Abstract
Radiation protection regulations have been established to reduce exposure of individuals to acceptable safe levels. These limits assume that people have similar responses to ionizing radiation and that there is no variation in individual radiation risk. The purpose of this research was to determine if apoptosis in lymphocytes can be used to assess individual sensitivity to ionizing radiation. Blood samples were taken from 54 males ranging in age from 19–85 years. Apoptosis was measured using modified flow cytometry based Annexin-FITC/7AAD and DiOC6/7AAD assays in different populations of lymphocytes (total mixed lymphocyte population, subset CD4+ or CD8+ lymphocytes) after exposure to in vitro doses of 0, 2, 4 or 8Gy (dose rate 0.1Gy/min). The variation in individual responses to radiation was large. The variation was the largest in the CD4+ lymphocyte subpopulation. Radiation-induced apoptosis decreased with age of donor demonstrating that as people age their lymphocytes may become relatively more resistant to radiation. This research shows that individuals have marked differences in their sensitivity to radiation and protection policies may someday need to be tailored for some individuals.
INTRODUCTION
In the nuclear power industry and other activities that use ionizing radiation such as medical diagnostics and therapy, limits are set to ensure doses are at a safe level. These limits assume that people have equal responses to ionizing radiation and that there is no variation in radiation risk. However, in radiotherapy, where patients receive large doses of radiation to their tumours and the surrounding tissue volume, it is well documented that all patients do not respond the same to a specific treatment plan. As a result, 5–10% of patients will have adverse reactions. In other activities that involve exposure to ionizing radiation, individual risk from low doses is extrapolated from risks observed from high dose exposures. However, the actual risk to an individual likely relates to their own intrinsic sensitivity to the radiation. Therefore, experiments that can determine the range of individual responses to radiation are useful for a better understanding of actual risk.
The ability to predict radiation sensitivity would benefit both medical and health physicists, and emergency responders (Kanda et al, 1999, Kleinerman et al, 2006). If radiation sensitive patients could be identified before they undergo therapy, a reduced dose regiment or alternative therapy could be prescribed (Barber et al, 2000). Furthermore, predicting radiation sensitive individuals would allow the radiation protection industry to tailor dose limits to an individual, reducing risk to the worker. If an individual has a decreased ability to detect damage caused by radiation, perhaps a low dose exposure would not be detected or repaired properly, turning a ‘safe’ exposure into a potential cancer causing event.
We postulate that this variation in radiation response is due to the individual's intrinsic cellular response to the damage created by radiation. These individuals could have altered genetic factors regulating DNA repair processes and/or controlling apoptosis. Therefore, this systemic predisposition results in a lack of ability for damaged cells to be eliminated properly or be repaired and consequently causes an adverse reaction. Understanding this phenomenon is crucial for radiation protection practices, since these radiosensitive individuals may also be at increased risk to high occupational or medical exposures. Recent evidence indicates that some cancer patients that exhibit adverse effects (toxicity) from radiotherapy may also have abnormal apoptotic responses in subpopulations of their lymphocytes.
Apoptosis is a form of programmed cell death that is the dominant mode of cell killing induced by radiation in human lymphocytes. Measuring levels of apoptosis in lymphocytes has been suggested as a method for estimating in vivo dose (Menz, 1997; Boreham, 1996) following an accidental exposure to radiation. Menz et al, researched the dose response of human lymphocytes when exposed to doses up to 8Gy of gamma radiation as measured by a flow cytometry apoptosis assay in 5 donors. Boreham et al, (1996) investigated doses of up to 1Gy using the in situ terminal deoxynucleotidyl transferase (TdT) assay and the fluorescence analysis of DNA unwinding (FADU) assay to quantify apoptosis in 10 donors. These studies found that apoptosis was detectable for doses as low as 0.05Gy and reproducible enough to act as a biological dosimeter.
The purpose of this study was to assess the feasibility of using apoptosis as an assay to investigate individual variability and sensitivity to radiation. The range of this variability was tested in an attempt to evaluate the importance of individual responses with respect to current radiation risk estimation and possible adverse reactions to medical procedures.
MATERIALS AND METHODS
Sample Collection
Blood was taken from two distinct groups of male donors. The first group of donors included previous patients at the Juravinski Cancer Centre, Hamilton, Canada, enrolled in the Predictive Assay clinical trial (Schnarr et al., in press). The Predictive Assay study took blood samples from 44 patients previously treated for prostate cancer and investigated in vitro lymphocyte radiation induced apoptosis. These patients ranged in age from 61–85. Exclusion criteria from this study included concurrent use of chemotherapy drugs or hormone therapy. Ethical approval for the study protocol was obtained through the Hamilton Health Sciences Research Ethics Board. The other donor cohorts consisted of 10 male volunteers, aged 19–47 with no previous history of cancer and were apparently normal.
