Abstract
This paper proposes the development of a novel coagulant for dust suppression in open-cast mines. Specifically, pretreated sodium lignin sulfonate and acrylic acid were first cross-linked, then the graft copolymerization of the intermediate product (the cross-linking product) and acrylamide was conducted and finally the resulting gelatinous substances were crushed. During the reaction process, N,N’-methylene-bis-acrylamide and ammonium persulfate were used as the cross-linking agent and initiator, respectively. Subsequently, the functional groups, crystalline structure, and thermal stability of the dust coagulant were examined by means of Fourier transform infrared spectra measurements, X-ray diffraction spectra measurements, and differential scanning calorimeter analysis. Moreover, single-factor experiments were conducted to explore the optimal synthesis condition. According to the experimental results, the coagulant achieved its optimal dust suppression performance under the following conditions: the mass ratio of lignin to acrylic acid was 1:3, the mass ratio of lignin to acrylamide was 2:7, the content of the cross-linking agent was 0.9%, the mass ratio of initiator to acrylamide was 2:100, the reaction temperature was set as 60℃, and the pH value was set as 7. Finally, the coagulant was measured for its swelling kinetics, viscosity, film-forming hardness, peeling strength, and ability to suppress dust. It can be concluded that the coagulant exhibits a very high standard of both dust suppression and wind resistance.
Keywords
Introduction
For economic and geographical reasons, not all Chinese coal mines are located underground, with approximately 10% being classed as open-cast mines (Du and Li, 2013; Khan and Javed, 2012). Open-cast coal mines have a number of advantages, such as full resource utilization, suitability for large-scale mechanized excavation, high labor productivity, favorable working conditions, and safer production practices. These advantages should lead to the future growth of open-cast mines (Konorev and Nesterenko, 2012; Lal and Tripathy, 2012). However, during the processes of coal production, stockpiling, and transportation, a considerable quantity of dust is produced and transmitted to a larger area due to the wind. This not only causes a huge loss of resources but also leads to serious pollution problems (Işiklan et al., 2009; Zhao and Zhang, 2015).
Currently, chemical dust suppressants are widely used as a way of dealing with this problem, and for a number of years researchers have conducted systematic studies on the related techniques and gained some interesting insights regarding the application of such suppressants to underground mines. For example, Mandal et al. (2012) used the mixture of aliphatic organic compounds and wetting agents for dust prevention in coal mines. Furthermore, Yan et al. (2016) sought the optimal formula for a dust suppressant through surfactant compounding and applied it to underground mines (Wu et al., 2005), and Wang et al. (2015a) used foams for dust prevention in coal mines (Lu et al., 2015). Although these dust suppressants can suppress the dispersion of coal dust effectively, they are not suitable for the open-air environment due to their poor wind resistance and low adhesion ability with regard to dust (Konorev and Nesterenko, 2012; Lu et al., 2015; Wang et al., 2015a). To address this problem, some researchers have also investigated dust prevention and control during the processes of stockpiling and transporting coal in open-cast mines. Dixon-Hardy et al. (2008) used the waste from refineries to prepare a specific product for dust prevention; however, although they achieved some very good results, the product often caused secondary pollutions. Du and Li (2013) optimized the formula for the compounding of inorganic salts for dust suppression in open-cast mines; however, although the raw materials were easy and convenient to obtain and the method was highly efficient, the prepared suppressant only lasted for a short duration and only had a minor impact on the dust. Yan et al. (2016) mixed a prepared copolymer with water using a certain ratio and then utilized the mixture for dust suppression. This mixture was environmentally friendly and easy to spray; however, when it made contact with dust it formed a film, which performed poorly with regard to wind resistance and was therefore ineffective in windy conditions. Moreover, the mixture’s suppression of coal dust only lasted for a short time, and if the monomers used in compounding are difficult to biodegrade, the use of such dust suppressants can cause secondary pollutions, which is a greater problem. Therefore, it is necessary to develop and apply a type of environmentally friendly dust suppressant that is suitable for dust prevention and control during the stockpiling and transportation of coal in surface coal mines.
