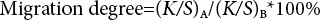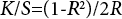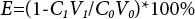Abstract
In this paper, two disperse dyes with different substituents were selected to dye polyester fabrics in decmethylcyclopentasiloxane (D5) medium. The structures and dyeing performance of the two dyes were studied and compared. The results showed that dyes with lower solubility in D5 medium had higher dyeing rates. DFT calculations showed that large steric hindrance of cyano groups on diazo components contributed to improving the dyeing rate. This was due to the generation of more monomolecular dyes by destroying π-π stacking interactions. These results provide theoretical guidance for improving the exhaustion and further promote the industrial development of disperse dye in a D5 dyeing system.
Introduction
Polyester fibers are aromatic polyester from a mineral oil source; they have become the highest consumed textiles in the world due to their excellent performance.1,2 However, the conventional dyeing processes for polyester usually cause serious pollution. The discharged wastewater contains unfixed dyes, dyeing auxiliaries, carcinogenic organ-ics, heavy metals, and so on. 3 Pollution has inhibited the development of the traditional textile industry. In response, people have reduced pollutant emissions in textile dyeing by using new technology or dyeing media. These developments include plasma dyeing, 4 digital printing, 5 supercritical CO2 dyeing,6-8 and other approaches. Even these technologies have certain limitations. For example, supercritical CO2 staining can dye synthetic fibers, but industrial production is still limited due to its high-pressure environment and expensive equipment. 9 Solvent dyeing, such as DMSO and carbon tetrachloride, can achieve dyeing without auxiliaries; however, they are not environmentally friendly, and are also harmful to the human body.
In our previous work, D5 was selected as an environmentally friendly medium for dyeing polyester fibers. D5 has advantageous properties such as being non-toxic, non-polar, easy to reuse, and stable to many chemicals. The chemical structure of D5 is shown in Fig. 1. The results showed that D5 will be an ideal dyeing medium to realize better dyeing performance without adding any auxiliary agent, and the medium can be recycled many times. Unfortunately, the absorption rate of disperse dyes in D5 system is low. The previous studies only focused on the influence of different chromophores on dyeing properties; up to now, the relationship between the substituents and dyeing performance has not been systematically studied.
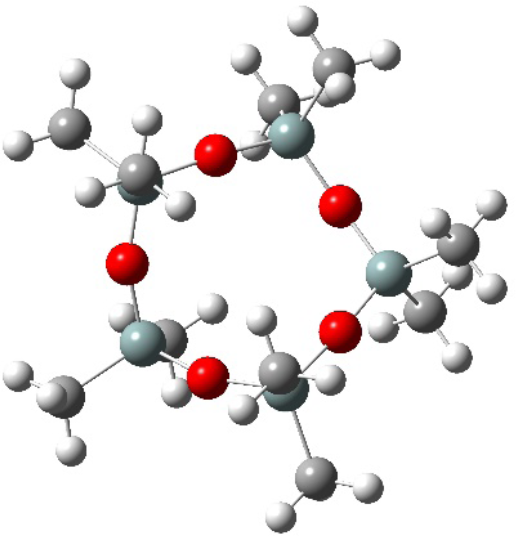
Chemical structure of D5.
It is well known that the use of suitable dyes can solve the low utilization rate of dyes and reduce the emission of dyes.10-12 For this reason, the selection of appropriate molecular structure of dyes was significant to improve the dyeing rate of dyes in the D5 dyeing system. In this work, two commercial dyes with different substituents were used to investigate the effect of dyes on the dyeing properties in the D5 dyeing system.
Experimental
Materials
Polyester fabrics (PET 100%, 8.3 Tex×2) were collected from Wanziqianhong Dyeing and Printing Co Ltd, Zhejiang, China. C.I. Disperse Orange 25 (dye A1) and C.I. Disperse Red 73 (dye A2) (as shown in Fig. 2) were received from Longsheng Dye Chemical Co Ltd, Shangyu, China. DMSO (Dimethyl sulfoxide, analytical reagent) was obtained from Aladdin (Shanghai, China). D5 (purity > 96%) was purchased from GE Toshiba Silicone Ltd, Jiande, China.
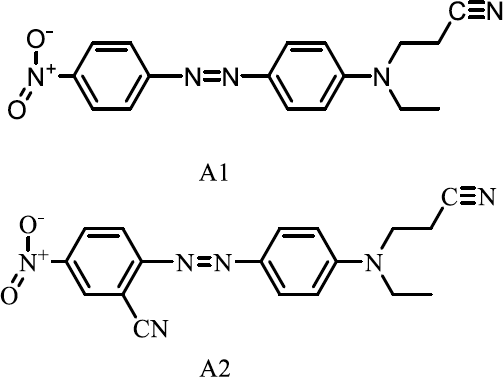
Molecular structures of commercial disperse dyes.
Dye Solubility in D5
The solubility of disperse dyes in D5 was measured at differ-ent temperatures. Firstly, the two disperse dyes were purified using ethanol. Then, excess disperse dyes were added in D5 at constant temperature for 60 min. and sampled at various temperatures. The dissolved dye in D5 was diluted by DMSO. Finally, the solubility of the dye in D5 was calculated according to the absorbance of the solution.
