Abstract
Persistent organic pollutants (POPs) are long-lived toxic organic compounds and are of major concern for human and ecosystem health. Although the use of most POPs is banned in most countries, some organochlorine pesticides are still being used in several parts of the world. Although environmental levels of some POPs such as polychlorinated biphenyls (PCBs) have declined, newly emerging POPs such as polybrominated diphenyl ethers (PBDEs) have been increasing considerably. Exposure to POPs has been associated with a wide spectrum of effects including reproductive, developmental, immunologic, carcinogenic, and neurotoxic effects. It is of particular concern that neurotoxic effects of some POPs have been observed in humans at low environmental concentrations. This review focuses on PCBs as a representative chemical class of POPs and discusses the possible mode(s) of action for the neurotoxic effects with emphasis on comparing dose-response and structure-activity relationships (SAR) with other structurally related chemicals. There is sufficient epidemiological and experimental evidence showing that PCB exposure is associated with motor and cognitive deficits in humans and animal models. Although several potential mode(s) of actions were postulated for PCB-induced neurotoxic effects, changes in neurotransmitter systems, altered intracellular signalling processes, and thyroid hormone imbalance are predominant ones. These three potential mechanisms are discussed in detail in vitro and in vivo. In addition, SAR was conducted on other structurally similar chemicals to see if they have a common mode(s) of action. Relative potency factors for several of these POPs were calculated based on their effects on intracellular signalling processes. This is a comprehensive review comparing molecular effects at the cellular level to the neurotoxic effects seen in the whole animal for environmentally relevant POPs.
Keywords
INTRODUCTION
Tens of thousands of chemicals are produced or handled in a modern industrial society. Some of these chemicals are generated unintentionally as contaminants in the course of various manufacturing processes. Both laboratory and epidemiological studies have shown that many of these chemicals are hazardous to human health if present in excessive quantities in our food or the environment in which we live. Persistent organic pollutants (POPs) are long-lived toxic organic compounds such as polychlorinated biphenyls (PCBs), organochlorine pesticides, and dioxins. These chemicals are major concern for human and ecosystem health due to their high degree of persistence. Although the use of most POPs are banned in most countries, some organochlorine pesticides are still being used in parts of the world, which could lead to the environmental pollution of the neighboring regions, or even the entire world. In addition, several tons of these POPs are still available for release into the environment (Lang, 1992, ATSDR, 1999). For example, it has been estimated that more than 1.2 × 109 kg of PCBs have been manufactured throughout the world; 3.7 × 108 kg of PCBs have already been released into the environment, and an additional 7.8 × 108 kg are still available or deposited in different ways (Lang, 1992). These POPs could be released with time, remain in the environment for long period of time due to their high persistence, and bioaccumulate in the food web. Even though concentrations of POPs in the natural environment have fallen, PCBs and other long-known POPs cannot be dismissed as being of no significance to human health (SEPA, 1998). Since PCBs represent a significant group of POPs, the focus of this review will be on this group of chemicals discussing the neurotoxic effects and possible mode(s) of action with emphasis on dose-response in vitro, in vivo, and structure-activity relationships.
Polychlorinated biphenyls (PCBs) belong to a group of pollutants called polyhalogenated aromatic hydrocarbon family. This family includes other pollutants such as polychlorinated dibenzo-p-dioxins, polychlorinated dibenzofurans (PCDF), polychlorinated diphenyl ethers, DDT, and polybrominated diphenyl ethers (PBDEs) (Figure 1). PCBs exist as mixtures containing up to 209 possible congeners and are ubiquitous environmental contaminants resulting from intensive industrial use and inadequate disposal over past decades (Erickson, 1986). PCB mixtures as well as congeners possess a surprising array of biological activity leading to toxicity (WHO, 1993). It is known that some PCBs and other halogenated hydrocarbons such as 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) produce some of their biological effects through a receptor-mediated response by binding to the cytosolic aryl hydrocarbon (Ah) receptor followed by induction of a number of genes (Safe, 1994; Okey et al., 1994). It has been proposed that non-ortho substitutions on the biphenyl ring (lateral substitutions at the meta- and para- positions) promote coplanarity and this is associated with the TCDD-like toxic effects of some coplanar PCB congeners (McKinney and Waller, 1994). Ah receptor involvement is associated with reproductive, immunologic, teratogenic, and carcinogenic effects of PCBs (Safe, 1994; Kafafi et al., 1993). The neurotoxic effects of PCBs, however, might not be entirely mediated through the Ah receptor (Schantz, 1996; Seegal, 1996; Tilson and Kodavanti, 1997, 1998; Kodavanti, 2004).

Structural similarities between polyhalogenated aromatic hydrocarbons and thyroxine (T4). The numbers on the structures indicate the possible position for halogens. o = ortho; m = meta; p = para; PCB = polychlorinated biphenyl; T4 = Thyroxine; PBDE = polybrominated diphenyl ether; PCDE = polychlorinated dipheyl ether; DDT = 1,1,1-trichloro-2,2-bis[p-chlorophenyl]-ethane.
HUMAN (EPIDEMIOLOGICAL STUDIES) EVIDENCE OF NEUROTOXICITY BY POPs EXPOSURE
There is sufficient evidence that PCBs can cause neurotoxicity in humans (see Table 1) and it is believed that in utero exposure is more important than lactational exposure in causing the neurotoxic effects. The toxicological properties of PCBs became evident after several well documented episodes of accidental exposures (Tilson et al., 1990; Rogan and Gladen, 1992; Sinks et al., 1992). There have been two major accidental PCB poisoning incidents; “Yusho” occurred in 1968 in Japan and “YuCheng” occurred ten years later in Taiwan. These incidents resulted in several health problems including chloracne, numbness, weakness in limbs, decreased peripheral nerve conduction velocities, developmental delays, and speech problems (Kuratsune et al., 1971; 1972; Rogan et al., 1988). The offspring of women who had ingested PCB-contaminated rice-bran cooking oil in Taiwan and Japan were perinatally exposed to PCBs. The Japanese children were hypoactive (Harada, 1976) while the Taiwanese children were hyperactive and had more behavior problems compared with unexposed controls (Chen et al., 1994). When the children were tested at ages between 7 and 12, the exposed Taiwanese children also had significantly lower verbal and full-scale IQs. They also showed prolongation of, and a significant reduction in the amplitude of auditory evoked potentials (Chen and Hsu, 1994). The YuCheng and Yusho incidents were also attributed to PCDF exposure, since rice oils were cross-contaminated with PCDFs and PCBs.
Evidence of neurotoxicity in humans and animals (monkeys, rats, and mice) following exposure to PCBs.
