Abstract
In the current study, synthesis of silver nanoparticles/chitosan/poly vinyl alcohol/poly ethylene glycol (AgNPs/Cs/PVA/PEG) hydrogel nanocomposite and finishing of cotton nasal tampons were reported via the γ-irradiation procedure. The uniform size distribution of nanoparticles and the presence of polymer nanocomposite were confirmed by energy dispersive spectroscopy (EDS) and field emission scanning electron microscope (FE-SEM), respectively. Z-average size of formed (Ag/Cs/PVA/PEG) hydrogel nanocomposite was about 96 nm. The water and simulated body fluid retention capacity of the modified tampons were assessed. The blood coagulation capability of the modified tampons was evaluated through Duke bleeding time test. The antibacterial evaluation of the modified tampons showed good antibacterial activity against
Introduction
Rhinoplasty and septoplasty are common surgeries that require nasal tampon usage. Nasal tampons can lead to serious infection such as toxic shock syndrome. On the other hand, nasal tampons should have soft surfaces to minimize mucosal adhesion, improve wound healing, and increase patient comfort. Therefore, a proper nasal packing can lead to create a moist and clean milieu for wound healing and avoiding dryness [1,2].
During the fast growth of nanoscience, the synthesis of metal nanoparticles (NP)/polymer composite was considered to be an emerging area of nanotechnology. Among the metal nanoparticles, silver nanoparticle (AgNP) has shown an enormous potential in different applications due to its interesting features, such as antimicrobial nature, biocompatibility, conductivity, and chemical stability [3,4]. Biomedical applications, filters, textiles, catalysts, sensors, and water treatment are some of the applications where the combination of all such features is reported [5,6]. Many synthetic procedures containing physical, chemical, and biological methods for reducing the silver salts and formation of AgNPs are available [7–11]. AgNPs can be obtained through the chemical synthesis, but these methods use toxic chemicals, which raises enormous concern for the environmental reasons. In this respect, the green synthesis of AgNPs using polysaccharide, tollens, poly oxomatalates, microorganisms, and irradiation was suggested. Irradiation technology is one such method, by which it is possible to synthesize nanoparticles without using any toxic chemical reducing agents or any complicated techniques. Unlike chemical methods, the irradiation technique is free from contamination and does not require any cross-linkers or chemical initiators. Therefore, this clean, environmental friendly and biocompatible method can be considered for the biomedical applications [3,7,12–14].
Uniform distribution, long stability and no tendency to agglomerate in an aqueous solution are the main indices of a successful synthesis method of nanoparticles [15]. Increase in the size or formation of the large clusters due to the agglomerate of small nanoparticles causes to reduce the effectiveness of these nanoparticles. Among the different methods for stabilization of metallic nanoparticles, the application of polymer as a good host matrix, in order to uniform dispensation and inhibition from agglomeration, has attracted much attention [3]. Dispersion of the previously prepared nanoparticles or in situ synthesis of metal nanoparticles in polymer matrix and simultaneous polymerization and metal reduction are the main possible fabrication approaches for the preparation of a polymer/metal nanocomposite [16–18]. Nanocomposites consisting of the metal nanoparticles and hydrogels, due to the three dimensional and hydrophilic nature and stabilization of the nanoparticles in the polymeric network, have attracted a lot of attention [19].
Among the hydrogel systems containing AgNPs, the blend of synthetic polymers like polyvinyl alcohol (PVA), polyvinyl pyrollidone (PVP), and biopolymers like chitosan (CS), carboxymethyl cellulose, and sodium alginate have been more investigated for the antibacterial purposes [5,20–23]. PVA, as a bio-friendly and water soluble polymer, has extremely low cytotoxicity. Therefore, it is frequently used in the biomedical applications, clothing, and household products. On the other hand, CS has shown remarkable potential as a biomedical polymer due to its interesting features such as antimicrobial and blood coagulation nature, biocompatibility, biodegradability, and scar prevention [24]. PVA and CS are used as the reducing agents of silver salts and stabilize AgNPs, too. In order to improve PVA/CS composite features, different types of polymers such as alginate, gelatin, and polyethylene glycol (PEG) have been utilized. The hydrogel composites which is made from PVA, CS, and PEG is not only biodegradable but also is more flexible [25,26]. The use of γ-irradiation as well as forming AgNPs can be utilized to form the hydrogel network. In addition to these, the γ-radiation process can be optimized to form a sterilized hydrogel system. If this procedure is done on the final product, therefore it can be used for various biomedical applications [27]. The new approach in textile engineering is the utilization of functional materials that are able to modify the conventional textile and achieve high performance textiles. The most widely used hydrogels in textiles are the antimicrobial hydrogels or hydrogels with the antimicrobial agents embedded in the polymeric networks. Among the hydrogels, CS, and PVA are more favorable [28].