As global environmental issues have become more serious, environmentally friendly and biodegradable materials have been increasingly sought and accepted by modern society. Dust prevention and control are no exceptions in this area (Ma et al., 2016) and although a variety of natural, degradable materials such as lignin, starch, and protein exist, they cannot be used directly as these natural polymers tend to have irregular structures and undesirable properties. Therefore, it is important to address the reconstruction of their structures through modification and subsequently make them widely usable (Gordobil et al., 2016). For example, lignosulfonate, a type of derivative in the sulfonation of lignin, is abundant in the effluents paper industry, the discharge of which can seriously contaminate the environment (Muranaka et al., 2016). Furthermore, because of its irregular structure, lignosulfonate can only be recycled for energy purposes; however, large amounts of active functional groups in lignin, such as hydroxy and benzene rings, will be wasted if lignosulfonate is only used for such purposes. Those working with other natural molecular materials are also grappling with similar problems. Consequently, in order to ensure these polymer materials can be used in an environmentally friendly manner, their natural molecular structures have to be modified by physical, chemical, or biological modification methods (Kim et al., 2016; Muranaka et al., 2016).
Currently, graft copolymerization is a simple but effective chemical modification method for enhancing and modifying molecular structures and functions (Ma et al., 2016; Sirirat et al., 2015), as the active functional groups in natural polymers can provide favorable reaction preconditions. In the presence of initiators, the graft copolymerization reaction can occur subject to the appropriate selection of grafting monomers. This means that the exceptional properties of the grafted polymer can be fully exploited and incorporated into the overall modification process, so that the end products can demonstrate an improved performance and be utilized more extensively (Muranaka et al., 2016; Wang et al., 2015b). The graft copolymerization technique has been widely applied in several fields including mining. For example, Cheng et al. (2015) adopted the graft copolymerization method to develop a super absorbent resin for fire suppression in mines. Furthermore, Zhao and Zhang (2015) synthesized and applied related materials for filling and grouting in mines. Inspired by these hydrogel products, polymer gels can dampen and adsorb the dust in mines effectively (Zhao and Zhang, 2015); moreover, as the water in hydrogels evaporates, the collected dust will eventually coagulate and thus demonstrate highly effective dust removal properties (Wang et al., 2016). However, in previous studies, the application of graft copolymerized products in the prevention and control of coal dust was insufficiently investigated, as most researchers focused on the film that was formed when spraying to suppress the diffusion of coal dust.
In this study, sodium lignin sulfonate (SLS), which is abundant in nature, was selected as the raw material; moreover, the amide groups were introduced in the reaction process for enhancing the adsorption of dust by micro hydrogen bonds in polymeric products. However, it was found that the mechanical performance of the initial product needs to be improved, and thus, acrylic acid (AA) was introduced for strengthening the end product’s mechanical performance so that it can act on the dust for a long time. Accordingly, this study prepared a novel hydrogel by conducting cross-linking and modification on SLS, AA, and acrylamide (AAM) through solution polymerization (Lai et al., 2011; Xiao et al., 2015). The wettability of the hydrogel surface and the molecular force in the hydrogel can improve its effect on coal dust. Furthermore, because of its outstanding mechanical performance, the end product can cope with severe weather. More importantly, the end product is a high-molecular polymer dominated by carbon chains, which is highly biodegradable and causes no pollution to the environment. Therefore, this coagulant has the potential to be widely applied in the suppression of coal dust.
The experiments
The materials
The reagents used in this experiment included SLS, N,N’-methylene-bis-acrylamide, AA, AAM, ammonium persulfate (APS), sodium hydroxide, calcium chloride, acetone, and distilled water. With the exception of the distilled water (self-prepared), all the reagents were analytically pure and purchased from Tianjin Kemiou Chemical Reagent Co., Ltd.
