Abstract
Objective
Direct evidence that dehydration results from scuba diving is scanty. Increased hematocrit (Ht) is a commonly used proxy measure for dehydration. This study sought evidence that an increase in Ht occurs over the course of a scuba dive in tropical conditions. As a secondary outcome, evidence was sought that the degree of Ht increase is correlated to pressure exposure.
Methods
Twenty male and 21 female scuba divers were recruited at a remote tropical dive site. Water temperature was 30°C (±1°C). Each diver gave venous blood relating to 1 dive only. Mean maximum dive depth was 13.6 m (±3.7 m [SD]) and mean duration 39.5 minutes (±4.5 minutes [SD]) using air as the breathing gas. Blood was taken at a mean of 12.4 minutes (±3.5 minutes [SD]) before diving and a mean of 16.2 minutes (±3.7 minutes [SD]) after diving. After centrifugation of microcapillaries, Ht was estimated on a visual plate reader.
Results
A paired Wilcoxon test showed evidence (P < .001) for a change in Ht. The mean difference between predive and postdive measurements was 0.0073 (95% confidence interval: 0.0104–0.0042), equating to a mean relative Ht increase of 1.78%. Similar results were found for the sexes individually. A correlation between maximum depth of dive and Ht increase was statistically significant, although the correlation itself was weak (P = .049, Spearman's r = .326).
Conclusions
There is evidence of a statistically significant increase in Ht over the course of a single warm-water scuba dive. This increase is small and is within the range of error associated with the techniques of Ht estimation employed in this study. Depth exposure was found to correlate with Ht increase. In view of the small magnitude of change in the Ht, there is no reason to amend protocols for fluid resuscitation of recreational scuba divers suspected to have experienced decompression injury in tropical locations.
Keywords
Introduction
Hematocrit (Ht) can be used as a proxy measure of hydration, with the benefit to the field worker that the procedure is simple to perform with limited equipment. Hematocrit is, however, an estimation of intravascular hydration only and makes no differentiation between fluid loss from the body and fluid shift between compartments. Being a ratio, its value reflects changes in both red cell mass and plasma volume. In health, the former is assumed to be constant, although for the recreational scuba diver this assumption may not necessarily apply, as splenic contraction has been recognized as part of the human response to diving 1 and to exercise. 2 The hematological changes prompted by this mechanism are known to return to baseline values within 10 minutes following a breath-hold stimulus3,4 and within 20 minutes of the cessation of maximal exercise. 5 Whether these findings apply to the non–breath-hold scuba diver undertaking moderate exercise at depth is unknown.
The activity of scuba diving offers a number of further mechanisms with potential to cause an increase of Ht. The hydrostatic effect of immersion is thought to oppose gravitational pooling of blood in dependent areas of the body and to increase venous return from the peripheries. In the face of this relative central fluid overload the release of atrial natriuretic peptide (ANP) is thought to prompt the removal of excess plasma volume by the kidney. 6
Based on this theory, Ht should increase upon return to surface pressure since, with the peripheral circulation restored, the ANP-invoked diuresis at depth should yield a relative hemoconcentration by the time of return to the surface. This diuresis of immersion is potentiated by immersion in cool water, 7 possibly on account of increased peripheral vasoconstriction in such conditions. Yet although increased ANP levels have been demonstrated in both deep chamber 8 and shallow water immersion 9 settings and have been associated with increased urine flow, the evidence for an accompanying increase in Ht is inconclusive. Previous studies reporting the Ht response to diving interventions have featured prolonged immersions, 10 repetitive breath-hold diving series, 11 high absolute pressures, 12 and dry chamber environments 13 ; some of these studies have observed Ht increases of 5% to 10%; others have reported no significant evidence of a change, but none can be said to have replicated the typical scuba diving experience.
Fluid shifts from the intravascular compartment to the extravascular space could also potentially cause an increased Ht in divers. The clinical entity of pulmonary edema of immersion 14 is a recognized expression of this phenomenon with respect to the lungs, although it is uncommon in tropical waters. 15 Whether scuba diving routinely provokes a more generalized, subclinical fluid shift remains unclear.
Divers may also be prone to increased water loss from the lungs in response to breathing dry gas. In a study of 6 saturation divers undergoing exercise at 37 atmospheres absolute while breathing helium and oxygen, Ronnestad et al 16 measured a respiratory water loss of 1.7 g·min−1. As a result of the pressure and the gas mix employed, the application of this finding to the recreational diving environment is not straightforward. It would seem, however, that the extra water loss incurred via this route is likely to be small.