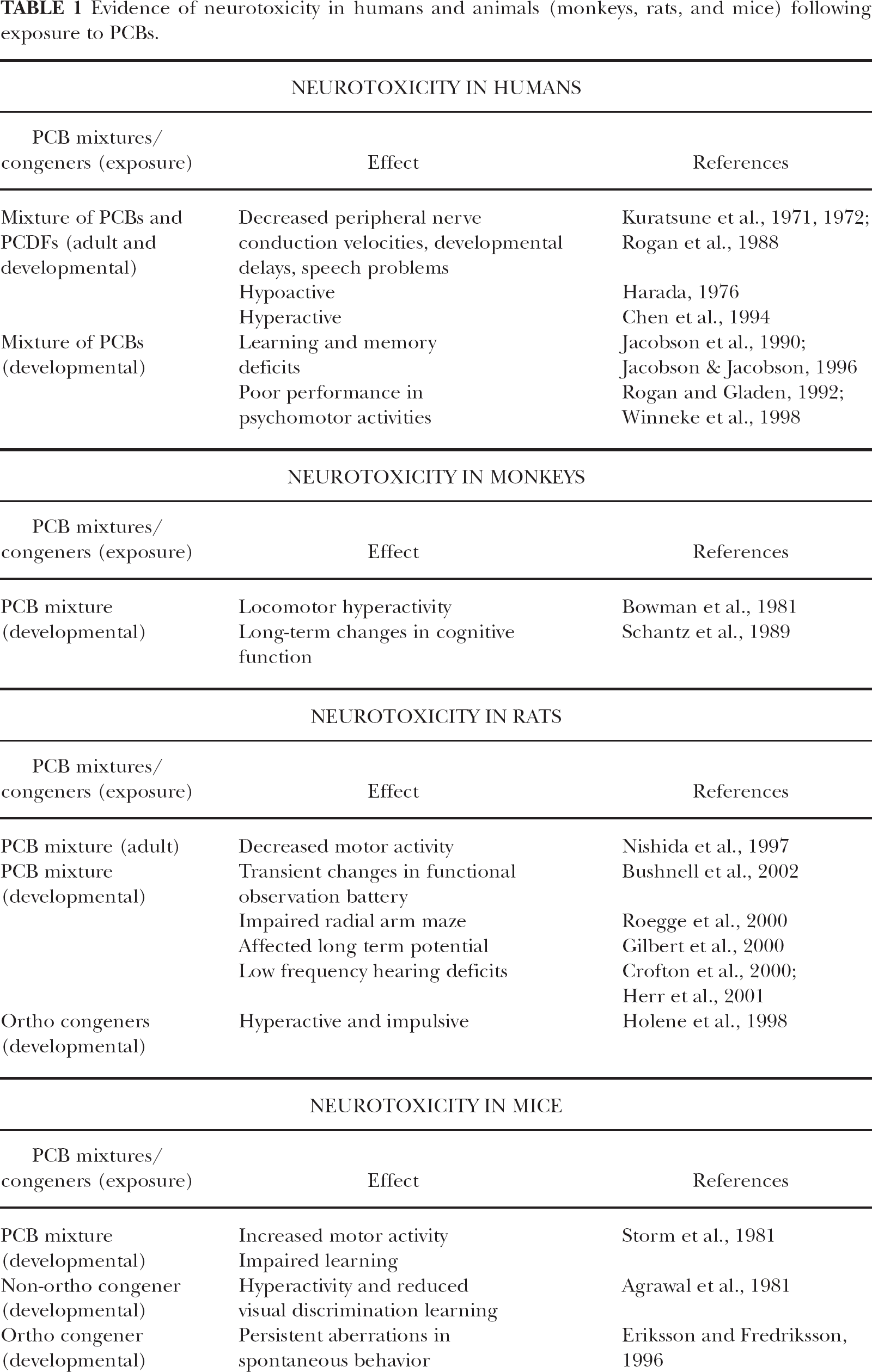



In addition to these accidental poisoning incidents, there were several epidemiological studies. Recently, Longnecker et al. (2003) compared PCB levels across ten epidemiological studies on human neurodevelopment. Jacobson et al. (1990) and Jacobson and Jacobson (1996) reported learning and memory deficits in children born to mothers, who had ingested fish, thought to be contaminated with PCBs. Rogan and Gladen (1992) reported that prenatal PCB exposure was associated with poorer performance on the Psychomotor Index from Bayley Scales of Infant Development in children from general population in North Carolina. Studies from The Netherlands (Patandin et al., 1999) also indicated that in utero exposure to “background” PCB concentrations is associated with poorer cognitive functioning in preschool children. Studies from Germany (Winneke et al., 1998; Walkowiak et al., 2001) also indicated that PCB exposure is associated with deficits in mental and motor scores in children up to 7-months of age. Studies from Faroese birth cohort in Denmark indicated that cord PCB concentration was associated with deficits in learning, however, these results may be confounded by mercury and suggest a possible interaction between these two neurotoxicants (Grandjean et al., 2001). These accidental poisoning incidents and epidemiological studies suggest that the nervous system, especially during development, is sensitive to exposure to PCBs and related chemicals.
EVIDENCE OF NEUROTOXICITY IN ANIMALS BY POPs EXPOSURE
Several studies using experimental animals support the results observed in humans (for reviews, see Seegal and Schantz, 1994; Tilson and Kodavanti, 1997; Kodavanti, 2004; and Table 1). Behavioral changes and learning deficits have been observed in monkeys (Schantz et al., 1989), rats (Pantaleoni et al., 1988), and mice (Eriksson and Fredriksson, 1996). Neurobehavioral changes were observed in adult animals following acute exposure (Nishida et al., 1997) as well as in animals exposed perinatally during development (Schantz et al., 1995). In general, neurotoxicity studies have used different PCB dosing (e.g., mixtures versus individual congener), different dosing regimens (gestational, lactational, perinatal or adults), and different dosing pattern (acute versus chronic or repeated), and these methodological factors are important in explaining some inconsistencies in the literature. In many of these animal studies, functional deficits were observed in the absence of reduced body weight or overt signs of toxicity. Animals that were prenatally, postnatally or perinatally exposed to PCBs showed many of the behavioral characteristics indicative of Attention Deficit Hyperactivity Disorder (AD/HD). For example, in utero exposure of mice led to increased motor activity and impaired learning (Storm et al., 1981), combined pre- and post-natal exposure lead to higher activity levels and reduced visual discrimination learning in rats (Agrawal et al., 1981). Holene et al. (1998) showed that male rats exposed to 2,2′,4,4′,5,5′-hexachlorobiphenyl (PCB153) during lactation became hyperactive and impulsive. Rhesus monkeys exposed throughout gestation and lactation showed locomotor hyperactivity during their first year of life (Bowman et al., 1981). Mice exposed during brain “growth spurt” on postnatal day 10 to 2,4,4′-trichlorobiphenyl and 2,2′,5,5′-tetrachlorobiphenyl exhibited persistent aberrations in spontaneous behavior while 2,3′,4,4′,5-penta — and 2,3,3′,4,4′,5-hexa-chlorobiphenyls did not (Eriksson and Fredriksson, 1996). Studies by Bushnell et al (2002) indicated that perinatal exposure to Aroclor 1254 caused only transient changes in some measures of functional observational battery without any changes in motor activity.
