Abstract
Silk fibroin (SF)-based biocomposites have attracted considerable attention for several decades, due to excellent biocompatibility and mechanical toughness. However, insufficient hydrophilicity and mechanical brittleness limit its practical application, especially in tissue engineering. In this study, by blending konjac glucomannan (KGM) with SF, along with the water-insoluble treatment of ethanol and ammonia, SF/KGM composite membranes were generated with good flexibility and water adsorption capacity. Scanning electron microscopy (SEM) showed that KGM/SF membrane surfaces were uniform and dense. Fourier transform infrared spectroscopy (FTIR) showed that the silk II structure was dominant. Contact angle and mechanical testing showed that the use of KGM with SF in membranes helped to form more flexible and hydrophilic membranes for potential use in biomaterials and devices.
Introduction
Natural polymer composites have drawn a great deal of interests in recent years because of their superior material performance and great potential for use in biomedical fields.1,2 Silk, extracted from the
SF possess excellent film-forming ability, which makes it an outstanding candidate for membrane science. Silk fibroin membranes are permeable to dissolved oxygen under wet conditions, similar to human skin, and has potential applications in wound dressings and artificial skin systems.7,8 However, SF membranes are soluble in water because its dominant form is the random coil structure. Treating SF with ethanol is a mature technique for improving the water solubility of SF products.7,9 Unfortunately, in the dry state, these pure SF films become hard and brittle over time, showing impressive stiffness but low elongation. Therefore, it is still necessary to modify the physical and mechanical properties of silk films to control their performance, and to mainly develop more flexible systems. Konjac glucomannan (KGM) is a pharmacologically-active heteropolysaccharide. It is a neutral and water-soluble linear copolymer derived from konjac tubers, having β-1,4-linked D-mannose, D-glucose, and acetyl groups randomly attached to sac-charide units.7,10 Biocompatibility studies have shown that membranes of konjac glucomannan cause little damage to red blood cells and has certain anticoagulant properties and blood compatibility. 10 KGM is also used as a stabilizer in dairy products. Chen and his workmates used konjac flour to prepare microporous gel carriers for immobilization of microbial cells and enzymes. 11 Wang et al. chemically modi-fied KGM and prepared a carrier for immobilized enzyme by insoluble treatment. 12 KGM, having high water holding capacity and biocompatibility, was used recently for making contacts lenses, homeostasis sponges, and wound dressing materials. In other words, KGM is a high value-added biomaterial that not only has good water retention, but also has biomedical use potential. Many researchers have turned their attentions to blend composite membranes for improving the properties of single membranes in terms of tensile strength, breathability, and water permeability.7,13
To overcome the limitations of single SF and KGM components, we hypothesize that combining the above two bioactive natural polymers can be used to create SF/KGM composite membranes with superhydrophilicity and flexibil-ity. Our results suggest that SF/KGM composite membranes present a uniform and dense surface, giving it good mechanical behavior. In addition, the hydrophilicity and flexibility of the composite membrane increased with the KGM content. The abundant hydroxyl group presence in KGM imparts water-retention abilities, and results in creation of a moist environment, promoting the development of SF/KGM composite membranes in biomedical applications such as wound dressings.
Materials and Methods
Preparation of SF and KGM Solutions
Preparation of SF/KGM Composite Membranes
The final solute of SF and KGM was set at 0.3 g. The KGM solution was added into the SF solution in various KGM/ SF ratios of 0:100, 25:75, 50:50, 75:25, and 100:0 (w/w), and followed by ultrasonic treatment (SCIENTZ-IID, Ningbo, China) for 10 min. The homogenous solution was poured into a petri dish (90-mm diameter, Sinopharm China), de-foamed, and allowed to dry naturally. Air-dried pure SF membrane was immersed in 75% ethanol solution and pure KGM membrane was immersed in 25% ammonia solution, 7 respectively as control groups. The SF/KGM composite membrane was immersed in ethanol/ammonia (75/25 v/v) at an LR of 3:1. After incubation for 2 h, the membrane was washed with deionized water, dried at RT, and stored at 4 °C.