This study sought to quantify Ht change in the setting of a single warm-water scuba dive. As a secondary measure, the correlation between Ht change and pressure exposure was explored.
Methods
This project was approved by the South Devon Local Regional Ethics Committee in the UK and was performed in accordance with the ethical standards of the Declaration of Helsinki (1964).
Twenty males and 21 females with a mean age of 20.7 years (±3.4 years [SD]) were recruited from volunteers on a tropical reef survey site. All gave informed consent for their participation. Using air as the breathing gas, divers at the site performed a maximum of 2 dives per day in accordance with the no-decompression practices of the Professional Association of Diving Instructors. A minimum surface interval of 4 hours was universally applied. For the purposes of the study a single sheltered dive site was used. The researchers had no control over the organization of the dive program; volunteers were simply approached on an ad-hoc basis when they were rostered to dive on the selected site.
Each volunteer gave 2 blood samples relating to a single dive, the first taken as late as possible before descent, the second as soon as possible after surfacing. Blood was collected directly into EDTA tubes of the Vacutainer system. No regimen of hydration was used beyond general advice to all divers to keep themselves well hydrated in tropical conditions. No fluid intake was allowed between sampling. The maximum depths of each dive were recorded from divers’ depth monitors. Blood samples were transferred to a closed cool box, which was kept in the shade until centrifugation and cooled by the addition of prefrozen water bottles. Centrifugation was carried out on the evening of the dive in a Hawksley 01410 microcentrifuge, the rotation speed of which was independently verified immediately prior to the study as being within the manufacturer's recommended tolerances. A minimum of 3 microcapillaries were prepared from each blood sample. These were centrifuged for 5 minutes prior to Ht estimation on a standard visual plate reader. All microcapillaries were prepared and read by the same researcher, who was not blinded to the study.
On the assumption of a SD for Ht of 0.025, 17 which is the same for both sexes, 18 a paired t test would require 15 subjects to detect an Ht change of 0.02 at a significance level of 5% and 80% power. To detect a change of 0.01 would require 52 subjects. To minimize the possibility that subjects with an initial Ht in the tail of a normal Gaussian distribution may react atypically to the exposure, sample pairs would be excluded if the initial Ht exceeded 2 SD from the same-sex mean in the study population.
Of the 41 sample pairs collected, 2 pairs yielded a pre-dive Ht greater than 2 SD from the population same-sex mean; 1 pair contained a hemolyzed sample and a fourth showed hemodilution over the course of the dive (the only sample pair to do so) and was excluded on suspicion that pre- and postdive samples had been incorrectly labeled. Thirty-seven sample pairs were thus included in the final analysis.
Results
The water temperature during the study period was 30°C at the surface, falling to 28.5°C over the depths at which the divers were active. Blood samples were drawn on the shore as late as possible before and as soon as possible after the dive. Dive characteristics are shown in Table 1. Delays were incurred by divers donning and removing dive equipment and traversing the reef between the shore and the point of descent.
Dive characteristics (n = 37)
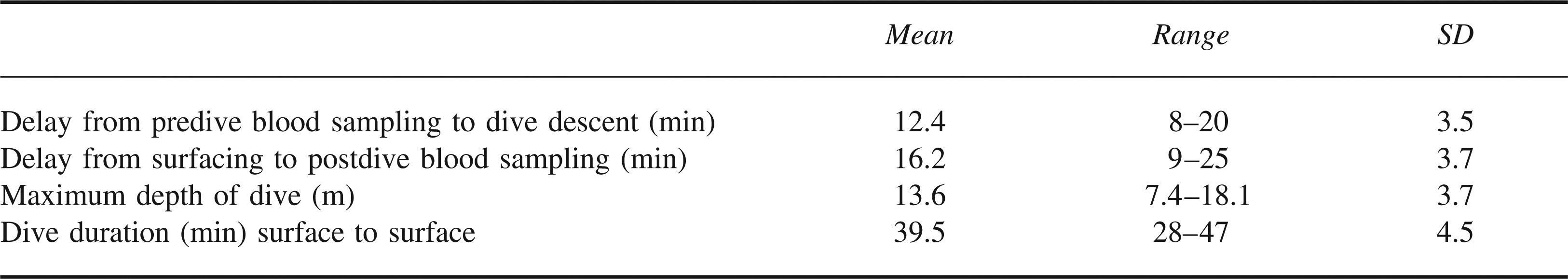
Distribution of maximum depths reached was bimodal, with peaks at 10 m and 17.5 m. There was no significant evidence of a correlation between dive duration and maximum dive depth (Spearman's r = −.241, P = .151). Mean values for pre- and postdive Ht are shown in the Figure.