Dyeing Process
A 2.0 g sample of polyester fabric was dyed with disperse dyes (0.5%, owf) using a dyeing machine (ShangHai Chain-Lih Automation Equipment Co Ltd, Shanghai, China). The liquor ratio was of 1:20. The polyester fabric was dyed at room temperature for 5 min. Then, the dyeing temperature was increased from 30 °C to 80 °C at 6 °C/min. The dyeing rate was changed to 3 °C/min. The temperature was raised to 140 °C and maintained for 3 h. Finally, the polyester fabrics were washed for 15 min. at 80 °C using the same bath ratio as in the dyeing process, and dried at normal atmospheric temperature.
DFT Calculation
To understand the relationship between structure and performance, dyes A1 and A2 were optimized and calculated using quantum chemical density functional theory (DFT) at B3LYP/6-311G++ (d, p) level.
Measurement of Migration Degree
The uncolored polyester fabric (A) was stitched together with the colored sample (B) and dyed for 60 min. at 140 °C and a concentration of 0.5%, according to the experimental staining method above. The two samples were cooled, washed, and dried. The migration degree (%) was calculated using Eq. 1.
Where (K/S)A and (K/S)B are the (K/S) values of samples A and B, respectively.
Dyeing Evaluations
The reflectance R of the dyed fiber was measured using the Hunterlab QX-E spectrophotometer. The color yield (K/S) value was then calculated according to Eq. 2.
The final uptake of disperse dyes on polyester fabric was calculated using Eq. 3.
Where E refers to the dye absorptivity; C0 and C1 refer to the concentration of dye (g/L) before and after dyeing, respectively; V0 and V1 refer to the volume of the initial dye bath (mL) and the volume of all the extraction solution (mL), respectively. Levelling of dyed fabrics is one of the most important factors to evaluate dyeing performance. It can be evaluated by ΔE, which is measured using the same device. The smaller the ΔE, the better the leveling property.
Colorfastness
Colorfastness was evaluated according to the respective international standard methods. Rubbing fastness was tested using the Crockmeter according to ISO 105-X12 (2016); Washfastness was tested by using soap fakes and sodium carbonate as soaping additives at a ratio of 1:50 and temperature of 95 °C for 30 min according to ISO 105-C03 (2010); Lightfastness was evaluated by comparing with standard wool fabric according to ISO I05-B02 (2014).
Results and Discussion
The interaction between solute and solvent in the dissolution process was extremely complex. Intermolecular forces included hydrogen bonding force, van der Waals force, polarizing force, and other forces. Some forces play a dominant role in the dissolution process. As shown in Fig. 3, the solubility of A1 and A2 in D5 medium increased with an increase of temperature. The solubility of the two dyes in D5 medium was clearly different due to different substituents. Compared with A1, A2 had lower solubility. These results showed that the oleophobic property of cyano groups can obviously weaken the interaction between dyes and D5. As a result, the solubility of A2 in D5 was lower.
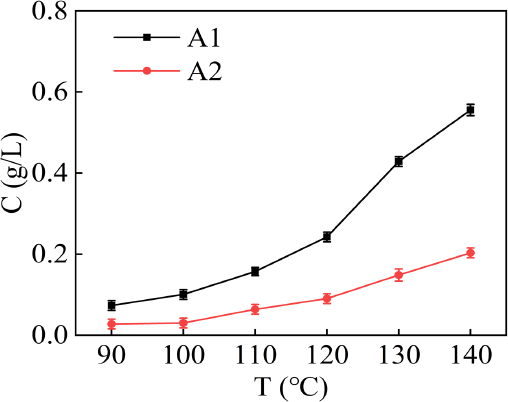
Solubility of A1 and A2 at various temperatures in the D5 medium.
Effect of the Dye Structure on Dyeing Rate
Dyeing equilibrium was achieved by keeping the dyeing system at 140 °C for at least 60 min. As shown in Fig. 4, with the increase of dyeing time from 30 to 90 min., the dyeing rate of A2 increased rapidly. It was significantly higher than that of A1. Comparing the solubility in Fig. 3 with the exhaustion in Fig. 4, the dye uptake of the dyes was inversely proportional to solubility, when the solubility reduced from 0.60 g/L to 0.19 g/L, the dye uptake increased from 50% to 80%. Compared with A1, the exhaustion of A2 increased by 59% after reaching dyeing balance. It was clearly found that dyes with lower solubility had a higher dyeing rate. This could be attributed to cyano groups on the diazo component of A2.
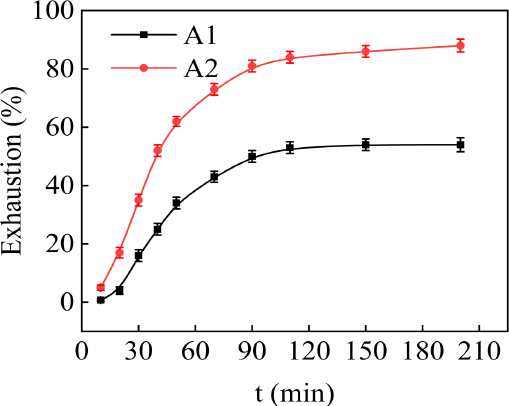
Effect of dyeing time on the exhaustion of the polyester fabrics.
