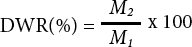Abstract
Protein-polysaccharide scaffolds are usually used in tissue engineering; however, matching regeneration rate with healing modes is a challenge because of unpredictable scaffold degradation profiles. We investigated the effect of the blend ratio on degradation behavior of silk fibroin (SF)/hyaluronic acid (HA) hydrogels by varying weight ratios. The hydrogels were incubated in 1 U/mL collagenase IA at 37 °C to create an in vitro model of proteolytic degradation. Samples were harvested at designated time points up to 21 days and scanning electron microscopy (SEM), digital photos, and mass loss were investigated. Control samples were incubated in phosphate-buffered saline. The results showed regular and controllable degradability. The higher the HA content, the stronger the degradability of the hydrogel. When the HA content increased from 20% to 40%, the mass loss of the hydrogel increased from 22% to 35% after 21 days of degradation.
Introduction
In the tissue repair process, biological scaffolds should meet the degradation rate needs for the specific tissue engineering application. Ideally, the rate of scaffold degradation should match regeneration rate with healing modes. 1 Meanwhile, the scaffold hydrolyzes or enzymatically degrades at a moderate rate, neither too fast to impede tissue growth, nor too slow to provide space for tissue repair. 2 The biological scaffolds used in clinical application, such as silicone oil and fluorinated gases, can only be utilized as short-term substitutes due to their toxicity to surrounding ocular tissues and their tendency to emulsify. 3 Therefore, it is necessary to prolong and control the degradation rate without introducing toxic polymers. 4
Hydrogels with similar components and structure to that of the target tissue extracellular matrix (ECM) can provide a suitable carrier and microenvironment for cell adhesion, proliferation, and differentiation through physical support and spatial structure. 5 Several hydrogel formats in a variety of applications utilized silk fibroin as the main ingredients due to its biocompatibility, mechanical tunability, and relatively slow degradation.6-10 Moreover, natural materials, such as hyaluronic acid (HA) and collagen, are biologically similar to native vitreous humor (which consists of up to 400 μg/ cm3 of HA and 532 μg/cm3 of collagen), 11 but have limited retention times in vivo due to relatively fast degradation. 3 The composite hydrogels, blending natural polysaccharide HA with silk fibroin to create suitable SF/HA through different methods, has been investigated.4,6,12,13 However, the systematic and in-depth study on the degradation of hydro-gels in collagenase solution is still insufficient.
In our previous study, 14 The SF/HA composite hydrogels derived from a CaCl2-formic acid (Ca2+-FA) solvent system was designed based on its good physical and chemical properties. Interesting, each polymer of the hydrogels not only retained its favorable properties, but also improved the mechanical properties such as flexibility, water-insolubility, and stability. So, the objective of this study was to explore the controllability and regularity of degradation of Ca+2-FA SF/HA composite hydrogels by changing the blend ratio of SF/HA.
Materials and Methods
Preparation of SF/HA Hydrogels
As in a previous study, raw silk (Huzhou, China) was boiled three times in 0.06% (w/v) Na2CO3 solution for 30 min. to remove sericin. Silk fibroin fibers and hyaluronic acid were dissolved with 5% (w/v) Ca2+-FA solvent acid (HA, 7.76 × 105 Da, Sigma, China) to get SF/HA solution.15,16 Changing the HA powder proportion, solutions with various mass ratios of SF/HA (60:40, 70:30, 80:20, and 100:0) were prepared in parallel. When a final concentration of 10 wt % SF/HA Ca2+-FA solution was obtained, the mixture was put into a dialysis tube (molecular cut of: 14 kDa, Viskase, USA) for 24 h to form SF/HA hydrogels. The SF/HA hydro-gel is frozen at –80 °C overnight and freeze-dried to obtain SF/HA scaffolds for further experiments.
In Vitro Degradation of SF/HA Hydrogels
Collagenase IA (Sigma, China) was dissolved in PBS (0.05 M, pH 7.4) at 1.0 U/mL. SF/HA hydrogels were cut into cylinders with a diameter of 14 mm, a height of 3 mm, and initial dry weight (mass) M1. The samples were cut into 50 × 50 mm squares after drying, wrapped with 160-mesh wire, and incubated in 1 U/mL collagenase solution (bath ratio 1:150) at 37 °C to create an in vitro model system of proteolytic degradation. Tree samples were harvested at each designated time point (after 1, 3, 7, 14, and 21 days). Harvested samples were dried at 105 °C after the deionized water thoroughly flushed. Weight (mass) was recorded as M2 to calculate the degradation rate of hydrogels. The PBS was used as the control group, in which the enzyme solution and PBS solution were replaced every three days. The DWR (Dry Weight Remaining Ratio) of SF/HA composite hydrogel was calculated using Eq. 1.
