Abstract
Objective
Exercise stress, immune status, and mood are interrelated. The stress of adventure racing is unique; exercise is very prolonged and competitive, with severe sleep deprivation and sustained cognitive demands, usually in arduous terrain and environmental conditions. The purpose of this prospective, descriptive study was to identify mood changes along with symptoms of illness and injury during and in the weeks following an international-level adventure race.
Methods
Mood, sleep, injury, and illness data were collected using questionnaires before, during, and for 2 weeks following New Zealand's Southern Traverse Adventure Race in November 2003.
Results
Mood was variable between athletes, but peaks of altered mood subscores were evident (P < .05) during the first 24 hours of racing, around race completion, and, as was hypothesized, 3 days after racing. Altered mood subscores resolved within 2 weeks. Symptoms of upper respiratory illness were most common immediately before (25/60, 42%) and after (28/49, 57%) racing, and largely resolved over the 2-week follow-up (5/27, 19%). Skin wounds and infections were common (43/49, 88%) immediately after the race but settled quickly. Pain was universal (100%), and musculoskeletal injury was common (38/48, 79%). Gastrointestinal complaints were common at the finish (8/49, 16%) and during the next 5 days but settled more quickly than upper respiratory symptoms.
Conclusions
Adventure racing of approximately 100 hours causes significant symptomatic injury and illness and mood state disruption, which generally resolve within a fortnight following racing. Disrupted mood and symptoms of illness and injury indicate athlete susceptibility to overreaching or overtraining without sufficient recovery.
Introduction
Adventure racing is a unique and popular form of stress. 1 It involves very prolonged competitive exercise, typically undertaken with severe sleep deprivation (ie, loss of sleep) and sustained cognitive demands (especially navigational), usually in arduous terrain and environmental conditions. Exercise includes running, trekking, kayaking, mountain biking, and rope skills for a total period from several hours to several days. Teams often consist of 4 athletes who travel together, making strategic decisions on navigation, pace, and sleep. Thus, wilderness multisport and adventure races provide unique challenges, require their own skill set, and apparently have distinct illness and injury profiles. 2 –7 Acute and chronic injury is frequent, 2 especially of the lower limbs. 8 Illness appears more common in those competing with injury. 2 The time course of recovery from this volume of sustained exercise and the associated afflictions has not previously been reported.
When athletes’ recovery is insufficient for their exercise impulse (in the context of their other stressors), they develop a short-term decrement in performance capacity known as “overreaching.” 9 –11 Sustained overreaching leads to the more protracted condition of “overtraining.” Both states are characterized by decreased exercise performance, altered immune function, and increased global disturbance of mood. 12 These changes may result from as few as 7 days intensified exercise with limited recovery. 11 Therefore, it seems plausible that competing in an adventure race would mandate significant physical and psychological recovery and that repeated racing with inadequate recovery might lead to symptoms similar to those seen in overtraining. However, with increasing sponsorship and prize money, athletes are doing more of these multiday races in a year and, while uncommon, can do more than 1 in a month. It is therefore important to develop an appreciation not only of the physiological, psychological, and health effects of such races, but also the requirements of recovery and recuperation.
Sleep deprivation degrades mood,13,14 but whether exercise does 14 or does not 13 exacerbate the mood disruptions is not entirely clear. Moreover, the aforementioned studies involved less exercise or sleep deprivation stress than is encountered in expedition-style adventure racing and tested those effects using people who were not highly trained or participating in self-motivated circumstances. Furthermore, athletes appear more susceptible to mood state changes in the early postrace period, 15 but the mood states during racing and recovery are undetermined. Measuring mood states has successfully identified athletes showing signs of distress due to intense training of high volume. 9 The mucosal immune system plays an important role in protecting the body from upper respiratory tract infection (URTI) and is affected by both exercise and psychological stress/anxiety. A reduction in salivary immunoglobulin A (SIgA) may be seen with either, and low levels of SIgA are associated with an increased risk of URTI, which is, along with low mood, a feature of overtraining. 16
The main purposes of this study were to record athletes’ mood states and record and categorize their symptoms of illness and injury in racing and recovery from an international-level adventure race. A secondary purpose was to identify the possible occurrence of “postrace blues” reported in anecdote at approximately day 3 after the race. We hypothesized that alterations in mood subscores would be evident around the third recovery day, specifically with an increase in depression and reduced vigor.