The preparation process
The synthesis of the coagulant for dust suppression in this study is now described in detail. First, the lignosulfonate was pretreated and then the mixture of SLS and calcium chloride was added to the aqueous solution (100 ml) at room temperature and stirred intensively for 10 min. A certain amount of AA, which had been neutralized, was then added to the mixed solution and stirred thoroughly. Next, the reaction temperature was adjusted to 45℃ and N,N’-methylene-bis-acrylamide was added to the mixed solution for a reaction of approximately 1.5 h. After the reaction was finished, the solution was cooled to room temperature and a certain amount of AAM was added to the product; simultaneously, the pH value of the solution was adjusted. Subsequently, the reactants were placed in a stirrer where nitrogen was added for protection, and APS (as the initiator) was also added after the reaction temperature had been set as 60℃. The mixture was then stirred and reacted for 2 h before the gel product was obtained. After being crushed, the gel product was evenly mixed with water according to a certain proportion, i.e. the coagulant was prepared. The coagulant was then purified. The crude product was placed in a vacuum drying oven and dried at 50℃ for 24 h. With acetone as the extractant, the dried crude product was placed in a Soxhlet extractor for 24 h before being dried again in a vacuum drying oven. This process led to the development of the pure product for spectral analysis.
The effects of single factors on the properties of end products
Illustration of the single-factor experiment.

AA: acrylic acid; AAM: acrylamide; APS: ammonium persulfate; SLS: sodium lignin sulfonate.
The Fourier transform infrared (FT-IR) spectroscopy
Having cross-linked the intermediate product and after the graft copolymerization of the end product, both sets of products were dried in a vacuum drying oven at 100℃ for 24 h. After drying, the substances were added to the mortar for grinding until they could pass through a 200-mesh sieve. These two substances were then measured in KBr pellets using the FT-IR spectrometer (Nicolet 380, Thermal Fisher Scientific, USA). By analyzing the infrared spectra, it is possible to investigate the changes to the functional groups of the products.
The X-ray diffraction (XRD) measurements
The dried samples were ground in a mortar and made into XRD scanning plates after the powder size met certain requirements. These XRD scanning plates were then scanned by the X-ray analyzer (Ultima IV, Rigaku Corporation, Japan). The scanning angle ranged from 5° to 75° and the scanning rate was set as 5°/min. The XRD spectra of the intermediate product and end product were obtained for comparison purposes.
The differential scanning calorimeter (DSC) analysis
The prepared gelatinous substances were dried in a vacuum drying oven and then, 8–10 mg of solid powder was placed under the DSC analyzer (Mettler-toledo, Switzerland) to investigate their thermal stability and thermal decomposition performance. During the TG analysis, the heating temperature was controlled within the range of 40–650℃ at a heating rate of 20℃/min, and the aluminum oxide (Al2O3) was used as the standard substance.
The performance tests on the end products
The swelling kinetics and viscosity tests
The prepared, pure grafted copolymer was dried in a vacuum dry oven at 50℃ for 24 h. After drying, certain amounts of the product were added to the beaker that contained enough water for investigating the swelling kinetics. The changes in the mass of the end product over time were measured and the swelling kinetics of the product was calculated according to the following equation

The correlation curves of the swelling kinetics and time were then plotted. The viscosity of the product was measured by a rotary viscosimeter. Then, using the product’s physical state at different time points as the independent variable, the relation between the viscosity variation and water absorbency was explored. This provided sufficient theoretical guidance for the subsequent practical applications.
The film-forming hardness and peel strength tests
According to the formulas used in single-factor experiments, Group 1–11 was selected for the 180° peel strength and film-forming hardness tests. The peel strength tests were conducted on a material tensile tester (CMT-2000, Sansitaijie Electrical Equipment Co., Ltd., China), at a peel-away speed of 100 mm/min. The prepared product was spread on the coal surface; then, under a natural wind, a cured film was formed after 5 h. The hardness of the cured protective film was measured by a Shore durometer and the correlation curves were plotted.