In the present research, in situ synthesis of AgNPs/CS/PVA/PEG hydrogel nanocomposites via γ-irradiation method and simultaneous surface modification of the cotton nasal tampons with this hydrogel system has been investigated and the new features of the modified tampons are presented.
Experimental
Materials
CS (medium molecular weight, 75–85% deacetylated, 200–800 cP, 1 wt% in 1% acetic acid) and silver nitrate (AgNO3, ACS reagent, ≥ 99.0%) were purchased from Aldrich Chemical Co. PVA (Mw = 72000) was obtained from Merck Chemical Co. PEG (Mw = 400), acetic acid (Merck), and all other chemicals were analytical graded and used without further purification. Cotton nasal tampons were supplied by Sigma, Germany.
Preparation of AgNPs/CS/PVA/PEG hydrogel nanocomposite on nasal tampons
Different concentrations of prepared samples.

Cotton tampons were put in polyethylene bags with the AgNO3/CS/PVA/PEG solution and irradiated by CO-60 gamma ray source model (GC-220) activity 13922 ci, to the total dose of 25 kGy with a dose rate of 3.41 Gy/s. Finally, the impregnated tampons were padded in order to remove the excess hydrogel on fabric and dried at the room temperature.
The modified tampons should be washed at 60℃ for 45 min in order to remove any hydrogel systems that are not bonded to the fabric and dried at the room temperature and kept in the desiccator.
The irradiated samples without AgNO3 (samples 7, 8, 9) were prepared as the blank samples for UV–vis spectrophotometer evaluation.
Characterization
The presence of the AgNPs and the morphology of the modified samples were determined by field emission scanning electron microscope (FE-SEM), and energy dispersive spectroscopy (EDS) (ZEISS, ∑IGMA, VP). The small centerpiece of the fabric sample (5 mm × 5 mm) was cut, mounted on a sample holder, and was coated without further preparation. The size distribution of nanoparticles in hydrogel dispersion was determined using a Zetasizer Nano ZS system (Malvern, UK). The hydrogel dispersion was diluted 200 times for using a dynamic light scattering (DLS) method. The UV–visible spectroscopic measurements were carried out using UV-1601 Ray Leigh UV–visible spectrometer in the range 250–600 nm.
Fourier transform infrared (FTIR, Nicolet, model: NEXUS 670) was utilized to characterize the chemical structure of the CS/PVA/PEG hydrogel and AgNPs/CS/PVA/PEG composites.
Measurement of the water and SBF retention capacity of the modified tampons
The water retention capacity (WRC) of the modified tampons was measured according to the standard method D2402-centrifuge procedure. The WRC of the fabric is earned from the difference in masses between the centrifuged and dry samples following equation (1)

A specimen is throughly wetted-out by immersion in water and centrifuged for 5 min at the acceleration of 9800 m/s2. At the end, the specimen is dried and reweighed [29].
Reagents for preparing SBF (pH 7, 1 L) [30].

The same procedure was done for measuring SBF retention capacity (SBF.RC) of the modified samples and SBF.RC of the samples were measured according to equation (2).

All presented values for WRC and SBF.RC are the mean value of three parallel measurements.