Methods
This was a prospective descriptive study conducted at the “Southern Traverse Adventure Race,” in Otago, New Zealand, in November 2003. The race was over a course of ∼411 km. It required athletes to race as continuously as possible in mixed-gender teams of 4 (racing concurrently) through wilderness terrain. Disciplines involved kayaking, coasteering (traversing rugged sections of coastline below the high-tide mark), mountain-biking, and running/trekking. Navigation between check points and transition areas was aided by maps and compasses. Sleep was obtained in discipline-transition areas and out on the course.
Participants
There were 2 participant groups. A primary group of 12 athletes from 3 teams was recruited in the weeks preceding the race, referred to as “science teams.” They were recruited from registered race entrants on the basis that they lived locally (within 300 km) and were studied before, during, and after the race for this study and a wider project on physiological effects. 1 Their experience and ability spanned that of the overall race population; one team (team A) all had international experience, another team had extensive Southern Traverse experience (team B), while the third team was comprised of adventure racing novices (team C). A second group consisted of athletes approached at the race registration and invited to participate in the study. Of 116 athletes who started the race, prerace data were obtained from 60 (52%), and all but 1 of these provided finish-line data. Twenty-seven athletes from both groups submitted repeated questionnaires for the 2 weeks following the race. Ethical approval was obtained prospectively through the University of Otago Human Ethics Committee, and athletes provided their informed consent in writing before participation.
Procedures
Illness, injury, and mood data were obtained using questionnaires. Questionnaires were first administered and completed 2 days before the race, once teams had undergone prerace registration. During the race, the science teams were intercepted on a daily basis, usually at check points and transition areas, for immediate completion of questionnaires. At the finish line, participants from both groups completed the questionnaires within 60 minutes of finishing. Athletes were invited at the postrace prize-giving ceremony to participate in the follow-up part of the study. They were given a pack including questionnaires for 2 weeks of follow-up. They were asked to complete the questionnaires at the same time each day and were reminded on the day of assessment by electronic mail. Questionnaires were completed on recovery days 1, 2, 3, 4, 7, 10, and 13 after most athletes finished the race.
Measurements
Mood state was assessed using the Brunel Mood Scale (BRUMS).17,18 The BRUMS was used because of its brevity and ease of administration compared to other longer mood scales because the athletes were required to complete it during the race. The BRUMS contains 24 adjectives relating to 6 elements of mood (confusion, depression, anger, fatigue, vigor, and tension; ie, 4 adjectives for each). Participants were asked to rate how they feel “right now” and respond on a 5-point Likert scale with verbal anchors of “not at all” (1), “moderately” (3), and extremely” (5). Thus, each element of mood could range from a score of 4 to 20. The validity and reliability of the BRUMS has been demonstrated in studies using athletes.18,19 Ratings of pain (locations, types, and intensity of most painful) were appended using the same scale. Participants also reported their duration of sleep in the previous 24 hours on this questionnaire.
Illness and injury questionnaires were those developed and used by the Australian Institute of Sport 20 and supplemented to also record musculoskeletal injury. They were further modified from a 3-point severity scale of subjective ratings of illness and injury to a 5-point scale to attain consistency of ratings with the BRUMS questionnaire for mood (to minimize confusion for athletes while responding to questions during the race). Symptoms were divided into 8 categories: upper respiratory, chest infection, flu, gastrointestinal stress, headache, eye irritation, skin, and musculoskeletal injury/ muscle soreness. Record of any medications taken, including analgesics, stimulants, or dietary supplement, was made to gauge their usage and to potentially assist with interpretation of other data.
Data Analysis
Descriptive data are presented as means ± SD unless stated otherwise. Repeated measures of analysis of variance were employed to investigate changes in each variable across time. The periods in which change was hypothesized to occur were identified a priori, hence planned (repeated) contrasts (using α = .05) with Bonferroni corrections were used as follow-up tests. Specifically, mood ratings after 1 day of racing were compared to those taken before the race (baseline) and those taken after 2 days. The purpose was to examine the effect of the greater exercise intensity over the first 24 hours of racing; also, the period when exercise intensity has undergone its major drop and sleep deprivation is minimal. Prerace ratings of mood, illness, and injury were compared to postrace (ie, recovery period) ratings. Ratings from the end of week 2 of recovery (recovery day 13) were compared to ratings taken through days 1, 2, 3, 4, and 7 of the recovery period.