To assess reproducibility, 10 donors had a replicate blood sample taken. The elapsed time between the original and replicate blood sample collection ranged from 1.5 to 12 months. All samples were processed anonymously in a blinded system to ensure patient confidentiality and eliminate processing bias.
Sample Preparation
A total of 20ml of blood was collected into sodium heparin venous blood collection tubes (Vacutainer®, BD Biosciences, Mississauga, ON, Canada) from consenting volunteers. The patient donors had blood taken at the Juravinski Cancer Centre and blood was transported in a standard insulated blood sample transport box at ambient temperature to McMaster University. The average elapsed time from collection to processing was about 3h. The volunteer donor subset had blood taken in the McMaster University Radiation Biology laboratory by a trained phlebotomist.
Lymphocytes were isolated from whole blood using Histopaque-1077 by centrifugation at room temperature for 30 minutes spun at 200g according to the manufacturer's instructions (Sigma-Aldrich, Oakville, ON, Canada). The isolated lymphocytes were washed twice with complete RPMI medium (17% Fetal Bovine Serum, 0.8% L-glutamine, 0.8% Penicillin Streptomycin). The cell number was determined by a Z2 counter (Beckman Coulter, Mississauga, ON, Canada) and the final cell concentration was adjusted to 5 × 105 cells/ml in complete RPMI. Lymphocyte cell culture from each patient was placed into 8 different 25cm2 flasks each containing 5ml of media.
Sample Irradiation and Preparation
Irradiations were performed using a 137Cs source (661keV) at a dose rate of 0.1Gy/min. Two flasks containing 5ml of cell sample in media for each dose point (0, 2, 4 and 8Gy) were cooled to 0°C and held on ice during and after the irradiation. When all irradiations were completed, the flasks were all warmed to 37°C simultaneously in a 95% humidified incubator containing 5% CO2 and then incubated for 44h. Post incubation, three samples of 2.5 × 105 cells from each flask were placed into 5ml tubes. The tubes were centrifuged at 300g for 5min and the supernatant was removed to leave a final remaining volume of 100 μl of media used to resuspend the cells for labeling.
Flow Cytometry
Samples were analyzed with a Beckman Coulter EPICS® XL Flow Cytometer (Beckman Coulter, Mississauga, ON, Canada). Apoptosis was assessed in lymphocyte samples using two well established endpoints: Annexin V-FITC (Beckman Coulter, Mississauga, ON, Canada) and 3,3'-dihexyloxacarbocyanine iodide (DiOC6).
For the Annexin V-FITC assay, a commercially available kit (Beckman Coulter, IM3614) was used. Apoptotic cells were defined as cells that stained positive for Annexin V-FITC binding on outer cell membranes. To each tube, the following antibodies were added: 10μl 7-amino-actinomycin D (7-AAD), 5μl Annexin V-FITC, 3μl CD4-RD1, 3μl CD8-ECD and 100ml 1× Binding Buffer (Beckman Coulter, Mississauga, ON, Canada). The tubes were incubated on ice for 15min in the dark. Post incubation, 300μl of 1× Binding Buffer (Beckman Coulter) was added to each tube.
For the DiOC6 assay, apoptotic cells were identified as those with mid-range fluorescence intensity. To each tube, the following antibodies were added: 10μl 7AAD, 3μl CD4-RD1, 3μl CD8-ECD (Beckman Coulter, Mississauga, ON, Canada) and 100μl PBS. The tubes were incubated in the dark at room temperature for 10min. Post incubation 500μl of DiOC6 solution (40nM of DiOC6 in PBS) preheated to room temperature, to ensure complete solubility, was added to each tube. Samples were left to incubate for 15min in the dark at room temperature.
Apoptosis levels in the CD4 and CD8 lymphocyte subpopulations were measured at four radiation doses (0, 2, 4, 8Gy) in duplicate. Three samples per flask were stained and measured on the flow cytometer resulting in 24 sample tubes being processed for each donor. Information was collected from 10,000 lymphocytes and characterized by size, density and fluorescence signal intensity for each labeled reagent per sample.
Statistical Analysis
To determine the statistical significance of the different characteristics of radiation induced apoptosis, the data was analyzed using: student's t-test, paired t-test, ANOVA, coefficient of variation and Pearson correlation coefficients. Reproducibility was quantified using Cronbach's alpha.
RESULTS
Individual Variation
To assess differences in individual responses, apoptosis was measured at 0, 2, 4 and 8Gy using two different apoptosis assays. The response of CD4, CD8 and the total lymphocyte populations were measured (Table 1). Variation was found in both apoptosis assays, but was larger with the DiOC6 assay. Inter-individual variation was seen in for the spontaneous apoptosis (0Gy) levels and cells that had been irradiated. The variation increased with dose and the largest differences were seen at 8Gy. Spontaneous apoptosis in the CD8 lymphocytes had the largest coefficient of variation, 0.44, in the Annexin assay and 0.63 with the DiOC6 assay (Table 2). The wide range of apoptotic responses at each dose point resulted in overlapping dose responses. Mean apoptotic values using the Annexin assay were 9–34% at 0Gy, 24–56% at 2Gy, 31–68% at 4Gy and 41–79% at 8Gy.
Apoptosis in lymphocytes from a radiation exposure of 2, 4 and 8Gy.