Where DWR is degradation rate of composite hydrogel, M1 is dry hydrogel mass before degradation minus the dissolve-loss in solution, and M2 is dry mass of the composite hydrogel after degradation. 17
Morphology and Structure of Hydrogels during in Vitro Degradation
After fully washing and drying the degraded SF/HA hydrogel taken out on day 3, 7, 14, and 21 with deionized water, they were fixed on the sample table with conductive adhesive and sprayed with gold. The surface morphology of the samples was observed with a jsm-6510lv scanning electron microscope (SEM, Zeiss, EVO MA 10/LS 10, Germany).
Results and Discussion
Mass Loss of SF/HA Hydrogels
As shown in Fig. 1a, the mass loss rate of all SF/HA hydrogels in PBS solution was less than 20%. The mass loss rate of SF/HA = 60/40 hydrogel was about 18%, SF/HA = 70/30 hydrogel was about 13%, SF/HA = 80/20 hydrogel was about 10%, and SF/HA = 100/0 hydrogel was only about 8%. This maybe associated with the increased of HA content. HA has strong water absorption and addition of HA increases moisture inside the hydrogel. The process of freeze-drying sublimates the ice crystals in the aerogel and makes porous scaffolds. The SF/HA = 100/0 hydrogel degraded relatively slowly compared with the blend composite hydrogels which added HA.
Mass loss of SF/HA hydrogels in various ratios in PBS (a) and collagenase (b).
As shown in Fig. 1b, with the decrease of HA content, the mass loss of the hydrogel in collagenase solution also decreased. The residual mass of SF/HA (60/40, 70/30, 80/20) hydrogels were about 65%, 72%, and 78%, respectively, and the residual mass reached about 90% when SF/HA = 100/0, which was like that in PBS solution. This may be due to lower HA content hydrogels containing less water, with the pore size after freeze drying being relatively small. Moreover, the collagenase solution immersed and absorbed inside the hydrogels decreased with the decrease of HA content.
According to the comprehensive analysis in Figs. 1a and b, the degradation rate of the hydrogel blends with HA was slower in PBS solution than in the collagenase solution. To further express the morphology changes of SF/HA hydrogels in the degradation process, the SF/HA blend hydrogels were photographed and recorded at every time point.
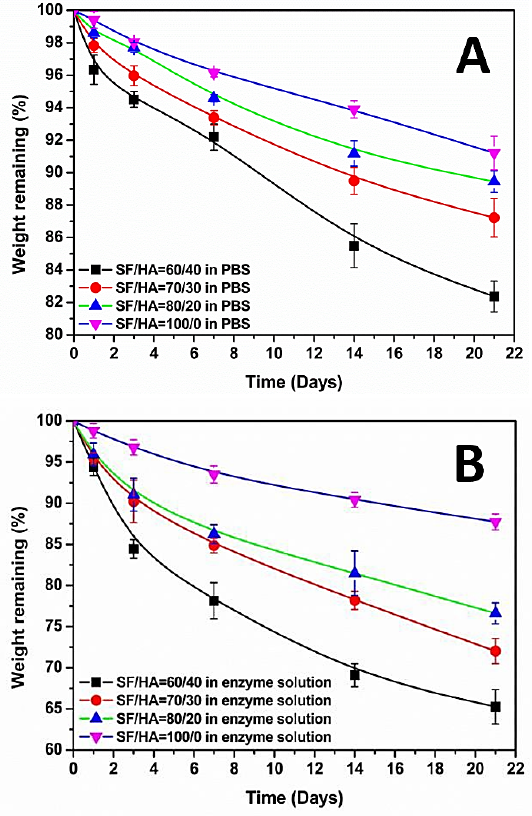
As shown in Fig. 2, none of the hydrogel blends showed obvious changes in appearance or sample diameter at day 1, 3, 7, 14. Sample diameter decreased slightly compared with that before degradation at day 21 in PBS solution. To some degree, the thickness changes of the degraded samples can refect the mass loss. The thickness of the hydrogels decreased and the pores size increased with degradation time. However, the samples of SF/HA = 100/0 hydrogel at various degradation time points were different from those of the blend hydrogel. As can be seen from Fig. 2, the surface of the samples of SF/HA = 100/0 hydrogel was relatively smooth and had light-colored ribbon stripes before degradation, but showed slight shrinkage and was transparent and brittle, and easy to break after degradation. Secondly, the morphology of the blend hydrogel changed significantly in the collagenase solution. The sample of SF/HA = 60/40 was slightly broken on the third day of degradation. The diameter and thickness were significantly reduced and the surface was grooved after 14 and 21 days of degradation. The morphology of SF/HA = 70/30 and SF/HA = 80/20 blends changed significantly on the 14th day, and the intermediate fracture of SF/HA = 80/20 blends may be related to the relatively high porosity. The appearance morphology of SF/ HA = 100/0 hydrogel after degradation in collagenase solution was not significantly different from the sample in PBS solution, which was consistent with the small difference in residual mass of the hydrogel in PBS solution and collage-nase solution mentioned above.