Results
Initial prerace data were collected on 60 (19 female) athletes (mean age, 36 ± 8 years). Twelve (5 female) athletes (mean age, 36 ± 11 years) were monitored closely during the race as part of the 3 science teams, but 1 female athlete withdrew after 36 hours due to gastrointestinal stress characterized by intractable nausea and vomiting, so these data are from 11 athletes. Postrace testing was performed on 49 (17 female) athletes (11 from science teams), 27 (9 female) of whom were monitored over a 2-week follow-up (8 from science teams). Race duration among finishing teams ranged from 96 to 125 hours. Slower participants were able to finish by being rerouted to a shortened course, thus providing a comparable duration and severity of physiological strain across competitors. Race duration for the science teams was distributed widely in accordance with their respective adventure racing experience—96 hours, 108 hours, and 116 hours, with the latter on a shortened course—yet their exercise intensity profiles estimated from heart rate were equivalent. As detailed elsewhere, 1 exercise intensity averaged ∼64% of aerobic power range across the first 12 hours, but settled to ∼41% by day 2 and thereafter. The work stress was supplemented by adverse weather, including hail and snow storms. Temperatures recorded at the closest meteorological stations within the course (albeit below 300 m) during racing ranged from 2.6°C to 22.3°C, and winds gusted to 71 km·h−1 (NIWA Science New Zealand). Elevation ranged from 0 to 1170 meters. At registration, medication and/or supplement use was reported by 29/60 athletes, with a total of 44 usages. Eleven of those were prescription medication, including nonsteroidal anti-inflammatory medication (NSAID; n = 2), β2 agonists (n = 1), and oral contraception (n = 4). There was no reported use of medications under restriction by the International Olympic Committee.
Sleep, pain, and mood data obtained from 49 athletes at both pre- and end-race sampling times were omitted from the results because the means for that cohort were within 10% of the means for the science teams and follow-up cohorts in all cases.
Sleep
Sleep duration was already compromised on the night preceding the race compared with the previous night (6.4 hours compared to 8.1 hours; n = 10; P < .001). Almost no sleep was obtained on the first night of racing (12 ± 12 minutes), and the mean throughout the race was only 1.2 ± 0.3 hours per day (daily average of 45 minutes, 85 minutes, and 82 minutes for teams A, B, and C, respectively). Sleep duration during early recovery days was only slightly longer than for the second night before racing and appeared to attain a stable 8-hour average by 1 week after racing (∼8 ± 1 hour; Figure 1a). Caffeine was taken in some form by 12/49 athletes, with pseudoephedrine taken by 1 individual. There was no other reported stimulant use.
