Abstract
Objective
Mountaineers face a variety of health risks at altitude including pulmonary edema; portable ultrasound may be used to diagnose high altitude pulmonary edema. This report tests the functionality of electronic equipment in a hypobaric test environment and the ability of remotely guided nonexperts to use ultrasound to evaluate respiratory status on Mt Everest.
Methods
Two ultrasound devices and associated video equipment were tested in a cooled (4°C–5°C) hypobaric chamber to 27 000 feet (8230 m) before travel to Mt Everest. The ultrasound system was connected via satellite phone to a video streaming device and portable computer to stream video through the Internet for remote guidance of a novice user by an expert. Pulmonary interstitial fluid was quantified by the presence of “comet tail” artifacts.
Results
There was no notable degradation in equipment performance in cold, hypobaric conditions; ultrasound confirmation of increased comet tails was noted in the chamber despite oxygen supplementation and the very brief exposure. Two pulmonary surveys of asymptomatic participants were completed by novice operators within 25 minutes on Mt Everest. The remote expert was able to guide and identify comet tails suggestive of intermediate pulmonary interstitial fluid. Image quality was excellent.
Conclusions
The tested ultrasound devices functioned nominally in cold, hypobaric conditions; acute changes in lung fluid content were noted in these conditions despite normoxia. We successfully used a satellite telemedical connection with a remote expert to guide thoracic ultrasound examinations at Advanced Base Camp on Mt Everest. Coupling portable ultrasound with remote expert guidance telemedicine provides a robust diagnostic capability in austere locations.
Introduction
A number of health problems, including pulmonary edema, may occur in recreational or experienced mountaineers during exposure to the hypobaric hypoxia of high altitudes. 1 The signs and symptoms of high altitude pulmonary edema (HAPE) are nonspecific, which hampers the clinical diagnosis and delays appropriate treatment. 2 Ultrasound is an effective diagnostic modality for a variety of clinical conditions. Recent investigations have suggested that ultrasound can be used to evaluate pulmonary edema by quantifying the appearance of “comet tails,” which are reported to result from increased fluid content around the visceral parietal interface. 3
Light-weight, portable, high-fidelity ultrasound machines could theoretically be used in the early diagnosis of pulmonary edema in remote settings. However, many portable electronic devices such as MP3 players and laptop computers have been plagued by failures in the cold, hypobaric conditions present at extreme altitude. In preparation for a research expedition to Mt Everest, the hypobaric chamber provided a controlled environment in which we could observe physiologic responses and test the reliability of the equipment under conditions that simulated the extreme mountain environment.
Scientists at NASA developed the technique of expert-guided remote ultrasound examinations to enable nonphysicians to perform medical evaluations on the International Space Station. 4 This report documents use of the same technique in a terrestrial setting where, under remote expert guidance, climbers performed thoracic ultrasound examinations on each other at Advanced Base Camp on Mt Everest. Pretrial evaluation of 2 high-fidelity, portable ultrasound systems was also done to test equipment functionality under these conditions and the acute pulmonary response to reduced pressure.
Methods
The experiments detailed herein were reviewed and approved by the Human Investigation Committee at Henry Ford Hospital in Detroit, MI. A commercially available, high-fidelity, portable ultrasound system (GE LOGIQ e; General Electric Medical, Milwaukee WI) and a prototype solid state system (GE LOGIQ e; General Electric Medical) equipped with a 12-5 linear probe were used for the ultrasound investigations. The super video graphics array (SVGA) output of the ultrasound system was fed to a video streaming device (DistanceDoc, Mediphan, Ottawa, Canada) to digitize and compress the video signal. The DistanceDoc USB output was coupled to a portable computer (Mac Book Air, Apple Computers, Cupertino, CA) for streaming through the Ethernet connection (ooVoo, New York, NY) to secure, remote viewing sites over a satellite phone link-up (Figure 1).
