the activity of NK lymphocytes (for review see Young and Ortaldo 2006) and inhibition of the function of the former cells may affect that of the latter cells in vivo (Fig. 4).

Schematic outline of the possible interactions between NK cells, macrophages, and tumor cells.
3. Cytokines produced by NK cells and macrophages
Notably, the results obtained in our studies indicated that neither the i.p. injection of mice with CGN nor addition of AG to the culture medium led to the complete abrogation of the cytotoxic activity of peritoneal Mφ even when the two blockers were used simultaneously. Likewise, both CMA and the anti-FasL antibody were unable to totally suppress the cytolytic function of the NK cell-enriched splenocytes (Table 4). These findings suggest that the residual cytotoxic activity of the two types of the effector cells may result from the production and secretion of additional cytotoxic and/or cytostatic factors likely to be involved in elimination of neoplastic cells. In fact, both NK lymphocytes and activated Mφ produce a number of cytokines which mediate anti-neoplastic functions of these cells, i.e. either directly suppress proliferation of and/or kill tumor targets (e.g., IL-1β, IFN-γ, TNF-α) or paracrinely stimulate neighboring cells to secrete cytocidal factors (e.g., IL-1β, IL-12, IFN-γ). In fact, researchers in Japan and China, after exposure of mice and rats to single WBI with X- or γ-rays at doses ranging from 0.04 to 0.25 Gy, detected up-regulated secretion and/or expression of mRNAs for IL-1β, IL-12, TNF-α (by Mφ), IFN-γ (by NK cells), and IL-2 (by splenocytes) (DeBlaker-Hohe et al. 1995, Miller et al. 2003, Liu et al. 1994b, 2001, Fu et al. 1996, 1997, Zhang et al. 1996, Gong et al. 1997, Zhang et al. 1998a,b, 1999, Bai et al. 1998, Ibuki and Goto 1999, Hashimoto et al. 1999, Shan et al. 2007, for review see: Al-Sarireh and Eremin 2000, Belardelli and Ferrantini 2002, Liu 2007). In contrast to single irradiations, almost no evidence exists in the literature on triggering of the expression of cytokines by multiple low-level exposures to ionizing radiation. Indeed, Pandey et al. (2005) were unable to detect any stimulation of the production of IFN-γ in splenocytes collected and assayed on the third day after completion of the fractionated (0.04 Gy per day for 5 days) irradiation of the C57BL/6 mice with γ-rays.
These observations were generally corroborated by the results of our own investigations in which peritoneal Mφ and NK cell-enriched splenocytes (Table 2 and 3) were assayed for their capacity to produce the selected cytokines using experimental procedures identical to the ones previously utilized by us for testing of the cytotoxic activity of these cells. Thus, tumor target cells (P815 or L1 for peritoneal Mφ and YAC-1 for NK cell-enriched splenocytes) or PHA (for splenocytes) were included in the incubation wells and then the cell-free supernatants were assayed by the ELISA methodology for the levels of IL-1β, TNF-α, IL-12 (synthesized by peritoneal Mφ), IL-2 (produced by splenocytes), and IFN-γ (secreted by NK cell-enriched splenocytes) (Cheda et al. 2008). In this study, we demonstrated that both single (Fig. 1) and fractionated (Fig. 2) irradiations of mice with total absorbed doses of 0.1 or 0.2 Gy X-rays significantly stimulated peritoneal Mφ to produce IL-1β (Table 2 and 3) (Cheda et al. 2008). Notably, the kinetics of the low-level X-ray-induced production of IL-1β was similar to that detected for the cytotoxic activity and production of NO by peritoneal Mφ obtained after single (Cheda et al. 2005, 2006, Janiak et al. 2006, Nowosielska et al. 2006a,b) or fractionated (Nowosielska et al. 2008) irradiations of mice with 0.1 or 0.2 Gy X-rays. Stimulation of the synthesis of IL-1β was accompanied by the enhanced production of TNF-α after both single (Fig. 1) and fractionated (Fig. 2) irradiations of mice, although the stimulatory effect of the latter exposures on the secretion of TNF-α was much stronger than that detected after the former exposures (Table 2 and 3) (Cheda et al. 2005, 2008, Janiak et al. 2006, Nowosielska et al. 2006a). The radiation-induced upregulated production of IL-1β and TNF-α coincided with the elevated secretion of IL-12 (Table 2 and 3) (Cheda et al. 2008). Moreover, both single (Fig. 1) and fractionated (Fig. 2) WBI of mice with 0.1 or 0.2 Gy X-rays significantly stimulated synthesis of IL-2 and IFN-γ in the unseparated and the NK cell-enriched splenocytes, respectively (Table 2 and 3) (Cheda et al. 2008). Importantly, the time course of the changes in the production of IFN-γ after a single but not fractionated irradiation of mice with either of the two low doses of X-rays closely resembled the kinetics of the previously demonstrated by us (Cheda et al. 2004a,b, 2006, Nowosielska et al. 2005, 2006a, 2008, Janiak et al. 2006) enhanced cytotoxicity of the NK cell-enriched splenocytes obtained from the similarly exposed animals.