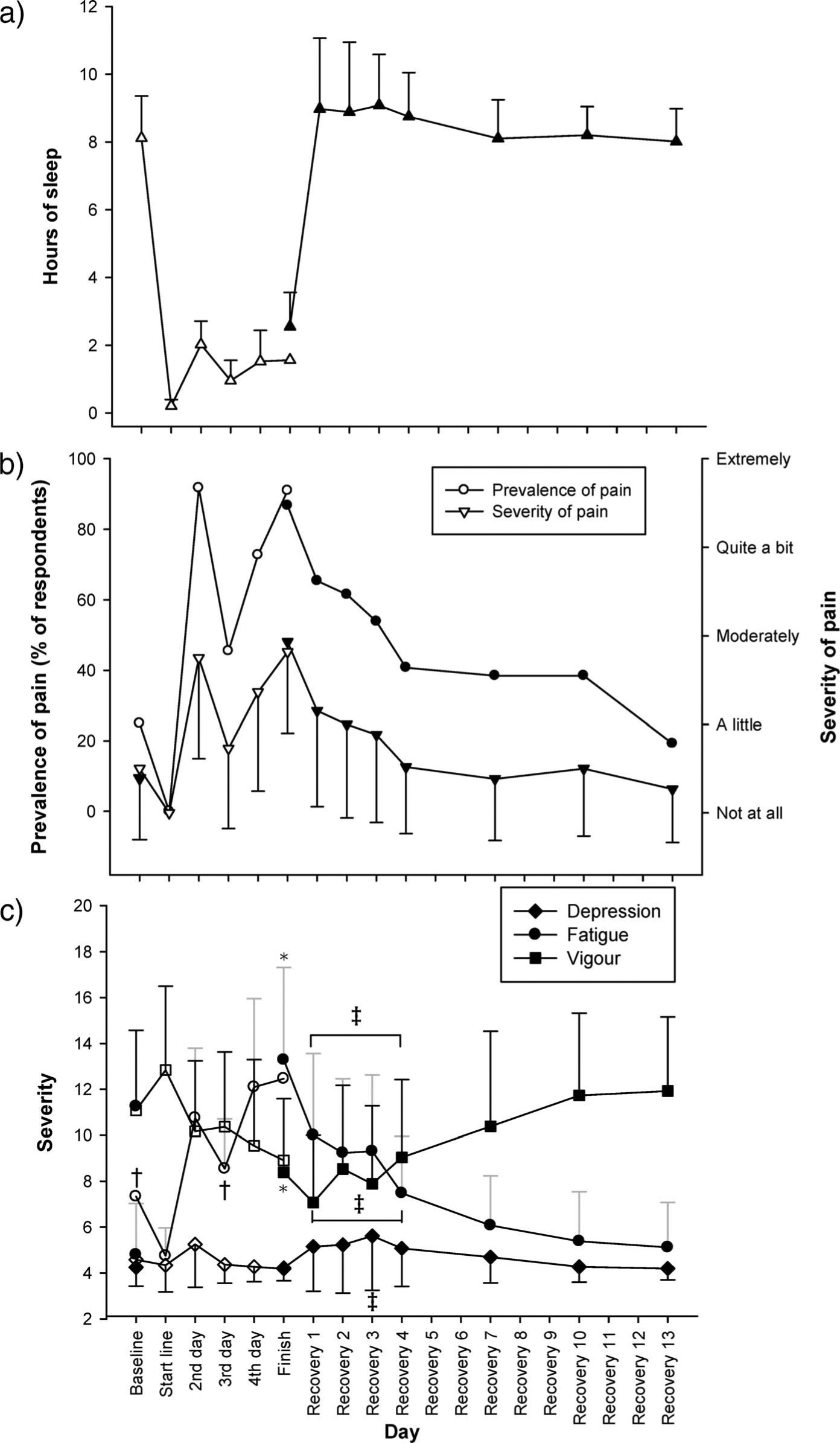
For 11 athletes (hollow symbol) sampled before, during, and at completion of the race, and for 27 athletes (including 8 of the previous 11; solid symbol) sampled across 2 weeks following the race. a, Reported sleep duration. b, Prevalence and mean severity of pain. c, Ratings of mood scores for fatigue, vigor, and depression. Data are means ±SD. * denotes P < .05 between prerace and finish; † denotes P < .05 relative to value on second day of racing; ‡ denotes P < .05 relative to value on recovery day 13. See text for details on sampling procedures and inferential analyses
Pain
Pain was recorded independent of injury because of its potential association with mood. Pain increased (P = .014) to become moderate after 1 day of racing in 95% of respondents and retained this prevalence and severity at race completion (P = .022; Figure 1b). Thereafter it diminished rapidly in prevalence and severity over the first 4 days of recovery and, by 2 weeks, was no more prevalent than before racing. At the finish line, 24/49 athletes reported taking NSAIDs and 8 athletes took simple analgesics.
Mood
Figure 1c shows the scores for fatigue, vigor, and depression pre-, during, and postrace. Anger and confusion scores are not illustrated because they were similar to the depression scores; their mean ratings all remained low and relatively stable (ie, below 6.5, where 4 and 8 denote “none” and “a little,” respectively). Ratings of tension were modest immediately prestart and had dropped (P = .003) to “none” at race completion. Across the race, fatigue increased and vigor decreased (both P < .05; Figure 1c), but the average scores remained at least 1 rating away from end-scale. In the 2-week follow-up period, depression scores were higher on the third recovery day than on all pre-, during, and postrace days; while this was not pronounced, it was statistically significant (P = .008 for comparison with the last recovery day). Fatigue was higher and vigor was lower during the first 4 recovery days compared to the last recovery day (Figure 1c; P < .05). Furthermore, in relation to the phenomenon of “postrace blues,” the recovery of mean mood scores was larger from the third to the fourth day following racing than it was from the first to the third day following racing for every mood subscore except tension (which remained below 5).
