Abstract
Objective
This exploratory study assessed a potential relationship between elevated carboxyhemoglobin (COHb) levels and acute mountain sickness (AMS) at 4300 m on Denali. Additional analysis assessed the relationship among COHb levels, AMS, and climber characteristics and behaviors.
Methods
Participants were screened for AMS with the Lake Louise Self-Report questionnaire and answered questions focusing on AMS symptoms, prevention, and previous altitude illness. Levels of COHb were measured by serum cooximetry. Additional questions assessed stove practices, climbing practices, and climber behaviors. Nonparametric statistical analyses were performed to examine potential relationships among COHb levels, AMS symptoms, and climber behaviors.
Results
A total of 146 climbers participated in the study. Eighteen climbers (12.5%) were positive for carbon monoxide (CO) exposure and 20 (13.7%) met criteria for AMS. No significant relationship was observed between positive CO exposure and positive criteria for AMS. Climbers descending the mountain were 3.6 times more likely to meet the study criteria for positive CO exposure compared with those ascending the mountain (P = .42). In addition, COHb levels were significantly higher for those descending the mountain (P = .012) and for those taking prophylactic medications (P = .010). Climbers meeting positive criteria for AMS operated their stoves significantly longer (P = .047).
Conclusions
No significant relationship between AMS symptoms and CO exposure was observed. This may have been affected by the low percentage of climbers reporting AMS symptoms, as well as limited power. Descending climbers had a 3.6 times increased risk of CO exposure compared with ascending climbers and had significantly higher COHb scores. Increased hours of stove operation was significantly linked to climbers who also met criteria for AMS.
Introduction
Since the advent of polar exploration in the past century, portable stoves have been used on high-altitude expeditions. Although invaluable, stove use increases the risk of carbon monoxide (CO) exposure and toxicity for individuals exposed to exhaust fumes. 1 –4 Interestingly, initial symptoms of CO poisoning are similar to symptoms of acute mountain sickness (AMS), and determining whether a climber has AMS, CO poisoning, or both poses a challenge to health care providers in the high-altitude setting. 4 –7
Anecdotal reports of CO poisoning in mountaineers have been documented by the National Park Service (NPS) on Denali (Mt McKinley) for at least 20 years. 8 In 1985, 2 American climbers were found severely impaired after cooking inside their tent at the high camp (5200 m). 9 In 1986, a fatal incident was reported involving 2 Swiss climbers cooking inside a sealed tent at advanced base camp (4300 m). 10 There are numerous anecdotal reports of climbers perishing from CO poisoning while climbing in the Himalayas.11,12 Descriptions of fatal CO poisonings also include exposures while cooking inside tents on camping trips in temperate climates and during military expeditions in subzero temperatures.1,5,13
Carbon monoxide has a binding affinity with hemoglobin 200 to 250 times greater than oxygen and a half-life of 4 to 6 hours at sea level. Normal basal levels of carboxyhemoglobin (COHb) range from <1.0% to 3.0% in nonsmokers and up to 10.0% in moderate smokers.6,14. Levels of COHb ≥3.0% but <10.0% in asymptomatic nonsmokers are abnormal and constitute CO exposure.2,6,14,15 At approximately 10.0% COHb in nonsmokers, mild to moderate CO intoxication occurs, causing severe headache, nausea, dizziness, and insomnia.6,14–16
From 1985 to present, 5% to 32% of all climbers on Denali were treated annually for symptoms of AMS.14,17 The number of climbers experiencing CO exposure or intoxication on Denali, however, is unknown and diagnosis is difficult considering a climber may have either AMS, CO toxicity, or both. 18 Additionally, the effects of CO toxicity (acute or chronic) and altitude hypoxia are possibly additive, as CO bound to hemoglobin increases hypoxic stress and renders a person at a “physiologically higher” altitude, which may precipitate AMS.4,5,19–22 For this study, it was hypothesized that a significant number of climbers with symptoms of AMS would also have elevated COHb levels.
Materials and methods
The Human Subjects Review Committee of the Human Subjects Division of the University of Washington reviewed and approved this research project.
All climbers reviewed a general information card explaining the research during the mandatory climber orientation meeting at the NPS headquarters in Talkeetna, AK, before flying to Kahiltna Base Camp (2200 m). Individuals interested in participating in the study signed a consent form, which the NPS headquarters held until completion of the study. The consent form required the participant's signature only and no personal identifying information.