Apart from effects on motor activity, PCBs have been shown to decrease cognitive function in rats, non-human primates, and mice. Holene et al. (1995) reported that developmental exposure to 3,3′,4,4′,5-pentachlorobiphenyl (PCB 126) and 2,3′,4,4′,5-pentachlorobiphenyl (PCB 118) at doses that did not produce maternal or fetotoxicity interfered with visual discrimination learning of rats in an operant task. Schantz et al (1995) reported that developmental exposure to ortho-substituted PCBs significantly interfered with acquisition of the spatial alternation task in females, but not in males. Studies also indicated that gestational and lactational exposure to a commercial PCB mixture, Aroclor 1254, impaired radial arm maze performance in male rats (Roegge et al., 2000) and affected long-term potentiation in the hippocampus (Niemi et al., 1998; Gilbert et al., 2000). In non-human primates, developmental exposure to commercial PCB mixtures (Aroclor 1016 or Aroclor1248) resulted in long-term changes in cognitive function (Schantz et al., 1989). Mice exposed to 2,2′,5,5′-tetrachlorobiphenyl on postnatal day 10 (during brain “growth spurt”) exhibited deficits in learning and memory while 2,3′,4,4′,5-penta- and 2,3,3′,4,4′,5-hexa-chlorobiphenyls did not (Eriksson and Fredriksson, 1996).
Other effects seen with developmental exposure to PCBs include sensory deficits. The most significant effect seen with developmental exposure to PCBs was deficits in hearing; the auditory threshold at low frequency (1 kHz) was significantly increased (Crofton et al., 2000; Herr et al., 2001). These effects of PCBs on hearing deficits seen at a very low frequency threshold in rats may not be of biological significance in humans. Herr et al (2001) also indicated that developmental PCB exposure has no significant effect on visual-, somatosensory- or peripheral nerve-evoked potentials. Geller et al. (2001) reported gender-specific effects of PCBs on neurobehavioral development on measures of acquisition and sensory function, as seen with some behavioral endpoints. These animal studies clearly indicated that developmental exposure to PCBs resulted in cognitive dysfunction, motor deficits, and hearing loss.
POSSIBLE MODE(S) OF ACTION
Although there is both epidemiological and experimental evidence that developmental exposure to PCBs causes motor and cognitive deficits in humans and animals (see Table 1), the underlying cellular or molecular mechanisms are not known (Kodavanti and Tilson, 1997, 2000; Tilson and Kodavanti, 1997). There are several potential mechanisms postulated for the developmental neurotoxicity of PCBs. This review will focus on three predominant hypotheses; changes in neurotransmitters, altered intracellular signaling processes, and thyroid hormone imbalance.
Dose-response effects of POPs on Neurotransmitters
It has been hypothesized that changes in neurotransmitters such as dopamine (DA) levels may be associated with the observed neurobehavioral changes. A study by Chou et al. (1979) in weaning mice indicated that approximately half of the surviving offspring exhibited a syndrome of rapid spinning and head bobbing when the moms were exposed to 3,3′,4,4′-tetrachlorobiphenyl (PCB 77) from gestational days 10–16, at a dose of 32 mg/kg/day. A similar observation was made by Agrawal et al (1981) indicating that mice exposed developmentally to PCB 77 had elevated levels of motor activity and these changes were associated with decreased DA concentrations and receptor binding sites in the corpus striatum. In contrast, studies by Seegal (1994) found that developmental exposure to commercial PCB mixture, Aroclor 1016, significantly increased DA concentrations in substantia nigra, caudate nucleus and nucleus accumbens, while there were no effects on DA metabolites, norepinephrine, serotonin or its metabolites. These observations were different from those seen after acute and repeated exposure of Aroclor mixtures in adults where significant decreases in DA concentrations were observed (Seegal et al., 1986, Seegal et al., 1991). However, in utero and lactational exposure to 2,2′,4,4′-tetrachlorobiphenyl (ortho substituted congener, PCB 47) resulted in a decrease in DA concentrations in frontal cortex and caudate nucleus that persisted into adulthood while exposure to PCB 77 (non-ortho substituted congener) resulted in increases in DA concentrations in the same brain regions (Seegal et al., 1997). The effects of PCBs were not restricted to dopamine function only. Morse et al. (1996a) reported that developmental exposure to a PCB mixture, Aroclor 1254, resulted in persistent serotonergic effects, while no effects on dopaminergic system were observed. Also, Juarez de Ku et al. (1994) reported that developmental exposure to a PCB mixture affected hippocampal and forebrain cholinergic system. Eriksson and Fredriksson (1996) reported decreases in cholinergic nicotinic receptors in mice with deficits in spontaneous motor activity and learning following exposure to PCBs during brain “growth spurt”.
These in vivo observations on neurotransmitters were accompanied by several in vitro studies to understand the mechanism and structure-activity relationships (SAR) of PCB congeners. Seegal and his colleagues focused on dopamine. As seen in vivo with ortho-substituted congeners and Aroclor mixtures, DA concentrations in PC12 cells also decreased in a concentration-dependent manner following exposure in vitro. Studies with 43 PCB congeners to understand SAR found that congeners with ortho- or ortho- and para-chlorine substitutions were most potent in decreasing DA concentrations in PC12 cells (Shain et al., 1991; Table 2). Further studies investigated the mechanism by which PCB congeners inhibited DA synthesis using neuroblastoma cells (N1E-N115), which lack the enzyme, aromatic amino acid decarboxylase. This decarboxylase enzyme converts 3,4-dihydroxylphenylalanine (DOPA), formed by hydroxylation of tyrosine, to dopamine. These studies indicated that PCBs appear to decrease cellular DA concentrations by inhibiting the activity of the rate-limiting enzyme, tyrosine hydroxylase (Seegal et al., 1991). Other studies with rat striatal slices indicated that ortho-substituted PCB congeners inhibited tyrosine hydroxylase activity in vitro (Choksi et al., 1997). However, these in vitro results were not confirmed by in vivo studies in adult rats where tyrosine hydroxylase immunoreactivity in forebrain or enzyme activity in striatum was not significantly altered following repeated exposure to Aroclor 1254 (Kodavanti et al., 1998a). In the same animals, however, neurotransmitter levels in striatum were slightly altered by PCB exposure. Recent studies also indicated that tyrosine hydroxylase expression was not affected by acute exposure to Aroclors 1016 or 1260 (Richardson and Miller, 2004). There are other studies indicating effects of PCBs on the high affinity uptake of the neurotransmitters in to rat brain synaptic vesicles and synaptosomes (Mariussen et al., 1999; Mariussen and Fonnum, 2001). These studies indicated that several ortho-substituted PCBs inhibited neurotransmitter uptake in to rat brain synaptosomes; dopamine uptake was inhibited more efficient that that of glutamate. On the other hand, non-ortho PCBs did not have any effect supporting the SAR of Shain et al. (1991). In addition, recent reports indicated that inhibition of vesicular monoamine transporter might be involved in the reductions of synaptosomal dopamine content by PCBs (Bemis and Seegal, 2004; Richardson and Miller, 2004). Based on these in vivo and in vitro studies, a possible role of neurotransmitters in PCB neurotoxicity could not be ruled out.
The reason for selecting different polychlorinated biphenyl (PCB) mixtures, polybrominated diphenyl ether (PBDE) mixures, congeners, dioxin, and furan to study the structure-activity relationships (SAR) † .






Note:
The numbers under congeners category indicate the position of chlorines on the biphenyl ring.
Source: See Erickson (1986), Safe et al. (1987), Barnes (1991), and WHO (1993). (Adapted from Kodavanti and Tilson, Neurotoxicology 18: 425–442, 1997).