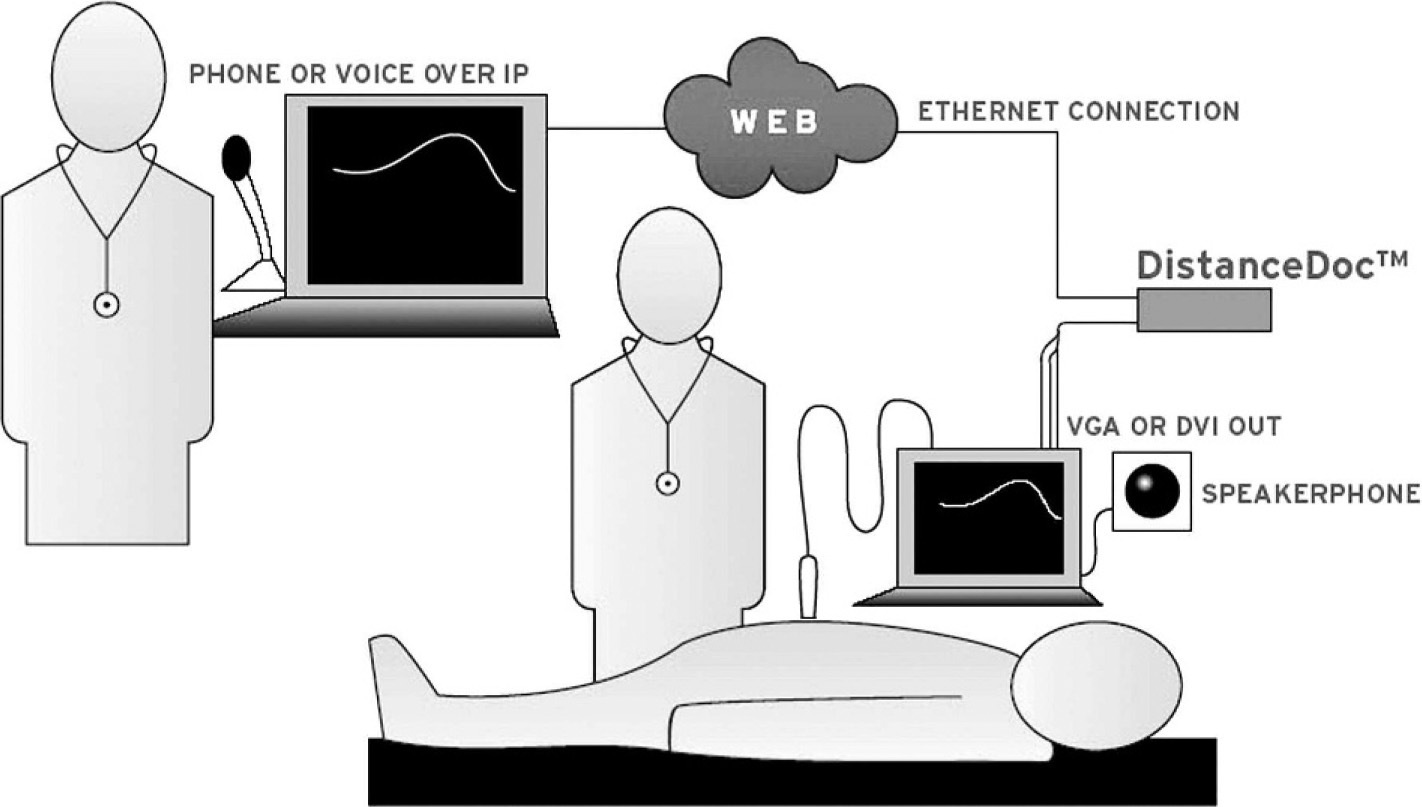
Remote expert guidance of thoracic ultrasound examinations was done over the Internet using a satellite phone connection. The video output of the ultrasound device was compressed with a streaming device (DistanceDoc; Mediphan, Ottawa, Ontario) for real-time viewing by the remote expert.