Illness and Injury
Symptoms of upper respiratory illness were common at baseline (25/60, 42%) and at race completion (28/49, 57%) and largely resolved over the 2-week follow-up period such that only 5 of 27 respondents were reporting symptoms at 2 weeks (19%; Figure 2a). Only 1 athlete reported the use of β2 agonist medication. The nature of vegetation and terrain in the race made skin injuries common. Skin wounds and infections were reported by most participants (43/49, 88%) immediately after the race. These tended to settle quickly beyond the third recovery day, such that at 1 and 2 weeks, only 10 and 2 of 27 participants, respectively, reported them. Eight of 49 (16%) participants reported gastrointestinal complaints at the finish line. Symptoms settled quickly, with only 2 participants symptomatic on the fourth recovery day. Nonetheless, they were debilitating; 5 teams reportedly withdrew due to gastrointestinal complaints in 1 or more team members.
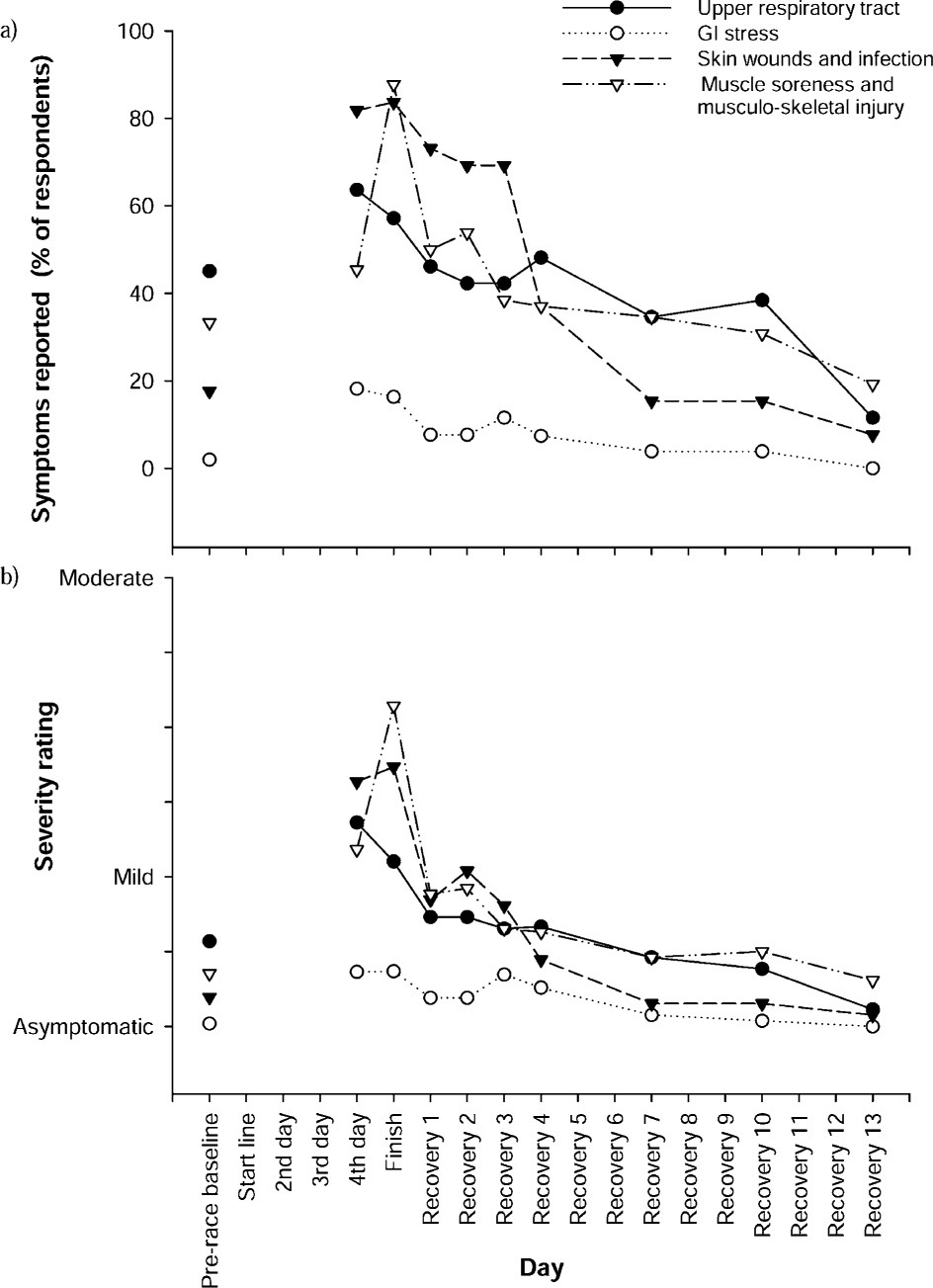
a, Prevalence, and b, mean severity of symptoms reported before (n = 60), during (n = 11), at the finish (n = 49), and for 2 weeks following the race (n = 27). Symptoms rated as being of mild severity equated to athletes being permitted to race as normal, whereas moderate equated to them having to modify their racing
Record was made of 3 athletes from the science teams (1 male, 2 females) who suffered a severe medical illness. One athlete was forced to withdraw during the race having suffered a fractured hand and intractable vomiting (a female from a science team). One was diagnosed with infectious mononucleosis on the 13th recovery day. The other athlete suffered a prolonged course of fatigue with disrupted thyroid function and was followed by an endocrinologist. His recovery was still incomplete at 12 months.
Of the 48 participants for whom musculoskeletal rating data were available at race completion, 38 reported a total of 49 musculoskeletal injuries. Thirty-seven (75%) injuries related to the lower limb, 14 to the foot/ ankle (28% of total), 15 to the leg/thigh (31%), and 8 (16%) to the knee. Within the 2-week follow-up period, there was a total of 27 injuries reported by 18 athletes, but at the 2-week point only 5 athletes were still symptomatic with injury.
Discussion