The results are presented for all 54 individual donors for two different assays (Annexin and DiOC6) in three different populations (all lymphocytes, CD4 and CD8). The data represent the mean (% apoptosis), standard deviation and coefficient of variation for each lymphocyte population.
Mean (% apoptosis) spontaneous apoptosis levels measure in 44 donors for two different assays (Annexin and DiOC6) in three different populations (all lymphocytes, CD4 and CD8).

The data represent the mean (% apoptosis), standard deviation and coefficient of variation for each lymphocyte population.
For each dose point, three replicate tubes were analyzed on the flow cytometer. Results were reproducible and consistent within the same replicate blood sample. The average coefficient of variation (σ/μ) for all 54 donors was 0.04 for the Annexin assay and 0.09 for the DiOC6 assay. The CD4 and CD8 lymphocytes showed greater variation within an individual, with coefficient of variation values of 0.05 for the Annexin and 0.11 for the DiOC6 assay. Therefore, the Annexin assay was more reliable.
When the specific subsets of lymphocytes were analyzed, different results were observed. CD4 lymphocytes had less radiation induced apoptosis then CD8 lymphocytes at all doses for both assays. CD4 lymphocytes had the greatest degree of variation between individuals with a coefficient of variation of 0.32 for the Annexin assay and 0.39 for the DiOC6 assay at 8Gy calculated from data given in Table 1. Multivariate analysis shows that apoptosis increases with the dose given to the sample (p=0.0001 for Annexin, p=0.003 for DiOC6) and varies depending on the subset of lymphocytes investigated (p=0.0001 for Annexin, p=0.008 for DiOC6).
Age Relationship
Age variation in the donor cohort ranged from 19 to 85. Spontaneous apoptosis was modeled with age (Figures 1–6). Pearson correlation coefficients were calculated to estimate the correlation. A weak positive correlation was found in the Annexin assay, and no correlation or trend was identified in the DiOC6 assay (Table 3). Overall, no significant relationship was found between age and spontaneous apoptosis, although there may be a trend for increased spontaneous levels in older donors using the Annexin assay (Figure 5).
Mathematical modelling found a correlation between age and radiation induced apoptosis at 8Gy data.