Hypobaric Testing
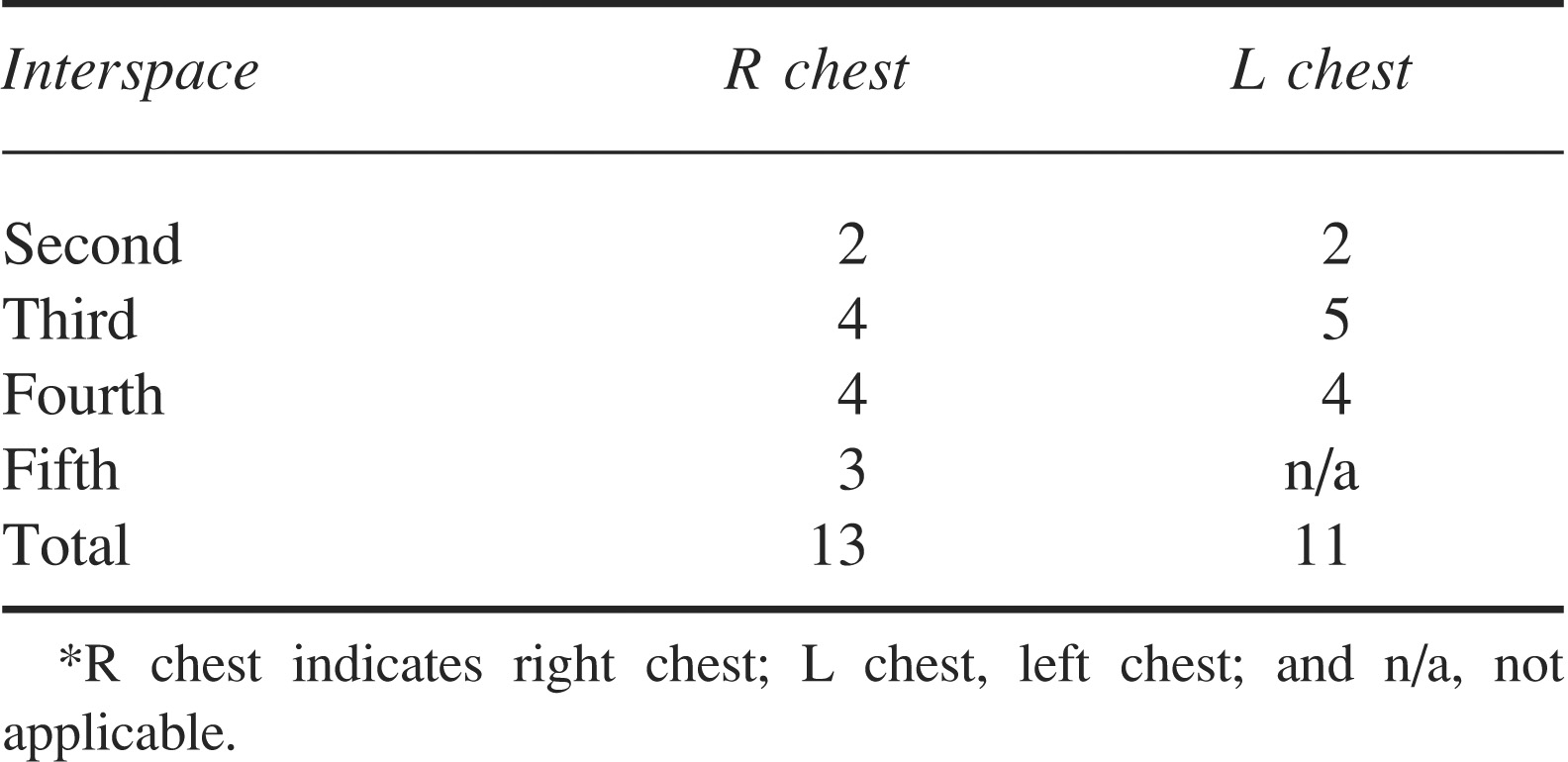
The electronic equipment was allowed to temperature equilibrate at 40°F for 1 hour before the decompression trial. Baseline scanning of a single participant's lung fields was done as previously described before the depressurization experiment. 5 Briefly, the second to fifth intercostal spaces on the right and second to fourth intercostal spaces on the left chest were scanned in parasternal, anterior axillary, midclavicular, and posterior axillary locations. Comet tail artifacts were defined by a well visualized, demarcated hyper-echoic signal originating at the visceral-parietal pleural surface; the number of comet tail artifacts per scanned field was quantified. The participant and ultrasound operator were then fitted with supplemental oxygen and decompressed to 27 000 feet (8230 m) at 5000 feet/min (1524 m/min) for the testing protocol.
The electronic equipment was cold-cycled twice at altitude to verify functionality; the testing protocol was conducted during a 1-hour period. Comprehensive ultrasound scans of both chest cavities were done to quantify lung sliding and to quantify comet tail artifacts. The equipment was then turned off and the chamber repressurized to nominal at 3000 feet/min (914 m/min). The electronic equipment was cycled after the run to verify continued functionality.