The purpose of this prospective study was to document athletes’ sleep, mood states, illness, and injury symptoms (self-reported) before, during, and in recovery from an expedition-style, international-level adventure race. This study is unique in that those factors were measured concurrently during racing and across the subsequent fortnight to provide a time course for recovery from a multiday adventure race. The main findings were that athletes were severely sleep deprived during racing (averaging about 1 hour of sleep per day for 5 days) but sleep durations postrace were not overly high; vigor and tension fell during racing while fatigue rose, yet depression, anger, and confusion remained low, indicating that mood was only modestly disrupted during multiday competitive exercise with severe sleep deprivation, and the recovery in mood scores, illness, and injury symptoms was mostly complete within 1 week after racing, but there was a small deterioration of mood on day 3 of recovery, as hypothesized.
Exercise stress, sleep deprivation, mood, and immune function are interrelated, albeit complexly. While it is important to delineate their effects using controlled experiments, it is also important to know the concurrent status of these factors for humans under severe physiological and psychological strain in the field, as during expedition-style adventure racing. Therefore, the main contribution of this study is in documenting these factors in an ecologically valid race setting using athletes who were motivated, committed, likely to have had personal goals for performance, and had responsibilities to the team. These psychological factors cannot be reproduced in an artificially controlled, laboratory-based environment and may be inherently different to those associated with military training exercises. 21 The data obtained from 12 athletes during the race provided the first record of symptoms during such prolonged, sustained exercise. These data thus provide an indication of the race-related development of altered mood, injury, and illness status and overcome sampling bias arising from the so-called “end-spurt effect,” 22 where athletes or experimental participants perform better and provide less disrupted ratings near completion of ultra-endurance exercise or sleep-depriving laboratory experiments. 22 These benefits of sampling during racing must be balanced against interruption of athletes’ performance; while this study aids ecological validity for self-reporting within racing and recovery, the imposition limits sample size and detail of data collected. Longer questionnaires with physical examinations may also give more information but would involve resistance during racing and noncompliance during recovery.