The blood clotting test of the modified cotton tampons
The blood clotting activity (BCA) of the modified samples was evaluated in comparison with the control sample. The BCA evaluation was performed according to Duke bleeding time method. BCA was evaluated as follows:
The ear lobe was pricked using the lancet and the blood was made to flow continuously. At the time of flowing blood, the stopwatch was started simultaneously. One fabric strip was blotted with blood drops after every 30 s time intervals. The same procedure was repeated for other modified and control strips. The time between starting stopwatch and the end of the bleeding was recorded as Duke bleeding time [31].
Antibacterial activity of the modified cotton tampons
The modified colony counting method (AATCC 100-2004) and parallel streak method (AATCC 147-2004) on
The reduction of the number of colonies (%) in the treated samples, in comparison with the control sample, was used to determine the antibacterial activity of the AgNPs/CS/PVA/PEG coated fabric. The antibacterial activity was determined using equation (3).

The average width of a zone of inhibition along a streak on either side of the specimen was calculated using equation (4):
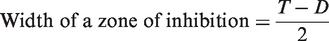
Results and discussion
Synthesis of the AgNPs/CS/PVA/PEG hydrogel nanocomposite
Hydrogen and hydroxyl radicals (H˙, OH˙), hydrated electrons (eaq−), and other chemicals such as H3O+ and H2O2 are the main products of γ-irradiated aqueous solutions (equation (5)).
OH˙ is an oxidizing agent, while H˙ and eaq− are the reducing agents. Therefore, H˙ and eaq− can reduce silver ions (Ag+) to neutral Ag atoms (Ag0) and other radical compounds participate in the crosslinking procedures of hydrogel network and formation of free radicals on the γ-irradiated cotton fabric (equation (6)). The combination of Ag0 atoms with trapped Ag ions in the polymer networks led to the formation of silver clusters (equation (7)) [33].

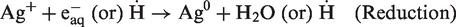

When these agglomerated Ag clusters are trapped in the hydrogel network (which are formed by the inter/intra molecular and H-bonds of carbonyl, hydroxyl, and amine groups of CS, PVA, and PEG polymer and hydroxyl group of cellulose), they are converted into the AgNPs.
On the radiation-induced crosslinking of PVA, CS, and PEG polymers, H and OH radicals play an important role in the crosslinking reaction. The crosslinking may be occurred by coupling of two free radicals obtained from the polymer radicals which are produced due to the hydrogen abstraction from the initial polymer chain by H and OH radicals (equations (8), (9), (10), (12)) or by the reaction of polymer radical (Pċ) prepared from the direct action of radiation (equations (11), (12)) [34].




The introduction of a polysaccharide such as CS would reduce the crosslinking density, crystallinity, and hydrogen bonds of PVA hydrogel. It is observed that the oxidizing species especially OHċ radicals play an important role in the crosslinking of PVA and degradation of polysaccharides. The presence of CS in PVA hydrogel matrix leads to decrease the probability of recombination and crosslinking density. Therefore, more free volumes form in the polymer network and more water can be absorbed in this hydrogel [27].
Earlier research confirmed that when cotton as a cellulosic fiber is subjected to γ-irradiation, the number of carboxyl and carbonyl groups by the influence of high energy γ-rays increases. Indeed, γ-irradiation, due to the formation of reducing and carboxylic groups, causes the depolymerization of cellulose chain and formation of the oxidative degradation of polymer chain [35].
So, due to the formation of active groups on the cellulose chains, crosslinking of hydrogel network to the cotton surface without any further chemical agents is possible. Figure 1 shows the schematic for the surface modification of cotton fabric with AgNPs/CS/PVA/PEG hydrogel nano composite via γ-irradiation.
