Abstract
We report the case of a previously healthy 32-year-old man presenting with severe headache on 2 separate expeditions to Cho Oyu (8201 m). No brain imaging was performed after the first expedition. On the second expedition, thrombosis of the superior sagittal sinus was detected. Investigations for hypercoagulable states, including polycythemia, were negative. He had no neurological symptoms except headache, vomiting, and slight drowsiness. In retrospect, there are strong indications that cerebral thrombosis caused his headache on the first expedition as well. Severe headache occurring at high altitude that persists despite adequate treatment for high-altitude cerebral edema should raise suspicion of a cerebrovascular disorder.
Introduction
High-altitude climbing involves several risk factors. High-altitude pulmonary edema and high-altitude cerebral edema (HACE) are potentially fatal medical conditions specific for this activity. Cerebrovascular disorders reported at altitude mimicking the symptoms of HACE include cerebral venous thrombosis (CVT), migraine, stroke or transient ischemic attack, and intracranial bleeding. 1 Differentiating between HACE and vascular catastrophes is often difficult in the field. We present a patient who developed CVT at high altitude.
Case report
On 2 separate occasions, in 2001 and 2004, the patient, a 32-year-old man, was heading expeditions to Cho Oyu (8201 m). He had no medical history or family history of venous thrombosis. He was an occasional smoker. As a former officer of an elite military unit, he was in good health with exceptional physical and mental fitness. Before his expeditions to the Himalayas, he had climbed beyond 6000 m on 4 occasions, with 6500 m as his highest elevation. He experienced no symptoms of altitude illness on any of these climbs.
Acute Headache at Cho Oyu in 2001
Day 22
The patient reached camp 2 (7200 m) in good health. His expedition team had acclimatized during a 10-day period between Kathmandu and Advanced Base Camp (ABC) (5700 m) and had spent another 12 days (including resting days) before reaching camp 2. Fluid intake for the patient varied from 2 to 4 L per day. The expedition conducted a medical project, investigating whether alterations in body weight could be used to assess water retention as a cause of altitude illness. At this point, the patient's weight loss, measured with a daily calibrated Salter Personal Scale model 621 (Salter Housewares Ltd, Kent, UK), was estimated to be 3.9% (range 0%–8.3%, median 3.9%) and he had a hemoglobin level of 17.0 g·dL−1 (range 15.0–18.0 g·dL−1, median 16.5 g·dL−1), which was measured with HemoCue Hb 201+ (HemoCue AB, Ängelholm, Sweden). Before he left for Nepal, the patient had a hemoglobin level of 15.0 g·dL−1.
Days 23 and 24
After spending the night at camp 2, the patient returned to ABC. He had not slept well, experiencing headache and nausea. The Lake Louise Consensus 2 was used to assess the degree of altitude illness. On this morning, the patient had a score of 10 (headache 3, gastrointestinal problems 2, fatigue 2, dizziness 0, and difficulty sleeping 3), consistent with severe acute mountain sickness. After an otherwise normal clinical examination by the expedition doctor, the patient received paracetamol 1000 mg for his headache and no other treatment. He recovered slightly after 24 hours in ABC; however, the headache persisted. He went to bed with retro-orbital pain, facial edema, and left-sided occipital headache. His blood pressure was 132/92 mm Hg, pulse was 90 beats/ min, respiratory rate was 20 breaths/min, and oxygen saturation was 72%. His vital signs were not significantly altered from the previous days and did not suggest serious pathology at this point, nor were any neurological findings present. The team chose to observe the patient in ABC.