The dust suppression performance test
Wettability data of the coal sample.

Proximate analysis results of the coal sample.
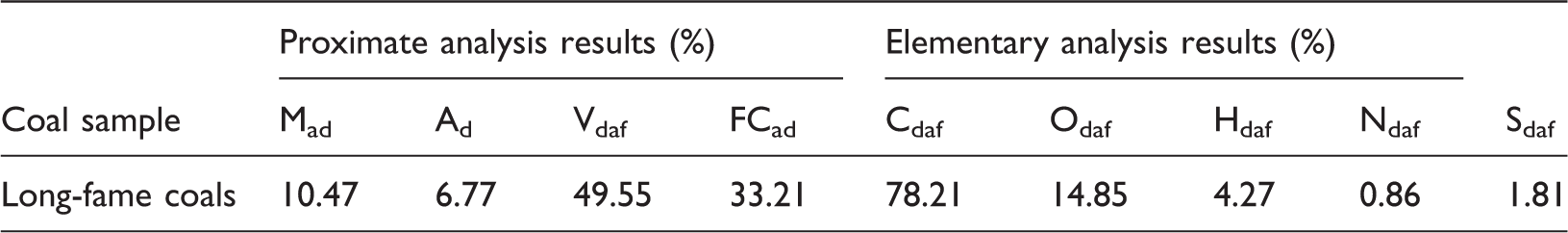
Mad denotes the moisture content; Ad denotes the ash content; Vdaf denotes the content of volatile component; FCad denotes the content of fixed carbon; Cdaf denotes the content of the dry ash-free carbon elements; Odaf denotes the content of the dry ash-free oxygen elements; Hdaf denotes the content of the dry ash-free hydrogen elements; Ndaf denotes the content of the dry ash-free nitrogen elements; Sdaf denotes the content of the dry ash-free sulfur elements.
Results and discussion
The analysis of the FT-IR spectra
Figure 1 displays the IR spectra of the intermediate product (i.e. the cross-linking product of SLS and AA) and end product (i.e. the above cross-linking product after being grafted by AAM), in which the blue curve denotes the spectra of the intermediate product and the red curve denotes the spectra of the end product. Several peaks can be observed in the FT-IR spectra of the cross-liking product. The peaks at 3365, 3308, 1630, 1562, and 870 cm−1 were induced by the vibration of amides in the cross-lining agent; specifically, the peaks at 3365 and 3308 cm−1 are correlated with the absorption of NH2, the peak at 1630 cm−1 is correlated with the vibration of C = O, the peak at 1562 cm−1 is correlated with the vibration of N–H, and the peak at 837 cm−1 is a vibration adsorption peak. The observation of these peaks can prove the existence of primary amide, suggesting that cross-linking agents were involved in the reaction. Additionally, the peaks at 1725 and 1200 cm−1 were induced by the absorption of AA; the former peak corresponds to the vibration of ester groups while the latter was induced by the vibration of the C–O–C bond. The observation of these peaks indicates that the AA successfully reacted with the cross-linking agent. By comparing the spectra of the intermediate product and end product, it can be found that the absorption peaks at 2927, 2856, 1662, 1408, and 666 cm−1 show obvious changes. The dual peak at 2925 and 2856 cm−1 was induced by the vibration of the C–H bond in an aliphatic series, suggesting the formation of aliphatic compounds. The absorption peaks at 1662, 1408, and 666 cm−1 are related to the vibration of C = O, C–N, and NH2 in AAM, suggesting the occurrence of a favorable graft copolymerization between the intermediate product and AAM.
FT-IR spectra of intermediate products and end products. FT-IR: Fourier transform infrared.