Dose-response effects of POPs on intracellular signaling
Intracellular signaling or signal transduction can be defined as a mechanism by which extracellular signals are transferred to the cytosol and nucleus of the cell. These signals are essential not only for the function of the nervous system, but also play a key role in nervous system development (Murphy et al., 1987; Girard and Kuo, 1990). Any interference with these processes would have the potential for profound effects on the function of neuron as well as their development. Growth factors, neurotransmitters, and hormones serve as first messengers to transfer information from one cell to another by interacting with specific cell membrane receptors. This interaction leads to activation or inhibition of specific enzymes and/or opening of ion channels, which results in changes in intracellular metabolism, leading to a variety of effects including activation of protein kinases and transcription factors. These intracellular pathways can be activated by separate receptors, and there is significant cross talk between the receptor signals, so they can control and modulate each other. In recent years, investigators have considered a number of signal transduction pathways as potential targets for neurotoxicants (Costa, 1998; Nihei et al., 2001; Limke et al., 2003). Neurotoxicants may alter any step beginning from the extracellular stimulus all the way to the transcriptional level leading to alterations in the structure or function of the nervous system. Disrupted intracellular signaling is a potential mechanism by which chemicals might alter nervous system function either directly or through alterations in nervous system morphology. Thus, brain function and/or morphology may be affected by neurotoxicants through changes in intracellular signaling at critical phases during development (Kodavanti, 2004).
It has been hypothesized that PCBs exert their neurotoxic effects by disrupting intracellular signaling/second messenger homeostasis. This hypothesis was based on the following reasons: a) the most significant neurotoxic effects of PCBs seen in humans are learning and memory deficits (Jacobson and Jacobson, 1996; Patandin et al., 1999); b) laboratory studies indicate that PCBs inhibit long-term potentiation (LTP, a form of synaptic plasticity) and impair learning/memory in vivo (Schantz et al., 1995; Niemi et al., 1998; Gilbert et al., 2000); c) LTP is often described as a physiological model for neuronal development, learning and memory (Lynch, 1998); d) second messengers such as calcium, inositol phosphates, PKC, arachidonic acid (AA), and nitric oxide synthase (NOS) have been shown to modulate LTP and play key roles in neuronal development (Lynch, 1998); and e) epidemiological studies indicate that infants born to mothers who consumed contaminated cooking oil in Yusho, Japan showed abnormal calcification of the skull indicating that PCBs/PCDFs might interfere with calcium metabolism (Yamashita and Hayashi, 1985).
A number of studies have been conducted both in vitro using neuronal cultures as well as brain homogenate preparations and in vivo which involved developmental exposure to a PCB mixture, Aroclor 1254, to understand the role of second messengers in the PCB-induced neurotoxicity. In vitro studies have been conducted with prototypic ortho (2,2′-dichlorobiphenyl, DCB) and non-ortho (3,3′,4,4′,5-pentachlorobiphenyl; PeCB or 4,4′-DCB) PCBs followed by an extensive analysis of structure-activity relationships with more than 35 PCB congeners and several analogs (Tables 2 –4). PCB-induced alterations in intracellular free Ca2+ ([Ca2+]i) measured with a fluorescent dye (Fluo-3AM) were reported for the first time; the ortho-substituted 2,2′-DCB was more effective than the non-ortho-substituted 3,3′,4,4′,5-PeCB (Kodavanti et al., 1993). The increase in [Ca2+]i was slow, and a steady rise was observed with time (Kodavanti et al., 1993). Follow up studies confirmed these observations in cerebellar granule neurons (Carpenter et al., 1997; Mundy et al., 1999; Bemis and Seegal, 2000), cortical neurons (Inglefield and Shafer, 2000; Inglefield et al., 2002), and in human granulocytes (Voie and Fonnum, 1998). Studies characterizing the mechanisms by which PCBs increase [Ca2+]i indicated that 2,2′-DCB was an inhibitor of 45Ca2+-uptake by mitochondria and microsomes with IC50 (concentration which inhibits control activity by 50%) values of 6–8 μM. 3,3′,4,4′,5-PeCB inhibited Ca2+-sequestration, but the effects were much less than those produced by equivalent concentrations of 2,2′-DCB. Further studies indicated that Aroclor 1254 inhibited both microsomal and mitochondrial Ca2+-sequestration uniformly in selected brain regions, however, inhibition of microsomal Ca2+-sequestration increased with age (Sharma et al., 2000). Synaptosomal Ca2+-ATPase, involved in Ca2+-extrusion process, was inhibited by 2,2′-DCB, but not by 3,3′,4,4′,5-PeCB (Kodavanti et al., 1993). Further structure-activity relationship (SAR) studies indicated that congeners that are noncoplanar inhibited 45Ca2+-uptake by microsomes and mitochondria while coplanar congeners did not (Kodavanti et al., 1996a).
The disruption of Ca2+-homeostasis may have a significant effect on other signal transduction pathways (e.g., inositol phosphate [IP] and AA second messengers) regulated or modulated by Ca2+. The congener 2,2′-DCB, but not 3,3′,4,4′,5-PeCB affected basal and carbachol (CB)-stimulated IP accumulation in cerebellar granule cells (Kodavanti et al., 1994). Further studies indicated that any modulation of CB-stimulated IP accumulation is due to Ca2+-overload, but not due to activation of PKC activity (Kodavanti et al., 1994; Shafer et al., 1996). AA is released intracellularly following activation of membrane phospholipases, and AA is an important second messenger in releasing Ca2+ from endoplasmic reticulum (Striggow and Ehrlich, 1997). Aroclor 1254 (a commercial mixture of PCBs) and 2,2′-DCB increased [3H]-AA release in cerebellar granule cells while 4,4-DCB did not (Kodavanti and Derr-Yellin, 1999); this is in agreement with previous structure-activity relationship studies on Ca2+ buffering and PKC translocation (Kodavanti and Tilson, 1997). A similar increase in [3H]-AA was observed with structurally similar chemicals such as PBDE mixtures (Kodavanti and Derr-Yellin, 2002). Further studies indicated that the [3H]-AA release caused by these chemicals could be due to activation of both Ca2+ dependent and -independent phospholipase A2 (PLA2).
One of the down-stream effects of perturbed Ca2+-homeostasis is translocation of PKC from the cytosol to the membrane where it is activated (Trilivas and Brown, 1989). [3H]-Phorbol ester ([3H]-PDBu) binding has been used as an indicator of PKC translocation. The congener 2,2′-DCB increased [3H]-PDBu binding in a concentration-dependent manner in cerebellar granule cells, while 3,3′,4,4′,5-PeCB had no effect with concentrations up to 100 μM. The effect of 2,2′-DCB was time-dependent, and also dependent on the presence of external Ca2+ in the medium. Sphingosine, a PKC translocation blocker, prevented 2,2′-DCB-induced increases in [3H]-PDBu binding (Kodavanti and Tilson, 2000). Experiments with several pharmacological agents revealed that the effects are additive with glutamate, and none of the channel (glutamate, calcium, and sodium) antagonists blocked the response of 2,2′-DCB (Kodavanti et al., 1994). Immunoblots of PKC-alpha and epsilon indicated that non-coplanar ortho-PCB decreased the cytosolic form and increased the membrane form significantly at 25 μM (Yang and Kodavanti, 2001).