Days 25 and 26
The patient's headache worsened during the night, and was described by the patient as severe and worse upon movement. His blood pressure the next morning was 148/112 mm Hg, pulse was 116 beats/min, respiratory rate was 20 breaths/min, and oxygen saturation was 78%. Ophthalmoscopy revealed probable right-sided papilledema and signs of bilateral retinal bleeding. Neurological examination proved inconclusive, with probable photophobia, slight ataxia, and drowsiness as the only findings. Because HACE was suspected, treatment was initiated with dexamethasone 8 mg (followed by 4 mg every 6 hours), acetazolamide 500 mg (followed by 500 mg every 12 hours), codeine 60 mg and paracetamol 1000 mg (given approximately 3 times daily). The patient was also given nifedipine 40 mg (followed by 40 mg every 12 hours), though high-altitude pulmonary edema was not suspected. He was evacuated by foot to base camp (4800 m), where he received 3 hours of hyperbaric treatment in a Vivesco Lifebag (Vivesco AS, Borgheim, Norway), equivalent to an altitude of 2500 m. Because of only minor improvement, he was evacuated to Kathmandu the next day.
Postdescent
The patient experienced a rather severe headache and diffuse lack of concentration for the next 6 to 8 weeks after returning to Norway. For the first 2 weeks he was not able to read a book or watch television. He complained of compromised short-term memory. The symptoms prevented him from rejoining his military unit until January 2002. By this time his condition had gradually recuperated and he was no longer symptomatic. No brain imaging was conducted. The incident was considered to be probable HACE. It was suspected that the responsibility of heading a high-altitude expedition, combined with sleep deprivation and hard physical strain, may have aggravated the condition.
Acute Headache at Cho Oyu in 2004
The patient returned to Cho Oyu as an expedition leader in September 2004. The team performed a slow acclimatization, trying not to exceed a gain in sleeping altitude beyond 350 m per day. They were cautious not to put themselves through more physical strain than absolutely necessary. No doctor accompanied this expedition; instead, the team communicated with the Norwegian National Hospital via a portable televideo communication unit. The first 20 days were uneventful and the patient acclimatized well up to camp 2 (7200 m).
Days 22 and 23
After spending the night at 7200 m, the patient returned to ABC (5700 m) to prepare for the summit. The next morning he woke up with a mild headache and diarrhea. He started treatment with acetazolamide 250 mg (followed by 250 mg every 12 hours), suspecting acute mountain sickness. Because of persistent diarrhea and possibly slight fever, his condition was thought to be complicated by a gastrointestinal infection. He therefore received ciprofloxacin 500 mg (followed by 500 mg every 12 hours) and fluids.
Day 24
After 48 hours of rest, the patient attempted to ascend toward camp 1. At the halfway point, he decided to return to ABC. He felt exhausted and had lost his appetite. No sign of neurological dysfunction was apparent to the other team members. He went to sleep feeling dehydrated, though he urinated twice during the night.
Days 25 to 28
Experiencing dyssomnia, the patient woke up with severe headache and vomited repeatedly. Evacuation was immediately conducted. In base camp, because HACE was suspected, administration of acetazolamide was continued and treatment with dexamethasone 8 mg (followed by 4 mg every 6 hours) was initiated. He was evacuated to Zangmu (2300 m). Over the next 2 days his condition improved. However, the headache was continuously present and worsened upon abrupt head movement. Except for the antibiotics, all medications were terminated.
Day 29
On the fourth day in Zangmu, the headache again worsened. The patient experienced nausea, vomiting, and slight diarrhea and was promptly evacuated to Norvic Hospital, Kathmandu.
Postdescent
Upon admission to Norvic Hospital, Kathmandu, the patient had a blood pressure of 120/80 mm Hg, pulse of 50 beats/min, and oxygen saturation of 96% on room air. He was afebrile and had normal heart, lung, and abdominal examination. Neurological examination, including ophthalmoscopy, was normal. Blood count showed no sign of infection. His hemoglobin and hematocrit levels were 16.3 g·dL−1 and 49%. Thrombocytes, bleeding time, activated partial thromboplastin time, and prothrombin time were all within normal values. No other pathology was detected, including urine analysis. A D-dimer test was not performed. The symptoms were interpreted as not classic for HACE. Because of the persistent headache, a cerebral magnetic resonance imaging was performed. The images revealed thrombosis in the posterior one third of the superior sagittal sinus (Figure 1). Additional findings included several cystic lesions above the right posterior aspect of the corpus callosum, near the interhemispheric fissure (Figure 2). Steroid treatment was continued and oral antithrombotic secondary prophylaxis with warfarin was initiated. The symptoms resolved, and the patient was discharged after 1 week.