Characterization of Composite Membranes
The morphology of the SF/KGM composite membranes was observed by scanning electron microscopy (SEM, Hitachi, S4800, Japan). Structure analysis was performed by Fourier-transform infrared FTIR (Vertex70, Bruker, Germany) spectroscopy. Tensile performance was performed using an Instron 5566 universal material testing machine, with a chuck sensor force of 1 kN, tensile speed of 10 mm/min, clamping distance of 30 mm, spline width of 10 mm, and sample dimensions of 10 × 50 mm. Water contact angle (WCA) analysis was carried out using a JY-820 (Chengde, China) instrument. The SF/KGM membranes (20 × 20 mm) were fixed on the measuring stage, and the static angle of each film was measured, photographed, and recorded. Surface WCA values greater than 90° were considered as hydrophobic surfaces, and those less than 90° were considered as hydrophilic surfaces.
Results and Discussion
Morphology
In general, all composite membranes had a uniform and dense surface (Figs. 1 A1–E1). However, the cross-section morphology of each group was different (Figs. 1 A2–E2). After ultrasonication, the SF can be evenly mixed (Figs. 1 B–D). The composite membranes had smooth surfaces with no obvious phase separation. The cross-section of the pure SF membrane was uneven and rough, and accompanied by a large number of cracks. SF is an amphoteric polymer with hydrophilic and hydrophobic chains arranged alternately. During air drying, due to the difference in water distribution and volatilization, the hydrophobic segments of SF membranes accumulate, forming a rough section. 2 In the dry state, the SF membrane is brittle and cracks are formed during sample preparation. Due to the poor mechanical properties of pure KGM film, the morphology was rough and cracked. With an increase in the KGM component of SF/KGM membranes, the water distribution was more even. 7 Therefore, the cross-sections of SF/KGM composite membranes were uniform and smooth (Figs. 1 B2-D2).

SEM of KGM/SF membrane surface and cross section. (A) Pure SF, (B) KGM/SF = 25:75, (C) KGM/SF = 50:50, (D) KGM/SF = 75:25, and (E) pure KGM. A1–F1 images were at a scale of 500 μm and A2–F2 images were at a scale of 10 μm.
Structure Analysis
Fig. 2A shows the FTIR spectra of the KGM membrane. The untreated pure KGM films showed a characteristic peak at 808 cm–1. A characteristic peak belonging to man-nose appeared at 874 cm−1, and a weak C=O vibration peak appeared at 1724 cm–1, indicating some acetyl groups in KGM. At 2884 cm–1, a vibration peak of methyl hydrogen (C-H) bond appeared, and at around 3310 cm–1, the oxygen-hydrogen bond (O-H) was observed. Pure KGM after alkali treatment showed a significant decrease in the C=O peak at 1724 cm–1, demonstrating the loss of the acetyl group. 12 , 14 The result shows that ammonia dehydrated the KGM membrane and removed acetyl groups, improving the water resistance of KGM. The presence of acetyl groups of KGM molecule restrained the formation of both intramolecular and intermolecular hydrogen bonds, and elimination of ste-ric resistance resulted by removing acetyl groups prompted the interaction between KGM molecules.