Mt Everest Trial
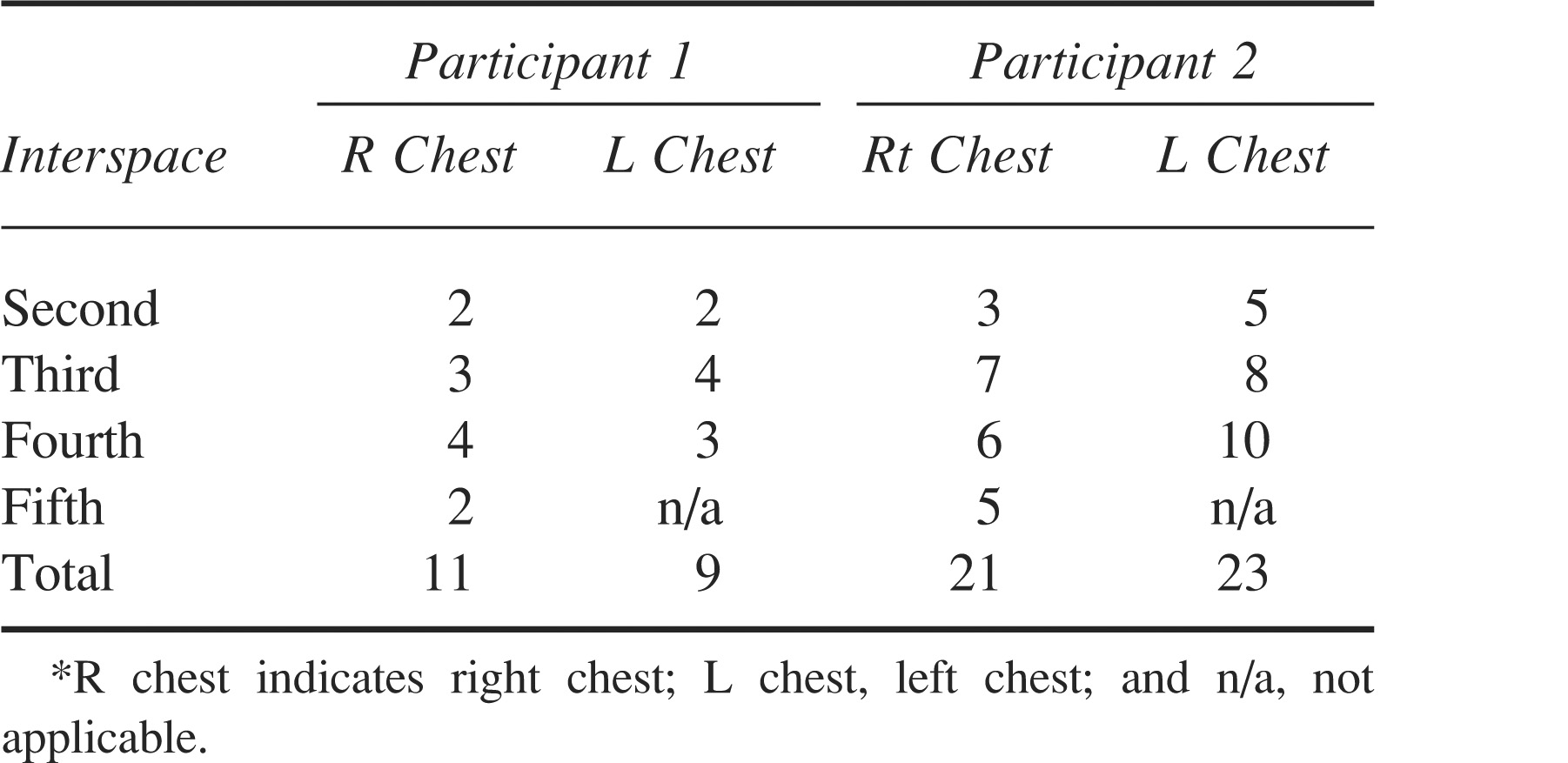
The ultrasound examinations were performed using the solid state prototype system in an expedition medical tent at Advanced Base Camp on Mt Everest (21 000 feet or 6400 m). The ultrasound participants were 2 young males (ages 24 and 20) who were in the early and late phases of acclimatization, respectively (Figure 2). The ultrasound operators were nonphysician, fellow climbers with no ultrasound experience. A rapid (2 minute) orientation to the ultrasound machine, probe orientation, remote commands, and examination conduct was given over a satellite phone by a remote ultrasound expert (S.A.D.) just before the examination.The remote expert communicated with the climbers using bidirectional audio, reviewed streaming video output from the ultrasound, and guided the examination with the aid of a cue card to which the climbers referred (Figure 3).