The model was a negative linear relationship. The slope of the line (% decrease in apoptosis/year of age) is given for each assay and lymphocyte subset. Correlation coefficients describing how well the data fits a negative linear relationship are also given.
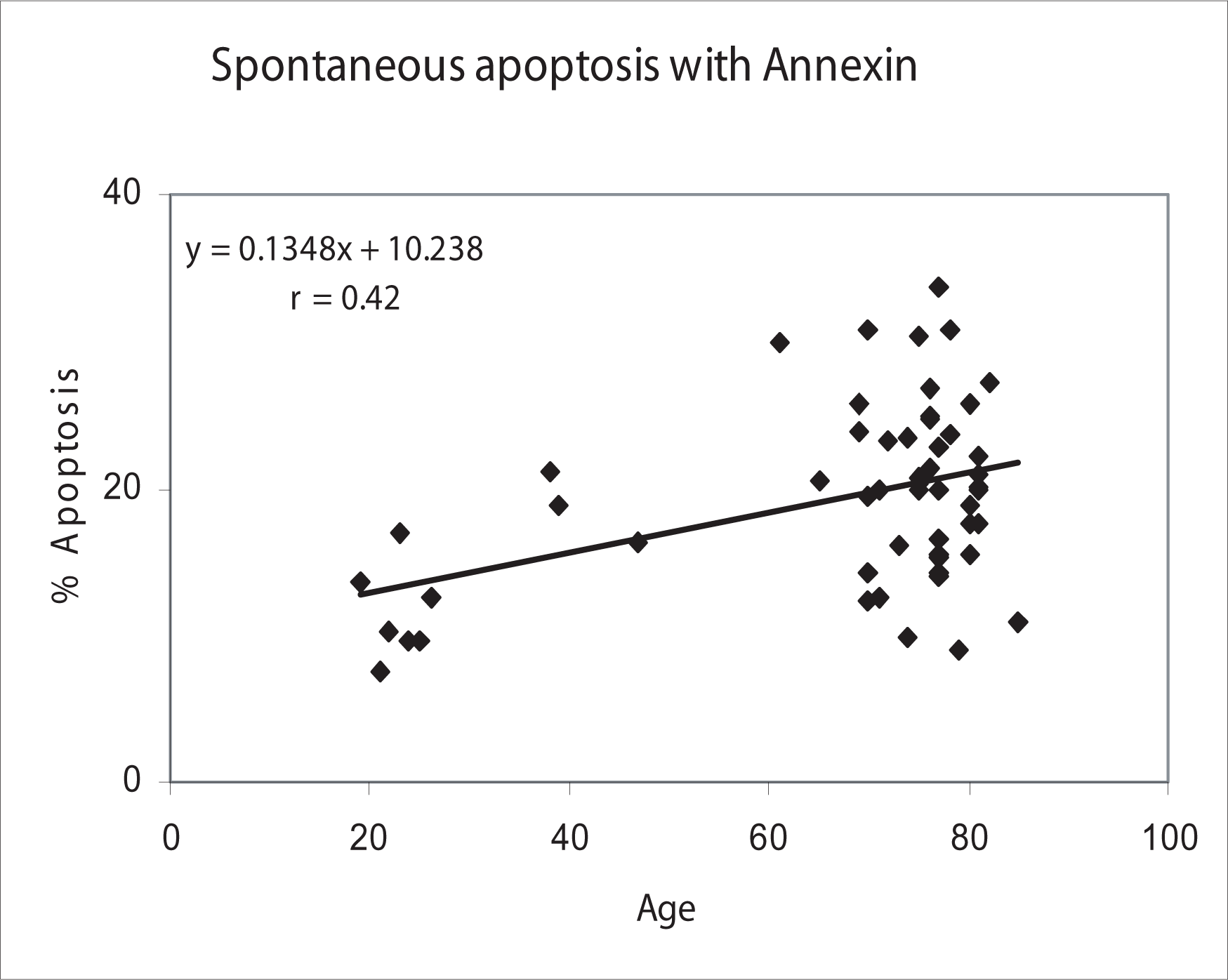
Spontaneous apoptosis levels in all lymphocytes correlated with age as measured by the Annexin assay.