Surface modification of cotton fabric with AgNPs/CS/PVA/PEG hydrogel nanocomposite via γ-irradiation.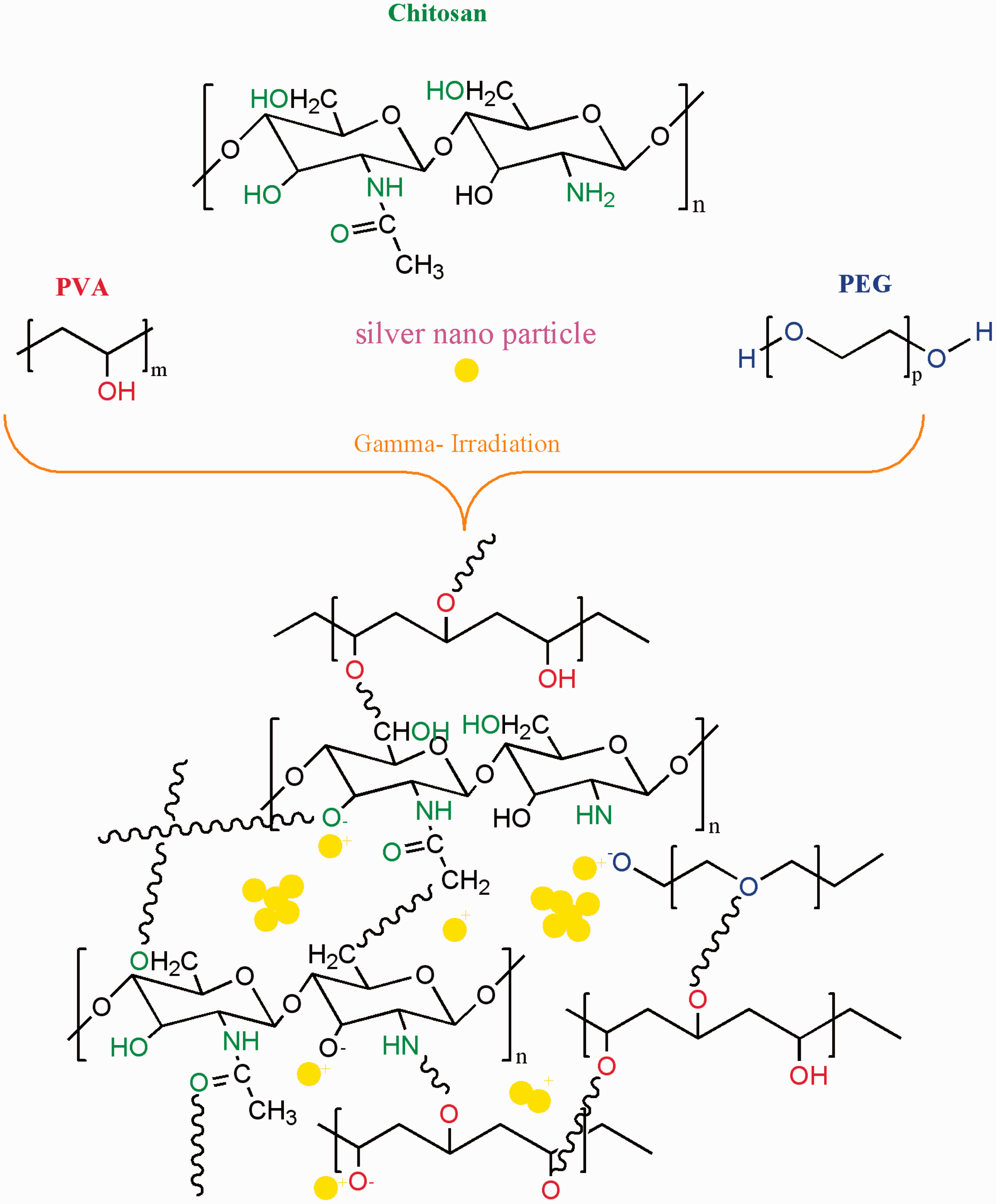
AgNPs have a surface plasmon resonance (SPR) absorption in the UV–visible region. When the size of AgNPs is much smaller than the wavelength of visible light, the colloidal solution has a yellow color and a sharp absorption peak in the 380–400 nm. The shift in absorption band is dependent on the chemical structure, shape, and size of the nanoparticles.
Figure 2 shows the color of γ-irradiated AgNPs/CS/PVA/PEG hydrogel colloidal solutions. The color of the samples changed from colorless to yellow, light brown, and reddish brown.
The effect of process parameters on the color of AgNPs/CS/PVA/PEG hydrogels.
According to Table 1, it seems that the increase in the amount of silver in the reaction solution has led to the color changes from yellow to red and reddish brown.