The analysis of the XRD spectra
Figure 2 displays the XRD spectra of SLS, the intermediate product and the end product, respectively. Some sharp diffraction peaks can be observed in the XRD spectra of SLS, which are indicative of the crystallinity, i.e. SLS is a type of crystal. The XRD spectra of the intermediate product display an obvious bread-shaped peak, suggesting the existence of polymer. Since the intermediate product is liquid, it cannot be judged whether it participated in the reaction. However, according to the XRD spectra, noncrystalline structures were produced, i.e. the intermediate product was involved in the reaction. Finally, the diffraction peak in the XRD spectra of the end product becomes sharper compared with that in the XRD spectra of the intermediate product. The results reveal that the intermediate product continued participating in the reaction and thus the spectra show more obvious absorption characteristics. However, the end product was still amorphous.
XRD spectra of intermediate products and end products. XRD: X-ray diffraction.
The analysis of thermal stability
Figure 3 shows the DSC curves of SLS, the intermediate product and the end product, respectively. The DSC curve of SLS shows an absorption peak at 90℃ and no fluctuations during the whole heating process. According to the DSC curve of the intermediate product, a small absorption peak can be observed at 350℃, suggesting that structural changes took place in the product; however, this peak is inconspicuous. The DSC curve of the end product presents a peak at 345℃, which indicates that the structural change at this temperature destroyed the formed molecular structure. Overall, the formed graft polymer is characterized by a high thermal stability.
DSC curves of the grafted copolymer. DSC: differential scanning calorimeter.
The effects of single-factor variation on test results
The effects of AAM content on percent grafting and grafting efficiency
Figure 4(a) shows the effects of AAM content on percent grafting and grafting efficiency. Both the percent grafting and grafting efficiency of the end product increase at the beginning and then decrease as the AAM dosage rises. This is due to the fact that as the content of AAM increases, free radicals and active functional groups in the solution increase correspondingly, which can enhance the percent grafting and grafting efficiency. When the dosage of AAM exceeds 7 g, the percent grafting and grafting efficiency are negatively correlated with AAM content. Since the initiating radicals were limited during the reaction process, even a small number of free radicals could occupy the locations of initiating radicals rather than being grafted onto the main chain. Thus, self-polymerization occurred, which reduced the number of initiating radicals for effective graft copolymerization and led to a decline in percent grafting and grafting efficiency.
Effect of acrylamide (AAM), acrylic acid (AA), temperature (T), ammonium persulfate/acrylamide (APS/AAM), pH on percent grafting and grafting efficiency.
The effects of AA content on percent grafting and grafting efficiency
Figure 4(b) shows the effects of AA content on percent grafting and grafting efficiency. When the dosage of AA is less than 6 ml, the percent grafting and grafting efficiency increase as the acrylic acid content increases. The content of free radicals in the solution increases correspondingly with the addition of AA, and the molecular structure becomes more compact following the improvement in both percent grafting and grafting efficiency. When the dosage of AA exceeds 6 ml, the percent grafting and grafting efficiency decrease as AA content increases. This is because the increase in AA content does not cause any change in the amount of initiating radicals, while more free radicals make them increasingly prone to self-polymerization; thus, certain amounts of free radicals are consumed, leading to a decrease in percent grafting and grafting efficiency in the end product.
The effects of pH value on percent grafting and grafting efficiency
Figure 4(c) shows the effects of pH value on percent grafting and grafting efficiency. Obviously, the pH value significantly influences the graft copolymerization reaction under an acidic environment. In an acid solution, the reactions of both free radicals and initiating radicals were greatly affected and the cross-linking agents were decomposed. Therefore, both the percent grafting and grafting efficiency are greatly reduced under strong acidic conditions. As the acidity decreases, the reaction environment becomes more moderate and the reaction is accelerated. When the pH value of the solution is equal to 7, the percent grafting and grafting efficiency of the end product reach the maxima; however, when the pH value exceeds 7, the percent grafting and grafting efficiency gradually weaken. Under alkaline conditions, the free hydroxyl radicals may hinder the process of graft copolymerization and change the structure of the end product to a certain degree. The graft copolymerization under alkaline conditions is less sensitive to the variation of pH value than that under acidic conditions.