Spontaneous apoptosis levels in all lymphocytes correlated with age as measured by the DiOC6 assay.
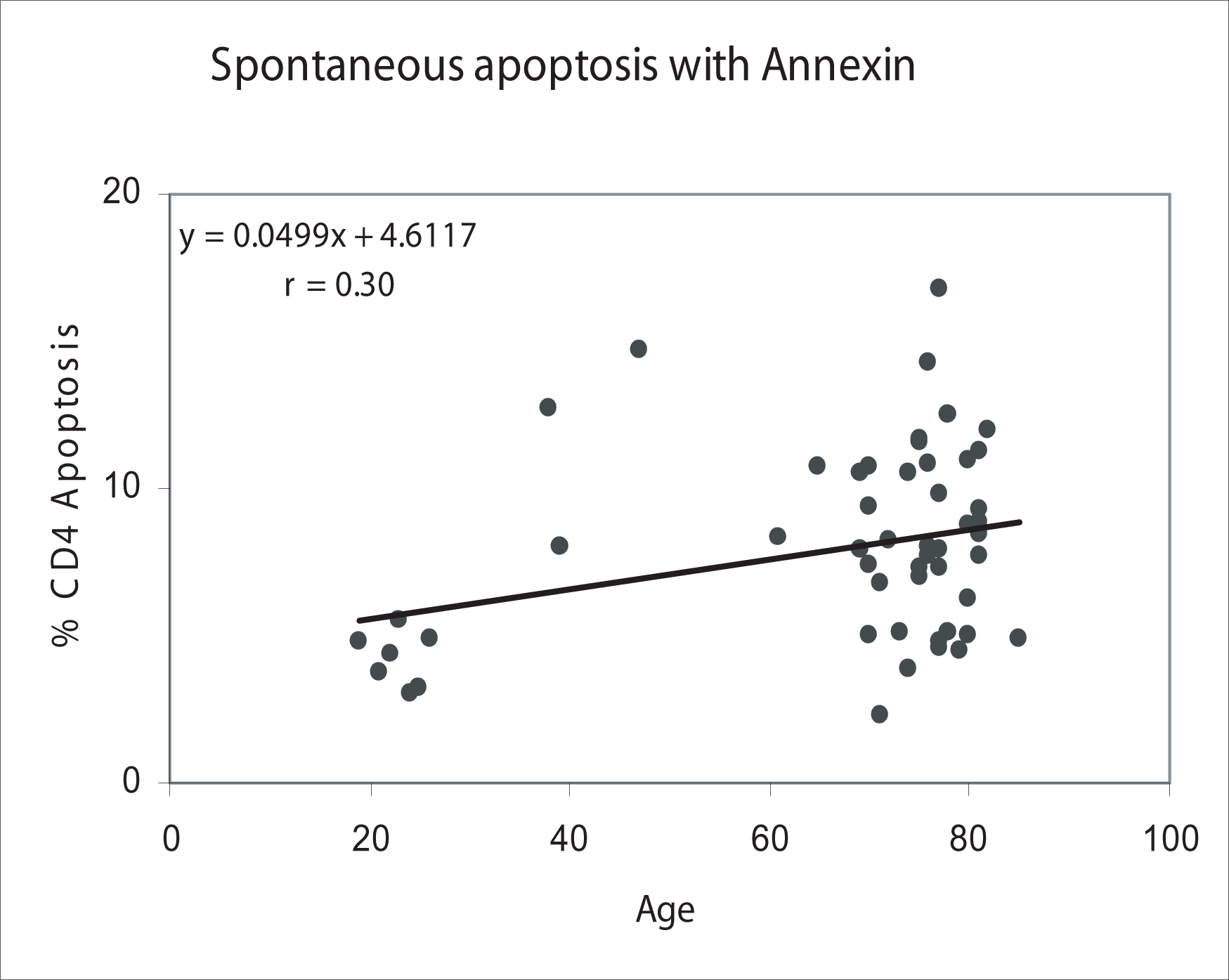
Spontaneous apoptosis in CD4 lymphocytes correlated with age as measured by the Annexin assay.

Spontaneous apoptosis in CD4 lymphocytes correlated with age as measured by the DiOC6 assay.