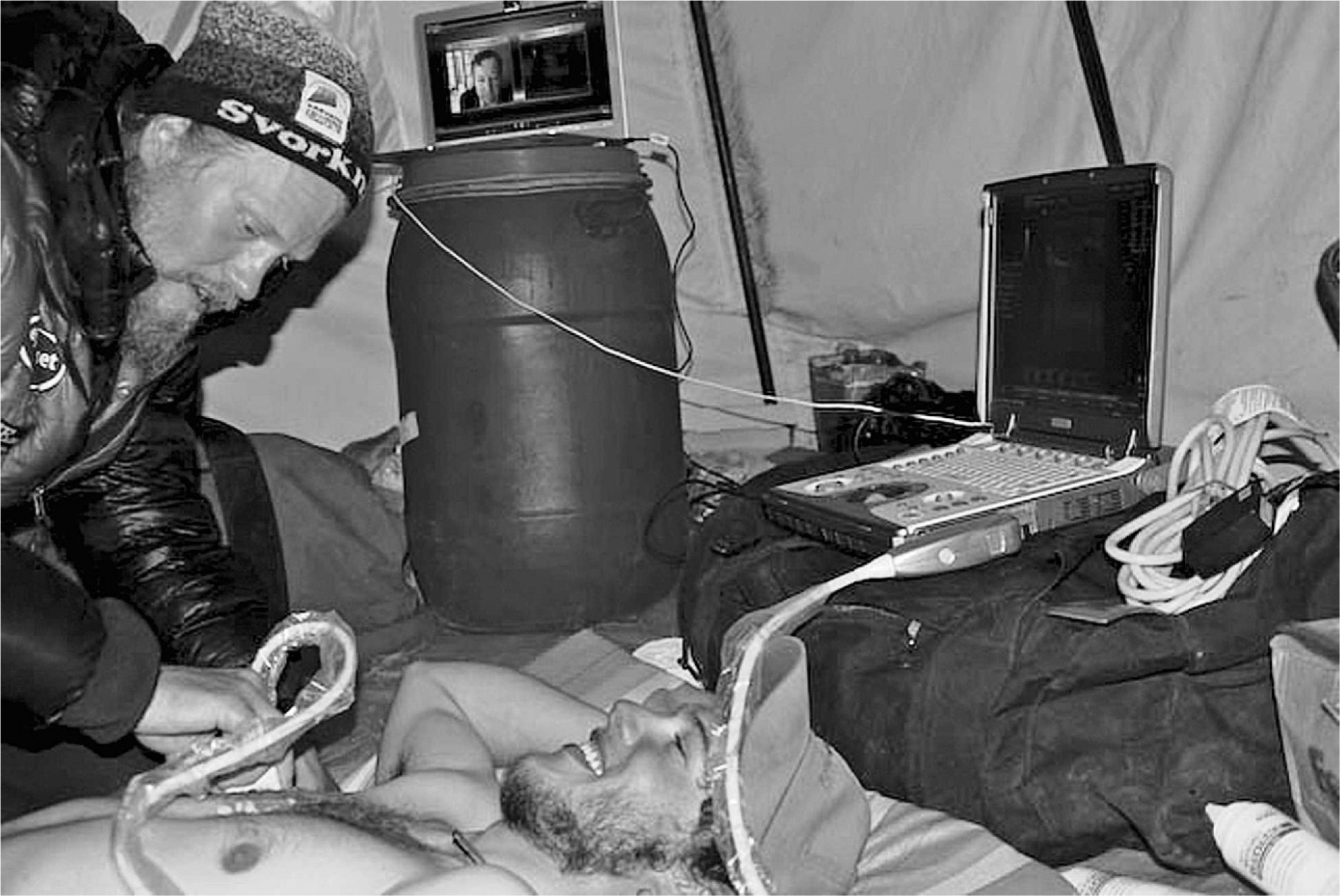
A nonexpert operator is performing a thoracic ultrasound examination on a fellow climber in a tent at Advanced Base Camp. The remote expert is seen on the computer screen in the background directing the examination.