The effects of the initiator dosage on percent grafting and grafting efficiency
Figure 4(d) shows the effects of an initiator dosage on percent grafting and grafting efficiency. It can be observed that, as the dosage of the initiator increases, the percent grafting and grafting efficiency of the end product increase initially and then decrease. When the mass fraction of APS to AAM equals 2%, both the percent grafting and grafting efficiency of the end product reach the maxima and the molecular structure becomes compact. When the mass fraction of APS to AAM is less than 2%, the number of initiating radicals increases gradually as the dosage of initiator increases, which can lead to the polymerization between the free radicals and main chain and thereby enhance the percent grafting and grafting efficiency. When the mass fraction of APS to AAM exceeds 2%, surplus initiation radicals exist in the solution as the concentration of initiator increases; thus, the probability of self-polymerization of free radicals increases greatly and the polymerization between free radicals and the main chain decreases correspondingly, leading to a reduction in effective percent grafting and grafting efficiency.
The effects of reaction temperature on percent grafting and grafting efficiency
Figure 4(e) shows the effects of the reaction temperature on percent grafting and grafting efficiency. One can observe that the percent grafting and grafting efficiency increase at first and then decrease as the reaction temperature rises, both of which reach the maxima at 60℃. When the reaction temperature is below 60℃, the percent grafting and grafting efficiency increase as the temperature rises. A low temperature affects the activity of free radicals and the rate of grafting reaction. As the temperature rises, the activity of free radicals increases and both the probability and rate of graft copolymerization increase, resulting in the enhancement of percent grafting and grafting efficiency. When the reaction temperature is over 60℃, the percent grafting and grafting efficiency decrease as the temperature rises. This is due to the fact that the decomposition rate of the initiating radicals can be accelerated as the temperature rises, while some cross-linking and self-polymerization reactions can occur during fast reaction and decomposition. This effect finally produces the low-efficiency network structure and reduces the percent grafting and grafting efficiency.
The performance tests of the end product
The swelling kinetics and viscosity tests
Using the method described above, the swelling kinetics of the dried dust suppressant and the viscosity of the product at different time points were measured and the results are shown in Figure 5. It can be observed that the product’s water absorptivity increases rapidly at first and then flattens. Specifically, the product’s water absorbency increases rapidly in the first 4 h of measurements, then increases slowly and reaches the maximum. The maximum water absorbency can reach as high as 80. The viscosity, another important index for measuring the coagulant’s properties, also exhibits changes with the increase of water absorbency. The viscosity presents no obvious variations when the water absorbency ranges from 20 to 60, which indicates the viscosity has no evident effects on the product’s dust suppression performance. However, as the product’s water absorbency further increases, the viscosity drops rapidly and the product begins to swell due to the increase of water absorbency, which can reduce the product’s dust absorbability and thus affect the coagulant’s performance. Therefore, it can be concluded that 20–60 is a suitable proportion range for the mixture of the product and water, and the water absorbency can be selected appropriately according to the practical application environment (such as climate and rainfall).
Test results of swelling kinetics and viscosity of the products.
The film-forming hardness and peeling strength tests
The film-forming hardness and peeling strength of the product were measured in single-factor experiments. The experiment products of Group 1–11 were selected and the variations of shore hardness and peeling strength were investigated for the products containing different proportions of acrylic acid and acrylamide. The results are shown in Figure 6.
Test results of film-forming hardness and peel strength of the end products.