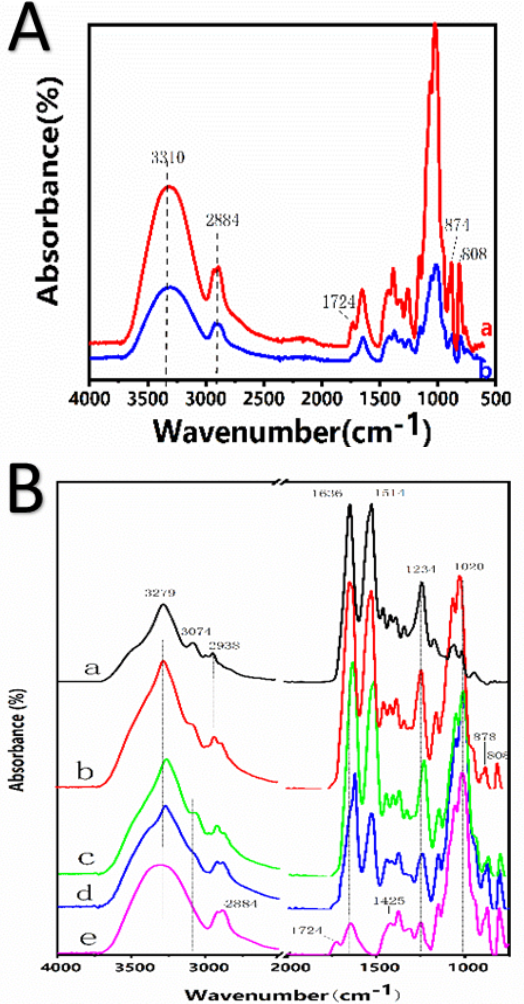
(A) FTIR of konjac glucomannan. (a) Untreated KGM film and (b) pure KGM membranes insoluble in water. (B) FTIR of KGM/SF composite membranes. (a) Pure SF film, (b) KGM/SF = 25:75, (c) KGM/SF = 50:50, (d) KGM: BSF = 75:25, and (e) pure KGM membrane.
As shown in Fig. 2B, the SF membrane showed strong FTIR absorption bands at 1636 (amide I), 1514, (amide II), and 1234 cm1 (amide III), corresponding to the water-stable (3-sheet structure. These results indicated that the successful crosslinking of ethanol made the composite water insoluble. The silk II structure of the SF macromolecule was dominant. The presence of ethanol in water can induce transformation of the SF secondary structure from random coil to a (3-sheet structure. 2 , 8 The introduction of KGM did not change the secondary structure of SF molecules.
Hydrophilicity
A water contact angle (WCA) test was used to assess the hydrophilicity of the composite membrane. Hydrophilicity of the material was determined by measuring the angle between the droplet and the plane. When the angle was acute, the material was hydrophilic and when the angle was obtuse, the material was hydrophobic. As shown in Figs. 3A-E and Table I, all samples were hydrophilic due to the large number of hydrophilic groups (-OH, -COOH) in both SF and KGM molecular chains.9,10
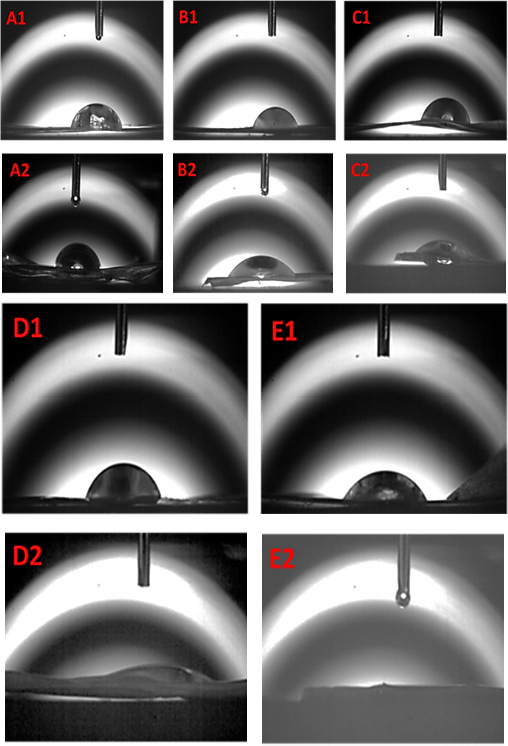
WCA testing of KGM/SF composite membranes. (A) Pure SF; (B) KGM/SF = 25:75; (C) KGM/SF = 50:50, (D) KGM/SF = 75:25, and (E) pure KGM. 1 means untreated and 2 means insoluble in water (treated).