These studies were further extended to understand the structure-activity relationship (SAR) of a number of PCB congeners and related chemicals. The reason for the selection of these chemicals is provided in Table 2. The results indicated several structural requirements to exhibit their activity in neuronal cells. Congeners with ortho-substitutions, especially together with lateral substitutions such as 2,2′,4,6-tetrachlorobiphenyl and 2,2′,4,6,6′-pentachlorobiphenyl, are most active (Tables 3 and 4). Congeners without ortho-substitutions such as 3,3′,4,4′-tetrachlorobiphenyl and 3,3′,4,4′,5-pentachlorobiphenyl, which assume more coplanar configurations tend not to be active (Tables 3 and 4). The data presented in Tables 3 and 4 clearly indicated that the potency of the PCB congeners in neurons is more directly associated with the structural configuration of PCB congeners than their hydrophobicity. The data also indicate that low lateral substitution, especially without para-substitution, or lateral content in the presence of ortho-substitution, may be the most important structural requirement for the in vitro activity of these congeners in neuronal preparations. SAR studies conclude that congeners that are noncoplanar increased PKC translocation while coplanar congeners did not (Tables 3 and 4; Kodavanti et al., 1995; Kodavanti and Tilson, 1997 and 2000; Fischer et al., 1998). This was further strengthened by the observations with structurally similar chemicals such as DDT, polychlorinated diphenyl ethers and polybrominated diphenyl ethers (Tables 3 and 4; Kodavanti et al., 1996b). Nitric oxide (NO), which is produced by NOS, is a gaseous neurotransmitter. NO has an important role as a retrograde messenger in LTP, learning and memory processes, and endocrine function (Schuman and Madison, 1994; McCann et al., 1998). The congener 2,2′-DCB, but not 4,4′-DCB, inhibited both cytosolic (nNOS) and membrane (eNOS) forms of NOS (Sharma and Kodavanti, 2002).
Comparative effects of persistent organic pollutants (POPs) in vitro on intracellular signaling (PKC translocation and microsomal Ca2+-buffering), neurotransmiiters (dopamine), and thyroid hormones (binding to transthyretin). Values shown are the mean concentrations, which caused a 50% change (increase or decrease). At these concentrations, there was no cytotoxicity. The numbers in parenthesis and bold indicate the relative potency against 2,2′,5,5′-tetrachlorobiphenyl based on the effects on PKC translocation.







Chauhan et al., (2000). Blank spaces indicate that the data are not available. NEO = no effect observed up to 100 μM.
Relative potency of POPs on protein kinase C (PKC) translocation, determined by measuring increases in [3H]PDBu binding in rat cerebellar granule cells in vitro. The congeners vary with different position and number of halogens.
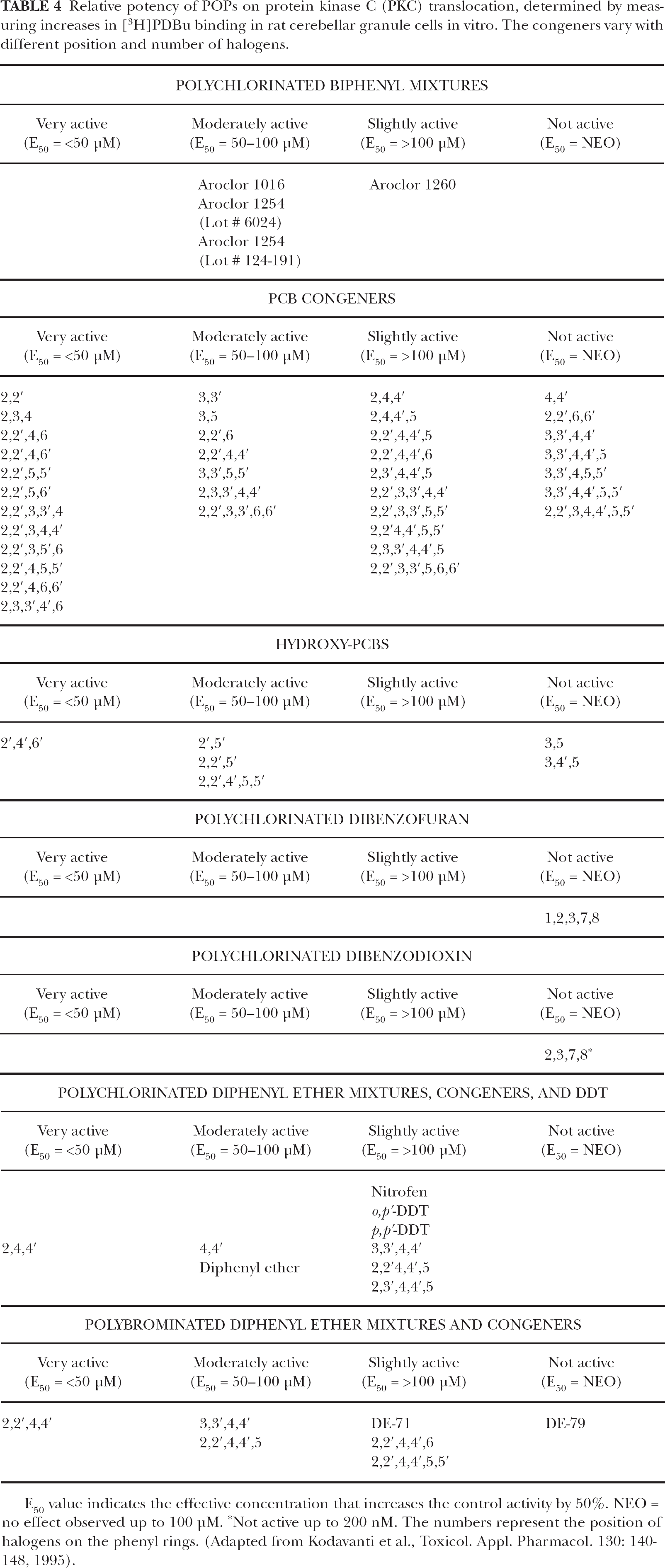






E50 value indicates the effective concentration that increases the control activity by 50%. NEO = no effect observed up to 100 μM.
Not active up to 200 nM. The numbers represent the position of halogens on the phenyl rings. (Adapted from Kodavanti et al., Toxicol. Appl. Pharmacol. 130: 140–148, 1995).
These in vitro studies clearly demonstrated that second messenger systems, involved in the development of the nervous system, LTP, and learning and memory, are sensitive targets for the ortho-substituted PCBs and related chemicals. PCBs (ortho-PCBs and commercial PCB mixtures) affected intracellular signaling events at low micromolar concentrations and shorter exposure periods, where cytotoxicity is not evident. These signaling pathways include calcium homeostasis and PKC translocation. The rise of intracellular free Ca2+ is slow, but steady following exposure. This free Ca2+ rise could be due to increased calcium influx, inhibited Ca2+ buffering mechanisms, and/or calcium release from intracellular stores by the products of membrane phospholipases. The increases in free Ca2+ levels may lead to the translocation of PKC. The coplanar non-ortho-PCBs have marginal effects on calcium homeostasis and no effects on PKC translocation. Literature reports indicate that at slightly higher concentrations, commercial PCB mixtures (Aroclors 1221 and 1254) have been known to alter neurite outgrowth in PC12 cells (Angus and Contreras, 1995) and hypothalamic cells (Gore et al., 2002). PCBs have also been shown to inhibit thyroid hormone-dependent extension of dendrites of cerebellar Purkinje cells (Kuroda, 2003). It also appears that ortho-substituted PCBs can kill cerebellar granule cells by altering membrane structure (Tan et al., 2004) and it is possible that this structural change could be secondary to changes in intracellular signaling by these chemicals.