It can be seen that the content of acrylamide strongly affects the film-forming hardness of the product. With higher acrylamide content, the product has a more compact structure and an enhanced interaction force, leading to an increase in film-forming hardness. With an optimal dosage of acrylamide, the shore hardness of the product can reach the maximum (85 HA). When the dosage of acrylamide exceeds the optimal content, the percent grafting and grafting efficiency decrease and the molecular structure gets looser; thus, the film-forming hardness declines. Because the cross-linking agent is the basis for the end product, the acrylic acid’s content can definitely affect the final film-forming hardness. The effects induced by the variation of the acrylic content are relatively smooth and less obvious than those induced by the variation of acrylamide content. By contrast, the content of the cross-linking agent (acrylic acid) has a more significant effect on the peel strength of the end product. As the acrylic acid increases, the cross-linking degree increases gradually, which provides additional free polymer chains for the subsequent graft copolymerization and thus increases the ductility of the end product. When the acrylic acid exceeds the optimal content, although the cross-linking degree increases, the effective cross-linking between lignin and acrylic acid decreases, i.e. the self-cross-linking of acrylic acid gradually plays a dominant role. Therefore, in cross-linking products, both the length and strength of the polymer chain drop. Hence, the molecular structure becomes looser and the intermolecular force is weakened dramatically. The acrylamide content also affects the peel strength of the end product to a certain degree; however, the effects are not obvious as a stable, cross-linking product has been produced. In conclusion, the content of acrylic acid has a more significant effect on the peel strength of the end product.
The suppression performances tests
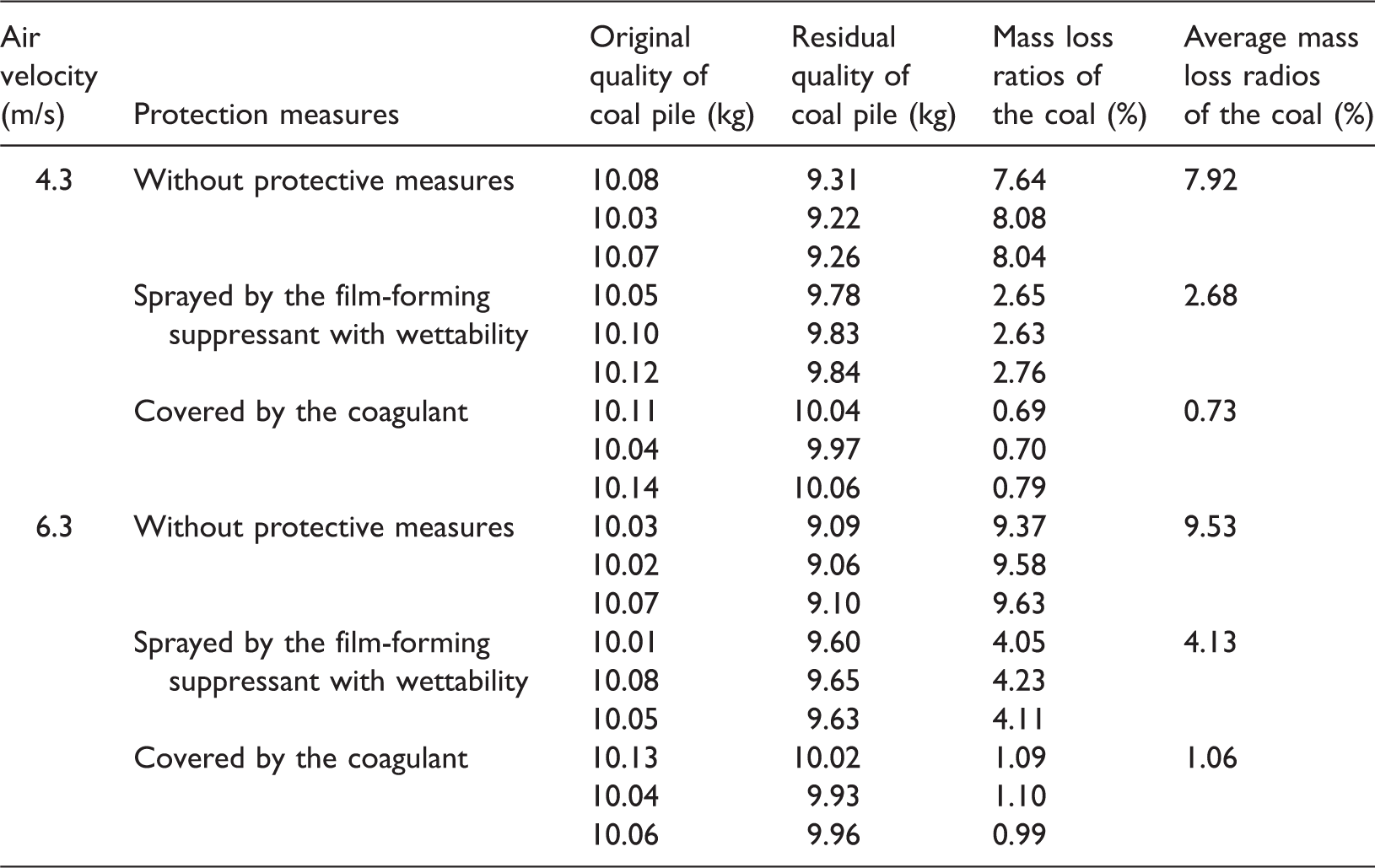
Finally, the mass loss ratios of two coal heaps under different wind speed conditions were measured to examine the dust suppression performance of the end product. The measurements were repeated several times and the average values were calculated and are listed in Table 3. Figure 7 shows the pictures of the coal heap before and after different treatments.
Pictures of the coal pile without and under different treatments. (a) Picture of the primary coal pile, (b) picture of the coal pile after being wetted, (c) picture of the coal pile after being coated with the coagulant, and (d) picture of the coal pile after the application of coagulant.