At the 4300-m high base camp on Denali, research team members recruited subjects after confirming that they had signed a consent form. Individuals who did not sign a consent form in Talkeetna were not recruited into the study.
All participants were screened for AMS with the Lake Louise Self-Report (LLSR) questionnaire (Appendix 1). The LLSR score was developed at consensus meetings in Lake Louise, Canada, in 1991 and 1993. Primarily developed for research use, the short and simple format, which is easy to complete in difficult situations, has led to its adoption by trekkers and mountaineers. The LLSR is sensitive enough to detect AMS, with sufficient specificity to prevent overdiagnosis. 23
Subjects and controls were asked 11 questions focusing on basic demographic data and climber behaviors that focused on potential CO exposure risk and individual risk for developing AMS (Appendix 2). Demographic, LLSR, and climber-behavior data were recorded by a research investigator and did not interfere with any assessment and treatment performed by the NPS medical staff.
Subjects’ COHb levels were measured with the GEM OPL Oxygenation Portable Laboratory cooximeter (Instrumentation Laboratory Inc, Lexington, MA). Approximately 0.3 mL of blood was collected by venipuncture from a peripheral vein on the hand or wrist by using a sterile technique and a 1-mL heparinzed tuberculin syringe. A 50-μL sample was injected into a disposable cuvette and analyzed with the cooximeter. Levels of COHb were recorded as a percent with a range of 0.1% to 75.0%. On Denali, the operation of the cooximeter took place inside the NPS first-aid shelter located at high base camp, which is heated and provided a relatively stable environment for operation. Optical quality control cuvettes were used before analysis of every blood sample to calibrate the instrument and maintain constant result reliability of blood samples analyzed.
Each participant's LLSR score, demographic data, and COHb level was assigned a unique identifying number. All data were anonymous and without link to participant name, consent form, or other personal identifying information.
Levels of COHb >3% (nonsmokers) or >10% (smokers) were considered positive for CO exposure. Levels below these cutoffs were classified as negative for COHb exposure. For this study, COHb levels >10% for nonsmokers and >20% for smokers were considered positive for CO intoxication.5,6,12–14,16
Data Analysis
By using SPSS software (version 12, SPSS Inc, Chicago, IL), analysis of frequency distributions and quantitative variables showed that nonparametric statistical techniques were indicated. Spearman rank order correlations were calculated for the quantitative variables in the study. Crosstabulations were produced for the AMS and CO exposure classification variables with all other categorical study variables that could potentially contribute to positive CO exposure or AMS (eg, age, days ascending, hours operating stove, gender, smoking status, history of AMS, use of medications, and ascending or descending the mountain). Mann-Whitney and Kruskal-Wallis tests were used to test for differences in the mean ranks of the quantitative variables. Logistic regression was used to explore models and identify predictive variables for both AMS and CO exposure classifications by using combinations of study variables.
Results
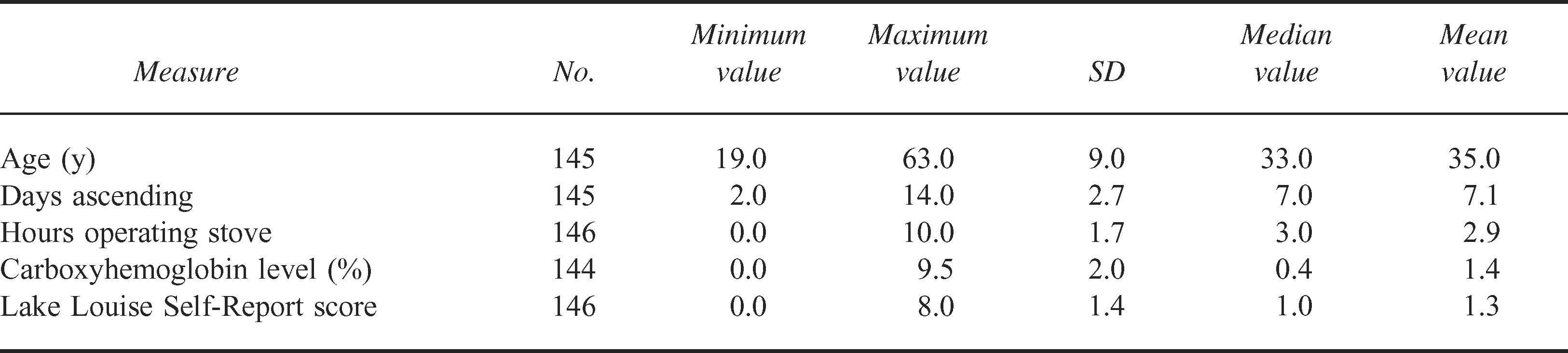
A total of 146 consented climbers (median age 33 years, men 84.8%, nonsmokers 86.2%) were enrolled in the study during May 2004. Most participants were ascending the mountain, and the median time to reach the 4300-m high base camp was 7 days (Tables 1 and 2).