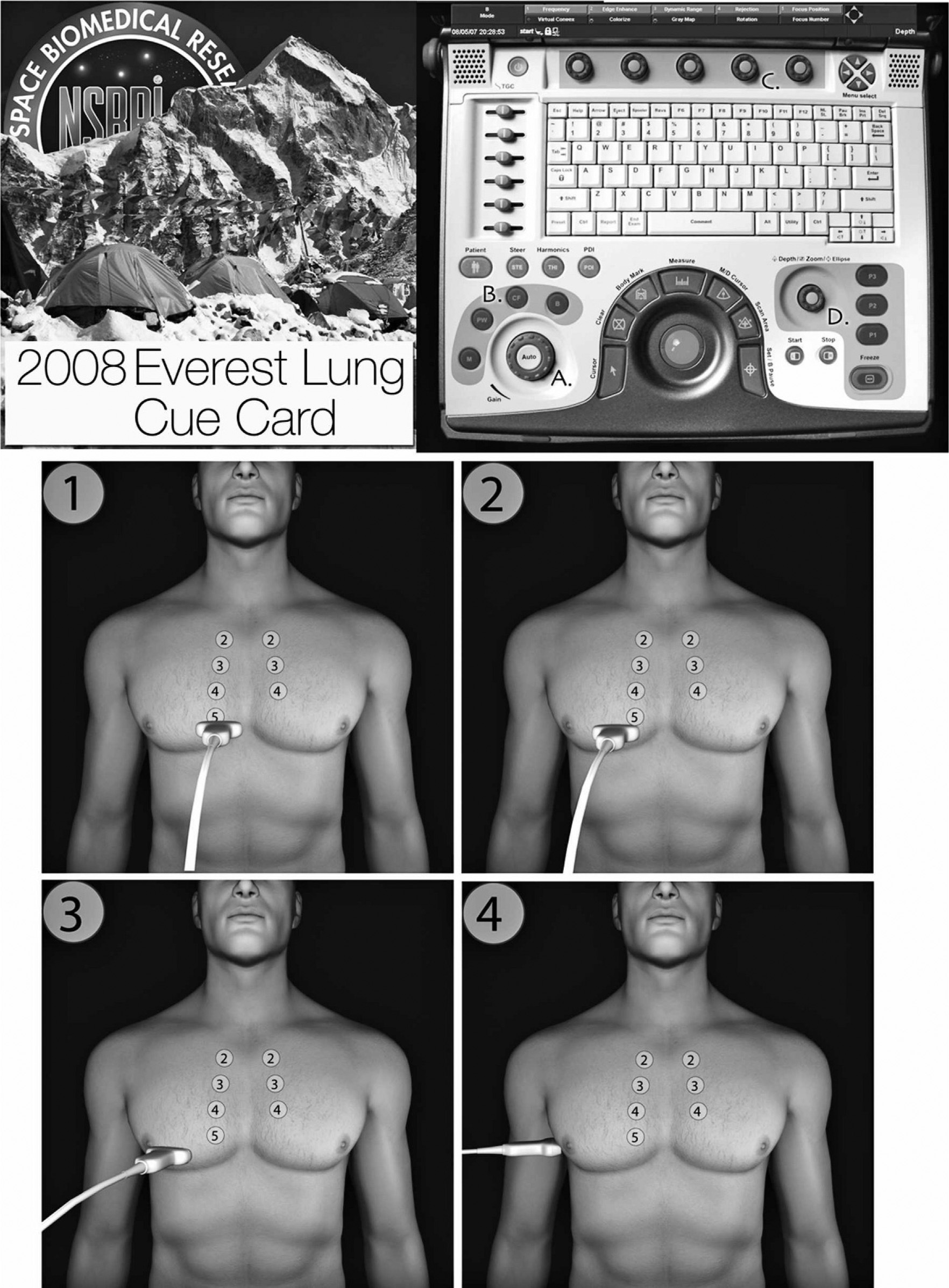
A cue card was developed to help the operators perform the thoracic ultrasound examination. The cue card used a color coded keyboard guide and external probe position starting points to streamline remote commands to the on-site operator.
A comprehensive ultrasound scan of the lungs was then performed. 5 Briefly, the second to fifth intercostal spaces on the right and second to fourth intercostal spaces on the left chest were scanned in parasternal, midclavicular, anterior axillary, and posterior axillary locations. Comet tail artifacts were defined by a well visualized, demarcated hyper-echoic signal originating at the visceral-parietal pleural surface. Real-time assessment of the ultrasound examination, and the total number of comet tail artifacts noted, was done by the remote expert; high-quality ultrasound images were stored locally on the ultrasound machine for later review by a blinded expert. The results were tabulated based upon anatomic location in the chest and totaled to provide an index of overall pulmonary interstitial fluid status.
Results
Hypobaric Testing
Altitude (27 000 feet; 8230 m) and temperature (4.4°C) remained stable during the 1-hour test protocol in the hypobaric chamber. The ultrasound participant and operator continuously used 100% oxygen under positive pressure during the entire chamber decompression trial and did not exhibit hypoxic or other clinical symptoms of mountain sickness. The solid state ultrasound device required significantly longer boot up times than the classic hard drive in both ambient and hypobaric conditions. The portable computer and video streaming device functioned equally well at low and high altitudes and showed no degradation in function in the cold.
Under normobaric, normoxic conditions there were no comet tail artifacts seen in any of the lung fields. However, under hypobaric conditions, comet tail artifacts appeared in all lung fields within 5 minutes of arriving at 27 000 feet (8230 m). This dramatic change was most notable in the lateral lung fields of the right lung and occurred even though the subject remained normoxic, and gradually resolved over 1 hour after repressurization to normobaric conditions (Table 1).