List of WCA of KGM/SF Composite Membranes
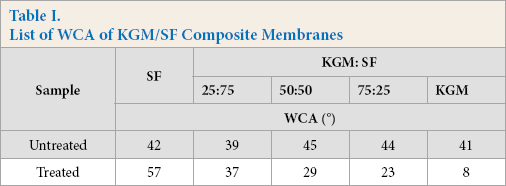
Hydrophilic biomaterials are of interest in tissue engineering because they can better support cell adhesion, proliferation, and promote tissue regeneration. 2 , 8 Untreated composite membranes are unstable and heterogeneous, impeding the penetration of water droplets. 15 SF/KGM composite membranes treated with the ethanol/ammonia mixture showed an acute WCA, indicating greater hydrophilicity, making them more useful in the biomedical field.
KGM has more hydrophilic groups than SF. 7 Therefore, the hydrophilicity increased with the increase of KGM in the SF/ KGM composite membranes. This tunable hydrophilicity expands the functional properties of SF-based composites, making them more compatible as biomaterials.
Tensile Testing
A functional composite material inevitably bears various stresses in practical application. Therefore, good tensile strength and proper flexibility are important to maintain the integrity of the composite membranes. 16 , 17 Pure KGM materials are either very stiff when dry or have poor shape when wet. 7 ,10-12 After blending with SF, the shape stability of the material was improved. In addition, tensile testing was conducted to study the tensile performance of SF/ KGM composite membranes when wet. The stress-strain curve of the composite membranes is shown in Fig. 4. KGM possessed stronger water-adsorption, and thus showed a significantly reduced modulus compared to the SF membrane. 7 The pure SF membranes were stiff and had low extension (< 20%), while the pure KGM membranes had low stiffness (< 0.5 MPa) and high extension. These inadequate mechanical behaviors limit their further development. By blending SF with KGM, the mechanical behavior of SF/KGM membrane was complementary. When the ratio of KGM reached 25%, the SF/KGM composite membrane could be stretched over twice its own length and still maintain good tensile strength. By continuing to increase the content of KGM, we found that the stress and strain of the SF/KGM composite membranes decreased, which may be due to the SF/KGM composite membranes absorbing a large amount of water, leading to the swelling of the composite membrane. The gap between the molecules was thereby widened, reducing the interaction between the molecules.
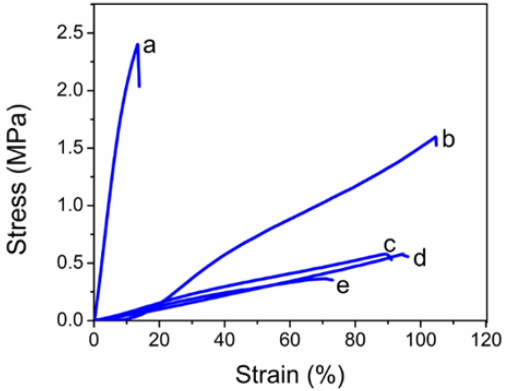
Tensile stress-strain diagram of water-insoluble KGM/SF composite membranes. (a) pure SF membrane, (b) KGM/SF = 25:75, (c) KGM/SF = 50:50, (d) KGM/SF = 75:25, and (e) pure KGM membrane.
Conclusion
In this study, SF/KGM composite membranes were successfully fabricated. The mechanical behavior and hydrophilicity of these membranes could be tuned by varying the ratio of SF to KGM. SF/KGM membranes with smooth and dense surfaces showed enhanced tensile strength and flexibility. Water contact angle (WCA) testing demonstrated the excellent hydrophilicity of SF/KGM composite membranes. This study provides a simple and effective way to prepare novel protein/polysaccharide-based biomaterials for potential biomedical applications.
Footnotes
Acknowledgments
The authors acknowledge funding support from the Wuhan Municipal Science and Technology Bureau (2019010701011388), the National Natural Science Foundation of China (51803154), and the Hubei Provincial Department of Education (D20191706).