Quantitative climber characteristics
Categorical climber characteristics
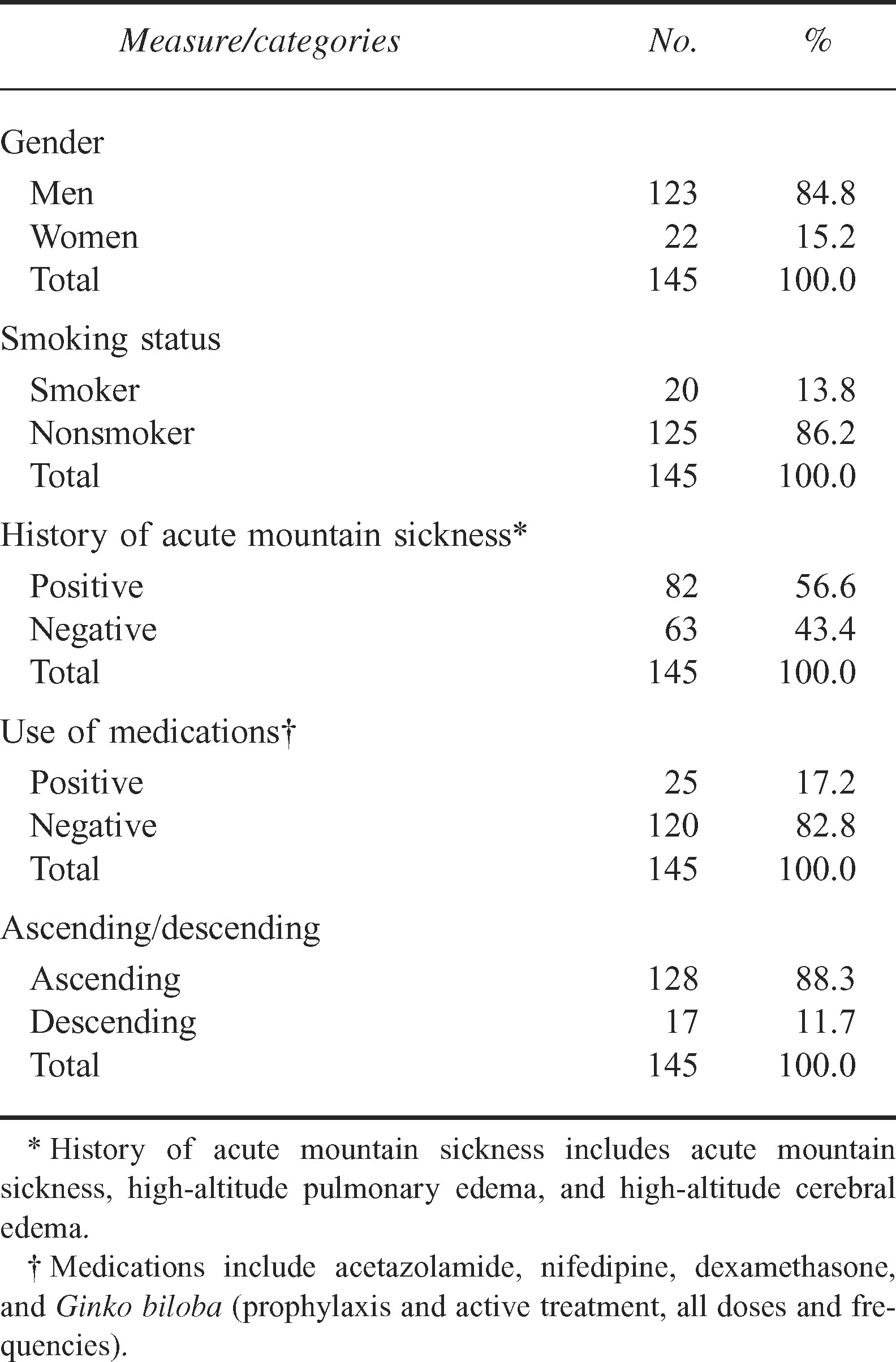
Fifty-six percent of participants reported having a history of AMS, and the majority denied taking any prophylactic medications to assist with acclimatization. Other median values for study variables include daily stove operation of 3 hours (range 0.0–10.0 hours), participant COHb level of 0.4% (range 0.1%–9.5%), and an LLSR score of 1.0 (range 1–5) (Tables 1 and 2).