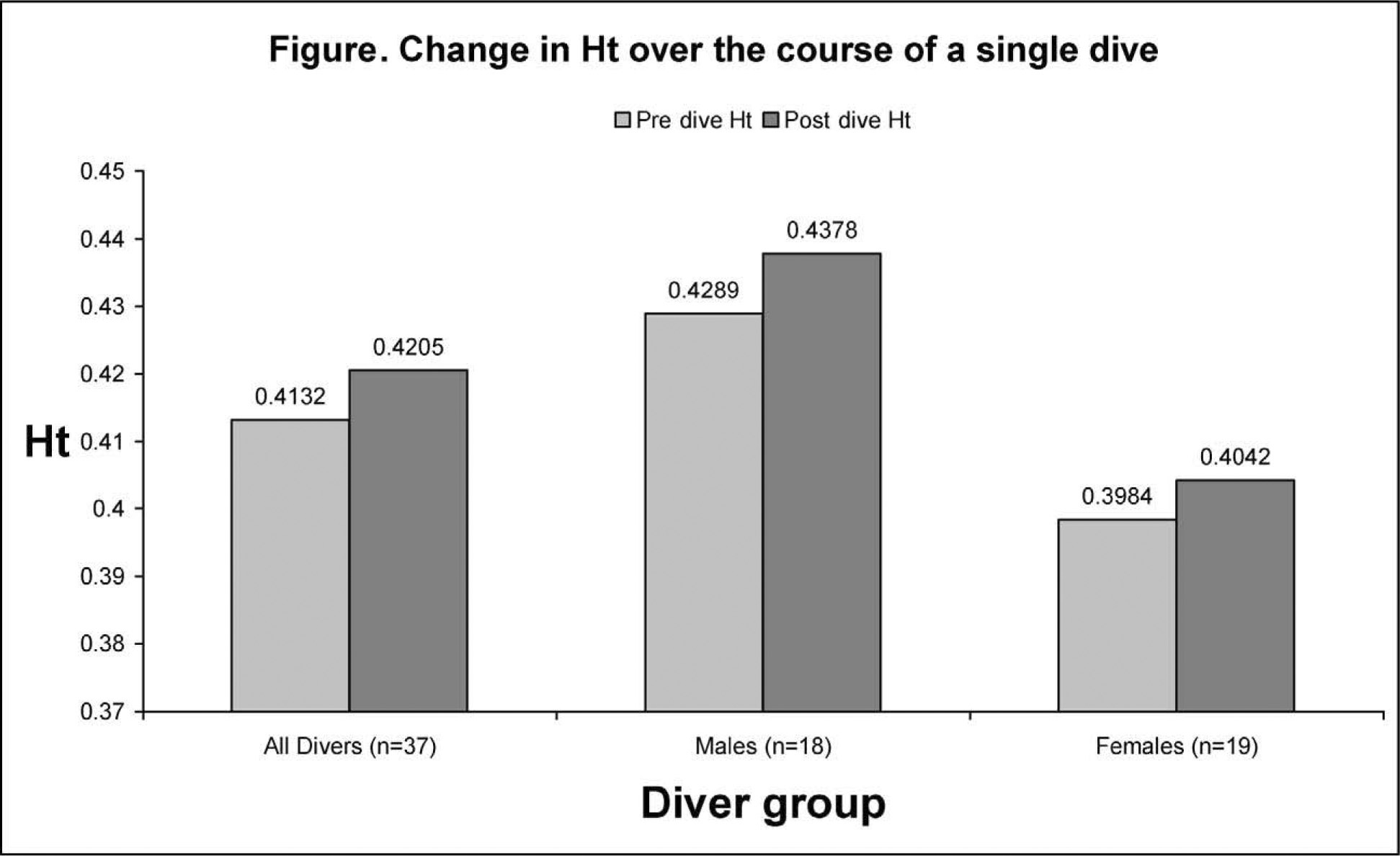
The change in Ht was calculated for each diver. Mean values are shown in Table 2. These values were not normally distributed. Comparison of pre- and postdive pairs was performed by nonparametric Wilcoxon test, as shown in Table 3. The test statistic (P < .001) gives statistically significant evidence of a difference in the pre- and postdive Ht readings for all diver groups.