Characterization of AgNPs/CS/PVA/PEG hydrogel nanocomposite
Due to the SPR effect of AgNPs, the physical features of the nanoparticles can be obtained through analyzing the spectral properties of AgNPs in the solution. The spectral response of AgNPs is shown in Figure 3.
Optical absorption spectra of AgNPs/CS/PVA hydrogel composites.
It can be clearly seen that the spectrum of reference hydrogel (without silver nitrate) does not show any characteristic peak which indicates the absence of silver nanostructures. However, the samples with Ag content exhibit surface plasmon resonant absorption around 400 nm indicating the presence of AgNPs. With an increase in Ag content (samples 3, 4 in comparison with samples 1, 2), a significant enhancement in the SPR intensity has been observed.
In order to assess the aggregation state of the nanoparticles, the effective size of the nanoparticles in the solution is measured by DLS technique. DLS showed a narrow distribution size with an average of 96 nm (Figure 4).
Particle size distribution of AgNPs/CS/PVA hydrogel composites (sample 6) analyzed by DLS method.
The FTIR spectra of PVA, PEG, CS/PVA/PEG hydrogel, and AgNPs/CS/PVA/PEG hydrogel were evaluated (Figure 5).
FTIR spectra of PVA, PEG, CS/PVA/PEG hydrogel, and AgNPs/CS/PVA/PEG hydrogel.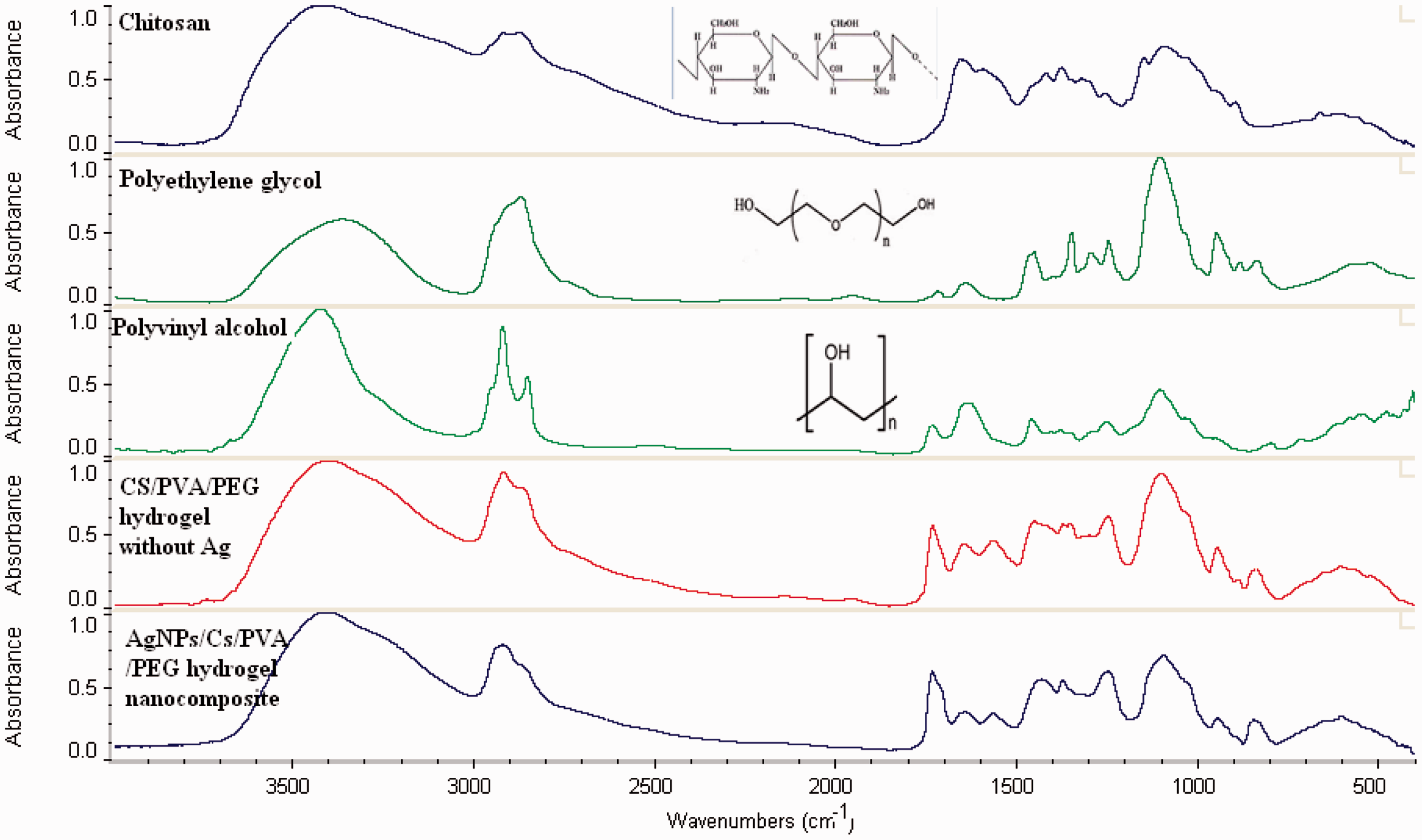
CS has the broad absorption peak at 3436 cm−1 belonging to N–H symmetrical vibration. Peaks at 2800 and 2900 cm−1 are due to the C–H stretch vibrations. The peaks observed at 1651 and 1322 cm−1 have been assigned to amide I and II. PVA has the peak around 2840–3000 cm−1, indicating the presence of the stretching C–H from alkyl groups. For CS/PVA/PEG hydrogel, a peak appeared at 3410 cm−1 indicates the presence of hydrogen bonds between NH2 (CS) and OH (PVA). Two bands observed at 2922 and 2871 cm−1 correspond to the CH2 stretching and C–H bond of chitosn–PVA and CS chains , respectively. There is an absorption peak at 1103 cm−1 related to –C–O–C– stretching vibration that confirms the crosslinking between the polymers. In the case of AgNPs embedded hydrogel, the Ag ions and electron rich groups of NH2 and OH groups formed co-ordination bond. This leads to the slight shifting frequency and increase of the intensity which confirmed the incorporation of AgNPs within the hydrogel network [4,20,25].