All participants used a portable stove, and 3 fuel types were reported: white gas (78.2%), isobutane (15.5%), and propane (6.3%). Ten climbers had daily exposure to portable heater units. The majority of climbers (95%) reported ventilating their cooking space (ie, outside, in a cook tent, in tent vestibule or tent). Of note, 23.5% of climbers cooked inside a ventilated igloo or tent.
Eighteen climbers (12.5%) tested positive for CO exposure and 20 climbers (13.7%) met criteria for AMS. No significant relationship was observed between positive CO exposure and positive criteria for AMS. No significant relationship was observed between CO exposure and age, gender, smoking status, history of AMS, or stove and fuel type.
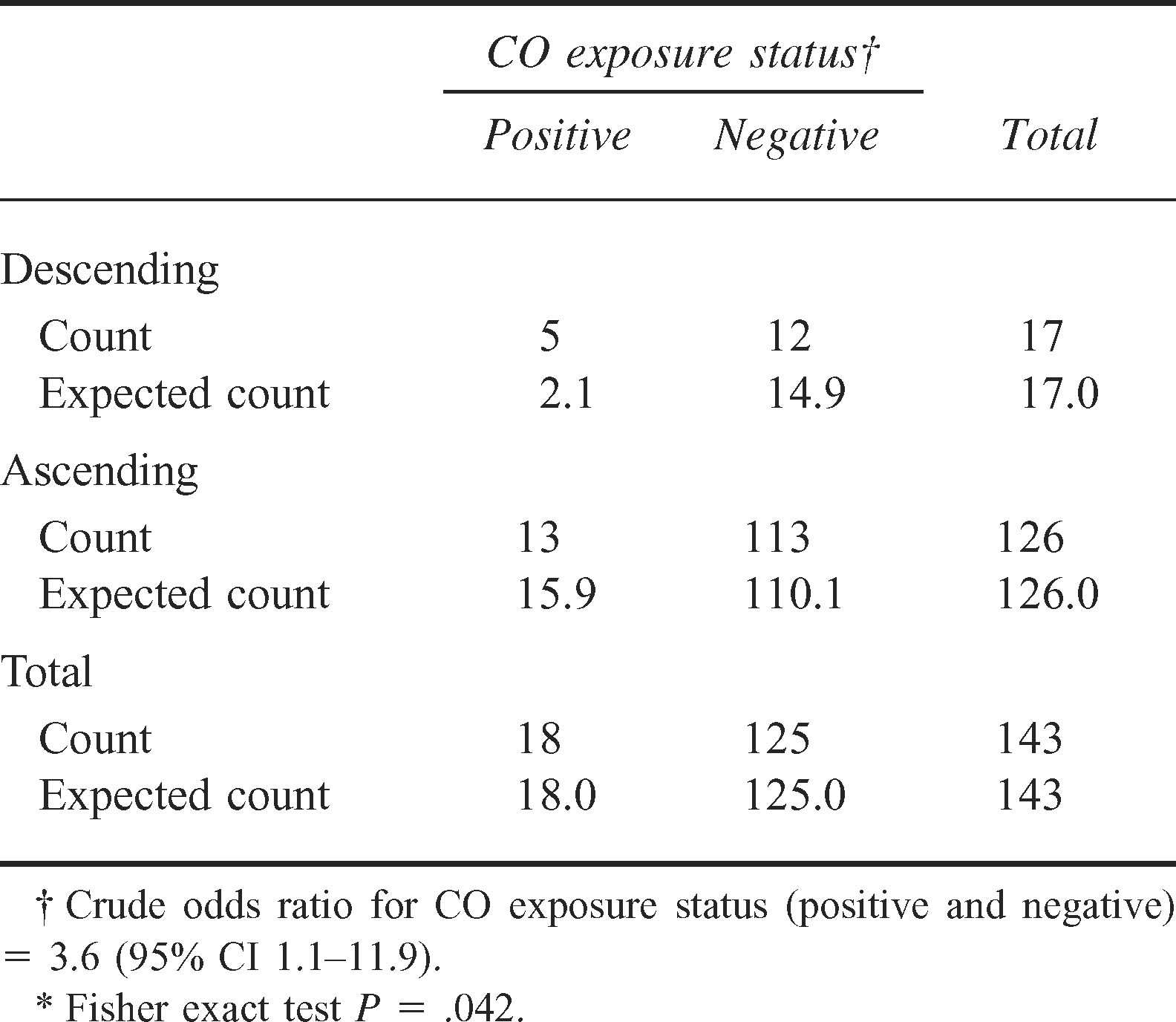
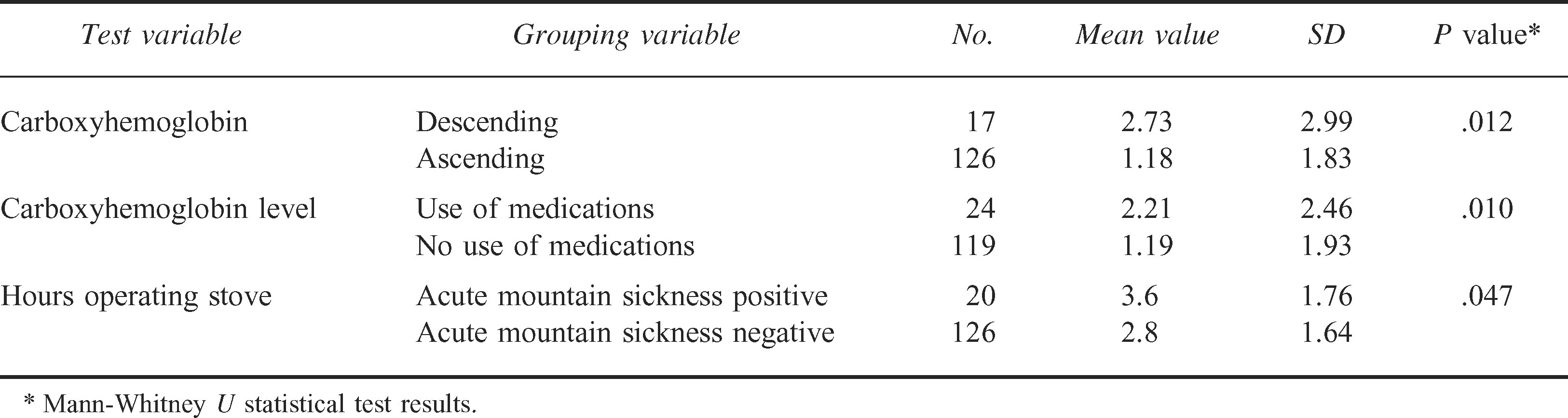
Climbers descending the mountain were 3.6 times more likely to meet the study criteria for positive CO exposure compared with those ascending the mountain (P = .042) (Table 3). In addition, COHb levels were significantly higher for those descending the mountain (P = .012) and for those taking prophylactic medications (P = .010) (Table 4). Also, climbers with positive criteria for AMS operated their stoves significantly longer (P = .047) (Table 4).
Carbon monoxide (CO) exposure status by descend ing or ascending crosstabulation*
Statistically significant findings
Discussion
Although 18 climbers tested positive for CO exposure and 20 tested positive for AMS, a significant correlation between elevated COHb level and a positive diagnosis of AMS was not demonstrated (P = .245). Poor weather, mandating acclimatization, as well as a small sample size likely hindered the ability to prove the study hypothesis.
Weather on Denali during May 2004 was highlighted by several low-pressure storm systems intermixed with high-pressure systems generating winds in excess of 80 mph as low as 4000 m. Because of poor weather, climbing teams took more time ascending and spent more time at the 4300-m high base camp. It quickly became apparent to both the research team and the NPS climbing ranger staff that most climbers arriving to the 4300-m high base camp were well acclimatized. During the month, only 4 (of an estimated 250) individuals were assessed by NPS first-aid staff at the 4300-m high base camp for symptoms suggestive of AMS. 24 The weather influence on acclimatization may also be supported by the fact that only 13.7% of study participants met positive criteria for AMS.
Because of the low power of this study, the relationship between CO exposure and AMS remains unknown but will likely become clearer with screening of larger numbers of climbers, perhaps through an entire climbing season on Denali.