Measurements of coal’s mass loss ratio under different conditions.
Conclusions
In this study, a type of coagulant that can be used to suppress coal dust in open-cast mines was synthesized. To prepare this coagulant, a SLS was firstly cross-linked with acrylic acid, and then the cross-linked product was grafted with acrylamide. The fact that the cross-linking reaction and graft copolymerization took place was confirmed by the FT-IR spectra of the intermediate product and end product. According to the XRD results, the crystal structure was gradually destroyed during the reaction process, so that the end product was an amorphous polymer. Additionally, the DSC analysis results demonstrated that the dust suppressant developed has a favorable thermal stability. Moreover, single-factor tests were conducted to investigate the effects of single factors on the performance of this dust coagulant. It can be concluded that the developed coagulant can achieve its optimal dust suppression performances when the following requirements are met: a 1:3 mass ratio of lignin to acrylic acid, a 2:7 mass ratio of lignin to acrylamide, a 0.9% content of the cross-linking agent, a 2% mass fraction of initiator to acrylamide, a reaction temperature of 60℃, and a pH value of 7. Moreover, the overall performance of the coagulant was also measured, and the following conclusions are drawn from the test results. The swelling kinetics of the coagulant is related to its viscosity; the appropriate water absorbency can be selected in accordance with the application environment; the prepared coagulant has a large film-forming hardness, as it can effectively prevent the coal from being dispersed by an external wind; and the coagulant has a strong dust adsorption ability, a modest peel strength, and a promising ability to suppress dust.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is funded by the National Natural Science Foundation of China (Grant no. 51474139), the SDUST Research Fund (Grant no. 2014JQJH106), the Key Research and Development Program of Shandong Province (Grant no. 2016GSF120002), the Qingdao City Science and Technology Project (Grant no. 16-6-2-52-nsh), the China Postdoctoral Science Foundation Funded Special Project (Grant no. 2016T90642), the China Postdoctoral Science Foundation Funded Project (Grant no. 2015M570602), the Open Fund of the Key Laboratory of Coal Mine Gas and Fire Prevention and Control of Ministry of Education (Grant no. 2015KJZX02), and the Qingdao Postdoctoral Applied Research Project (Grant no. 2015194).