Spontaneous apoptosis in CD8 lymphocytes correlated with age as measured with the Annexin assay.
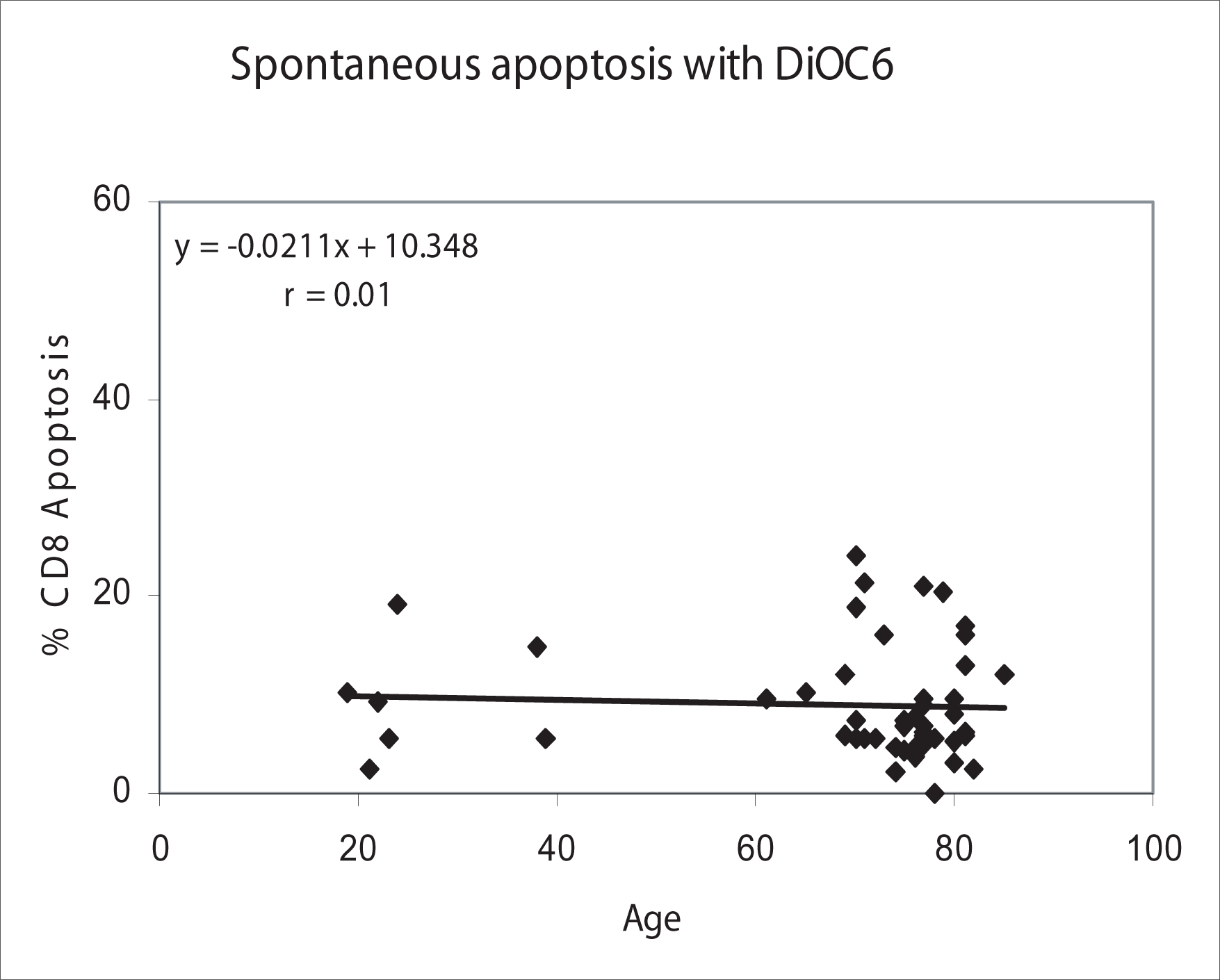
Spontaneous apoptosis in CD8 lymphocytes correlated with age as measured with the DiOC6 assay.
Radiation induced apoptosis was modeled with age (Figures 7–12). The resulting trend was fit with a negative linear relationship. The average radiation induced apoptotic response decreased as the age of the donor increased. The slopes of the linear models varied depending on the apoptosis assay used (Annexin or DiOC6) and lymphocyte subpopulation (all, CD4 or CD8 lymphocytes). Multivariate analysis showed that both the type of assay (p=0.05) and lymphocyte subset (p=0.03) were significant when determining the decrease in radiation induced apoptosis per year of age. The strength of the radiation induced apoptosis correlation varied between different lymphocyte subsets (Table 3). The CD4 lymphocytes showed a strong correlation with age (r=0.78), the all lymphocyte population slightly weaker (r=0.63 and r=0.69) and CD8 lymphocytes had the least significant trend (r=0.51 and r=0.31).
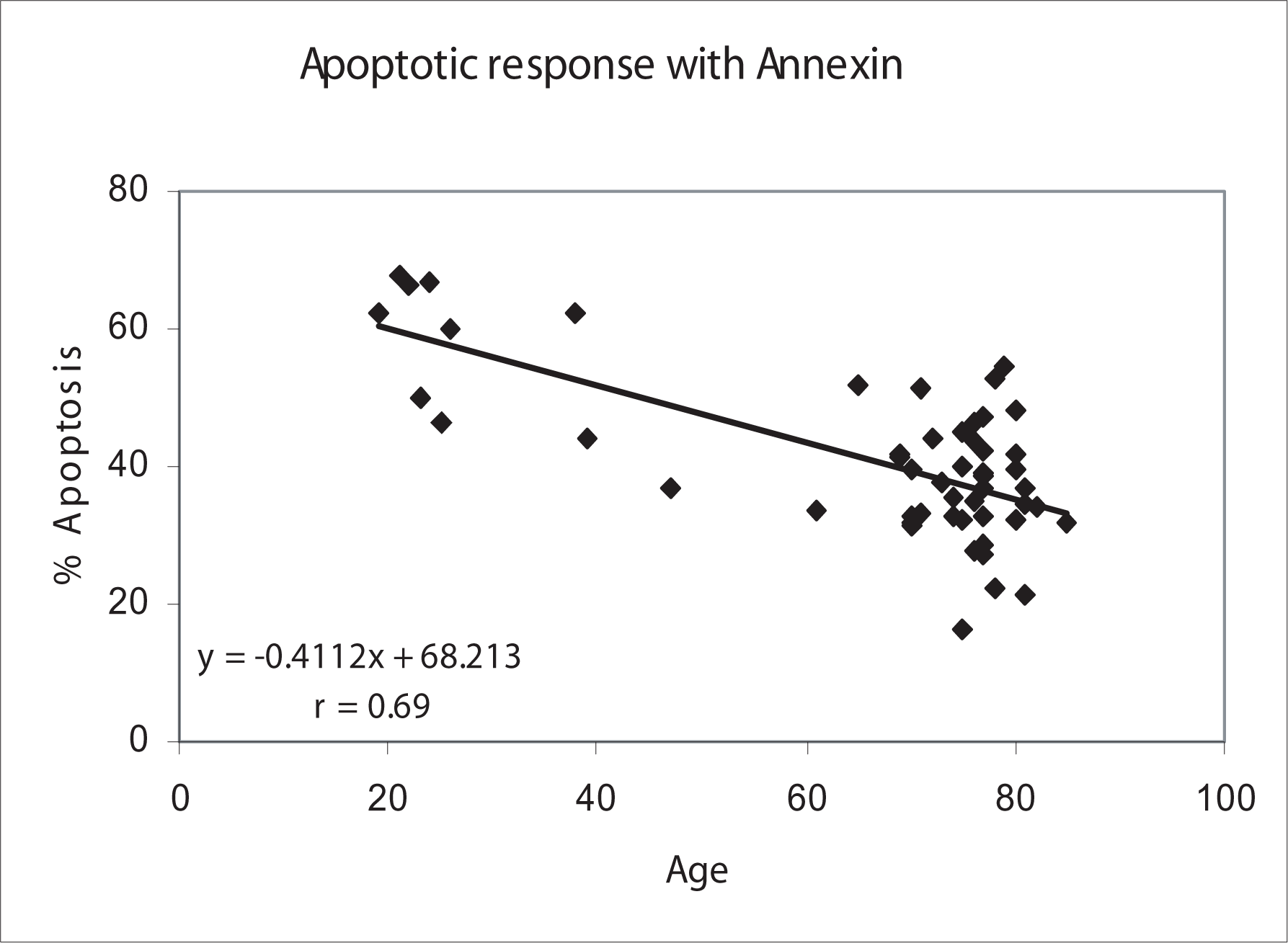
Radiation-induced (8 Gy) apoptosis in all lymphocytes correlated with age using the Annexin assay.