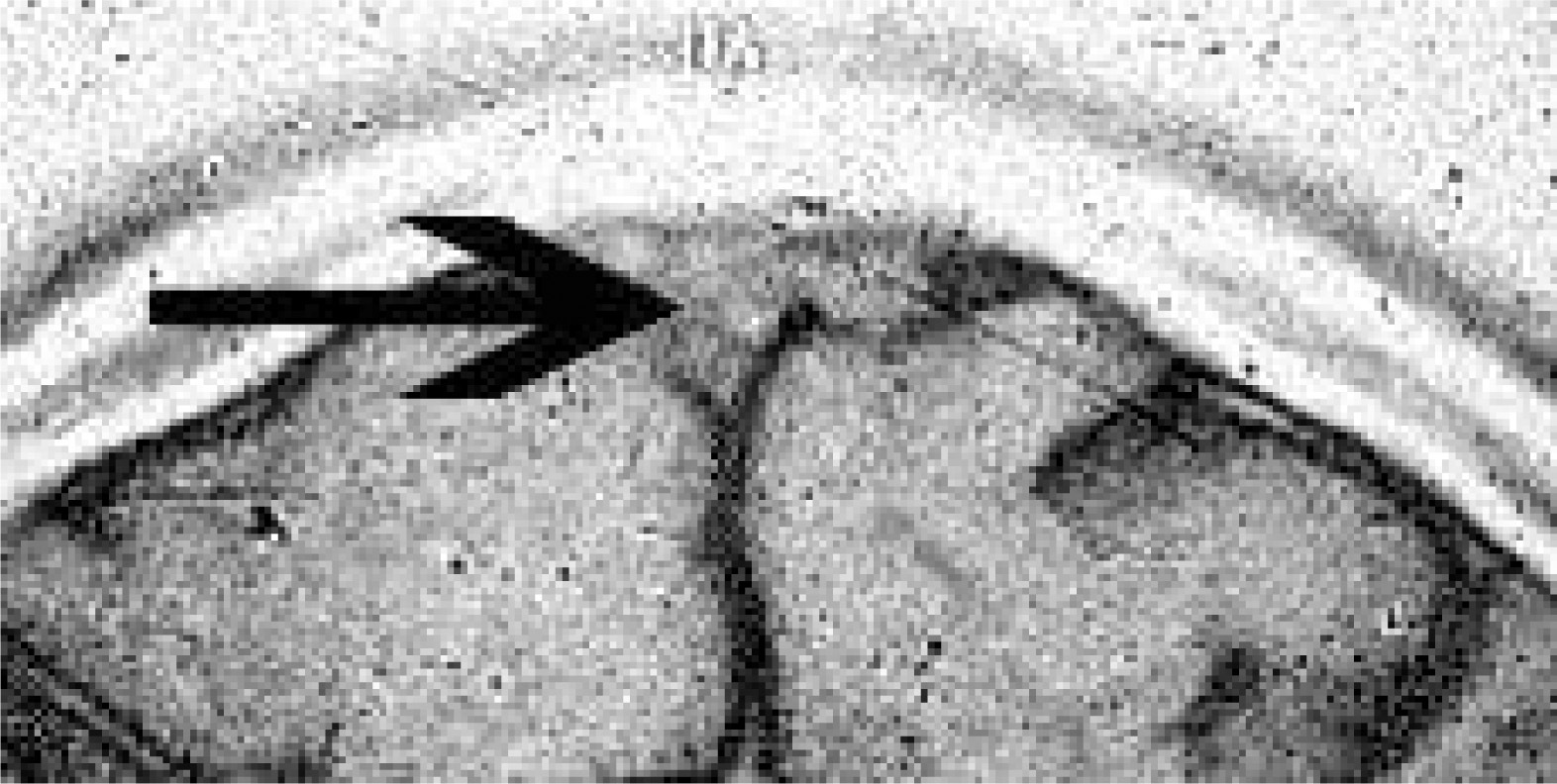
Thrombosis of the superior sagittal sinus at day 8 after onset of symptoms in 2004.