In vivo effects of PCBs on intracellular signaling have been studied with a commercial PCB mixture, Aroclor 1254, following adult or developmental exposures. Following exposure to adult rats, PCB mixture produced changes in second messenger systems that are similar to those observed after in vitro exposures (Kodavanti et al., 1998a) and concentrations that altered second messenger systems in vitro are achievable in brain in vivo (Kodavanti et al., 1998b). Following developmental exposure (given orally from gestational day 6 through postnatal day 21), Arolcor 1254 did not alter maternal body weight or percent mortality but caused a small, transient decrease in body weight gain of offspring. Both calcium homeostasis and PKC activities were significantly affected following developmental exposure to Aroclor 1254 (Kodavanti et al., 2000). Developmental exposure to PCBs also caused significant hypothyroxinemia and age-dependent alterations in the translocation of PKC isozymes; the effects were significant at postnatal day (PND) 14 (Yang et al., 2003). Immunoblot analysis of PKC-alpha (α), -gamma (γ), and -epsilon (ε) from both cerebellum and hippocampus revealed that developmental exposure to Aroclor1254 caused a significant decrease in cytosolic fraction and an increase in particulate fraction. For some isozymes, the ratio between the two fractions was increased in a dose-dependent manner. Thus, the patterns of subcellular distributions of PKC isoforms following a developmental PCB exposure were PKC isozyme- and developmental stage-specific (Yang et al., 2003). The changes in PKC and other second messengers were associated with changes in transcription factors such as Sp1 and NF-kB indicating changes in gene expression following developmental exposure to PCBs (Riyaz Basha et al., 2006). PCBs have been shown to suppress thyroid hormone-dependent gene expression (Iwasaki et al., 2002). Considering the significant role of PKC signaling in motor behavior, learning and memory (Murphy et al., 1987; Chen et al., 1997), it is suggested that altered subcellular distribution of PKC isoforms at critical periods of brain development may be associated with activation of transcription factors and subsequent gene expression, and may be a possible mechanism of PCB-induced neurotoxic effects. PKC-α, γ, and ε may be among the target molecules implicated in PCB-induced neurological impairments after developmental exposure.
Further studies focused on the structural consequences of changes in the intracellular signaling pathway following developmental PCB exposure. Detailed brain morphometric evaluation was performed by measuring neuronal branching and spine density. In utero and lactational exposure to PCBs affected normal dendritic development of Purkinje cells and CA1 pyramids in animals at postnatal day 22 (Mervis et al., 2002). The branching area was significantly smaller in the PCB-exposed rats. At adulthood, there was continued neurostructural disruption of the CA1 dendritic arbor, but the branching area of the Purkinje cells returned to normal levels. Developmental exposure to PCBs also resulted in a significantly smaller spine density in hippocampus, but not in cerebellum. This dysmorphic cytoarchitecture could be the structural basis for long-lasting neurocognitive deficits in PCB-exposed rats (Mervis et al., 2002).
Previously, Pruitt et al (1999) reported a reduced growth of intra- and infra-pyramidal mossy fibers following developmental exposure to PCBs. Nguon et al. (2005) also reported that perinatal exposure to PCBs differentially affected the development of cerebellum in male and female rats. Considering the importance of intracellular signaling in neuronal development (Kater and Mills, 1991), these studies indicate that developmental exposure to a PCB mixture resulted in altered cellular distribution of PKC isoforms, which can subsequently disrupt the normal maintenance of signal transduction in developing neurons. The perturbations in intracellular signaling events could lead to structural changes in the brain. These findings suggest that altered subcellular distribution of PKC isoforms may be a possible mode of action for PCB-induced neurotoxicity.
Dose-response effects of POPs on hormonal imbalance
The function and development of the brain are influenced by hormones. Thyroid hormones (TH) have profound effects on neurological function in people of all ages. TH increase the rate of neuronal proliferation and stimulate differentiation (Lauder, 1977). Once neurons are formed, they follow an orderly pattern of migration to the appropriate areas in the brain. TH deficiency has been shown to cause disorganization of brain (Nicholson and Actman, 1972; Nunez, 1984). TH also stimulate formation and development of neuronal processes, both axons and dendrites (Legrand et al., 1982; Nunez, 1984; Kimura-Kuroda et al., 2002). TH are synthesized as mostly T4 (thyroxine) and small amounts of T3 (3,3′,5-triiodothyronine) in the thyroid gland by the uptake of iodide from the blood and are stored as amino acid residues of thyroglobulin. The T4 and T3 are released into circulation for transport to different organs. TH synthesis and secretion are regulated by a sensitive feedback mechanism by hypothalamic-pituitary axis (Figure 2). In the circulating human blood, T4 and T3 bind to the proteins such as thyroxine-binding globulin (TBG), transthyretin (TTR), and albumin with decreasing order of affinity. However, the high affinity TBG is not present in rodents, birds, amphibians or fish (Capen, 1992). Although TTR is not the major T4 transport protein in humans, TTR is thought to be essential for T4 transport to brain in humans (Herbert et al., 1986). In addition, TTR is the only TH binding plasma protein that is synthesized by both in liver and brain. TTR is reported to serve a role in mediating the delivery of T4 across the blood-brain barrier and the maternal to fetal transport through placenta (Schreiber et al., 1995; Southwell et al., 1993). TTR also plays an essential role in the determination of free T4 levels in the extracellular compartment of the brain, which is independent of T4 homeostasis in the body (Schreiber et al., 1995). Free TH in circulation is excreted through UDP glucuronidation (Figure 2). Once circulating T4 reaches different organs of the body, they are deiodinated by deiodinases releasing T3. Type I deiodinase is prevalent in liver while type II deiodinase is prevalent in brain (Visser et al., 1982; Kaplan et al., 1983). These hormones bind to the nuclear TH receptors to elicit their functional role.

Disruption of thyroid hormone homeostais by polychlorinated biphenyls and the possible mechanisms. The solid arrow in blue shows the normal process for TH production and clearance. The solid arrow in red shows the predominant pathway for the effects of PCBs on TH homeostasis. The binding of PCBs to transport proteins in blood not only results in the reduction of circulating TH levels, but also gets transported to the organs to elicit an adverse effect. TRH, Thyrotropin-releasing hormone; TSH, Thyroid-stimulating hormone; TBG, Thyroxine-binding globulin; TTR, Transthyretin; T4, Thyroxine; UDPGT, UDP-glucuronyltransferase.