Radiation-induced (8 Gy) apoptosis in all lymphocytes correlated with age using the DiOC6 assay.

Radiation-induced (8 Gy) apoptosis in CD4 lymphocytes correlated with age using the Annexin assay.

Radiation-induced (8 Gy) apoptosis in CD4 lymphocytes correlated with age using the DiOC6 assay.

Radiation-induced (8 Gy) apoptosis in CD8 lymphocytes correlated with age using the Annexin assay.

Radiation-induced (8 Gy) apoptosis in CD8 lymphocytes correlated with age using the DiOC6 assay.
Dose Response
Using the apoptosis values found at 0, 2, 4 and 8Gy, dose response curves were made for each of the different lymphocyte groups (Figures 13–15). Average values for all 54 individuals were plotted with the standard deviation represented as error bars. Large variation between individuals resulted in large standard error bars on the graph and is indicative of varied sensitivity or resistance among individuals.

A mean dose response curve for induction of apoptosis in all lymphocytes for 54 donors with standard error bars.

A mean dose response curve for induction of apoptosis in CD4 lymphocytes for 54 donors with standard error bars.

A mean dose response curve for induction of apoptosis in CD8 lymphocytes for 54 donors with standard error bars.
Apoptosis Assay Correlation
Apoptosis can be characterized by a number of different biochemical and physiological changes in the cell. In order to determine if apoptosis was being measured accurately, two different assays were used. The results of the two apoptosis assays were compared to determine the correlation. Apoptosis at each dose point as measured by the Annexin was compared to apoptosis as measured by the DiOC6 assay and Pearson correlation coefficients were calculated. The CD4 and all lymphocyte populations showed a strong correlation between the two assays (r=0.90). The CD8 lymphocytes displayed a weaker correlation (r=0.77).
Reproducibility over time
Reproducibility was analysed using apoptosis levels from the 10 patients who had multiple blood draws. Cronbach's alpha was calculated to quantify reproducibility and reliability between blood samples using the Annexin assay. Spontaneous levels of apoptosis were also very reproducible and reliable (α ≥ 0.7) in the CD4+ cells and CD8+ cells. The all lymphocyte population was weakly reliable (α=0.69). Apoptotic response was reproducible in all cell populations for the doses of 2, 4 and 8Gy (Table 4).
Reproducibility of apoptosis at different doses as measured by the Annexin assay in 10 donors.