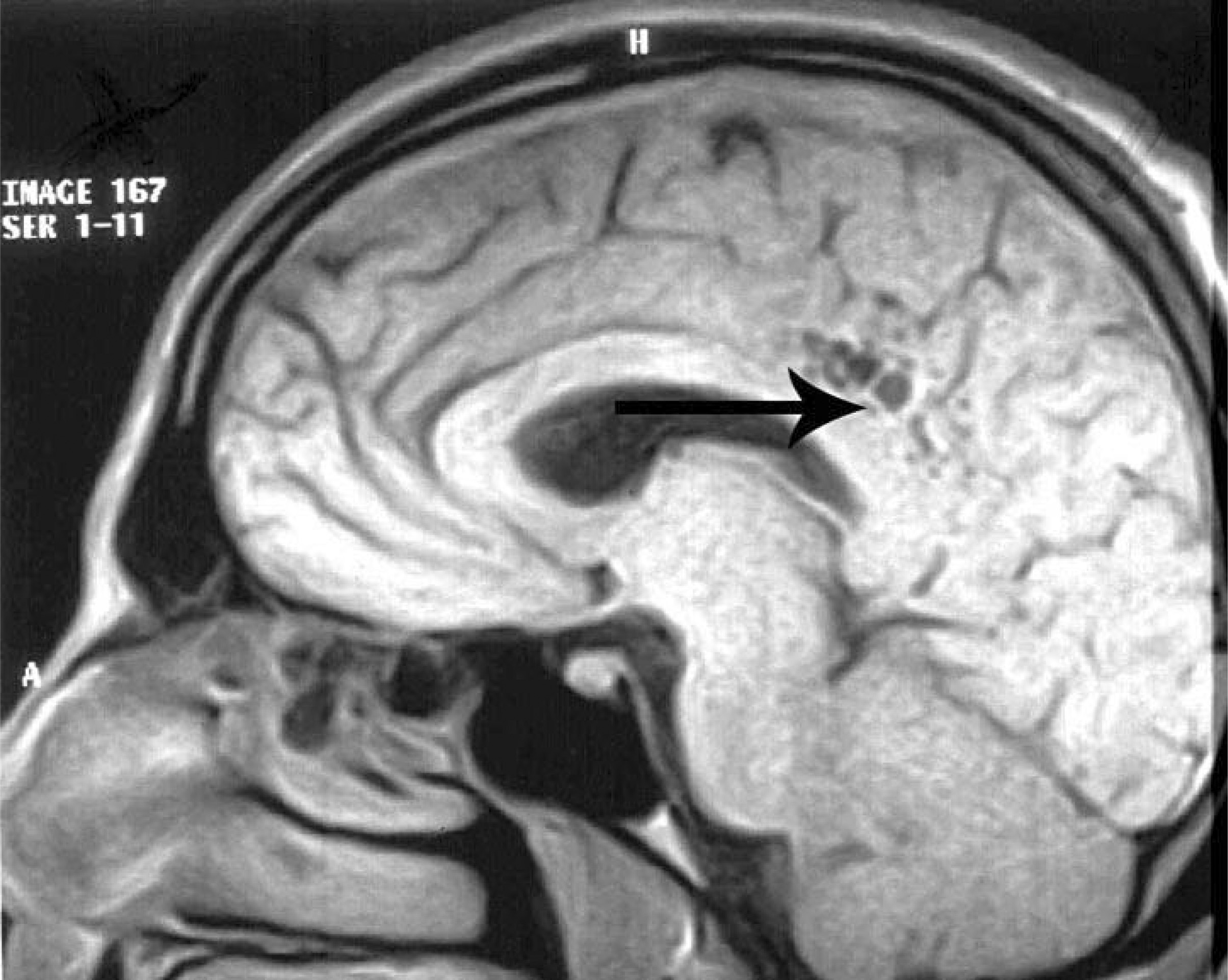
Cystic lesions in the distal drainage area of the superior sagittal sinus at day 8 after onset of symptoms in 2004.