Many environmental contaminants including POPs alter TH function, either inhibiting the thyroidal system or mimicking it. Among the pollutants, PCBs and dioxins have dramatic effects on TH function in humans (Koopman-Esseboom et al., 1994; Langer et al., 1998; Nagayama et al., 1998), wildlife species (Brouwer et al., 1989; Van den Berg et al., 1994) and in experimental animals (Morse et al., 1993; Goldey et al., 1995; Zoeller et al., 2002). PCBs have been shown to decrease circulating TH levels during development (Morse et al., 1993; Crofton et al., 2000) and in adult animals (Porterfield, 1994; Kodavanti et al., 1998a). PCBs have also shown to decrease thyroid hormones in rat brain and stimulate type II deiodinase activity in brain (Morse et al., 1996b). Many studies have been performed to elucidate the mechanism(s) involved in POP-induced alterations in TH levels. In general, the decrease in circulating TH levels by chemicals could be at least by 3 known mechanisms. First, the chemical can directly affect the thyroid gland to decrease the synthesis of TH (Collins and Capen, 1980). Second, reduced TH levels can be caused by enhanced biliary excretion of T4 due to the induction of UDP-glucuronyltransferases (UDPGT; Van Birgelen et al., 1995). Third, the chemical may displace natural ligand, T4, binding to TTR (Lans et al., 1993; Morse et al., 1996b; Chauhan et al., 2000). Although all these three mechanisms might be responsible for the decreases in circulating TH by POPs (parent compound and metabolites), high affinity binding of POPs to TTR might be the key factor in eliciting the biological effects. Binding of POPs to TTR has been hypothesized to result in (1) a selective retention of POPs in plasma, (2) facilitated transport of the POPs over the placenta to the fetal compartment, and (3) decreased maternal and fetal plasma T4 levels by competition with the natural ligand, T4 (reviewed by Brouwer et al., 1998). In addition, POPs will be reaching the target organ such as brain by facilitated transport where POPs can bind to the target sites and elicit biological effect.
Studies by Chauhan et al. (2000) indicated the importance of ortho-substitution on PCB-binding to TTR. Hydroxy-PCBs have high affinity to TTR compared to the parent PCBs. The PCB congeners with di-meta substitution in one or both rings, which most closely resembling the diiodophenolic ring of T4, showed the highest TTR binding activity. PCB congener patterns with single meta substitution in one or both rings, which more closely resembling the monoiodophenolic ring of T3 showed significantly lower TTR binding activity. The binding potencies of several ortho-PCBs seem to follow the biological activities such as decreases in PC12-cellular dopamine content and PKC translocation in neuronal cells (Table 3). Among the PCBs, differential effects on circulating TH were observed based on the coplanarity. PCB congeners that are non-coplanar such as 2,2′,4,4′,5,5′-hexachlorobiphenyl (PCB153) decreased circulating T4 levels to a greater extent (>90%) than coplanar PCBs such as 3,3′,4,4′,5-pentachlorobiphenyl (PCB 126) (Craft et al., 2002). However, PCB 126 has high binding affinity towards TTR compared to PCB 153 (Chauhan et al., 2000). As shown in the Figure 2, both coplanar and noncoplanar PCBs enter the hepatic portal system through gastro-intestinal absorption from the food. Since liver has high content of Ah-receptor proteins, coplanar PCBs bind to these proteins with high affinity and only small amount returns to the circulation. On the other hand, most of the noncoplanar PCBs return to circulation since they have very low or no affinity for the Ah-receptor proteins. Although coplanar PCBs have strong affinity for TTR, the concentrations in the target organs could be very low due to their sequestration in liver. In support of this, Saghir et al (2000) reported that the accumulation of 14C-PCB169 in liver was 3-fold higher compared to 14C-PCB153 and serum levels of 14C-PCB153 were about twice as high as those of 14C-PCB169. Accumulation of 14C-PCB153 in brain was 4- to 9-fold higher than that of 14C-PCB 169. Recent evidence also suggests that binding of PCBs to TTR might play a key role in decreasing circulating TH. Meerts et al. (2000) reported that perinatal exposure to hydroxylated PCB 107 (4-hydroxy 2,3,3′,4′,5-pentachlorobiphenyl) resulted in 89% decrease in total T4 levels in the absence of any increase in hepatic T4-UDP glucuronyltransferase or type I deiodinase activity. They have also demonstrated that the 14C-labeled PCB was bound to TTR in both maternal and fetal plasma. Hallgren and Darnerud (2002) reported that T4 decreases is mainly due to disturbances in serum transport, caused by binding of in vivo formed PCB and PBDE metabolites. Further, Kuriyama et al. (2003) reported exposure to PCB118 during development resulted in decreased circulating T4 levels by 42% at weaning without any changes in liver weight gain or induction of EROD activity. In other studies, microsomal inducers including phenobarbital, PCB, 3-methylcholanthrine, and pregnenolone-16α-carbonitrile caused induction of T4-UDP glucuronidation to a similar extent while similar doses caused decreases in serum T4 levels to a different extent (Hood et al., 2003) suggesting that mechanisms other than T4-UDP glucuronidation are responsible for decreases in circulating T4 levels. These studies led to insight that binding of PCBs to TTR may play an important role in PCB-induced decreases in circulating thyroid hormones and that the neurotoxic consequences following exposure to these chemicals may not be dependent on Ah-receptor activation. Even though in humans thyroxine binding globulin is the main TH transport protein in the blood, TTR still plays a role in mediating the delivery of T4 across the blood-brain barrier, transporting T4 into the cerebrospinal fluid and transferring maternal T4 to the fetal through placenta (Calvo et al., 1990; Southwell et al., 1993). In order to exert biological effect, TH should bind to the thyroid hormone receptors (TR) in the nucleus. Although some in vitro studies showed interference of PCBs with TR thereby affecting TH-sensitive genes (Bogazzi et al., 2003) and TR-mediated transcription (Miyazaki et al., 2004), other in vivo studies showed that PCBs exert TH-like effects in the fetal brain but do not bind to TR (Gauger et al., 2003). On the other hand, Kumura-Kuroda et al. (2005) reported that hydroxy metabolites of PCBs inhibit TH-dependent extension of cerebellar Purkinje cell dendrites. Additional studies are needed to confirm the role of TH in PCB-induced neurotoxicity.
Table 3 shows the concentrations, which altered neurochemcal parameters by 50% of control activity. It is obvious that there is a good correlation among the effects of PCB congeners on all three neurochemical endpoints. For PKC translocation, 2,2′,5,5′-tetrachlorobiphenyl seems to be the most active congener. Using this congener as a base, the relative potencies of other PCB congeners and structurally related chemicals were calculated (Table 3). Since the effects of PCBs on intracellular signaling, neurotransmitters, and thyroid hormone homeostasis occurred at biologically and environmentally relevant concentrations, these potential mechanisms might be involved in the neurotoxic effects of PCBs. The reasons are: (1) PCBs affected intracellular signaling, neurotransmitters, and thyroid hormone homeostasis at low micromolar concentrations; (2) Low micromolar concentrations accumulated in rat brain regions; (3) intracellular signaling, neurotransmitters, and thyroid hormone homeostasis are crucial for nervous system development and neuronal function; (4) PCBs affected neuronal development in vitro and in vivo, and caused learning and memory deficits in humans and animals.