DFT Calculation and Migration Degree
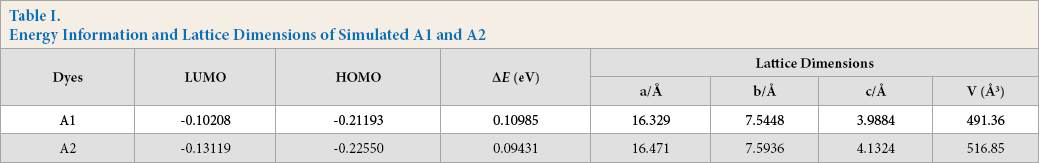
To further explain the relationship between structures and dyeing properties, the electrostatic potential energy diagram and lattice dimensions of A1 and A2 were simulated and calculated using DFT methods at B3LYP/6-311G++ (d, p) level. As shown in Fig. 5, the cyano group (-CN) possessed strong electronegativity. The hydrogen group showed weak positivity on the dye molecule. As shown in Table I, the molecule size of A2 was slightly larger than that of A1, which made it easy for A2 to generate more dye mono-molecule. It is known that the monomolecular state is beneficial to dye diffusion in the fibers.
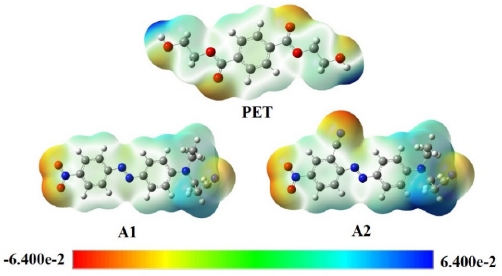
Electrostatic potential energy diagram of PET and A1 and A2 dyes. Fig. 5. (Orange color indicates the most negativity; blue color means the most positivity).
As shown in Fig. 6, the migration degree of A1 was signifi-cantly lower than that of A2. It further indicated that A2 had weak intermolecular force and was easy to diffuse into fibers as well as exude out from the surface. The migration degree was consistent with the dyeing rate.
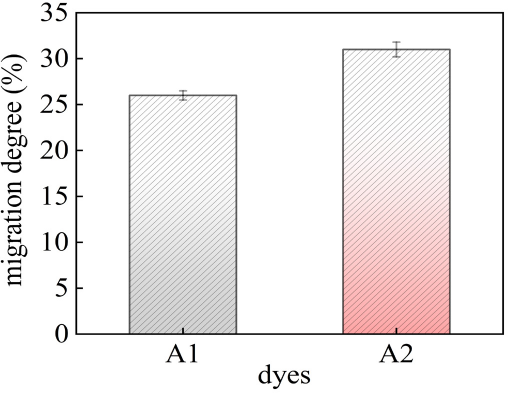
Migration degree of A1 and A2.
Fastness Properties of Dyes on Polyester Fabrics
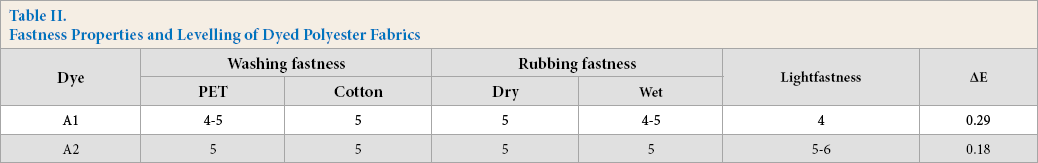
The results of colorfastness testing for dyed fabric at a dye concentration of 0.5% are shown in Table II. Dyes A1 and A2 exhibited excellent washing and rubbing fastness. A2 had better lightfastness than A1 on polyester fabrics. This was attributed to a low HOMO-LUMO energy gap for dye A2 as shown in Table I. Additionally, the fabric dyed with A2 had a lower ΔE value than that of A2, suggesting that the dyes with a cyano group had better levelling.
Energy Information and Lattice Dimensions of Simulated A1 and A2
Fastness Properties and Levelling of Dyed Polyester Fabrics
Conclusions
In this paper, the relationship between structures and dyeing properties of disperse dyes was investigated in D5 medium. The results showed that the solubility of disperse dyes was inversely proportional to the dyeing rate. The dye containing cyano groups had a better dyeing rate and levelling properties. Large steric hindrance of cyano groups promoted the formation of dye mono-molecule to achieve a better dyeing rate by destroying π-π stacking interactions. The results provide a potential theoretical basis for the development of suitable disperse dyes for a D5 dyeing system and promote the continuous development of the textile industry.
Footnotes
Acknowledgements
This work was supported by the Opening Project of Key Laboratory of Clean Dyeing and Finishing Technology of Zhejiang Province (QJRZ1901), National Key Research and Development Program of China (2017YFB0309600), and Key Research and Development Program of Xinjiang Production and Construction Corps (2019AA001).