Mean absolute and relative changes in hematocrit (Ht)
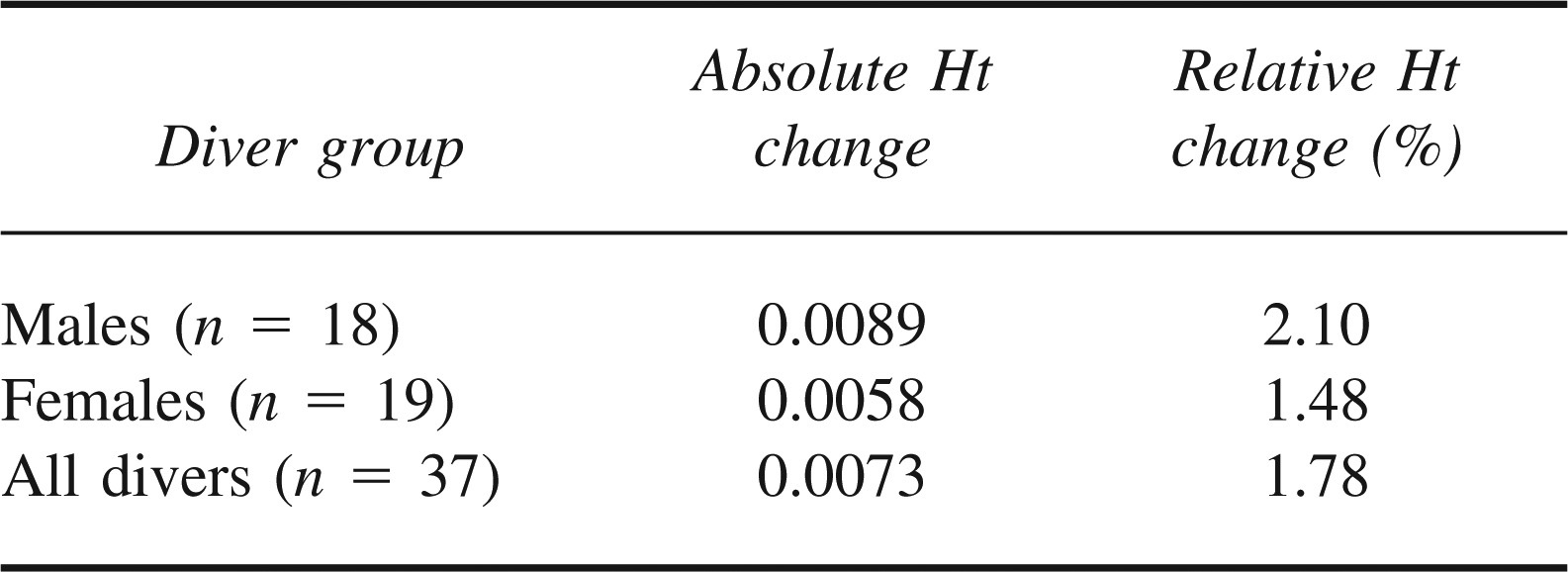
Comparison by Wilcoxon test of pre- and postdive hematocrit (Ht)
A correlation was sought between maximum depth of dive and change of Ht. Two-tailed analysis was statistically significant (P = .049), but the coefficient of .326 (Spearman) indicated a weak correlation only.
Discussion
Our findings provide evidence that there is an increase in Ht over the period of a single tropical scuba dive. But although statistically significant, the mean relative change of 1.78% is small. When not diving, volunteers were occupied in alternative activities on the site, occasionally involving physical activity in hot, outdoor environments. This prevented the establishment of a valid control group, and a comparative baseline rate against which to judge the small change observed was consequently not elaborated.