Characterization of surface modified tampons with AgNPs/CS/PVA/PEG
FE-SEM micrographs of the untreated and treated tampons with AgNPs/CS/PVA/PEG hydrogel nanocomposite are depicted in Figure 6(a) and (b), respectively. Hydrogel nanocomposites are clearly seen attached to the surfaces of the treated fabrics in Figure 6(b), whereas the untreated fabric has the smooth surface (Figure 6(a)).
FE-SEM micrographs of the untreated (a) and treated cotton fabric with AgNPs/CS/PVA/PEG hydrogel nanocomposite (b,c), the EDS spectrum of the Ag nanoparticles (d), and the elemental mapping of Ag on fabric (e).
The incorporation of AgNPs in the treated cotton tampon was also verified by EDS. Figure 6(d and e) presents the EDS spectrum of AgNPs and the elemental mapping of Ag on the treated cotton tampon, repectively, which can show the incorporation of AgNPs in the hydrogel matrix.
In order to investigate the absorption feature of the modified cotton tampon, the WRC and SBF.RC of the treated fabrics were evaluated. There are many factors that can influence on the absorption capacity of hydrogels, such as the hydrophilicity feature of materials, crosslinking density, temperature, pH value, and ionic strength [36].
The water and SBF retention capacity of the cotton fabric treated with hydrogel nanocomposite as a function of CS and silver nitrate concentrations are shown in Figure 7(a and b), respectively.
WRC (a) and SBF.RC (b) of the modified cotton fabric with AgNPs/CS/PVA/PEG hydrogel nanocomposite.
It is worth noting that the samples with more CS contents show the larger water/SBF retention capacity. This may be attributed by the effect of γ-irradiation on the polymers. CS biopolymer with the decrease in probability of radical recombination leads to reduce the crosslinking density and consequently increase of the free volumes in the polymer network. It causes the entering of large amount of water/SBF into the hydrogel network. On the other hands, due to the ionic charge of CS and osmotic pressure, more amount of water/SBF can be absorbed. The retention capacity of the fabrics in SBF solution was appreciably decreased compared to the values measured in distilled water.