The number of comet tails in 4 anatomic regions (parasternal, midclavicular, anterior axillary, and posterior axillary) was counted during 2 breath cycles in each area and tabulated. Comet tails were defined as a hyper-echoic signal, which originated at the visceral-parietal interface and extended into the field*
Mt Everest Trial
During the ultrasound scanning protocol, both climbers showed no signs of respiratory distress and maintained stable oxygen saturations of 84%. The testing protocols required 20 to 30 minutes for set-up and the examination itself. The portable computer, ultrasound machine, and video streaming device functioned without abnormality in the reduced temperature (ranging between 4°C and −7°C) and hypobaric conditions without degradation of performance.
There was a video signal delay of less than 1 second during both ultrasound sessions, which did not impact the conduct of the examinations or data interpretation. The average satellite bandwidth use for an ultrasound scanning session was 25 Mb.
The minimally trained, nonexpert ultrasound operators were readily guided through the ultrasound protocols without difficulty. Each pulmonary scanning location was viewed for 2 breath cycles and required approximately 30 to 45 seconds to acquire. The locally stored digital imaging and communications in medicine (DICOM) images were of excellent, diagnostic quality. The image quality of the remotely viewed examinations was good to excellent; thoracic structures, including the ribs and intercostal musculature, and dynamic findings, including lung sliding and comet tails, were readily visible (Figure 5). The remote experts could easily determine the absence of a pneumothorax by demonstrating lung sliding as well as quantify comet tail artifacts as an index of pulmonary Figure 4 The quality of the video-streaming signal during the thoracic ultrasound examinations enabled the remote expert to guide the examination and quantify the number of “comet tail” artifacts during the procedure. This screen grab of an actual examination shows a rib with acoustic shadowing on the right portion of the screen and the visceral-parietal interface in the midportion of the screen with a comet tail artifact extending downward.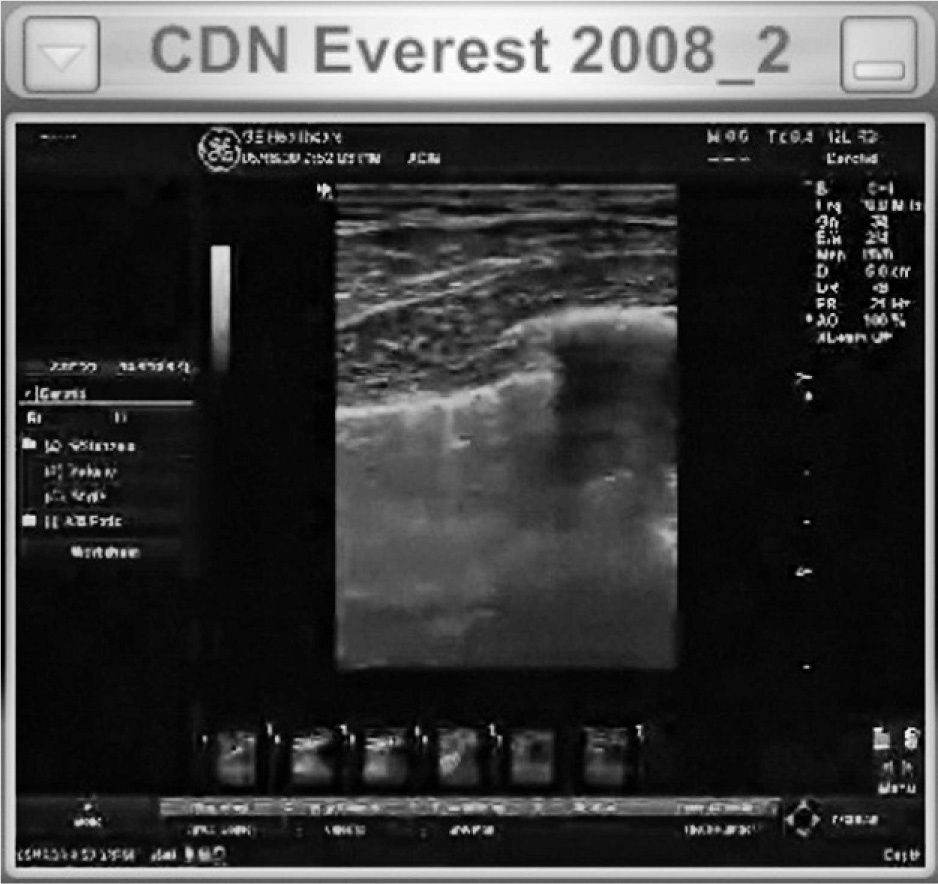
Comet tail artifacts were noted in all lung fields and were most pronounced in the lateral anatomic regions. Both subjects reported mild shortness of breath on exertion; subject 2 had a reduced acclimatization period*
Discussion
High-altitude illness may result from short-term exposures to altitudes in excess of 6560 feet (2000 m). This illness comprises a spectrum of clinical entities ranging from mild and self-limited syndromes such as acute mountain sickness to more life threatening forms such as HAPE and high altitude cerebral edema.2,6 The rate of ascent, the altitude attained, the amount of physical activity at high altitude, and individual susceptibility are contributing factors to the incidence and severity of high-altitude illness. 2
The incidence of acute mountain sickness varies from 1% to 30% in reported series. 6 The incidence of HAPE is considerably less common, ranging from 0.5% to 2%. 6 A recent series from Mt Everest suggested the incidence is less than 2% of acclimatized climbers. 7
The pathophysiology of HAPE is not well understood. It is a noncardiogenic form of pulmonary edema resulting from a leak in the alveolar capillary membrane. 7 There are various mechanisms believed to be responsible, although the dramatic clinical efficacy of pulmonary vasodilators both for prophylaxis and treatment has clearly implicated pulmonary hypertension from hypoxic vasoconstriction as the key pathophysiological insult. 7