4. NK cell- and macrophage-mediated activities after in vitro irradiation
In contrast to the single and fractionated WBI of mice with low doses of X-rays, we showed that the in vitro irradiations of NK cell-enriched splenocytes and peritoneal Mφ did not significantly affect the NK cell- and Mφ-mediated functions (Table 5). This observations suggests that the above described anti-tumor properties of both single and fractionated irradiations with low total doses of X-rays result from the stimulated interaction of many components of the complex immune surveillance system rather than from boosting of function(s) of a single cell population.
Effects of the in vitro irradiation of NK cell-enriched splenocytes (NK cells) and the IFN-γ- and LPS-stimulated peritoneal macrophages (Mφ) with low doses of X-rays.

Control – sham-exposed NK cells or Mφ; 0.1 Gy – NK cells or Mφ exposed to single irradiation with 0.1 Gy X-rays; 0.2 Gy – NK cells or Mφ exposed to single irradiation with 0.2 Gy X-rays.
IV. IMPLICATIONS
Results of the epidemiological and experimental studies presented in this paper indicate that low-level exposures to X- and γ-rays may suppress the development and progression of tumors and that these effects can be associated with stimulation by such irradiations of anti-neoplastic functions of the immune system. This type of the radiation-evoked hormetic effect (for review see: Luckey 1980, Webster 1993, Calabrese and Baldwin 2002, Pollycove 2007, Feinendegen et al. 2008) may have several implications.
Firstly, the described experimental data provide clues for, at least partial, explanation of the inhibitory effects of the low-level exposures to the low-LET radiation on the development and progression of tumors. Indeed, these low-dose data contradict the commonly applied LNT model-related assumption that the underlying biological processes that function after low radiation doses are essentially the same as those that function after higher radiation doses (for review see: Averbeck et al. 2006, Tubiana and Aurengo 2006, Tubiana et al. 2006a,b, Tubiana 2008, 2009).
Secondly, after careful preclinical and clinical trials, low-level exposures to X- or γ-rays could be employed as a new modality in the treatment of cancer. Indeed, the half or total body exposures to low doses of X-rays have been already tested in experimental therapeutic protocols as complements to the standard chemo- and/or radiotherapy of ovarian and colon cancer, as well as non-Hodgkin lymphoma (Choi et al. 1979, Sakamoto et al. 1997, for review see Cuttler and Pollycove 2003). As indicated recently by Tubiana in his review of the current data (Tubiana 2009), the risk of the second primary malignancies associated with therapeutical application of the total doses of radiation lower than 0.1–0.2 Gy is negligible.
Thirdly, the results of the investigations described above as well as other supporting radiation-adaptive-response data may in a broader perspective contribute to relaxing the current stringent radiation protection regulations. This includes regulations that govern diagnostic and therapeutic applications of ionizing radiation as well as those related to occupational and environmental radiation exposures. Current regulations are based on the controversial LNT hypothesis (ICRP 1990, 2006; Mossman 2009) which imposes avoiding exposures to even very low doses and dose rates of radiation. This practice not only elevates the costs of the enforcement of the protective measures, but may also have – opposite to the intentions – adverse health effects (for review see Jaworowski 2000, 2009).