Forasmuch as Na+, K+, Mg2+ , Ca2+, Cl−, HCO3−, HPO42−, and SO42− are the major ions present in SBF solution or blood plasma [30], the swelling phenomenon of ionic hydrogels (such as CS) due to a screening effect of the additional cations causes a non-perfect anionic electrostatic repulsion and led to the decrease of the osmotic pressure difference between hydrogel network and surrounding solution [37].
In order to evaluate the impact of AgNPs, it can be seen that the modified tampon with hydrogel containing AgNPs have a lower water/SBF retention capacity than the reference sample (WRC ∼ 22%). Because of the formation of many crosslinks between the AgNPs and the electron rich O and N atoms of ether and amino groups of hydrogel nanocomposite, the fluid penetration into the polymer network is reduced and it causes the decrease of the amount of water/SBF retention capacity in the related sample [4,20].
When blood vessels are cut or damaged, the loss of blood from the system must be stopped as soon as possible. Application of CS as a hemostatic biopolymer on medical textiles can be effectively used in the medical treatment to reduce the blood loss and induce faster wound healing. The blood coagulation ability of the treated samples was investigated based on Duke bleeding time. As it is shown in Table 3 and Figure 8, the number of blood drops and the blood coagulation time of the modified tampons with hydrogel nanocomposite containing 1% and 2% CS is reduced in comparison with the control sample.
Blood clotting test on control (a), fabric treated with 1% (b), and 1.5% chitosan (c). Blood coagulation test on the treated fabric.

The comparative study of blood coagulation activity of the samples revealed the effectiveness of CS on the bleeding time. There are several reports that refer to the hemostatic function of CS [38]. This cationic biopolymer aggregates the red blood cells which have negative charge and reduces the blood clotting time. In addition to the hemostatic effect of CS, AgNPs were beneficial to the reduction of the blood coagulation time [39].
Figure 9 shows the antibacterial activity studies of the modified tampons with AgNPs/CS/PVA/PEG hydrogel nanocomposite. Figure 9(a) shows the bactericidal efficiency of modified cotton fabric against gram-positive Antimicrobial studies of the modified samples against 
Antibacterial finishing of nasal tampons was assessed via AATCC 100 as a quantitative method. The obtained results confirm that this functional finishing on textile could lead to the formation of about 99% and 98% antibacterial effect against
The interaction between positively charged CS molecules (the protonated NH3+ groups) and negatively charged bacterial cell membranes is the most acceptable antibacterial mechanism of CS, it cause to the membrane wall permeability and internal osmotic imbalances and finally, the inhibition of microorganisms growth [40]. On the other hands, AgNPs have the ability to anchor and subsequently penetrate the bacterial cell wall, as well as the formation of free radicals by AgNPs in contact with the bacteria damage the cell membrane, and the release of silver ions by the AgNPs can interact with the thiol groups of vital enzymes of microorganism and inactivate them and ultimately lead to cell death [41].
Conclusion
We have developed a green method for in situ synthesis of AgNPs/CS/PVA/PEG hydrogel nanocomposite. In fact, the formation of hydrogel nanocomposite, and synthesis of AgNPs applying of cotton nasal tampons were performed in a single step by the in situ reduction in a γ-irradiation method. The results showed the AgNPs were synthesized without using any reducing agent. The Z-average of hydrogel nanocomposite was about 96 nm. The WRC and SBF.RC of the modified nasal tampons increase with the increase of the CS concentration. The blood coagulation studies revealed that the modified tampons with hydrogel nanocomposite reduced the bleeding time. Furthermore, this product showed an excellent potential for the antibacterial application such as biomedical textiles, wound dressing, and surgical gown. This method can be considered to be a green approach for the synthesis of silver hydrogel nanocomposite.
Footnotes
Acknowledgement
We would like to thank Dr Nasim Kashef from Tehran University (Department of Microbiology, School of Biology), for her help in the antibacterial evaluation.