Digital photos of various ratios of SF/HA blends degraded by PBS and collagenase solutions at multiple time points.
Morphology Change of SF/HA Hydrogels
The pore structure changes of SF/HA blends with different proportions were further observed by SEM during degradation. As shown in Fig. 3a, slight changes took place in the pore walls with varying content of HA in PBS solution. Even after degradation for 21 days, there was no significantly collapse observed in the pore structure, suggesting that the HA played a negative role in the stability of the blends in PBS solution. For degradation of SF/HA = 60/40 blend hydro-gel in PBS solution, the pore wall gradually thinned and many small holes were observed on the pore wall. With the degradation time extension, the small holes became larger, the pore wall became thinner, and the pores were connected by microfibers. In SF/HA = 70/30 and SF/HA = 80/20 blend hydrogels, the pore walls of the hydrogel section were relatively thick, and there were more pores on the pore walls at the 14th day of degradation. The SF/HA = 100/0 hydrogel surface became smoother during the degradation process. With the extension of degradation time, cracks appeared on the smooth surface, and larger pores were observed on the hydrogel surface. The degradation behavior of the hydrogel in the collagenase solution was detected as shown in Fig. 3b. The pore walls began to thin and collapse after 7 days degradation in the HA blended hydrogel, presenting a network structure. With the degradation time increased, the pore walls of the samples became thinner in SF/HA = 70/30 and SF/HA = 80/20. After 21 days, the pore walls of the samples collapsed and fractures were observed. The surface of the SF/HA = 100/0 hydrogel began to become smooth on the 3rd day after degradation, and cracks also appeared on the surface, showing little difference from the morphology of the samples degraded in PBS solution. The results indicated that the degradation rate of SF/HA hydrogel was different in different proportions. In contrast, the hydrogel with the addition of HA showed a higher degradation rate. The higher the content of HA, the higher the degradation rate. SF/HA = 100/0 hydrogel had little difference in degradation degree between PBS solution and collagenase solution due to its high crystallinity, which was consistent with the results of weight change of degradation. Therefore, the degradation rate of SF/HA blend hydrogel can be adjusted by the content of HA, and the degradation rate of the hydrogel is controllable.
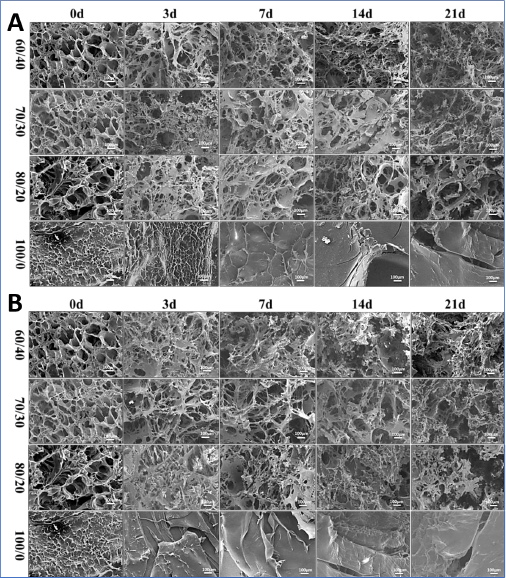
SEM images of various SF/HA blend ratios degraded by PBS (a) and collagenase (b) at multiple end points.
Conclusions
In this study, we built a new process of SF/HA hydrogel. During degradation, the samples with high content of HA showed rapid degradation, with the excessive residual mass occurring in the PBS solution after 21 days degradation. SEM revealed that the pore wall of the hydrogel became thinner. Many mini-pores were formed on the pore wall during the degradation process. More importantly, the higher HA content leads to more free water molecules absorbed in its internal pores. After freeze drying, the pore size of the hydrogel was relatively larger and the porosity was higher, which was determined by SEM. These features would contribute to effective degradation in vitro. The results could significantly contribute to the regulation of the degradation rate of SF-based composites to match tissue regeneration rates in regenerative medicine.
Footnotes
Acknowledgements
This work was supported by the Wuhan Municipal Science and Technology Bureau (2019010701011388)and by the open foundation of Shanxi Province Key Laboratory of Oral Diseases Prevention and New Materials (KF2020-06).