In addition to the mechanisms mentioned above, PCBs have been shown to alter the cholinergic system (Juarez de Ku et al., 1994) and cause oxidative stress (Mariussen et al., 2002; Lee and Opanashuk, 2004). It has also been demonstrated that inhibitors of nitric oxide synthase and phospholipase A2 led to a significant reduction of oxidative stress and cell death (Mariussen et al., 2002). Although studies on these systems are limited, both of them have a significant role in the function of the nervous system. Additional studies are needed to test whether one or more of these mechanisms are responsible for the effects of PCBs in the nervous system. Figure 3 shows the proposed mode of action for the neurotoxic consequences following exposure to PCBs. It is obvious from Tables 3 and 4 that polyhalogenated aromatic hydrocarbons (PAHs) can be separated as non-coplanar and coplanar PAHs. Both parent compounds and their hydroxy metabolites have differential effects on the three proposed mechanisms. Non-coplanar parent compounds affected intracellular signalling, thyroid hormone homeostasis, and neurotransmitters. The effects on intracellular signalling include calcium homeostasis, inositol phosphate and ryanodine receptors, membrane phosholipases, and protein kinases. The neurotransmitters include dopamine and serotonin. The hydroxy metabolites of PCBs affected intracellular signalling endpoints with equal potency, but affected thyroid hormone homeostasis to a greater extent (Table 3). On the other hand, coplanar parent compounds have effects only on thyroid hormones but no effects on neurotransmitters and intracellular signaling. The coplanar metabolites have been shown to affect intracellular signaling and thyroid hormones. In the case of non-coplanar PAHs, all three proposed mechanisms might be working together to cause neurotoxic response while coplanar PAHs could be working through one or two of the proposed mechanisms to cause neurotoxic response (Figure 3). In any case, the changes in these proposed pathways could lead to changes in the gene expression of several transcription factors (e.g., Sp1, Ap1, and NF-kB) that may lead to altered neuronal plasticity and development, and finally lead to a neurotoxic response such as motor dysfunction or learning and memory deficits.

Possible mechanism(s) for developmental neurotoxicity of persistent organic pollutants (e.g., PCBs). The polyhalogenated aromatic hydrocarbons (PAHs) can be separated as non-coplanar and coplanar compounds. Non-coplanar parent compounds and hydroxy metabolites affected intracellular signalling, thyroid hormone (TH) homeostasis, and neurotransmitters. On the other hand, coplanar parent compounds have effects only on TH while no effects on neurotransmitters and intracellular signaling. The coplanar metabolites have been shown to affect intracellular signaling and TH. In the case of non-coplanar PAHs, all three proposed mechanisms might be working together to cause neurotoxic response while coplanar PAHs could be working through one or two of the proposed mechanisms to cause neurotoxic response. The changes in these proposed pathways could lead to changes in the gene expression of several transcription factors that may lead to altered neuronal plasticity and development, and finally lead to a neurotoxic response such as motor dysfunction or learning and memory deficits.
FURTHER CONSIDERATIONS
Recently, there has been growing recognition of an apparent increase in the incidence of developmental disabilities. About 17% of children under 18 years old (nearly 12 million) in the USA suffer from deafness, speech deficits, cerebral palsy, delay in growth and development, emotional or behavioral problems, or learning disabilities (Boyle et al., 1994) and this number is increasing (CDC, 2003). Improved reporting and differing diagnostic criteria may explain some but not all of the trends (Schettler, 2001). Although genetic factors have a key role in these developmental disabilities, environmental chemicals may also be causative factors for various developmental disorders of the brain, such as Attention Deficient Hyperactive Disorder (ADHD) and/or autism.
Developmental neurotoxicity involves alterations in behavior, neurophysiology, neurochemistry, and/or gross dysmorphology of the nervous system occurring in the offspring as a result of chemical exposure in utero and during lactation. Compared to adults, the developing organism has its own physiologic characteristics, uptake characteristics, and possible inherent susceptibilities. The developing nervous system may be uniquely sensitive to toxicants, since there is rapid growth of the brain, known as “brain growth spurt”. In rodents, this period is entirely neonatal (the first 3–4 postnatal weeks). In humans, the “brain growth spurt” begins during the third trimester of pregnancy and continues throughout the first years of life. During this period, the brain goes through several developmental processes starting from neurogenesis to proliferation, migration, and differentiation. Chemical exposure during any of these processes could have detrimental effects on the establishment of normal brain structure, current brain functioning and indirectly on subsequent brain functioning in adulthood. Intracellular signaling pathways are crucial for the normal function and development of the nervous system. The intracellular second messengers generated in this process offer key steps in the mode of action for developmental neurotoxicity of a number of chemicals including PCBs (see Figure 1 for other structurally related chemicals) and provide good scientific basis for more quantitative and biologically defensible human health risk assessment.
Polybrominated diphenyl ethers (PBDEs) are good examples where information obtained from structurally related chemicals such as PCBs could be applied. PBDEs are used as flame-retardants in textiles, electrical equipment, plastics, and building materials. PBDEs are structurally similar to PCBs with ten substitutions for halogens and 209 possible congeners based on position and number of halogens (Figure 1). All PBDE congeners are non-coplanar in nature because of the structural configuration. Based on SAR results with PCBs, one can predict that PBDEs should be active in the nervous system. In agreement with this prediction, PBDEs have been shown to affect signal transduction pathways (Kodavanti and Derr-Yellin, 2002; Kodavanti, 2003; Kodavanti and Ward, 2005; Kodavanti et al., 2005; Reistad and Mariussen, 2005), alter circulating thyroid hormones (Hallgren et al., 2001), and cause learning and memory deficits in mice and rats (Eriksson et al., 2001; Viberg et al., 2004; Dufault et al., 2005). Although human evidence for the developmental neurotoxicity of PBDEs is not available, there is considerable public health concern since PBDEs have been detected in human blood, adipose tissue, and breast milk and the levels are rapidly rising in wildlife, humans, and the environment (Gill et al., 2004). Due to the continued use of PBDEs in consumer products and their bioaccumulative nature, attention must be paid for the potential health risks associated with exposure to these and other structurally related chemicals (McDonald, 2005). PBDEs have similar effects on the nervous system like PCBs and other structurally related chemicals (Eriksson et al., 2001; Kodavanti and Derr-Yellin, 2002; Kodavanti, 2003). PCBs and PBDEs have been shown exert neurobehavioral effects (Eriksson et al., 2001) and cause changes in intracellular signaling pathways in neuronal cells (Kodavanti et al., 2004) at equimolar doses/concentrations suggesting a common mode of action for these chemicals. Considering the structural similarities of PBDEs and PCBs and the known health effects of PCBs in humans, these two groups of chemicals (may be other structurally related chemicals) could conceivably work through the same mechanism(s), to cause developmental neurotoxicity.
Footnotes
ACKNOWLEDGMENTS
The author is grateful for the graphic assistance of Mr. John Havel and for the editorial comments of Mr. Tom Ward of USEPA, Dr. Frode Fonnum of Norwegian Research Establishment, Dr. Jae-Ho Yang of Catholic University of Daegu, Dr. Nigel Walker of NIEHS, and Dr. Wayne Bowers of Health Canada on an earlier version of this manuscript. This manuscript was reviewed by the NHEERL, U.S. Environmental Protection Agency, and approved for publication. Mention of trade names or commercial products does not constitute endorsement or recommendation for use. The opinions expressed by the authors are not to be misconstrued as U.S. Environmental Protection Agency policy.