A significant relationship between increased COHb levels and positive CO exposure for those descending the mountain was not expected and may represent a previously unknown risk factor for high-altitude mountaineers. Increased CO exposure while descending may be attributed to several factors. For example, descending climbers may spend more time in their tents when resting and be prone to operating their stoves in locations that could increase CO exposure. Descending climbers may also be less active and more sedentary when not actively descending, thus increasing their risk and decreasing perception of CO exposure.5,18 The observance of elevated COHb levels in descending climbers in this study also raises the question of whether or not high-altitude mountaineers chronically accumulate CO over time because of low atmospheric pressures, the prolonged COHb half-life at altitude, prolonged daily stove use, a progressive decrease in activity level (especially after a summit attempt), and other undetermined factors.6,12,18,19 Further research investigating the potential increased half-life of COHb at high altitude and the possible phenomenon of chronic CO accumulation by climbers operating portable stoves at high altitude is also needed.
The significant relationship between operating portable stoves for longer hours and positive AMS may suggest that CO exposure may be contributing to or mimicking AMS.5,7,12 Significantly elevated COHb levels in participants taking medication for acclimatization also raises the question of whether specific medications may increase endogenous COHb levels or enhance CO exposure and warrants additional investigation. 6
There are several potential limitations to this study. Most important is the possibility of inaccurate readings by the portable cooximeter when analyzing COHb levels in participating climbers. Although every effort was made to operate the cooximeter at a stable temperature, and optical control cuvettes were used to calibrate the machine before reading every blood sample, the variable and extreme environment on Denali at 4300 m may have caused inaccurate measurements of COHb levels. Also, per conversations with Instrumentation Laboratory Inc, the GEM OPL cooximeter has not been evaluated for performance at high altitude. The research project did not determine when participants had last operated a stove but only how many hours they had operated one in the past day. Because of the short half-life of COHb and the random recruiting of participants into the study, individuals may have had higher COHb levels before or after being tested. Because many climbers routinely do not seek care for mild symptoms that they perceive to be due to AMS (eg, headache, nausea, insomnia), additional CO-exposed subjects may have been missed. Both of these problems may have led to underreporting of elevated COHb levels in the population tested. Also, symptoms for the LLSR and all demographic information were self-reported and subject to recall bias.
Factors including poor weather, an overall decreased rate of ascent in climbers, improved acclimatization of subjects, and low sample size likely contributed to the study hypothesis not being proven. The findings of this study, the first of its kind to assess on-site serum COHb levels in climbers at altitude, demonstrate that CO exposure in high-altitude mountaineering does occur and merits additional research. The relationship between CO exposure and AMS remains unclear, but continued research and testing of additional subjects may result in a better understanding of the risk of CO exposure and its role in high-altitude illness, thus improving mountaineers’ chances of accomplishing climbing objectives safely at high altitude. At the time of this publication, plans for additional testing at the 4300-m camp on Denali investigating the relationship between CO exposure and altitude illness in climbers are underway.
Footnotes
Funding
This work was supported by equipment grants from Instrumentation Laboratory Inc (Lexington, MA) and K2 Sports Inc (Vashon Island, WA) and by research grants from Rocky Mountain Wilderness Medicine (Boise, ID) and Idaho Academy of Family Practitioners (Boise, ID).
Acknowledgments
The authors wish to thank the following individuals and institutions for their assistance: Ted Epperly, MD, and Family Medicine Residency of Idaho; Rob Hilvers, MD, and Rocky Mountain Wilderness Medicine; Kemala Thompson and Instrumentation Laboratory Inc; Lucy Tyrell and the Denali National Park and Preserve; Lyn O’Doran and the Human Subjects Division of the University of Washington; Tor Anderson; Peter Hackett, MD; Robert Schoene, MD; Kendra Witt; Meg Purdue; Lance Taysom, RN; John Evans; the Denali National Park climbing rangers and the park rangers based at the Talkeetna Ranger Station; Neva Santos and the Idaho Academy of Family Practitioners; Suzanne Allen, MD; Jim Roscoe and Roscoe Steel and Culvert Company; Ken Schiele; Renny Jackson and the Teton National Park climbing rangers; John Cook, MD, and Nancy Cook; Dana Design and K2 Sports Inc; and MSR Inc and Chris Johnson, MPH, Epidemiologist, Cancer Data Registry of Idaho.
Appendix
Lake Louise Self-Report questionnaire
23
Study questionnaire*