Sleep
Sleep duration was already compromised on the night before racing, perhaps reflecting race preparation time (route, equipment, food) or difficulty sleeping before competition. Sleep deprivation during racing was predictably severe (about 5 hours of sleep in 100+ hours) to an extent greater than in most military training or reported previously for adventure racing, 3 yet sleep duration in the ensuing days was not correspondingly high (Figure 1a). The magnitude of sleep deprivation is an important consideration when interpreting other variables. Sleep deprivation even across 1 day produces mood changes, 14 immune dysfunction, 23 a more catabolic neuroendocrine profile, impaired thermoregulation, 24 and inconsistent but generally negative effects on some physical and cognitive capabilities.25,26 The adverse effects can increase disproportionately to the sleep loss, at least for mood and psychomotor performance,21,27 although this was not evident in our study (discussed below). Such effects may be due in part to lack of creatine in the brain, since creatine supplementation lessens the sleep deprivation–induced impairments of mood and cognitive performance. 28
Obvious limitations with self-reporting sleep are recall bias and recognition of sleep actually occurring. Whereas sleep may have been underreported, major discrepancies were not seen when comparing recall within a team, and reporting accuracy would have been aided by athletes typically falling asleep rapidly and sleeping for predefined periods (when racing). Melody and colleagues 3 reported that three quarters of a larger sample (n = 155 respondents of 250 athletes in race) slept at least 3 hours per 24-hour day during a race lasting 7 to 10 days (based on reporting by athletes and support crew during and after the race). The lesser sleep deprivation in their study may reflect sampling variation and/or race duration, indicating that races lasting greater than 5 days require considerably more sleep, although there may also have been a change in racing sleep strategy over the 8 years between the 2 races.
Mood
Vigor and tension scores dropped across the race, while fatigue increased, and anger, depression, and confusion showed little change. This extent 29 and pattern 30 of mood state disruption is sometimes observed with sleep deprivation by itself. The effects of exercise in conjunction with sleep deprivation, although not fully resolved, may be such that mood states improve acutely with an exercise bout, 14 whereas low exercise volumes (intensity or duration) have little effect on mood 29 and higher volumes impair mood. 14 The current field-based data are interesting in this regard because the exercise volume was larger than has been imposed in the aforementioned laboratory-based studies, yet mood state disruptions were not more severe. Fatigue and vigor showed by far the largest effect, yet fatigue scores at race completion in this study averaged only 14 on a scale from 4 to 20, and most of the increase during racing occurred on the first day (ie, before sleep deprivation). On this day, exercise intensity was substantially higher than on subsequent race days (64% vs 41% of heart rate range 1 ). The modest fatigue at completion seems unlikely to reflect a bias due to the end-spurt effects seen in laboratory studies of sleep deprivation with and without exercise, 14 since the time course was relatively stable across the race (Figure 1c). Therefore, the lesser disruption in mood in the present athletes could be the result of the race environment being more self initiated and autonomous. This should be considered when investigating or interpreting the relationship between sleep deprivation and mood state.
Recovery of mood state disruption was such that the fatigue and lack of vigor normalized between days 4 and 7 into recovery. However, the third recovery day was pertinent; it was the only day on which depression scores were elevated relative to the 2-week, postrace point and showed a demarcation in recovery profile for anger, confusion, vigor, and tension (Figure 1c).
Illness
A high prevalence of respiratory conditions in adventure racing 7 and wilderness multisport 5 has been reported. Respiratory symptoms have also been linked to immune dysfunction (especially a reduction in SIgA) and overtraining in athletes.16,31 In the current study, respiratory symptoms were reported in 28/49 (57%) athletes at the finish line, which is higher than the rates reported by Townes et al 7 (18%) and Rogers et al 5 (9%). The lower prevalence reported by Townes et al 7 presumably reflects their recruitment from the medical tent at the finish line, where athletes would report only if they were concerned about their symptoms at that time. The study by Rogers et al 5 was a 2-day staged, multisport race, which is useful in helping to understand illnesses common across the sport as a whole, but has fundamentally different requirements to those of the continuous, expedition-style, multiday adventure race. Thus, direct comparisons between studies need to be made cautiously, but prevalence of illness appears to be relatively high with expedition-style racing.