Norway 2004
One month later, the patient was clinically healthy with no signs of neurological sequelae. Further laboratory investigations revealed an elevation of liver enzymes. Most notable was an alanine aminotransferase level of 816 U·L−1 (normal range 10–70 U·L−1). All other standard laboratory tests were within normal limits. By November 2004 his enzymes had returned to normal. Serology for hepatitis A, B, and C was negative. It was thought that the transient liver enzyme elevation may have been due to treatment with ciprofloxacin. Investigations for thrombophilia, including antithrombin, protein C, protein S, activated protein C resistance, factor V Leiden and the prothrombin gene 20210GA mutations, lupus anticoagulant, anticardiolipin antibodies, and homocysteine were all negative on 2 separate occasions.
After 3 months, a repeat cerebral magnetic resonance imaging showed nearly complete dissolution of the thrombus. Cerebral magnetic resonance imaging angiography detected a low-caliber right transverse sinus, interpreted as a normal variant. No cerebrovascular abnormalities were detected. The antithrombotic prophylaxis was continued for a total of 5 months.
Discussion
The patient experienced 2 similar incidents of severe headache at high altitude 3 years apart on the same route and at the same altitude. Clinical symptoms that were initially interpreted as HACE appeared at high altitude and notably worsened after descent on both expeditions. In 2001 the headache and concentration problems persisted for several weeks after descent. No brain imaging was performed.
The cystic lesions revealed by magnetic resonance imaging in 2004 (Figure 2) were interpreted as sequelae of previous venous infarction, located at the distal drainage area of the superior sagittal sinus. Not commonly found with fresh infarction, and visible only days after the latest incident, the focus probably represented sequelae of an earlier vascular incident. Thus, in retrospect there were substantial indications both clinically and radiologically that the symptoms in 2001 were indicative of CVT.
Saito and Tanaka, 3 Fujimaki et al, 4 and Song et al 5 have reported CVT occurring at high altitude, mainly involving the superior sagittal sinus. Boulos et al 6 reported a case of sagittal sinus thrombosis at high altitude in a man with congenital protein C deficiency. Previous reports of cases from the field concluded that inherited risk factors for either hypercoagulability or reactive polycythemia were the major pathological contributions. Torgovicky et al 7 reported a case of CVT in a young woman working in a high-altitude chamber. Oral contraceptive use was seemingly the only risk factor for hypercoagulability. Bendz et al 8 showed that acute exposure to hypobaric hypoxia per se may activate coagulation to induce a hypercoagulable state. None of the risk factors previously described were present in the patient in the current study, though a slight degree of dehydration cannot be excluded. In general, the physiological response to hypoxia by proper acclimatization has not been shown to involve important changes in platelet function or clotting factors; however, an association to acute mountain sickness cannot be excluded. 1
The HACE-like symptoms of CVT in the patient in the current study suggest that differentiating cerebral thrombosis from other neurovascular conditions may be extremely difficult in the field. The patient had no focal neurological deficits or seizures, which were reported by Terazzi et al 9 to be commonly associated with CVT. It is noteworthy that none of the previous case reports described any coexisting peripheral venous thrombosis, suggesting that patients with CVT may portray no other symptoms than those occurring from affected cerebral vessels.
The true prevalence of CVT during high-altitude climbing is unknown, most likely because the majority of events with severe headache at high altitude are interpreted as HACE, and brain imaging is not routinely performed.
The present case report suggests that high-altitude climbing per se is a risk factor for the development of CVT, though its occurrence is probably rare. Whether the etiology is primarily a high-altitude–induced hypercoagulable state of unknown origin or a result of cerebrovascular disturbances are issues that need further investigation. The lesson from this case report is to recognize that severe headache not responding to adequate medical management for HACE may represent a cerebrovascular disorder, particularly if the headache persists after descent.
Footnotes
Acknowledgments
The authors wish to thank Drs Einar Husebye and Ken Zafren for technical consultations.