DISCUSSION
Individual variation in apoptotic response was found to be significant between the 54 male donors that were investigated. In all three lymphocyte populations, variation was seen at all dose points. The apoptotic response of the 54 individuals investigated ranged from 41–79% at 8Gy. This range of response overlaps with both the 2Gy (24–56%) and 4Gy (31–68%) dose response ranges. Therefore, a dose response for each donor at each dose point would be required to assess the relative radiation sensitivity compared to the mean response of all donors.
Variation in spontaneous background apoptosis levels of individuals was also large. Background (spontaneous) apoptosis in the Annexin assay ranged from 9% to 34%. These spontaneous levels might be related to genetically or environmentally driven endogenous processes that influence the cells response to handling and culturing during the procedure. In order to test the reproducibility of apoptosis within an individual, 10 donors had blood taken on two different occasions. Blood samples were processed the same way and the apoptotic response compared. Apoptosis as measured by the Annexin assay was found to be relatively reproducible at all doses in all individuals. Apoptosis, although varied between individuals, appears to be relatively consistent in an individual over the time span of a couple months.
The phenomenon of inter-individual variation to radiation is known. Many studies have shown large in vitro inter-individual variation and small intra-individual variation for radiation-induced apoptosis in lymphocytes (Boreham et al, 1996; Menz et al 1997; Barber et al, 2000). However, Menz et al, (1997) questioned the correlation between in vivo and in vitro apoptosis induction, and the differences in renewal, replacement and apoptosis kinetics between the two systems so in vitro results might not reflect actual in vivo sensitivity.
It is difficult to fully understand the response of all humans to radiation as the sample size of the population that has been studied to date is still relatively small. Most of what is known about radiation effects and risks comes from animal studies, a few industrial radiation accidents, or data from Hiroshima and Nagasaki (ICRU 2002). However, many medical diagnostic and therapeutic procedures use ionizing radiation and individual or population responses to these doses would be useful to understand (Daniak and Ricks 2005). Radiation therapy patients are generally given 2Gy a day fractions to their tumour, and a smaller dose to the surrounding tissue depending on the treatment plan. In the clinic, these patients respond to standardized radiation treatments with a range of clinical symptoms to their normal tissue or organs. Some patients respond well to the treatment, while others may have acute toxicity (erythema of skin) at the time of treatment, or late toxicity (necrotic tissue, loss of organ function) developing many months after therapy. Since these people are responding with a range of responses to a known similar dose of radiation, individual variation is a concern for the patient and oncologist.
Currently, exactly what contributes to an individual's radiation sensitivity is not known. Schmitz et al, (2003) have studied the radiation response of lymphocytes. They have concluded that the extent of apoptotic induction at spontaneous levels or by radiation differs in all lymphocyte subpopulation and between individuals. They hypothesize that these differences could be the result of a number of controlled functional polymorphisms, and are linked to the genes that decide whether a cell commits to apoptosis or repair.
Other research on the relationship between age and apoptosis has been conducted. A decrease in levels of radiation induced apoptosis with increasing age was reported in studies investigating CD3, CD4 and CD8 lymphocytes (Wistop et al, 2005; Ozsahin et al 1997). The data from these studies agreed with the data found in this study. In the research reported here, we show a strong correlation between CD4+ cells and age (r=0.63–0.78) which is greater than any other previously published results (r=0.3) (Ozsahin, 1997). However, the work of Schmitz et al, (2003) found a different trend. They found a positive relationship with age and radiation-induced apoptosis (r=0.38) for 63 individuals within the CD8 lymphocytes. This area of work needs further examination before a conclusion can be reached but differences in protocols, procedures, and cell populations could account for some of the apparently contradictory results. What is agreed upon is that the age relationship is important and perhaps normalization for age would have to occur before an individual's actual sensitivity could be predicted (Radojcic and Crompton 2001).
Over the last decade research into the direct effects of radiation on humans has been performed. With this research we now have a better understanding of the differences between individuals. The results of this study and other studies similar in nature should not be overlooked with respect to estimating individual risk. Radiation does not induce the same biological response in all individuals. Large inter-individual variation occurs in a population when you are looking at the effects of radiation on apoptosis. How do we know that the same amount of variation is not occurring with respect to long term risk? Since our regulation and radiation protection principals, and most therapeutic plans for cancer treatment, assume we are all similar, these results support the contention that these practices might someday need reconsideration. Inevitably, our future understanding of individual radiation responses will allow us to tailor therapies and implement radiation protection practices specific to an individual.
Footnotes
ACKNOWLEDGMENTS
We would like to acknowledge the expert technical assistance of Nicole McFarlane for her outstanding skills in flow cytometry, Jan Barclay and the Clinical Trials department at the Juravinski Cancer Centre for accruing and consenting patients and Ashley Hodgins for her technical assistance. This research was supported by the Canadian Association for Radiation Oncologists (Abbott – CARO Uro-Oncologic Radiation Award) the CBRN Research and Technology Initiative (CRTI).