Respiratory symptoms associated with overtraining are likely to be of inflammatory, infectious, or allergic etiology. 32 A high prevalence of exercise-related bronchoconstriction has been reported in wilderness-environment racing,2,5,7 but we are unaware of any research yet supporting the notion that upper respiratory symptoms after severe exercise load are linked to transient immunocompromise, (resulting in increased viral shedding, latent virus reactivation, or an increased susceptibility to allergy-mediated disease) in this setting. Irrespective of etiology, it seems likely that respiratory symptoms would generally not be problematic for participation or performance in this race, because symptom severity was not high and exercise intensities and altitudes were generally modest.
Vomiting and diarrhea are common in adventure racing; 33% of competitors affected by illness in the 1995 Eco Challenge experienced diarrhea, 3 and in the present study, 16% of respondents reported gastrointestinal symptoms. As already mentioned, 1 athlete from a science team withdrew because of vomiting. Causes for these symptoms are not readily apparent, but could include infective, osmotic, and central factors. Quality of drinking water is an issue in adventure racing, because total water turnover is relatively high and water is routinely obtained en route. Infective causes should also include a consideration of endotoxaemia, which may be related to heat, exercise intensity, and duration, particularly given the high sustained intensity throughout the first day of racing. 1
Injury
Records of illness and injury in adventure racing has previously been published. A study by Melody et al 3 from the Eco Challenge in 1995 showed a high proportion of symptoms relating to feet and skin that was mirrored in the present study. They were not assessed medically. In another study at Primal Quest 2002, Townes et al 7 published data collected from athlete presentation to the medical tent. There, skin injury was also common and represented almost half of medical consultations. Townes et al 7 showed a comparatively low prevalence of musculoskeletal injury, indicating that although symptoms of pain and injury were likely to be present, they do not necessarily present to a medical facility. Medical problems concerning skin complaints (especially blisters) and the illnesses categorized as respiratory/ altitude sickness were much more likely to be reported. However, differences in data collection between that study and this one prevent the direct comparison of prevalence of illness or injury.
In the present study illness and injury were self-reported. While this raises obvious study limitations relating to diagnosis and pathology, there are also benefits in this methodology. It is the perception of these factors that is important in the context of the sleep-deprived individual with high pain ratings and altered mood state.33,34 Additionally, complications of clinical conditions make primary diagnoses difficult to collate; for example, does someone with endotoxaemia with vomiting get labeled as “dehydration” or “gastrointestinal stress”? By self-reporting on the symptoms of illness and injury and their severity, a measure of the athlete's perception of a problem is made. The definition of “injury” or “illness” in this study is simply the identification of a symptom by the athlete and subsequent rating of its severity. In some circumstances, this may be “less inaccurate” than incorrectly defining causative pathology. It is also important to note that without attributing diagnoses or medical management to the symptoms recorded, these data should not be used to formulate a medical management plan for adventure racing. To do so would risk catering for symptoms that might never present to a medical service.
The study by Melody et al 3 reported a particularly high prevalence of dehydration (41%), consistent with their race setting. The role, or indeed, presence of hypohydration in adventure racing will presumably depend on water availability and ambient conditions, although this must also be viewed with some caution, particularly in the context of popular sports medicine guidelines for endurance exercise per se. For instance, our participants drank ad libitum and finished the race hypervolaemic, with normal urine specific gravity and serum sodium concentrations. 1 Yet, the position of the American College of Sports Medicine 35 has been that thirst cannot be relied on to provide complete restoration of water lost by sweating. Importantly, the research underpinning those guidelines and the target audience are not of multiday adventure racing. Clinically, drinking ad libitum may be a safer and performance-effective practice in endurance sports, particularly with increasing duration and thus lower exercise intensities and sweat rates and higher usage of NSAIDs, 36 all of which characterize this race circumstance. Interestingly, the new (2007) position of the American College of Sports Medicine 37 may not conflict with the notion of ad libitum drinking. In the future, measuring thirst may be a more appropriate tool in self-reported scales.
Conclusions
Sleep, mood, injury, and illness reports were obtained before, during, and for 2 weeks following an expedition-style adventure race. These data showed that although athletes underwent severe sleep deprivation in racing, their mood states were not substantially disrupted other than in fatigue and lack of vigor, which developed quickly in association with a high exercise intensity on day 1. Furthermore, pain, injury, illness, mood, and nightly sleep durations recovered relatively quickly (mostly within 1 week) following racing despite a minor worsening of mood 3 days postrace.