Constraints of manpower in this remote location did not permit the independent preparation and reading of microcapillaries; it is thus possible that lack of blinding has exaggerated the findings and that the true difference is smaller than reported. Microhematocrit estimation is, furthermore, subject to a natural error on account of a variable quantity of plasma that is trapped within the microcapillary in the red cell column. The magnitude of this error in healthy subjects is on the order of 1.5%. 19 All centrifugation occurred at the same time of day, in the same location, and in similar ambient temperatures. But although a cool box was employed and was intermittently recharged with prefrozen water bottles, the lack of a refrigerator meant that a uniform storage temperature could not be guaranteed. It is possible that this too may have influenced results, as red cell hemolysis is known to occur as a result of excessively warm storage conditions. 20 The mean increase in Ht observed in this study is not so great that it cannot be explained by the combined error from these sources. We conclude that if hemoconcentration does indeed occur over the course of a single warm-water, recreational dive, the degree of Ht change is small and less than has been reported in some previous physiological experiments.
Fluid resuscitation of any diver suspected of having suffered decompression injury is recommended as standard treatment, regardless of location,21,22 and is still to be recommended in tropical environments, where fluid losses associated with elevated ambient temperatures are likely to be high. But in such tropical conditions, and where the diver has performed only a moderate recreational dive, the degree of hemoconcentration attributed to the dive exposure itself may be assumed to be small. Commercial rehydration solutions, administered orally, contain appropriately balanced electrolytes for enhanced absorption and are the treatment of first choice in such cases. 23
This study did not attempt to identify the physiological mechanisms responsible for the observed increase in Ht. The hematological effects of splenic contraction have been shown to return to baseline within 20 minutes of maximal exercise; given the delay of 16.2 minutes in this series between surfacing and postdive blood sampling, it seems likely that the contribution of splenic contraction to the observed increase in Ht will be small.
Sweat production due to immersion in thermoneutral seawater (34.5°C) has been calculated to be 0.05 kg after 2 hours of exposure. 24 The sea temperature in our study was below this level and exposures were shorter. In view of these circumstances it is unlikely that sweating was responsible for the Ht changes observed. Divers’ choice of insulation for repeat dives at the site gave no clear evidence as to their perceptions of cold, since some simply wore bathing suits while others habitually wore thin neoprene. Some who adopted wetsuits complained of overheating at the surface. It is possible that wetsuits were worn for protection from abrasion and stings rather than for reasons of thermal insulation. Nonetheless, cool water diuresis may have played a role in the mean Ht increase observed, since increased rates of urine output have been reported at temperatures within 2.5°C of thermoneutral. 7
The possibility of intercompartmental fluid shift requires comment. All participants were diving on a daily basis with a rest day every seventh day. Divers were approached for participation in the study on an ad-hoc basis, with no record made of their recent diving activity. Although minimum surface intervals were never less than 4 hours, our results could still be affected if recent, preceding diving exposure had caused an intercompartmental fluid shift requiring a prolonged period for correction.
The weakness of the correlation between depth and Ht increase may reflect the limited nature of depth data available for analysis in this study. Few divers wore sophisticated dive computers, and, consequently, the only depth data consistently available concerned the maximum depths reached. The dive site for this study featured a reef, which gave way entirely to sand at depths >10 m. For divers who recorded a depth of <10 m, the maximum depth figure may be regarded as a good indicator of the depth at which most of the dive was performed. But divers who ventured deeper than 10 m were generally morning divers who were gaining depth in order to avoid a reverse profile exposure on their second dive later in the day. Having satisfied this requirement, they tended to ascend to the reef. It is probable that the maximum depths they recorded indicate more severe dive exposures than were actually incurred; it is thus possible that the true correlation between depth and Ht increase is stronger than was found in this study. Our principal finding of only a small change in Ht during recreational diving should therefore be interpreted with caution when deeper diving is performed. To assess adequately the changes incurred by deeper exposures, a larger study would be required, incorporating a monitoring system capable of recording a continuous depth-time profile for each participant. Monitoring of this complexity exceeded the resources available for this project.
Footnotes
Acknowledgments
We extend our thanks to Hilary Sanders and Steve Shaw at the University of Plymouth for their statistical support, to Ann Tabra at Torbay for her hematology tuition, to Gary Smerdon for proofreading and suggestions, and most of all to Coral Cay Conservation, which allowed us access to the dive site and without whose generous cooperation the study could not have been completed.
*
Presented at the Underwater and Hyperbaric Medical Society Annual Meeting, Las Vegas, NV, June 16–19, 2005.