The diagnosis of HAPE is complicated by the austere environment in which it occurs, with a lack of trained medical personnel or equipment. Patients will exhibit an increased respiratory rate with a reduction in oxygen saturation. Auscultation of the chest will often reveal fine rales, which are distributed over the entire lung field. Pneumothorax and cardiogenic pulmonary edema should be excluded by history and physical examination.
Small clinical trials have suggested that ultrasound may be used to evaluate pulmonary edema by quantifying the presence of comet tail artifacts at the lung surface that result from increased interstitial fluid around the visceral-parietal interface. Fagenholtz used a portable ultrasound device to confirm the presence of comet tails suggestive of pulmonary edema in high altitude climbers in Pheriche, Nepal. 3 The authors suggested that the quantity of comet tail artifacts in a standardized ultrasound examination of multiple lung fields bilaterally correlates with the severity of HAPE and appeared to decrease in magnitude as HAPE clinically improved.
The identification of comet tails in the pilot study, suggestive of interstitial edema within a few minutes of exposure to acute normoxic hypobaria, was quite surprising and raises the possibility that hypobaria per se could alter pulmonary interstitial pressure. This theory was suggested by Levine et al 20 years ago. 8 The presence of comet tails in this circumstance raises serious questions about its utility in the specific diagnosis of HAPE, and this will need to be tested more directly in a carefully controlled trial.
Ultrasound is a powerful diagnostic modality for a variety of applications. The technique of ultrasound is operator-dependent and requires a prolonged learning period to master. Remote expert guidance was developed by NASA scientists to speed the learning curve and disseminate expertise to enable ultrasound to be used successfully for space medical contingencies. Cue cards that demonstrate proper anatomic positioning of the probe, combined with keyboard shortcuts, allow a remote guidance expert to direct a novice operator to perform advanced ultrasound techniques. 9
The ultrasound operators in this mountain trial were not familiar with ultrasound and received less than 2 minutes of orientation with the technique before performing the pulmonary ultrasound examinations. Simple lay commands, combined with remote viewing of the ultrasound video stream, enabled the operators to obtain diagnostic quality pulmonary ultrasound images. Although the 2 ultrasound participants in this trial were asymptomatic, they both exhibited numerous comet tail artifacts in both lung fields, consistent with moderate pulmonary interstitial fluid accumulation. Whether such information could be useful to titrate medical therapy or to guide treatment options if symptoms arose will have to be tested in a dedicated fashion.
Point of care ultrasound has expanded dramatically in recent years because of improvements in technology, clinical needs, and innovation by the operators. This report documents the first uses of remote guidance to facilitate the use of pulmonary ultrasound by a nonphysician to evaluate pulmonary status in a remote environment. If borne out by clinical testing, this technique could readily be used to exclude HAPE. “Just in time” training, combined with remote expert guidance, has been successfully used by NASA on the International Space Station to perform a variety of complex ultrasound examinations. Robust portable ultrasound devices can now provide high-fidelity diagnostic capabilities in remote locations. Telemedical coupling of an ultrasound device with a distant expert enables this novel technology to be expanded to a variety of applications to improve diagnosis and treatment.
Footnotes
This research was supported by the National Space Biomedical Research Institute through NASA NCC 9-58 and respectively submitted to Wilderness & Environmental Medicine from Mt Everest base camp, altitude 5340 m.